Abstract
Objective
Sepsis remains a critical global healthcare challenge with high mortality rates, where traditional diagnostics lack sensitivity and standardized treatments show limited efficacy due to patient variability. While artificial intelligence (AI) has advanced sepsis diagnosis and management, systematic bibliometric analyses are scarce, particularly regarding recent research trends from 2022 to 2025. This study aims to reveal research development patterns and frontier dynamics of AI applications in sepsis diagnosis and management, providing strategic guidance and decision support for researchers, clinicians, and policymakers in this rapidly evolving field.
Methods
Web of Science data related to AI in sepsis published from 2005 to 2025 were retrieved. Multidimensional bibliometric and visualization analyses were conducted using VOSviewer, CiteSpace, and Bibliometrix packages.
Results
AI applications in sepsis research demonstrated exponential growth since 2016, peaking at 352 publications and 1363 citations in 2024. The United States dominates both publication volume (487 papers) and citation impact (12,984 citations), while China ranks second in output (457 papers) but shows significantly lower impact (3929 citations). Research clustered into six major directions encompassing intelligent prediction, molecular mechanisms, and deep learning early warning systems. Post-2022 analysis revealed emerging keywords including “ARDS (Acute Respiratory Distress Syndrome),” “MIMIC-IV,” and “immune infiltration,” signaling shifts toward multi-omics integration and precision medicine. The most highly cited studies focused on sepsis phenotypic subtyping (776 citations) and AI treatment strategies (619 citations).
Conclusion
AI applications in sepsis research are transitioning from algorithm validation toward clinical application, developing in the direction of explainable AI and precision sepsis care. The research trajectory evolves from “proof of concept” toward “ensuring clinical utility.” These findings provide strategic guidance for healthcare systems, highlighting the importance of interpretable models, multi-center validation, and infrastructure development for next-generation precision sepsis management.
Introduction
Sepsis is a life-threatening organ dysfunction caused by dysregulated host response to infection, often leading to shock and representing a major global critical care challenge. Recent studies show sepsis affects approximately 50 million patients annually worldwide with nearly 20% mortality, making it a critical clinical problem in intensive care medicine. 1 Traditional diagnosis using Sequential Organ Failure Assessment (SOFA)/quick Sequential Organ Failure Assessment (qSOFA) scores and inflammatory markers (C-reactive protein, activated partial thromboplastin time) faces significant limitations: SOFA requires laboratory data, qSOFA has poor sensitivity for early detection, and inflammatory markers lack specificity.1–3 Treatment management faces challenges from patient heterogeneity (pathogen type, immune status, comorbidities) causing variable therapeutic outcomes,4–6 with delayed decision-making further increasing clinical uncertainty. 6
The rapid advancement of artificial intelligence (AI) technologies has opened new frontiers in comprehensive sepsis management, with researchers increasingly exploring machine learning applications for clinical decision support. AI technology integrates multimodal vital signs with sophisticated algorithms to achieve breakthrough advances in early warning, precision diagnosis, and dynamic management. Barton et al. 7 developed an XGBoost model predicting sepsis 48 h early using basic vital signs with superior accuracy (AUC = 0.830). Komorowski et al. 8 applied reinforcement learning for individualized fluid and vasopressor recommendations, showing the lowest mortality when physician prescribing aligned with AI recommendations.
Despite these promising developments, current AI applications in sepsis management face critical limitations that impede widespread clinical adoption. Most existing models demonstrate poor generalizability across different healthcare institutions due to variations in data collection protocols and clinical workflows, while the “black box” nature of advanced algorithms creates transparency challenges that hinder clinician trust and regulatory approval. 9 The explosive growth in AI-sepsis research has generated extensive literature encompassing diverse methodological approaches and clinical applications. However, traditional narrative and systematic reviews face inherent limitations in capturing broader research landscapes and identifying emerging trends across this rapidly evolving field. 10
Bibliometric analysis offers a powerful quantitative approach to address these analytical gaps by systematically examining academic literature through statistical methods, enabling researchers to identify trends, map collaboration networks, and analyze citation patterns within scientific fields through data-driven approaches. 11 While bibliometric analysis of AI applications in critical care medicine has revealed geographical disparities in research contributions and identified key research hotspots, 12 specialized analyses focusing exclusively on AI applications in sepsis remain scarce, particularly those capturing the most recent developments through 2025. This gap is significant since sepsis represents a distinct clinical entity with unique pathophysiological mechanisms and therapeutic considerations that differ from other critical care conditions.1,13 More importantly, the field has undergone rapid transformation in recent years—driven by new diagnostic criteria (Sepsis-3), the Coronavirus Disease 2019 (COVID-19) pandemic, and advances in explainable AI—changes that have not been captured by previous bibliometric studies.
This study provides the first comprehensive bibliometric analysis specifically dedicated to AI in sepsis spanning two decades (2005–2025), systematically searching Web of Science Core Collection for sepsis AI literature. Unlike previous broader analyses, we employ a multi-dimensional analytical framework using three complementary tools—VOSviewer, CiteSpace, and R “Bibliometrix"—to construct knowledge maps and collaboration networks that reveal not only publication patterns but also the critical paradigm shift from algorithm development to clinical implementation. Our analysis uniquely captures the post-2022 acceleration phase, identifying emerging trends in multi-omics integration, explainable AI adoption, and the transition from “proof of concept” to “ensuring clinical utility.” This analysis provides strategic insights for researchers and policymakers navigating this rapidly evolving field.
Data sources and methods
Study design and setting
This was a retrospective descriptive bibliometric analysis conducted at Guangdong Medical University and The First People's Hospital of Foshan between January 2025 to June 2025. The study analyzed published literature in the field of AI applications in sepsis diagnosis and management spanning a 20-year period.
Data source and search strategy
In this paper, all bibliographic data are derived from the Web of Science Core Collection database,14,15 which encompasses academic publications across more than 250 different disciplines worldwide, and whose effectiveness has been demonstrated by numerous researchers who have conducted bibliometric analysis in the past. The time frame for the bibliometric analysis spans from April 1, 2005, to May 15, 2025. The search query used is as follows: TS = (“Artificial Intelligence” OR “Machine Intelligence” OR “Computational Intelligence” OR “Computer Vision System” OR “Knowledge Representation” OR “Machine Learning” OR “Deep Learning” OR “Neural Network” OR “Intelligent System” OR “Pattern Analysis” OR “Prediction Method” OR “Predictive Learning Model” OR “Generative Artificial Intelligence” OR “Chatbot” OR “ChatGPT”) AND TS = (“Sepsis” OR “Septicemia” OR “Bloodstream Infection” OR “Blood Poisoning” OR “Pyemia” OR “Pyaemia” OR “Systemic Inflammatory Response Syndrome” OR “SIRS” OR “Bacteremia” OR “Septic Shock”) AND TS = (“Diagnos*” OR “detect*” OR “predict*” OR “prognos*” OR “Early Warning Score” OR “early detection” OR “screening” OR “mortality predict*” OR “risk assess*” OR “risk stratif*” OR “decision making” OR “treatment” OR “management” OR “intervention” OR “monitoring" OR “outcome*” OR “clinical decision” OR “precision medicine” OR “personalized medicine” OR “biomarker*”). Only English-language documents from the aforementioned search results were included in the analysis.
Data analysis and statistical methods
The retrieved bibliographic records were systematically processed and exported as Excel and plain text files, capturing essential metadata including titles, authors, keywords, institutional affiliations, geographic regions, cited references, journal names, and publication dates. Comprehensive bibliometric analysis was conducted using three complementary software platforms: VOSviewer 16 (version 1.6.18) for network visualization and clustering analysis, CiteSpace17,18 (version 6.1.R6) for citation analysis and burst detection, and the Bibliometrix package19,20 in R for temporal trend analysis. Traditional statistical computations were performed using IBM SPSS 19.0 and GraphPad Prism 5.0 where appropriate. Network analysis parameters required careful calibration to balance comprehensiveness against interpretability. The minimum thresholds were systematically configured as follows: Author collaborations (≥4 occurrences), institutional collaborations (≥7), national collaborations (≥10), author-keyword co-occurrences (≥6), author co-citations (≥40), journal co-citations (≥75), and journal occurrences (≥5). These parameters were determined through iterative testing to optimize network clarity while preserving meaningful connections. Lower thresholds produced overly complex networks with excessively weak connections that obscured major collaboration patterns, while higher thresholds eliminated important secondary connections and fragmented network structure. Visual inspection of alternative threshold settings confirmed that primary clustering patterns and major collaborative relationships remained consistent across reasonable parameter ranges, though formal sensitivity analysis across all threshold combinations was beyond the scope of this study. In all network visualizations, node sizes correspond to frequency or citation counts, link thickness indicates collaboration strength or co-occurrence frequency, and cluster colors represent thematic groupings or collaborative communities. The analytical workflow integrated multiple complementary approaches: VOSviewer generated collaborative network maps for countries, institutions, authors, journals, and keywords, revealing collaboration patterns and knowledge architecture. CiteSpace conducted journal dual-map overlay analysis, co-citation network analysis, burst citation detection, and keyword burst analysis to identify emerging trends and developmental pathways. Bibliometrix provided temporal analysis of keyword evolution and research theme development over time. Statistical validation of network clustering employed modularity analysis (Q > 0.3) combined with silhouette coefficient analysis (>0.7) to ensure reliable identification of research communities. Where comparative analysis was conducted, results were evaluated using appropriate statistical tests (t-test or ANOVA) with significance set at P < 0.05. Quantitative data are reported as mean ± standard deviation where applicable, though the primary analytical framework relied on network-based metrics and visualization methods appropriate for bibliometric research.
Results
Publication and citation analysis
Figure 1(a) presents the annual publication output and citation trends in AI-based sepsis diagnosis and management research from 2005 to 2025. Prior to 2016, both publication volume and annual citation frequency remained relatively insignificant without notable growth trends. However, post-2016 witnessed marked increases in both metrics, with particularly dramatic surges between 2020 and 2024—reaching 352 publications and 1363 citations in 2024 alone. Furthermore, this study conducted polynomial regression analysis on cumulative annual publication volume, illustrated in Figure 1(b), with excellent goodness-of-fit (R² = 0.998). The regression curve demonstrates robust growth trajectory in AI-based sepsis diagnosis and management research.

Trends in the number of published articles and citation counts in the field of artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025. (a) The annual publication quantity and citation frequency in the field of AI-based sepsis diagnosis and management from 2005 to 2025. (b) The annual publication quantity, cumulative publication quantity, and their polynomial fitting curves for research on AI-based sepsis diagnosis and management from 2005 to 2025.
Countries/regions analysis
Table 1 summarizes the geographic distribution of research output in AI-based sepsis diagnosis and management, ranking the top ten countries/regions by publication volume and citation frequency. The USA leads with 487 publications and 12,984 citations, followed by China with 457 publications and 3929 citations. England ranks third with 94 publications and 3760 citations. Other significant contributors include Germany (68 publications, 1418 citations), Australia and the Netherlands (each with 53 publications, 1266 and 2461 citations, respectively). Figure 2(a) presents the global research landscape through network visualization using proportionally sized, color-coded national nodes representing publication volumes, with connecting lines indicating collaborations. The largest blue-purple node (USA) is followed by China, England, and Germany, matching the country ranking in Table 1. The USA emerges as the primary hub for international collaborations, with strong connectivity to England and Germany. While China demonstrates high publication output, its collaborations remain limited primarily to the USA. Figure 2(b) provides three-dimensional representation of geographical distribution, with brown connecting lines illustrating collaborative relationships. The most frequent academic exchanges occur between China and the United States, while Europe's complex interconnections establish it as another important academic hub.

Diagram of the network of national collaborations in the field of artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025. (a) The contributions of different countries to the research field of AI-based sepsis diagnosis and management can be represented by a visual diagram, with each country represented by a specific color band. The connections between these color bands are represented by lines, representing academic exchanges. The thickness of these lines is directly related to the frequency of scholarly exchanges between the countries involved. (b) Countries/regions collaboration map.
Ranking of the top ten major countries/regions in the field of artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025.

Author analysis
Table 2 ranks the top 10 scholars in AI-based sepsis research by publication volume and citation impact. Leading contributors include Kamaleswaran, Rishikesan (Duke University; 26 publications), Nemati, Shamim (University of California San Diego; 14 publications), and Clermont, Gilles (University of Pittsburgh; 12 publications). For citation impact, Singer, M (University College London) leads with 560 co-citations, followed by Vincent, Jl (Université Libre de Bruxelles), and Seymour, Cw (University of Pittsburgh), both exceeding 300 co-citations. Notably, USA scholars occupy 7 of the top 10 co-citation positions.
Ranking of the top ten major authors in the field of artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025.

Figure 3(a) illustrates the collaborative relationships among authors, where node size corresponds to publication count and connecting lines represent co-authorship relationships. Through network analysis, authors are categorized into eight distinct color-coded clusters, with detailed compositions provided in Supplemental Table S1. The analysis reveals clear geographic and collaboration patterns: USA-dominated clusters (blue, red, pink) exhibit high inter-cluster connectivity and active scholarly collaboration among centrally located research groups. The blue cluster, centered on Kamaleswaran, contains the highest-publishing authors and maintains extensive connections with the red cluster. Regional clusters show more focused collaboration patterns—the brown cluster (Taiwan/Asia-focused), purple cluster (Europe), green cluster (China), and cyan cluster display predominantly intra-cluster collaboration with limited international connections. The network visualization reveals that peripheral clusters maintain collaborative relationships primarily within domestic partnerships, while central clusters demonstrate greater integration in the global research network. When collaboration intensity metrics are overlaid on the original network (Figure 3(a) to produce Figure 3(b)), the red, pink, and blue clusters exhibit the densest co-author networks, demonstrating particularly intensive research collaboration patterns within these groups.

Author collaboration and citation network map in the field of AI-based sepsis diagnosis and management research from 2005 to 2025. (a) The diagram displays co-occurring authors in artificial intelligence (AI)-based sepsis diagnosis and management research, with nodes colored to represent distinct author clusters. Node size indicates the frequency of co-occurrence, and links depict the relationships among co-occurring authors. (b) The figure overlays the presentation of heat based on Figure 3(a), displaying the frequency of recent publication volume through different shades of color. (c) The diagram illustrates co-cited authors in research on AI-based sepsis diagnosis and management, with node size indicating citation frequency. This visualization, created using VosViewer, succinctly captures and analyzes the interconnectedness of cited authors in this research area. (d) The visual analysis depicts papers from the top ten authors ranked by frequency of occurrence from 2005 to 2025. In the visual analysis, the length of the line indicates the duration of continuous publication, the size of the dots indicates the number of papers in chronological order, and the color of the dots indicates the frequency of citations.
Co-cited authors are defined as scholars jointly referenced by multiple publications, typically indicating their engagement in related or complementary research domains within the same field. Analysis of these co-citation relationships can effectively reveal disciplinary research frontiers and uncover hidden connections across different specialties. As illustrated in Figure 3(c), the co-citation network demonstrates four distinct research clusters: The red cluster comprises Zhang Zh, Singer M and Rudd Ke; the blue cluster is predominantly represented by Collins Gs, Churpek Mm and Lundberg Sm; the green cluster consists of Henry Ke, Desautels T and Rhee C; while the yellow cluster primarily features Kumar A, Levy Mm and Dellinger Rp. This clustering pattern not only validates the methodological coherence within each research trajectory but also visually maps the intellectual structure of AI-enhanced sepsis research, revealing both concentrated specialization areas and potential interdisciplinary bridging opportunities.
Figure 3(d) displays publication timelines from 2014 to 2025, with line length indicating publication duration, dot size representing annual output, and color shade reflecting citation frequency. Clermont shows the longest continuous publication record, while Kamaleswaran achieved high output in 2021, 2023, and 2024 despite starting in 2018.
Institution analysis
Table 3 presents the top 10 institutions by publication volume and citation frequency in AI-based sepsis diagnosis research. Emory University leads in productivity (46 publications) while the University of California, San Francisco tops citation impact (1474 citations). Beyond these leading American institutions, China demonstrates significant research activity with four institutions in the publication rankings: Zhejiang University (29), Central South University (28), Southern Medical University (20), and Huazhong University of Science and Technology (19). England contributes notable citation influence through Imperial College London (1150 citations) and Cambridge University (783 citations). This distribution reveals clear US institutional dominance, with American universities occupying the remaining six publication positions and eight citation positions among the top 10 rankings.
Ranking of the top ten major institutions in the field of artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025.

Figure 4(a) displays the collaborative network structure through color-clustered institutional relationships organized into eight distinct clusters, with detailed characteristics outlined in Supplemental Table S2. The institutional collaboration patterns reveal distinct regional characteristics: Chinese institutions cluster predominantly in red and brown groups, Taiwanese institutions form the purple cluster, and South Korean institutions comprise the pink/yellow clusters. These regional clusters show strong intra-cluster connectivity but limited inter-cluster linkages with foreign institutions such as Emory University and Harvard Medical School. Conversely, USA-dominated clusters (blue, green, orange, cyan) exhibit robust inter-institutional connections, demonstrating intensive international academic exchange. The blue cluster encompasses top US institutions including Harvard Medical School, MIT, and Johns Hopkins University, while the green cluster contains Stanford University, UCSF, and Imperial College London, reflecting established USA-UK research partnerships.

Institutional collaborative network mapping in AI-based sepsis diagnosis and management research from 2005 to 2025. (a) The graph illustrates the co-occurrence of research institutions, with node size representing the frequency of their occurrence together and connections indicating co-occurrence relationships. The size of each node indicates how frequently research institutions collaborate, while the links represent instances of their joint occurrences. (b) The figure illustrates recent contributions of institutions to AI-based sepsis diagnosis and management research relative to their overall output. A red bias indicates increased influence, while a blue bias suggests decreased activity in the field. The color scale reflects the ratio of keywords during the two-year period from early 2021 to early 2023, highlighting institutions with significant impacts or reduced involvement in this study.
Figure 4(b) enhances this analysis by integrating temporal dimensions, revealing that recent entrants—University of Pittsburgh, Harvard Medical School, MIT, and green cluster institutions—demonstrate comparable scholarly interaction levels with established players.
Journal analysis
Table 4 summarizes the top 10 journals by publication volume and citation frequency in AI-based sepsis diagnosis and management. Scientific Reports leads with 67 articles, followed by Frontiers in Medicine (49 articles) and Frontiers in Immunology (47 articles). For citations, Critical Care Medicine ranks first (3075 citations), followed by JAMA (1922 citations, IF 63.5), Critical Care (1647 citations), Intensive Care Medicine (1511 citations), and PLOS ONE (1222 citations), while New England Journal of Medicine (989 citations, IF 96.3) ranks sixth.
Ranking of the top ten major journals in the field of artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025.
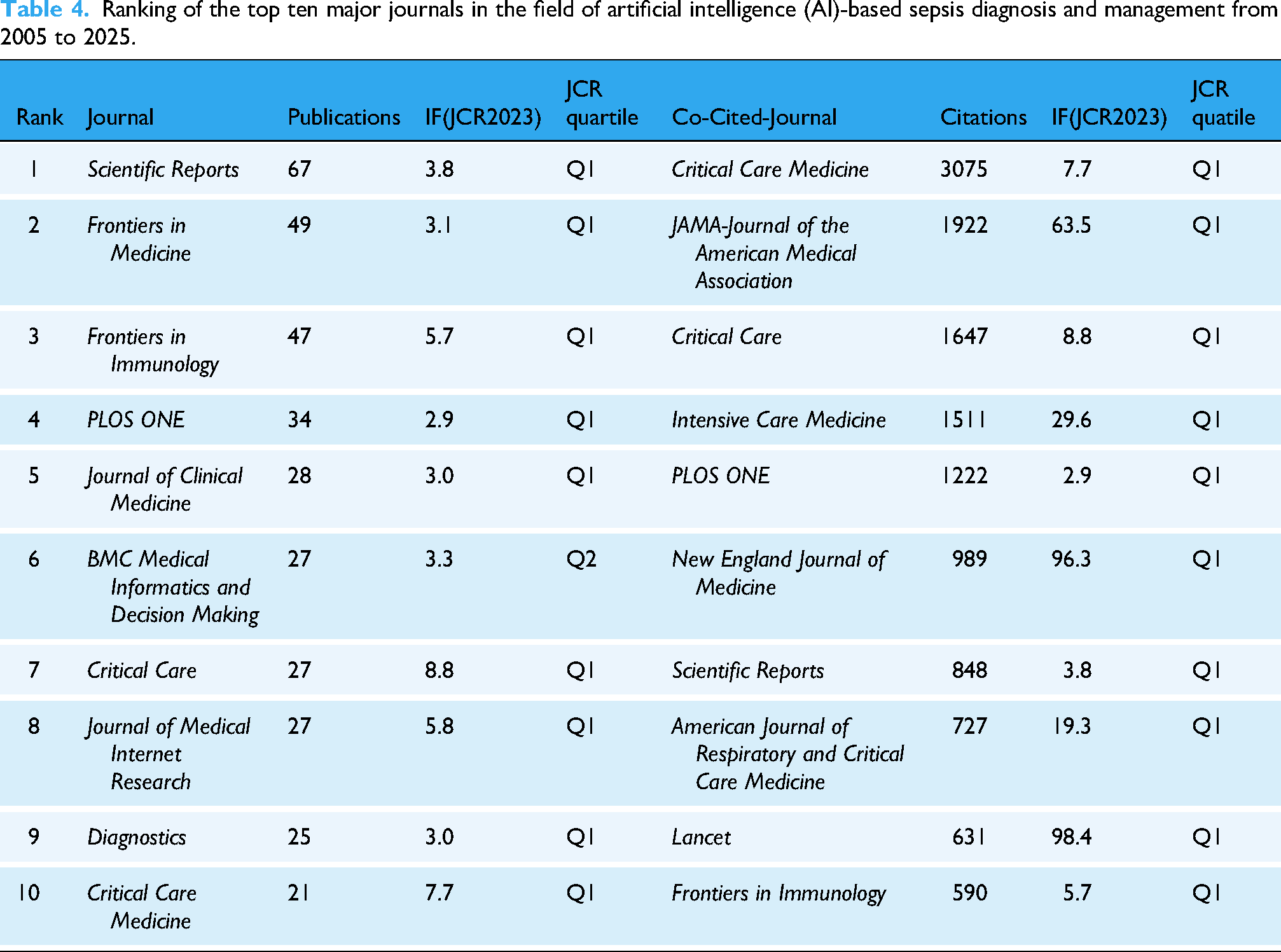
Figure 5(a) displays the journal network organized into seven distinct thematic clusters based on research focus, with detailed characteristics presented in Supplemental Table S3. The clustering analysis shows that journals group according to their research emphases in AI-sepsis research: The blue cluster focuses on multidisciplinary sepsis and critical care studies, the red cluster specializes in infection-immunity-critical care mechanisms, the yellow cluster concentrates on clinical decision support systems, the green cluster emphasizes healthcare information technology implementations, the cyan cluster highlights medical computing algorithm-hardware co-design, the orange cluster centers on real-time ICU intelligentization research, and the brown cluster focuses on diagnostic technology digital transformation.

Network visualization of journal publication volume, collaboration, and citation relationships in the field of artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025. (a) Visualization analysis of the collaboration network of journals in VOSviewer. Journals in different clusters are distinguished by nodes of different colors, with node size representing their frequency of occurrence. (b) The graph depicts the co-citation relationships among research journals, with node size indicating the frequency of co-occurrence and connections indicating co-citation relationships. Node size reflects the significance and influence of journals in the network.
Figure 5(b) presents the journal co-citation network, categorizing publications into four major research paradigms with characteristics detailed in Supplemental Table S4. The clustering patterns reveal distinct research approaches: The blue cluster includes highly cited clinical journals focusing on sepsis and septic shock management advancements, the red cluster comprises journals emphasizing AI-driven innovations and multimodal medical data integration, the green cluster represents journals focusing on multi-omics immune response analysis and computational immunology breakthroughs, and the yellow cluster consists of journals investigating clinical-microbiological aspects of infectious diseases. This clustering pattern reflects the diverse methodological approaches and research priorities within AI-based sepsis research, spanning from clinical management to computational innovation.
Figure 6(a) presents journal publication volumes according to Bradford's Law, with core journals including Scientific Reports, Frontiers in Medicine, and Frontiers in Immunology occupying important positions. Figure 6(b)'s dual-map overlay illustrates inter-journal citation relationships and research foci evolution through directional citation flows from citing journals (left) to cited journals (right). The prominent yellow-green connecting lines reveal significant interdisciplinary citation tendencies: Journals specializing in Molecular, Biological, and Immunological research primarily reference Molecular, Biological, and Genetic literature, while Medicine, Medical, and Clinical-oriented journals exhibit comprehensive citation patterns extending to Health, Nursing, and Medicine practice publications. Figure 6(c)'s correlation heat map indicates increased activity in Heliyon, BMC Medical Informatics, and PLOS ONE in recent years (2005–2025).

Publication, citation network, and heat map analysis of journals in the field of artificial intelligence (AI)-based sepsis diagnosis and management, 2000–2025. (a) Bradford's Law can be applied to academic journals in the field, as evidenced by the grey-shaded area in the figure. This area encompasses the journals that have made significant contributions to the field, arranged in descending order based on the number of articles. The visualization overlays the publication volume of journals, with color intensity indicating the volume of publications. (b) The dual map visualizes journals related to AI-based sepsis diagnosis and management, where clusters of citing journals are shown on the left side and cited journals on the right side. Colored trajectories between them represent citation relationships. (c) The heatmap illustrates the correlation among journals, grouping those with high content similarity during similar periods and displaying their publication trends over time on the timeline.
Keywords analysis
Keywords serve as pivotal indicators of research focus and core theme representations, making keyword analysis indispensable for comprehending research landscapes and predicting developmental trajectories. Table 5 presents the top 20 keywords by frequency and co-occurrence strength, with “machine learning” (1459 occurrences), “sepsis” (1182 occurrences), and “artificial intelligence” (488 occurrences) as the most prevalent terms.
Ranking of the top twenty major keywords in the field of AI-based sepsis diagnosis and management from 2005 to 2025.

AI: artificial intelligence; COVID-19: coronavirus disease 2019.
Figure 7(a) further visualizes the evolutionary trends of research hotspots in this field. It can be seen that before 2020, terms related to the integration of data science and clinical practice received relatively more attention, such as “Mortality prediction,” “Precision medicine,” and “Personalized medicine.” The impact of sepsis on neonates and children was also a hot topic of discussion, including terms like “Neonatal sepsis,” “Preterm infants,” and “Children.” By 2025, research focus shifted toward AI-driven sepsis diagnosis and management, emphasizing “Machine learning algorithm,” “Artificial intelligence,” “Early warning system,” and “Sepsis prediction.”

Keyword heatmap, correlation, and co-occurrence analysis of artificial intelligence (AI)-based sepsis diagnosis and management studies from 2005 to 2025. (a) The heatmap shows the keyword prevalence in AI-based sepsis diagnosis and management research, which is classified into different groups based on the prevalence of keywords in similar time periods and differentiated by color. (b) The figure demonstrates the correlation of keyword popularity, clustering keywords with similar peak periods of popularity together. (c) The keyword co-occurrence network illustrates the connections between studied keywords in AI-based sepsis diagnosis and management. Nodes, categorized by color, represent different clusters of keywords. Node size indicates the frequency of co-occurrence, while connections between nodes depict relationships among keywords. (d) A heat display based on Figure 7(c) is superimposed on the graph, showing the recent attention level of different keywords by different color shades.
Figure 7(b) shows keyword heatmap analysis divided into eight clusters based on correlation proximity. From top to bottom and left to right, the first purple cluster focuses on medicine-bioinformatics intersection (“antimicrobial resistance,” “risk-warning systems,” “transcriptome”). The next two clusters concern clinical evaluation and prediction in sepsis-related critical medicine (“SOFA,” “in-hospital mortality,” “sepsis prediction,” “deep neural network,” “diagnosis”). The fourth and fifth clusters focus on medical information management (“artificial neural network,” “electronic medical record,” “electronic health record,” “biomedical language processing”). The sixth cluster addresses bacterial infection diagnosis and management (“bacterial infection,” “bacteremia”). The final two clusters cover big data and AI applications in sepsis-related diseases (“cluster analysis,” “deep learning,” “predictive analytics,” “random forest”).
Figure 7(c) and (d) reveal the co-occurrence relationships and strength among keywords, uncovering latent connections and emerging trends in AI-sepsis research. The analysis identifies “machine learning” as the core keyword with the strongest co-occurrence intensity, followed by “sepsis,” reflecting the field's primary methodological and clinical focus areas. The keyword network segregates into six distinct thematic clusters (detailed in Table S5), each representing a specialized research domain. These clusters demonstrate a clear evolution from traditional clinical sepsis management (green cluster) toward sophisticated AI-driven approaches, encompassing machine learning applications in critical care (yellow cluster), deep learning-based early warning systems (red, light blue, and brown clusters), precision medicine implementations (dark blue cluster), and healthcare big data analytics (purple cluster). The co-occurrence strength distribution, when integrated with the visual analysis from Figure 5(b), reveals concentrated research attention on bridging clinical practice with technological innovation, particularly evident in the prominence of terms such as “intensive care unit,” “acute kidney injury,” and “precision medicine” within the network structure.
Figure 8 concisely presents the trend of discussion popularity of key keywords over time since 2005 from a temporal dimension. The horizontal axis represents the frequency or popularity of keyword occurrences, while the vertical axis serves as a measurement indicator in the temporal perspective, plotting significant keywords within four quadrants. Of particular note are the keywords in the upper-right and lower-right quadrants. The upper-right quadrant includes keywords with a longer development history and higher popularity in the field, such as “sepsis prediction,” “feature selection,” and “clinical deterioration.” Although the terms in the lower-right quadrant emerged later, they have also attracted substantial attention in the field, with representatives including “acute kidney injury,” “machine learning,” and “artificial intelligence.”

The x-axis represents relevance, while the y-axis indicates development level. Keywords in the upper-right quadrant show the highest relevance and significant development.
To track recent hotspots and trends in this field, we conducted a comparative statistical analysis of keywords after 2022 and those before 2022 to highlight the research focus of the field post-2022. Figure 9(a) superimposes an intensity analysis on the basis of Figure 7(c) to show the change in keyword popularity after 2022. The color gradient from blue to red reflects the proportion of the occurrence times of the keyword in 2022–2025 to the total occurrence times. It can be seen that terms such as “ards,” “mimic-iv,” “immune infiltration” and “prognosis” have become new hotspots in this research field from 2022 to the present. We also divided these keywords into two time periods: 2017–2021 as “basic” and after 2022 as “hotspots.”

Keyword hotspot analysis in the field of artificial intelligence (AI)-based sepsis diagnosis and management research after 2022. (a) Overlay analysis on top of Figure 7(c) shows the percentage of frequency of keyword appearances in the AI-based sepsis diagnosis and management research field since 2022. (b) Principal component analysis of the AI-based sepsis diagnosis and management study revealed the popularity of the keywords. Keywords are categorized according to the year of appearance and are represented by two different colored circles. (c) Keywords were analyzed for heat level using the random forest method. The color distribution of the two columns to the right of each keyword row represents the dynamics of popularity over time. (d) Volcano plot analysis of keywords after 2022, with the horizontal coordinate (FC) indicating the change in logarithmic multiplicity between the two sets of keywords, and the vertical coordinate (p-value) reflecting the statistical significance of the differences. (e) The Sankey diagram illustrates the evolving trends and connections among keyword popularity across different periods. The thickness of the bands and links corresponds to their respective levels of popularity and correlation strength. It is worth mentioning that the periods covered are not evenly distributed but decrease sequentially based on their temporal proximity. This approach is taken to provide a more precise and insightful representation of recent trends.
The results of principal component analysis are shown in Figure 9(b). There is some overlap between the two groups, but obvious separation, indicating that the similarity within each group is not significant in temporal development, which proves the validity of the classification and shows that significant changes have occurred in the development and evolution of keywords in this field. In Figure 9(c), the analysis results of keyword popularity using the random forest method highlight keywords with high popularity since 2022, including “diagnostic biomarker,” “immune cell infiltration,” “biomarker,” “immune infiltration” and “nomogram.” Figure 9(d) is a volcano plot of the statistical comparison of keywords in this field between after 2022 and before 2022. The points located in the upper right quadrant represent keywords with significant up-regulation and statistically significant differences, and the analysis results are basically consistent with those of Figure 9(c). Sankey diagram 9E illustrates the temporal distribution and evolutionary pathways of keywords across three distinct periods, demonstrating quantitative changes in research focus over time. As early as 2005–2016, the research in this field focused on exploring sepsis-related diseases using basic medical methods. Analysis of keyword evolution across temporal periods reveals distinct developmental phases. Early research (2005–2016) focused on basic medical approaches to sepsis-related diseases. The intermediate period (2017–2021) showed increased frequency of AI and data modeling terms. Post-2022 analysis demonstrates emergence of new high-frequency keywords including “data mining,” “electronic medical record,” and “immune infiltration.”
Highly cited references analysis
Citation frequency serves as a pivotal indicator of academic paper quality and scholarly impact, with highly cited publications representing research frontiers and seminal achievements. Systematic analysis of these influential works enables efficient identification of field developments and critical breakthroughs.
Table 6 summarizes the 15 most-cited publications in this domain. The most prominent is Seymour et al.'s 21 study “Derivation, Validation, and Potential Treatment Implications of Novel Clinical Phenotypes for Sepsis” (776 citations), published in JAMA-Journal of the American Medical Association. Using machine learning-based phenotyping, investigators identified four distinct clinical phenotypes (α, β, γ, δ) with significant variations in inflammatory markers, organ failure patterns, and mortality rates (γ phenotype: 40% mortality peak). Crucially, the research revealed phenotype-specific therapeutic responses to intravenous fluids and vasopressors, suggesting phenotype-targeted precision medicine could optimize sepsis management outcomes.
Ranking of the top 15 major highly cited references in the field of AI-based sepsis diagnosis and management from 2005 to 2025.

MIMIC: Medical information mart for intensive care; ICU: intensive care unit.
The second most-cited publication is Komorowski et al.'s 8 study “The Artificial Intelligence Clinician Learns Optimal Treatment Strategies for Sepsis in Intensive Care" (619 citations), published in Nature Medicine. Compared to actual clinical decisions, AI-recommended treatment strategies significantly reduced patient mortality (absolute risk reduction: ∼3%) while aligning more closely with international guideline standards.
Another notable study is Nemati et al.'s 2018 publication “An Interpretable Machine Learning Model for Accurate Prediction of Sepsis in the ICU" 22 (446 citations), published in [Critical Care Medicine]. The authors developed InSight, an interpretable machine learning model analyzing physiological parameters from electronic health records to predict sepsis onset six hours before clinical diagnosis (AUC = 0.83), demonstrating superior performance compared to conventional scoring systems like SOFA and Systemic Inflammatory Response Syndrome (SIRS).
Co-citation analysis categorized research topics into 12 distinct clusters (Figure 10(a)), revealing the comprehensive research landscape of AI applications in sepsis management. Cluster #0 “machine learning” demonstrated the broadest coverage as the core research direction, serving as the methodological foundation for the entire field. The remaining clusters represent specialized research domains: Cluster #1 “artificial intelligence” focuses on algorithmic development and optimization; Cluster #2 “intensive care unit” addresses clinical implementation environments; Cluster #3 “prediction model” concentrates on forecasting methodologies; Cluster #4 “biomarker” explores diagnostic indicators and molecular signatures; Cluster #5 “electronic health records” emphasizes data infrastructure and integration; Cluster #6 “hospital mortality” examines outcome prediction and risk stratification; Cluster #7 “critical care” covers broader intensive care applications; Cluster #8 “septic shock” addresses the most severe sepsis manifestations; Cluster #9 “risk factors” investigates predisposing conditions and vulnerability patterns; Cluster #10 “deep learning” represents advanced neural network approaches; and Cluster #11 “clinical decision support” focuses on translating AI insights into actionable clinical guidance. Through directional arrow relationships, Cluster #0 served as the methodological foundation for the four most influential application-oriented clusters: Cluster #5 “electronic health records,” Cluster #6 “hospital mortality,” Cluster #7 “critical care,” and Cluster #11 “clinical decision support.”
Highly-cited references’ citation and co-citation analysis in artificial intelligence (AI)-based sepsis diagnosis and management from 2005 to 2025. (a) References are grouped based on their likeness, where smaller numbers denote larger clusters, with #0 denoting the most substantial cluster. Node size reflects the frequency of co-citations, while the connections between nodes depict co-citation associations. (b) The connectivity among the primary 25 citation bursts is demonstrated, depicting the citation interconnections among these articles with arrows. (c) The diagram illustrates the 25 primary references characterized by pronounced bursts of citations, denoted by red spikes on the timeline. These spikes signify sudden surges in citation counts, signaling pivotal moments of emerging crucial questions or solutions within the field.
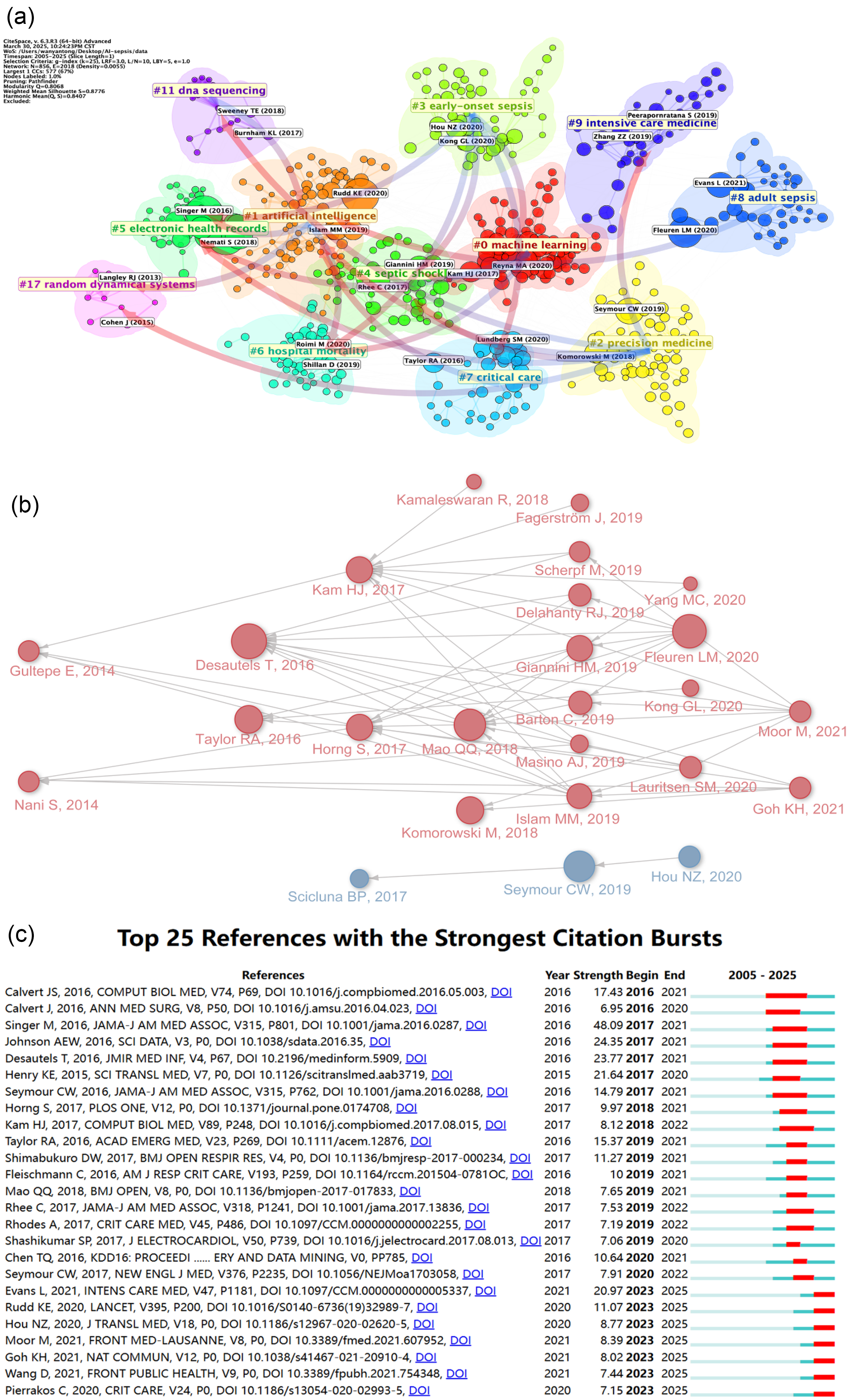
Figure 10(b)'s co-citation network reveals that Desautels et al.'s 2016 study “Prediction of Sepsis in the Intensive Care Unit With Minimal Electronic Health Record Data: A Machine Learning Approach” provided valuable insights for subsequent research. Mao et al.'s 2018 multicentre validation study and Fleuren et al.'s 2020 systematic review and meta-analysis further explored this direction, making milestone contributions alongside research achievements by Seymour CW, Kam HJ, and others.
Figure 10(c) presents citation burst analysis results, with the most prominent burst intensity (48.09) attributed to Singer et al.'s 2016 study “The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3)", 1 published in JAMA-Journal of the American Medical Association, followed by Johnson AEW et al.'s 2016 “MIMIC-III, a freely accessible critical care database" 23 (Scientific Data; burst intensity: 24.35). Several publications demonstrate ongoing citation bursts, including Evans et al.'s 2021 24 “Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021" (Intensive Care Medicine; 20.97) and Rudd et al.'s 25 2020 global epidemiological study (The Lancet; 11.07).
Discussion
This comprehensive bibliometric analysis reveals several novel insights into AI applications in sepsis that distinguish it from previous bibliometric studies in critical care or general medical AI. Our extended temporal analysis (2005–2025) captures a critical transformation missed by earlier studies: The field's evolution from technical feasibility demonstrations to clinical utility focus, particularly the post-2022 acceleration driven by multi-omics integration and explainable AI demands. Unlike broader AI-healthcare bibliometric analyses, our sepsis-specific focus uncovers unique patterns such as the field's response to Sepsis-3 definitions and the COVID-19 pandemic's catalytic effect on acute respiratory distress syndrome (ARDS)-related research.
Temporal development trajectory and critical turning points
Sepsis represents a major challenge in critical care medicine. The application of AI technologies in sepsis diagnosis and management has rapidly evolved, significantly transforming clinical decision-making paradigms.9,26 Through systematic bibliometric analysis, this study constructs a comprehensive developmental landscape from 2005 to 2025, revealing distinct phased characteristics. As illustrated in Figure 1(a), 2016 emerged as a critical turning point: Prior to this year, annual publications remained below 50 articles with weak citation patterns, but from 2016 onwards, research demonstrated a significant upward trend, achieving polynomial growth during 2020–2024 (R² = 0.998, Figure 1(b)), reaching a peak of 352 publications and 1363 citations in 2024. Notably, the annual growth rate during 2022–2025 reached 35%, substantially higher than the average level during 2016–2021, marking the field's entry into an accelerated development phase. This accelerated development trend may be closely related to several factors. First, the publication of The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) in 2016 restructured diagnostic criteria, promoting standardization of clinical data annotation and establishing foundations for AI algorithm development. 1 Second, the COVID-19 pandemic significantly accelerated digital transformation in critical care medicine, 27 evidenced by the notable increase in keywords such as “ARDS” shown in Figure 9(a). Sepsis, as a core clinical problem in severe infections, naturally became a focal area for clinical AI applications. 28 Additionally, the proliferation of open-source medical databases represented by Medical information mart for intensive care (MIMIC-III) (2016) and its successor MIMIC-IV (2020) provided rich training data for algorithm development. 23 Notably, the citation burst analysis revealed in Figure 10(c) demonstrates citation patterns that align closely with the field's developmental trajectory. Singer et al.'s 2016 Sepsis-3 consensus achieved the highest burst strength (48.09), while Johnson et al.'s MIMIC-III database paper showed significant burst activity (24.35), reflecting the academic community's deep recognition of the value of fundamental work in diagnostic standardization and data resource construction within the context of rapid AI technological advancement. This paradigm shift driven by both fundamental standard innovation and public health crises has not only reshaped the scientific problem spectrum of sepsis AI research but also profoundly influenced the dissemination pathway choices for academic achievements. When the research scale exhibits exponential expansion, knowledge production systems inevitably achieve self-regulation through quality screening mechanisms, providing a key entry point for understanding subsequent journal publication characteristics.
Academic journal publication patterns and quality centralization
The explosive growth curve shown in Figure 1 conceals the academic community's redefinition of outcome certification channels. Analysis of journal publication patterns reveals that the AI sepsis research field is experiencing a unique “quality centralization” phenomenon, reflecting underlying principles of knowledge production and dissemination in this domain. The journal quality distribution shown in Table 4 demonstrates that 90% of the top ten journals by publication volume are Q1-tier, while all top ten journals by citation count are Q1-tier. This high degree of concentration is not accidental but reflects the special challenges faced by medical AI research: The dual requirements of technical complexity and clinical rigor. 29 We believe the fundamental reason for this quality centralization lies in the interdisciplinary nature and high-risk characteristics of research in this field, compelling researchers to seek recognition in top-tier journals with rigorous peer-review mechanisms to ensure research scientific validity and clinical safety.
This concentration pattern is further validated by Bradford's Law analysis shown in Figure 6(a). Bradford's Law, a fundamental principle in bibliometrics, describes how scientific literature distributes unevenly across journals within any research field, with a small core of highly productive journals publishing a disproportionately large number of relevant articles, while a much larger number of journals contribute only a few articles each. 30 Our analysis confirms the stability of this concentration trend in AI sepsis research, where core journals dominate the publication landscape. More importantly, this centralization pattern may serve a quality screening function, ensuring that only research with genuine clinical value and technological innovation can obtain publication opportunities in these core journals. More deep-level journal clustering analysis (Figure 5(a) and (b)) reveals another important characteristic of knowledge production in this field: A highly topic-journal matching pattern exists between research content and publication platforms, reflecting the continuous deepening of disciplinary specialization. Technology innovation research tends to choose journals like Nature Medicine that emphasize methodological breakthroughs, while clinical mechanism research concentrates in specialized authoritative journals like Critical Care Medicine. This differentiation is not simply journal preference but reflects differential needs for validation standards and readership among different research paradigms.31,32
Particularly noteworthy is the close citation relationship between molecular-biological-immunological fields and molecular-biological-genetic fields shown by the yellow-green connections in Figure 6(b), indicating active knowledge exchange between these domains. Combined with keyword evolution analysis in Figure 9, this cross-disciplinary communication may reflect an ongoing shift in research focus in this field: The developmental trend from traditional clinical phenotype description toward molecular mechanism analysis. The emerging trends shown in Figure 6(c) hold more prospective significance—the rising popularity of interdisciplinary journals such as Heliyon and BMC Medical Informatics and Decision Making signals that this field is breaking through traditional disciplinary boundaries and forming new knowledge production and dissemination models. The deeper implication of this trend is that AI sepsis research is catalyzing a new type of academic publication ecosystem that emphasizes organic integration of technological innovation with clinical application rather than pure intra-disciplinary dialogue. We predict that the emergence of this interdisciplinary publication model will further accelerate the field's evolution from technical validation toward clinical translation.
Global research distribution and international collaboration networks
This field demonstrates not only rapid expansion in knowledge production but also a complex geopolitical academic ecosystem in its scholarly influence distribution. From the global output distribution perspective, AI applications in sepsis diagnosis and management exhibit distinct regional differentiation characteristics and collaborative pattern differences. The United States and China form a bipolar dominance pattern, with the United States maintaining clear leadership in both publications (487 papers) and citations (12,984), while China establishes a strong second position with 457 publications but demonstrates a substantial influence gap with 3929 citations (Table 1). This “competitive-output, lower-impact” phenomenon relative to the US not only manifests in macrostatistics but also reflects profoundly in structural differences within academic collaboration networks. Author collaboration network analysis (Figure 3(a)) further reveals the microscopic mechanisms of this regional differentiation. The blue cluster centered on Kamaleswaran (primarily Emory University) and the red cluster centered on Clermont (primarily University of Pittsburgh) exemplify the American “institutional resource aggregation effect,” forming core groups through preferential attachment mechanisms. 33 In contrast, the green cluster dominated by Chinese scholars exhibits “geographic-cultural cohesion characteristics,” showing regional clustering based on geographic proximity and cultural patterns. 34 Data analysis reveals two major structural factors contributing to the China-US influence gap despite China's substantial publication volume. First is the difference in collaboration network orientation—Chinese institutions show significantly higher domestic collaboration ratios compared to the international collaboration patterns characteristic of United States institutions (Figure 4(a)). Second is strategic differentiation in research focus—Chinese research predominantly focuses on algorithm optimization (such as XGBoost parameter tuning), while American research leans more toward multi-center clinical validation, exemplified by applications using the MIMIC-III database (released 2016) for foundational studies and the updated MIMIC-IV database (released 2020) for recent validation research. Despite China's substantial publication volume ranking second globally, this research pattern difference manifests in stratified publication strategies: Chinese papers predominantly appear in specialized technical journals such as “Frontiers in Medicine” (IF = 3.1), while American research, maintaining both higher publication volume and superior citation impact, concentrates in comprehensive top-tier journals such as “JAMA” (IF = 63.5), with the latter's broad readership significantly enhancing citation potential. In contrast, certain countries demonstrate “small but refined” research characteristics, achieving disproportionate academic influence relative to their publication volume. The Netherlands exemplifies this pattern, generating 2461 citations from only 53 publications—a research efficiency that suggests a “boutique research model” focused on high-impact contributions rather than volume-based strategies. The four research direction differentiations shown in the co-citation network of Figure 3(c)—pathophysiology, predictive modeling, AI decision support, and evidence-based medicine—further confirm the field's transformation from “universal technology exploration” toward “specialized intensive cultivation.” The high activity of red, pink, and blue clusters in Figure 3(b) reveals that different research clusters are in a critical transition period from technical validation toward clinical translation, thus demonstrating more intensive collaboration intensity. Particularly noteworthy is the positive change in institutional participation patterns in this field during 2022–2025. According to temporal dimension analysis in Figure 4(b), top-tier institutions such as Harvard Medical School, MIT, and University of Pittsburgh that joined later rapidly established collaboration intensity comparable to existing research networks. This rapid integration capability forms an interesting contrast with the previously analyzed regional collaboration limitations, indicating that despite overall collaboration patterns still exhibiting regional differentiation characteristics, top-tier research institutions possess the ability to rapidly construct cross-regional academic networks. This phenomenon aligns with theoretical predictions about preferential attachment mechanisms in network science, 35 where institutions with high reputation and resource advantages more easily attract new collaboration partners,36,37 providing important reference for future collaborative model evolution in this field.
Core research hotspots and high-impact literature analysis
Understanding the global research landscape enables further analysis of currently most influential research achievements and hotspot directions, helping grasp the core developmental trajectory of this field. High-impact literature co-citation network analysis (Figure 10(a)) reveals that current core hotspots in AI sepsis research are developing around three key dimensions. First, data-driven clinical decision support systems have become an important research direction, clearly evidenced by Seymour et al.'s 2019 phenotyping study receiving 776 citations and Komorowski et al.'s 8 AI treatment strategy research receiving 619 citations (Table 6). The high impact of these studies suggests an emerging shift from simple risk prediction to treatment strategy optimization. Beyond these landmark studies, this transition is evidenced by recent developments: Sendak et al.'s real-world clinical integration of sepsis prediction models demonstrated measurable clinical impact, 38 while Milano et al. documented how AI-enhanced bundle adherence achieved a 14.0% relative risk reduction in mortality, 39 representing concrete evidence of the transition from “diagnostic AI” toward “therapeutic AI” applications. Particularly in personalized treatment domains, machine learning models have begun applications in therapeutic drug monitoring and other precision medicine scenarios, providing more individualized treatment plans for patients. 5 Second, breakthrough progress in explainable AI technology demonstrates increasingly growing importance. Nemati et al.'s 2018 explainable model research receiving 446 citations (Table 6) precisely demonstrates the significance of this trend. Explainable AI models not only excel in real-time sepsis risk assessment but also provide interpretable information for understanding sepsis risk. 22 This interpretability is crucial for promoting broader clinical integration, as AI model predictions must possess features that clinicians can understand to cultivate their trust and acceptance of predictions. The shift toward explainable AI is supported by multiple converging lines of evidence beyond the foundational work of Nemati et al. Lauritsen et al. 40 developed an explainable AI model achieving 211 citations, demonstrating that interpretable approaches can maintain predictive performance while providing clinicians with understandable decision pathways. Chen and Hernández 41 further advanced this direction through sensitivity analysis-based explainable models for sepsis detection. The clinical necessity driving this trend is emphasized by Sadeghi et al., 42 who reviewed explainable AI applications in healthcare and highlighted that model interpretability is crucial for clinical adoption, regulatory approval, and maintaining physician trust in automated decision support systems. Bomrah et al.'s 43 scoping review specifically addressed the step toward explainability in sepsis prediction models, demonstrating the field's recognition of this critical need. Third, citation burst analysis in Figure 10(c) shows that Evans et al.'s 2021 sepsis management guidelines maintain high burst strength (20.97), indicating that deep integration of AI technology with clinical guidelines is becoming an important development direction. Practical application cases such as the Sepsis Watch system developed by Duke University Health System demonstrate this integration. This deep learning model updates risk scores hourly and identifies high-risk patients every 5 min. Its integration improved guideline adherence, thereby reducing mortality and improving overall care quality. 38 Research indicates that increased bundle compliance significantly reduced mortality with a relative risk reduction of 14.0%. 39 The central position of Cluster #0 “machine learning” and its connection patterns with other clusters shown in Figure 10(a) reflect that multi-center validation and clinical translation research are becoming new standards for evaluating research value. This transformation drives the field's shift from pursuing algorithm performance toward emphasizing clinical practicality and generalizability, although model generalization capability on external datasets still faces challenges. 44
Future development trends and technological evolution directions
Based on comprehensive analysis of citation burst patterns in Figure 10(c), knowledge network evolution trends in Figure 10(b), and keyword evolution data in the Figure 9 series, we can identify four important development directions likely to emerge in the next five years. The first development direction involves further refinement of multimodal data fusion and dynamic monitoring systems. The rationality of this trend is evidenced by the persistent high-frequency appearance of core keywords such as “machine learning” (1459 occurrences) and “electronic health record” (204 occurrences), along with technical accumulation from existing research such as Komorowski team's reinforcement learning systems. Recent multiple studies indicate that AI-based continuous monitoring systems, including multimodal data integration technologies incorporating wearable devices, are providing real-time predictive capabilities for sepsis-related complications, enabling timely interventions.9,45 This provides direct evidence supporting clinical practice needs for continuous, personalized monitoring. It is anticipated that AI systems will further enhance real-time dynamic treatment strategy adjustment capabilities based on existing foundations, predicting patient responses to specific therapies through personalized treatment plans while optimizing therapeutic effects and minimizing adverse reactions. 9 The second potential direction involves technical integration of explainable AI and precision medicine. The significant rise of keywords such as “immune infiltration” after 2022, combined with concrete research developments, indicates that multi-omics integration with AI is becoming a research priority. Recent studies demonstrate this trend: Tu et al. applied single-cell transcriptomics to reveal immune infiltration patterns in sepsis, 46 while Scicluna et al. 47 demonstrated blood genomic endotyping for sepsis classification, achieving 365 citations and establishing a foundation for precision medicine approaches that extend beyond the phenotyping work of Seymour et al. 21 This trend reflects growing clinical demands for explainability, with multiple studies emphasizing the importance of explainable models in healthcare, capable of helping medical professionals trust AI-generated predictions. 48 Clinical literature indicates that explainable AI models can extract 168 features hourly and perform real-time sepsis prediction training, demonstrating the technical reality of transformation from simple prediction accuracy toward interpretable individualized treatment. 48 Although maintaining model performance while enhancing interpretability remains a technical challenge, the clinical value of this direction has gained widespread recognition. The third noteworthy direction involves technical support system construction for multi-center collaborative research. Although “multi-center” keywords did not reach significant prominence in our analysis, the international collaboration network patterns shown in Figure 2(a) and inter-institutional cooperation relationships in Figure 4(a) indicate growing needs for cross-institutional data integration and model validation. The emphasis on clinical translation is evidenced by multiple implementation studies beyond theoretical development. Wang et al. conducted a methodological systematic review specifically focused on validation and performance of sepsis real-time prediction models, highlighting the field's maturation toward rigorous external validation. 44 Additionally, studies have demonstrated federated learning approaches addressing the practical challenges of multi-center collaboration while preserving patient privacy—a critical requirement for clinical implementation.49,50 This trend may drive exploration of privacy protection technologies (such as federated learning) applications in this field to address practical issues such as legal, security, and privacy concerns faced in inter-institutional data sharing. 50 The fourth emerging direction involves deepened applications of natural language processing technology in medical text analysis. The appearance of “natural language processing” keywords in Figure 7 and the continued popularity of “electronic medical record” reflect the possibility of this trend. Research published in authoritative journals confirms the technical feasibility of this direction, demonstrating that mining unstructured clinical records can significantly improve algorithm accuracy compared to early warning systems using only clinical indicators, with better predictive effects 12 to 48 h before sepsis occurrence. 51 Although large language models have not yet become significant hotpoints in our keyword analysis, considering the rapid development of AI technology and actual needs for medical text processing, this direction deserves continued attention. The common characteristic of these development directions is increased emphasis on clinical practicality and patient benefit realization.
Internal logic and driving forces of technological evolution
To more deeply understand the formation mechanisms of these future trends, it is necessary to analyze the internal logic and driving factors of technical evolution. From a technical evolution perspective, AI applications in sepsis research are undergoing a profound paradigm shift, reflecting the medical AI field's transition from policy-driven to demand-driven development models. The keyword co-occurrence networks in Figure 7(c) and (d) clearly demonstrate this transformation process: The evolution trajectory of the yellow cluster centered on “machine learning” and the red cluster focused on “deep learning” reveals that early research primarily focused on proving technical feasibility of various algorithms in sepsis prediction, with researchers enthusiastic about comparing performance differences among different algorithms such as random forest, support vector machines, and neural networks. 52 However, as basic technical validation has been largely completed, research focus has begun shifting toward solving actual clinical problems. 43 The sudden rise of explainable AI exemplifies this transformation—the keyword relevance-development level analysis in Figure 8 shows “machine learning” positioned in the lower right quadrant, indicating its status as an emerging but widely attention-receiving hotspot. This is not purely technical progress but researchers’ positive response to the practical problem of low clinical acceptance of “black box” models. 42 More noteworthy is the change in deep learning technology application patterns: From initially pursuing complex network structures and high-dimensional feature extraction, gradually transitioning toward developing lightweight, interpretable deep learning architectures suitable for medical scenarios. 41 Keyword burst analysis in Figure 9 shows that while large language model-related terms are still in exploratory stages, their potential in processing unstructured medical texts suggests this field may be approaching the starting point of the next round of technological innovation. 53 This internal logic of technical evolution indicates that future development directions will focus more on deep integration of technology with clinical needs rather than pure algorithmic innovation. Driven by this fundamental transformation in technical orientation, research methodology has also begun experiencing corresponding profound changes.
Research paradigm shifts and the rise of validation studies
This methodological transformation in research reveals that the field is experiencing a strategic shift from exploratory research toward validation research, with the core being researchers’ deep reflection on AI model generalization capability and practicality. The Sankey diagram in Figure 9(e) clearly shows the temporal evolution of research paradigms: From basic medical method exploration during 2005–2016, to AI technology clinical integration during 2017–2021, to multi-omics data fusion and clinical translation stages after 2022. The emergence of validation research paradigms is not accidental, with this trend confirmable from multiple dimensions. First, the significant rise in popularity of keywords such as “prognosis” and “biomarker” in Figure 9(a) reflects the shift from algorithm development toward clinical effect validation. Second, the evolution trajectory of high-impact literature in Table 6—from early proof-of-concept research by Desautels et al 54 to recent large-scale multi-phenotype validation research by Seymour et al. 21 —clearly demonstrates the research community's collective attention to model generalization capability. This methodological transformation reflects a deeper issue—the success criteria for medical AI are shifting from technical metrics toward clinical utility. The continued popularity of electronic health record-related research (as shown in the purple cluster in Figure 7(c)) further confirms this judgment: Researchers increasingly recognize that data quality and standardization issues are fundamental bottlenecks constraining AI clinical applications. The high popularity of standardized database keywords such as “mimic-iv"(the updated version released in 2020) in Figure 9(a) reflects researchers’ urgent needs for data standardization and external validation. From a more macroscopic perspective, this research paradigm transformation actually marks AI sepsis research's critical leap from “proving feasibility” toward “ensuring usability.” This paradigm evolution manifests clearly in the temporal keyword analysis shown in Figure 9(e). The progression from basic medical method exploration (2005–2016) to AI technology integration (2017–2021) represents the field's methodological maturation. During the intermediate period, researchers introduced AI and data models into both laboratory and clinical settings, promoting in-depth development of experimental and clinical research in sepsis diagnosis and management. This phase was characterized by building decision support systems through machine learning to assist medical decisions, with particular attention to vulnerable populations such as preterm infants and sepsis prediction applications. The post-2022 period marks a significant shift toward precision medicine approaches. The integration of single-cell and tumor omics into big data models has enabled AI diagnosis and management systems to become more closely aligned with emerging technologies and clinical scenarios. This evolution helps clinicians more accurately predict, diagnose, and formulate individualized treatment plans, as evidenced by the prominence of terms such as “data mining,” “electronic medical record,” and “immune infiltration” in recent literature. This temporal progression reflects the field's maturation from proving technological feasibility toward ensuring clinical utility and patient benefit.
Evolution of clinical application-oriented research hotspots
Examining this field's development from a clinical application perspective helps understand the ultimate direction of technological progress and methodological transformation. Clinical application-oriented research hotspot evolution reflects profound changes in sepsis diagnosis and management concepts, with the core transformation being the shift from disease-centered to patient-centered approaches, and from standardized treatment to precision medicine. 55 The volcano plot analysis and random forest heat analysis in Figure 9(c) and (d) show explosive growth of keywords such as “immune infiltration” and “diagnostic biomarker” after 2022. Superficially, this appears as results of new technology proliferation such as single-cell sequencing, 46 but more deeply reflects fundamental changes in clinicians’ understanding of sepsis heterogeneity. 21 Keyword relevance cluster analysis in Figure 7(b) further confirms this transformation: The green cluster surrounding “sepsis” integrates multi-dimensional clinical elements including biomarkers, prognostic assessment, and acute kidney injury, indicating that traditional diagnostic and therapeutic models based on clinical symptoms and routine laboratory indicators can no longer meet individualized treatment needs. The significant burst of the “ards” keyword in Figure 9(a) similarly reflects this conceptual transformation: As basic sepsis diagnostic technology has relatively matured, researchers have begun recognizing that what truly affects patient prognosis is the cascade reaction of organ dysfunction rather than initial infection itself. The clinical significance of this transformation lies in its driving AI applications’ evolution from passive risk assessment tools toward active treatment decision support systems. 56 While the COVID-19 pandemic catalyzed the surge in related research, more importantly, it accelerated the entire critical care medicine field's acceptance and application of systematic, holistic treatment concepts. 57 From a long-term perspective, these clinically-oriented research hotspots predict that AI applications in sepsis will increasingly emphasize clinical effectiveness and patient benefit rather than pure technological advancement. This transformation in application philosophy may bring revolutionary improvements to sepsis diagnosis and management. 58
Translational implications for clinical adoption
These emerging research patterns—particularly the emphasis on explainable AI, multi-omics integration, and clinical effectiveness—offer concrete guidance for healthcare systems preparing to adopt AI-based sepsis management tools. Our findings reveal key insights for healthcare systems implementing AI in sepsis management. The shift toward explainable AI models reflects a practical reality: Clinicians need to understand how AI tools make decisions before trusting them with patient care. Nemati et al.'s interpretable machine learning model achieved 446 citations precisely because it addressed this clinical need for transparency. Healthcare leaders would likely find greater clinical acceptance with AI tools offering clear decision-making processes rather than focusing solely on predictive accuracy metrics. The emergence of multi-omics integration keywords such as “immune infiltration” after 2022 signals a research direction toward precision sepsis care based on individual patient molecular profiles. This keyword trend evolution, combined with the high citation impact of genomic endotyping studies (Scicluna et al., 365 citations), suggests that next-generation AI tools will require integration of advanced laboratory capabilities. Healthcare systems should anticipate the need for upgraded laboratory infrastructure and clinician training programs that prepare staff for AI-assisted personalized treatment selection, as these technologies transition from research applications to clinical implementation. The pattern of high-impact research emerging from established international collaboration networks, as demonstrated in our institutional analysis (Figure 4(a)), provides insights for clinical adoption strategies. The most cited studies in our analysis consistently originate from multi-institutional collaborations rather than single-center developments, suggesting that externally validated AI tools may offer greater reliability for clinical implementation. Healthcare systems should prioritize AI tools with demonstrated multi-center validation over those developed and tested in isolated environments, as the former are more likely to perform consistently across different clinical settings and patient populations. The field is evolving beyond diagnostic prediction toward treatment guidance. Komorowski et al.'s AI clinician study (619 citations) gained attention because it moved from identifying sepsis risk to recommending specific treatments. Healthcare administrators should prepare for this evolution by considering how clinical workflows can accommodate AI-generated treatment recommendations alongside existing evidence-based sepsis protocols, while ensuring that such integration enhances rather than disrupts established care bundles.
Limitations
While Web of Science Core Collection provides comprehensive coverage of high-quality academic literature, this analysis may not capture research from other databases or non-English publications. Although network analysis thresholds were selected through iterative optimization rather than exhaustive sensitivity analysis, this approach ensures robust identification of meaningful collaboration patterns while maintaining computational efficiency for large-scale bibliometric datasets. Given the rapid evolution of AI applications in sepsis research, our findings reflect the state of knowledge only at the time of data collection. However, our 20-year analytical timeframe (2005–2025) provides substantial historical depth for identifying meaningful developmental patterns and emerging trends. Additionally, bibliometric analysis focuses on quantitative publication metrics, which may not fully capture the practical clinical impact of specific AI technologies. Citation analysis may also exhibit bias toward review articles and meta-analyses, which typically receive higher citation counts than original research studies due to their synthetic nature. The integration of multiple analytical tools (VOSviewer, CiteSpace, and Bibliometrix) helps enhance the reliability of our findings.
Conclusion
This study reveals rapid development of AI applications in sepsis diagnosis and management since 2016, with accelerated growth during 2022–2025. The United States leads in both citation impact and publication volume, with China ranking second in publication output but showing a significant citation impact gap. Research concentrates heavily in Q1 journals. The field is transitioning from algorithm validation to clinical implementation, risk prediction to treatment decision-making, and “black box” to explainable AI. Emerging hotspots including “immune infiltration” and “ARDS” after 2022 indicate multi-omics integration trends. The research trajectory suggests evolution from “proof of concept” toward “ensuring clinical utility.” These findings provide strategic guidance for healthcare systems implementing AI technologies, emphasizing the importance of explainable models, multi-center validation, and infrastructure preparation for precision sepsis care.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251396574 - Supplemental material for Global trends, collaboration networks and knowledge mapping of artificial intelligence evolution in sepsis diagnosis and management: A bibliometric analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076251396574 for Global trends, collaboration networks and knowledge mapping of artificial intelligence evolution in sepsis diagnosis and management: A bibliometric analysis by Zhiyong Lin, Jingyi Deng, Shiming Chen, Jiawen He, Zhiquan Li, Jierong Mo, Wenmin Tang, Lianshan Guo, Ran Zhan, Peiyi Liu, Shuirong Chen, Jun Jiang and Tianen Zhou in DIGITAL HEALTH
Footnotes
Acknowledgements
We thank everyone who contributed helpful comments and suggestions during the preparation of this manuscript.
ORCID iDs
Ethical approval
This study was conducted in accordance with institutional research guidelines at Guangdong Medical University and The First People's Hospital of Foshan. Ethical approval was not required as this research involved exclusively secondary analysis of publicly available published literature from bibliographic databases, with no collection of primary data, no human subject involvement, and no access to personal or identifiable information. This determination aligns with standard international guidelines for bibliometric research that uses only published, publicly accessible academic literature.
Contributorship
Zhiyong Lin, Jingyi Deng, and Shiming Chen: Conceptualization, data curation, formal analysis, investigation, methodology, writing—original draft.Zhiquan Li and Shuirong Chen: Data curation, investigation, software, visualization, writing—original draft. Jierong Mo and Lianshan Guo: Formal analysis, investigation, software, validation, writing—original draft. Ran Zhan and Peiyi Liu: Methodology, software, validation, writing—original draft. Jiawen He and Wenmin Tang: Resources, validation, visualization, writing—original draft. Jun Jiang: Conceptualization, funding acquisition, project administration, resources, supervision, writing—review & editing. Tianen Zhou: Conceptualization, funding acquisition, project administration, resources, supervision, writing—review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by The Basic and Applied Basic Research Fund of Guangdong (Regional Cooperation Fund Project, 2022) [grant number No. 2022A1515140033] and The Medical Research Fund of Guangdong [grant number No.B2025307] and The 14th Five-Year High-Level Medical Key Specialty Project of Foshan. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data supporting the findings of this study are included in the article and Supplemental Materials. The bibliometric dataset extracted from Web of Science Core Collection (2005–2025) can be obtained from the corresponding authors upon reasonable request. Due to database licensing restrictions, researchers with institutional Web of Science access can replicate the dataset using the detailed search strategy provided in ![]() .
.
Guarantor
Jun Jiang and Tianen Zhou accept full responsibility for the work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
