Abstract
Background
Fatigue, impaired sleep quality and daytime sleepiness, common in neurodegenerative and immune-mediated diseases, are debilitating and have serious societal and economic implications. Currently, measurement of these symptoms largely relies on self-reported questionnaires, which are burdensome for patients and lack sensitivity, granularity and reliability.
Methods
Building on a preceding feasibility study and qualification advice of the European Medicines Agency, the Clinical Observational Study of the European project Identifying Digital Endpoints to Assess FAtigue, Sleep and acTivities of daily living in Neurodegenerative disorders and Immune-mediated inflammatory diseases (IDEA-FAST) investigates the relationship between digital and clinical parameters of the target concepts of fatigue, reduced sleep quality and daytime sleepiness.
Results
Between 2022 and 2025, 2000 people are being recruited at 24 European sites – 500 with Parkinson's disease, 500 with inflammatory bowel disease, 200 with each of the following diseases: Huntington's disease, rheumatoid arthritis, systemic lupus erythematosus, primary Sjögren's syndrome and 200 healthy volunteers. Participants are followed over a 24-week period with four visits, each including a 1-week assessment phase at home using CE-certified digital health (including active and passive) technologies. The latter collect information on physical activity, physiology, cognition as well as social interaction and behaviour as core dimensions of the target concepts.
Conclusion
This study will help to develop reliable, valid and efficient digital endpoints of fatigue, impaired sleep quality and daytime sleepiness for use in future clinical studies and trials.
Keywords
Introduction
Fatigue, impaired sleep quality and daytime sleepiness are highly prevalent in neurodegenerative and immune-mediated disorders including Parkinson's disease (PD1,2), Huntington's disease (HD3,4), inflammatory bowel disease (IBD 5 ), rheumatoid arthritis (RA6,7), systemic lupus erythematosus (SLE8,9) and primary Sjögren's syndrome (PSS10,11). These symptoms have a large impact on physical, mental and social well-being of the affected persons, and considerable societal and economic impact.4,12–15 Existing measurement tools are based largely on self-reported questionnaires asking about the severity of the symptoms ‘on average’ or ‘at their worst’ over a typical recall record between 1 and 4 weeks. Such questionnaires lack sensitivity and reliability of capturing the fluctuations of symptoms. Additionally, it is burdensome and resource intensive especially when such questionnaires need to be administered repeatedly. To effectively develop treatments for these symptoms, more accurate and reliable parameters (here called intermediate clinical endpoints 16 ) are urgently needed, especially as, to our best knowledge, there is no such marker for the assessment of these symptoms currently available.
It was shown that fatigue, impaired sleep quality and daytime sleepiness (as the target concepts of the proposed study) are associated with changes in the following four core dimensions: biophysiology, cognitive performance, physical performance and social behaviour. For instance, biophysical changes, such as autonomic deficits, have been shown to be common among people with fatigue. 17 Cognitive changes such as impaired short-term memory, reduced attention span and poor concentration are commonly reported by patients with fatigue, and impaired performance of neuropsychological tests have been linked to fatigue. 18 Fatigued persons often report reduced levels of physical activities, 19 and there is evidence that fatigue translates into altered movements, such as sit-to-stand and stand-to-sit transitions, during the day. 20 Reduced social interactions because of fatigue are often identified by affected persons as key impacts of this disabling symptom. 21 It is noteworthy that the changes in the four core dimensions of the concepts of interest presented here are similar among fatigued persons with chronic diseases regardless of their underlying chronic conditions.22,23 These observations suggest that there may be a common fatigue ‘biosignature’ in chronic conditions including neurodegenerative diseases (NDD) and immune-mediated inflammatory diseases (IMID) 24 that can be detected with digital intermediate clinical endpoints. With the advances in digital health technology (DHT), it is now possible to monitor parameters of these core dimensions continuously or semi-continuously in the natural living environment. Figure 1 illustrates how to address the target concepts of fatigue, impaired sleep quality and daytime sleepiness with core dimensions and examples of how to assess these core dimensions with DHT-derived parameters.

Mapping between the IDEA-FAST Clinical Observational Study target concepts, core dimensions and a non-exhaustive list of digital parameters that could be used to assess each core dimension.
Such technological advances raise the possibility of identifying digital endpoints of fatigue, impaired sleep quality and daytime sleepiness. 25 The Innovative Medicines Initiative (IMI)-funded project IDEA-FAST (Identifying Digital Endpoints to Assess FAtigue, Sleep and acTivities of daily living in Neurodegenerative disorders and Immune-mediated inflammatory diseases, EC Grant Agreement 853981; www.idea-fast.eu) aims to identify objective, scalable digital (monitoring) endpoints related to fatigue, impaired sleep quality and daytime sleepiness in people with NDD and IMID, enhancing the development of parameters that can be used in clinical trials. A pilot study has been run at four sites previously26–29 and we received qualification advice on the study design from the European Medicines Agency (EMA 24 ). The objectives of the study are presented in Table 1.
Objectives of the IDEA-FAST Clinical Observational Study (COS).

IMID: immune-mediated inflammatory disease; MCID: Minimal Clinically Important Difference; MPID: Minimal Participant-reported Important Difference; NDD: neurodegenerative diseases.
Method
Participants and overall study design
The IDEA-FAST Clinical Observation Study (COS) is a prospective, European, multinational, observational study investigating the relationship between digital and clinical parameters of fatigue, impaired sleep quality and daytime sleepiness in people with PD (N = 500), HD (N = 200), IBD (N = 500), RA (N = 200), SLE (N = 200) and PSS (N = 200), as well as healthy volunteers (HV, 200). The total sample size is 2000 participants.
A total of 24 European sites are involved. The study started in June 2022 and the planned overall study duration is 41 months. A total of four clinical study visits, accompanied by a DHT use period of 7 days, respectively, are planned for every participant (see next paragraphs for details). The first DHT use period includes a run-in phase of 3 days. The individual mean study duration is 24 weeks. The DHT use periods are always in the usual environment of the participant.
All sites have received approval from the local ethics committees and all procedures are in agreement with the Declaration of Helsinki. The study has been registered before study start in the German Clinical Trials Register (DRKS00027946 30 ). In- and exclusion criteria are presented in Supplementary Table 1.
Sample size determination
The total sample size (n = 2000) was chosen to have adequate numbers of participants for the planned identification and evaluation analyses across all disease indications.
Estimating training sample size needed for the identification analysis (i.e., building (a) digital biomarker(s) via a machine learning approach) is challenging without a previously collected large dataset in the same domain which can be empirically tested to determine algorithm performance as a function of sample size. Therefore, we aimed to strike a useful balance between the size of the identification analysis set and ensuring that sufficient independent data was reserved for evaluation analyses.
Sample size calculations for evaluation analyses (i.e., exploration of the psychometric properties of the digital biomarker(s) identified in the identification analyses) were based on test-retest reliability because of the absence of a gold standard for comparison of objective fatigue, daytime sleepiness and sleep quality measures. For test–retest reliability, we assumed an intra-class correlation coefficient (ICC) null hypothesis of 0.75 and an alternative hypothesis of 0.87 and estimated that 49 participants would be needed to demonstrate with 80% power that observed ICC is significantly greater than 0.75 with a one-sided 5% type-1 error rate. 31 Evaluation analyses other than reliability were approached as an estimation problem, i.e., guided by precision of estimates, and 50 participants per cohort was deemed sufficient for the desired precision of estimates of convergent validity and meaningful changes. The assignment of participants to the evaluation set will be performed dynamically throughout the study and stratified by disease cohort, geographic region, biological sex, age group, and baseline fatigue group.
Clinical study visits
The four clinical study visits consist of two in-hospital visits (Visit 1 and Visit 4: Visit 1 + 24 ± 3 weeks) and two remote visits (Visit 2: Visit 1 + 8 ± 2 weeks; Visit 3: Visit 1 + 16 ± 2 weeks). At Visit 1, basic demographics and medical history are collected. During all clinical study visits, participants fill in several patient-reported outcomes (PROs) on fatigue, sleep quality and daytime sleepiness, and their perception of severity and change over time (Patient Global Impression Scale and Patient Global Impression of Change) before and after the DHT use period. Table 2 provides details about instruments used to assess fatigue and sleep, and respective recall periods, and Supplementary Table 2 provides a full overview of clinical assessments and questionnaires used, including disease-specific instruments to characterise patient subgroups. The validation status of the most relevant PROs on fatigue, sleep quality and daytime sleepiness, which will be used in this study, is presented in Supplementary Tables 3 and 4 for each of the diseases investigated in IDEA-FAST. Participants and research staff will also assess feasibility and usability of digital tools.
Clinical fatigue and sleep assessment in the IDEA-FAST Clinical Observational Study.
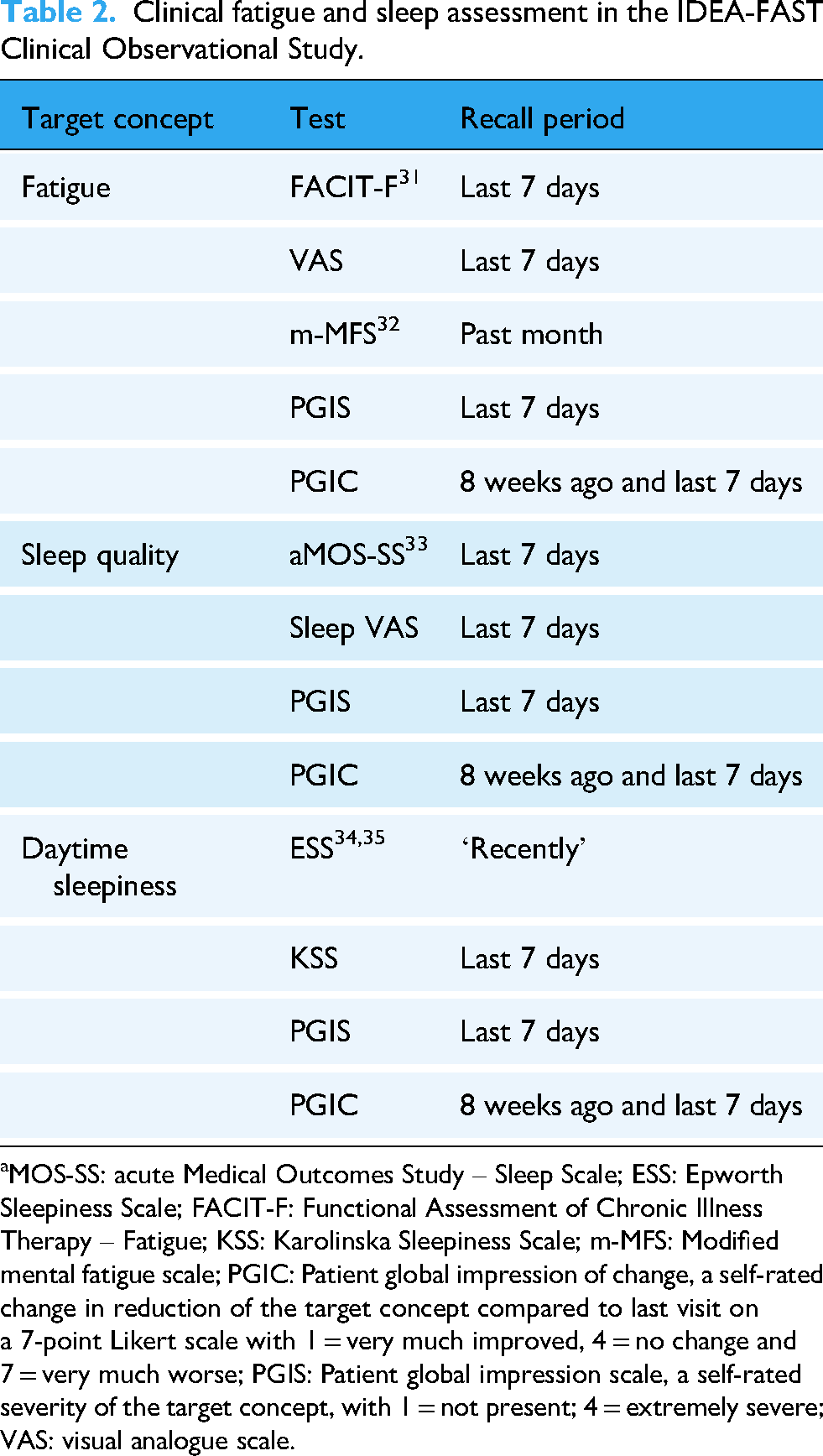
aMOS-SS: acute Medical Outcomes Study – Sleep Scale; ESS: Epworth Sleepiness Scale; FACIT-F: Functional Assessment of Chronic Illness Therapy – Fatigue; KSS: Karolinska Sleepiness Scale; m-MFS: Modified mental fatigue scale; PGIC: Patient global impression of change, a self-rated change in reduction of the target concept compared to last visit on a 7-point Likert scale with 1 = very much improved, 4 = no change and 7 = very much worse; PGIS: Patient global impression scale, a self-rated severity of the target concept, with 1 = not present; 4 = extremely severe; VAS: visual analogue scale.
Digital health technology use periods
Each clinical study visit is accompanied by a 7-day DHT use period. During these periods, participants are asked to do ecological momentary assessment of fatigue, sleep quality and daytime sleepiness, using a combination of an electronic diary (e-diary) with rating scales. Moreover, information about actual mood, anxiousness and pain, and activity aspects is collected. Based on the experience made in the IDEA-FAST pilot study, a frequency of 3 times per day (morning/afternoon/evening) was selected. 32 Sleep quality is reported once daily in the morning for the previous night's sleep. Table 3 provides detailed information on the questions asked in the e-diary. As another digital data collection approach requiring the user’s active contribution, participants are asked twice per day to assess cognitive performance (CANTAB app, CamCog, UK) on their own smartphone.
Questions of the e-diary used in the IDEA-FAST Clinical Observational Study.
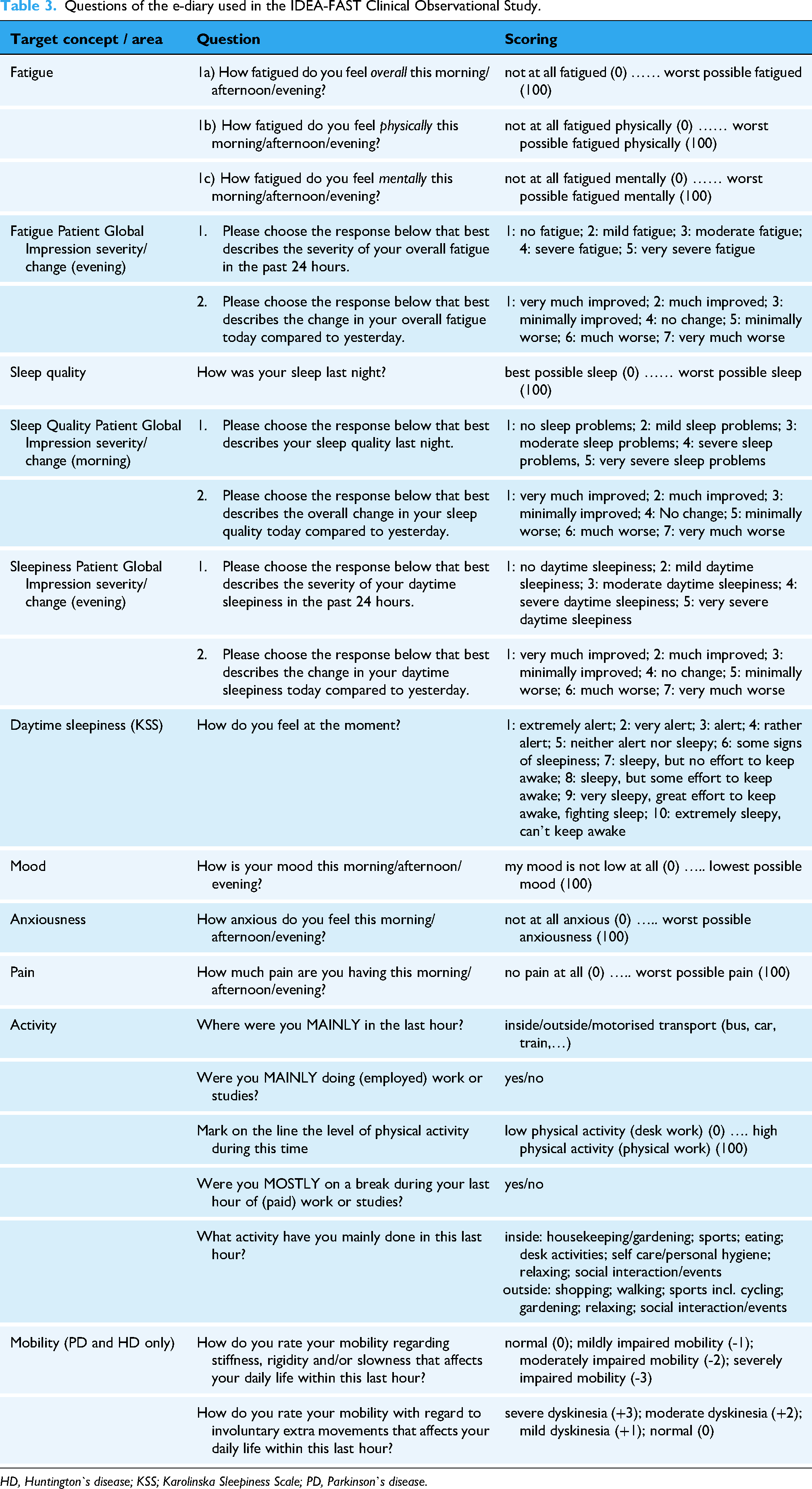
HD, Huntington`s disease; KSS; Karolinska Sleepiness Scale; PD, Parkinson’s disease.
DHT for the IDEA-FAST COS were selected based on participant-reported ease-of-use and data quality, which was tested in a pilot study with 14–40 participants per disease group and healthy volunteers, 27 as well as on qualitative interviews. 29 Figure 2 gives an overview of digitally assessed core dimensions of the target concepts used during the study.

Overview of digitally assessed core dimensions of the target concepts used during the IDEA-FAST Clinical Observational Study.
Digital data collection without requiring the active contribution of the user is done via the assessment of typing dynamics (WildKey app, LASIGE, Portugal 33 ) and social interaction parameters (WildKey Social app, LASIGE, Portugal 33 ) on their own smartphone. The AX6 inertial measurement unit (IMU) at the lower back (Axivity, UK34,35) measures movement, the Vital Patch positioned on the chest (VitalConnect, UK36,37) records an electrocardiogram, movement and temperature, and the VTT BedSensor, a pressure-sensitive device positioned under the mattress (VTT and eLive Ecosystem, Finland38,39), captures vital parameters as well as movement during sleep. For detailed information on all DHTs used in this study and the parameters collected (see Supplementary Table 5).
Study outcomes
The primary outcome is the strength of the relationship between the severity of daily self-rated fatigue (rated on a scale of 0–100 in the e-diary) and digital parameters over all cohorts.
Secondary outcomes are
the strength of the relationship between self-rated daytime sleepiness (Karolinska sleepiness scale, KSS, as reported in the e-diary) and sleep quality (0–100 in the e-diary) with digital parameters over all cohorts; the strength of the relationship between self-rated FACIT-Fatigue, weekly fatigue visual analogue scale (fVAS) or acute Medical Outcomes Study –Sleep Scale (MOS-SS) and digital parameters over all cohorts; the strength of the relationship between self-rated fatigue, sleep quality, and daytime sleepiness and digital parameters within the specific disease cohorts; the definition of minimal clinically important differences (MCID) and changes (MCIC) of digital parameters for fatigue/daytime sleepiness/sleep quality based on the information provided by participants (Patient Global Impression of Severity (PGIS), Patient Global Impression of Change (PGIC); Likert scales from 1–7 as reported in e-diary, 1 point change is considered a meaningful change)
We will also ask a subset of participants for feedback on the acceptability and usability of the technologies, via semi-structured interviews.
Analysis overview
The study employs a structured data management and analysis approach integrating clinical, DHT-derived and PRO data. Clinical data are captured using paper-based CRFs and subsequently entered into a secure electronic case report form system, while DHT data follows standardized, secure transfer pipelines to the IDEA-FAST data management platform. Data integration ensures pseudonymization and GDPR compliance, enabling secure access for analysis. Handling of missing data is described in detail in the supplementary material.
The study participants will be assigned for analysis into an identification analysis set (IAS) and an evaluation analysis set (EAS). In the IAS, advanced statistical and machine learning methods will be applied. The analysis will be focused on identifying digital endpoints to reflect the daily fatigue, sleep quality and daytime sleepiness. Candidate digital biomarkers will be identified across all disease cohorts as well as within IMID and NDD cohort groups separately, as well as within each specific disease cohort. The input digital features can be either raw signals (e.g., ECG), low-level derived features (e.g., 1-min heart rate) or high-level derived features (e.g., resting heart rate). The modeling task will consist of finding the optimal combination of digital measures and the related optimal time window that best estimate the response measures.
The primary focus will be using supervised learning methods such as lasso regression, random forest or extreme gradient boosting. Deep learning methods applied to raw signals or low-level features will also be explored such as long short-term memory recurrent neural networks. The response measure will be the self-reported fatigue, daytime sleepiness or sleep quality each modeled separately. The preferred models will be able to reflect daily self-reported measures, per the study's primary objective, but other timescales may also be explored. The supervised learning problem will be formulated as both being able to predict the reported scores (preferred) or being able to predict a participant's change from baseline in the score. In the latter case, baseline of the response measure will be defined as the mean of the timepoints from the first valid DHT use period and baseline for the digital measures will be defined separately for each digital measure using data from the first valid DHT use period.
An initial model will be built with only covariates and baseline measures and then subsequent models will be built including digital measures to see if they have any additional predictive value. A cross-validation procedure will be applied to the training set for hyperparameters tuning using a stratified five-fold cross validation. Data aggregation, normalization, features selection and modelling steps will be performed during the training procedure. Models will be evaluated with performance metrics and scatter plots comparing the observed response variable and the predictions. In addition to supervised methods, unsupervised approaches such as factor analysis may be explored.
Fifty participants in each cohort will be held-out for the final evaluation analysis, which will focus on characterizing the properties of the digital endpoints including the test-retest reliability, construct validity and sensitivity to change. Access to the EAS of participant data is tightly controlled to closely mimic an independent study. Exploratory analyses will probe additional symptoms, subgroup characteristics and correlations with PROs, aiming to refine digital endpoints as robust monitoring biomarkers for cross-disease application. 24
Discussion
The IDEA-FAST Clinical Observational Study represents a large, ambitious and novel approach to measure fatigue, impaired sleep quality and daytime sleepiness, debilitating symptoms prevalent across neurodegenerative (NDD) and immune-mediated diseases (IMID). By leveraging advances in digital health technology (DHT), this study seeks to move beyond the limitations of traditional self-reported parameters, offering a more granular and objective assessment of these symptoms.
Key strengths of the IDEA-FAST COS include the scientific advice of the European Medicines Agency for the study protocol presented here, 24 and the conduct of a feasibility study that served as the basis of the DHT battery used in this large multicenter study.26,27 Moreover, the inclusion of six disease groups and healthy volunteers can be seen as relevant strength, enabling the exploration of both cross-disease and disease-specific digital endpoints. The integration of diverse DHT, including smartphone-based apps, physical activity and mobility tracker, ECG and a bed sensor (Figure 2) reflects the study's commitment to capturing a multidimensional perspective on the core dimensions of the target concepts. By correlating digital parameters with self-reported and clinical parameters, the study not only aims to identify reliable digital endpoints but also intends to quantify their clinical relevance through evaluating the minimal important difference from a patient and clinical perspective. 40 Analysis is structured such that a subset of participants in each disease group are randomly assigned to a hold-out evaluation analysis set for final evaluation of the developed digital endpoints. Thus, the large sample size of this study allows for both the exploration and identification of digital endpoints together with their independent evaluation. The inclusion of qualitative interviews ensures that the usability and acceptability of digital tools are critically evaluated, a key factor for future implementation in clinical trials or real-world settings.
Challenges include ensuring participant compliance with the use of DHT and managing the vast heterogeneity of data across disease groups. We also do not span the entire year for the individual assessment, which may hinder intra-individual analysis of seasonal fluctuations in the symptoms of interest. However, we do not consider this to be a specific objective of this study.
Conclusion
In conclusion, IDEA-FAST COS has the potential to redefine how fatigue, impaired sleep quality and daytime sleepiness, and related symptoms are measured and understood using digital devices and ecological momentary assessment. If the study succeeds in identifying objective, scalable digital (monitoring) endpoints of these symptoms, this will significantly enhance the development of parameters that can be used in clinical trials. This, in turn, can pave the way for more personalized symptom management in neurodegenerative and immune-mediated conditions.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261428155 - Supplemental material for IDEA-FAST clinical study protocol: Identifying digital end-points of fatigue, sleep quality and daytime sleepiness in N = 2000
Supplemental material, sj-docx-1-dhj-10.1177_20552076261428155 for IDEA-FAST clinical study protocol: Identifying digital end-points of fatigue, sleep quality and daytime sleepiness in N = 2000 by Walter Maetzler, Stefan Avey, Andrea Pilotto, C Janneke van der Woude, Christopher A Lamb, Ralf Reilmann, Teemu Ahmaniemi, Nadir Ammour, Svenja Aufenberg, Luisa Avedano, Neil Basu, Simon Beniston, Sofia Bonomelli, Susanne T de Bot, Diogo Branco, Ian Bruce, Christopher Neil Bull, Neshat Chareh, Meenakshi Chatterjee, Hector Chinoy, Jade Chisholm, Hayley Connolly, Francesca Cormack, Leonor Correia Guedes and Cliona Cowhig, Dina De Sousa, Silvia Del Din, Monique Devillers, Inês M Dias, Colin Bone Dodds, Mary Doona, Annika Dvinskis, Joaquim J Ferreira, Konstantinos Fourlakis, Ulli Funken, Kay Goulden, Johanna Graeber, Miriam Grande Gordon, Hanna Graßhoff, Tiago Guerreiro, Tina Hagen-Hurley, Clint Hansen, Sinead Harney, Ailsa Hart, Chloe Hinchliffe, Sophia Hinz, Marthe Alver Hovsbakken, Michael Jackson, Lauren John, Hanna Kaduszkiewicz, Jérôme Kalifa, Niamh Kelly, Nick Kennedy, Norelee Kennedy, Juha M Kortelainen, Laura C. M. Kuijper, Samuel Labrecque, Yasmin Laidler, Tanja Lange, Maria Boge Lauvsnes, Jimmy K. Limdi, Ellen Lirani-Silva, Victoria Macrae, Corina Maetzler, Mayca Marín, Dario Masullo, Tania Nightingale, Alessandro Padovani, Miles Parkes, Emma Paulides, Clémence Pinaud, Zaireen Pios, Costantino Pitzalis, Alexandra Prodan, Rana Zia Ur Rehman, Gabriela Riemekasten, Andrea Rizzardi, Matthew W Roche, Lynn Rochester, Robbin Romijnders, Stefan Schreiber, Katarina Schwarzová, Christian Selinger, Klaus Seppi, Louise Stockley, Sreedhar Subramanian, Kai Sun, Nick Taptiklis, Gerlynn Tiongson, Kasper F van der Zwaan, Evert-Ben van Veen, Louise Warren, David Wenn, Grzegorz Witkowski, Alison Yarnall, Frederic Baribaud, Geert Van Gassen, Nikolay V. Manyakov, Wan-Fai Ng, in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076261428155 - Supplemental material for IDEA-FAST clinical study protocol: Identifying digital end-points of fatigue, sleep quality and daytime sleepiness in N = 2000
Supplemental material, sj-docx-2-dhj-10.1177_20552076261428155 for IDEA-FAST clinical study protocol: Identifying digital end-points of fatigue, sleep quality and daytime sleepiness in N = 2000 by Walter Maetzler, Stefan Avey, Andrea Pilotto, C Janneke van der Woude, Christopher A Lamb, Ralf Reilmann, Teemu Ahmaniemi, Nadir Ammour, Svenja Aufenberg, Luisa Avedano, Neil Basu, Simon Beniston, Sofia Bonomelli, Susanne T de Bot, Diogo Branco, Ian Bruce, Christopher Neil Bull, Neshat Chareh, Meenakshi Chatterjee, Hector Chinoy, Jade Chisholm, Hayley Connolly, Francesca Cormack, Leonor Correia Guedes and Cliona Cowhig, Dina De Sousa, Silvia Del Din, Monique Devillers, Inês M Dias, Colin Bone Dodds, Mary Doona, Annika Dvinskis, Joaquim J Ferreira, Konstantinos Fourlakis, Ulli Funken, Kay Goulden, Johanna Graeber, Miriam Grande Gordon, Hanna Graßhoff, Tiago Guerreiro, Tina Hagen-Hurley, Clint Hansen, Sinead Harney, Ailsa Hart, Chloe Hinchliffe, Sophia Hinz, Marthe Alver Hovsbakken, Michael Jackson, Lauren John, Hanna Kaduszkiewicz, Jérôme Kalifa, Niamh Kelly, Nick Kennedy, Norelee Kennedy, Juha M Kortelainen, Laura C. M. Kuijper, Samuel Labrecque, Yasmin Laidler, Tanja Lange, Maria Boge Lauvsnes, Jimmy K. Limdi, Ellen Lirani-Silva, Victoria Macrae, Corina Maetzler, Mayca Marín, Dario Masullo, Tania Nightingale, Alessandro Padovani, Miles Parkes, Emma Paulides, Clémence Pinaud, Zaireen Pios, Costantino Pitzalis, Alexandra Prodan, Rana Zia Ur Rehman, Gabriela Riemekasten, Andrea Rizzardi, Matthew W Roche, Lynn Rochester, Robbin Romijnders, Stefan Schreiber, Katarina Schwarzová, Christian Selinger, Klaus Seppi, Louise Stockley, Sreedhar Subramanian, Kai Sun, Nick Taptiklis, Gerlynn Tiongson, Kasper F van der Zwaan, Evert-Ben van Veen, Louise Warren, David Wenn, Grzegorz Witkowski, Alison Yarnall, Frederic Baribaud, Geert Van Gassen, Nikolay V. Manyakov, Wan-Fai Ng, in DIGITAL HEALTH
Footnotes
Acknowledgements
We would like to thank Jenelyn Carbonell, Juan de la Revilla, Pia Görrissen, Mohammed Halidu, Shyla Jagannatha, Thrusha Jain, Andrew King, Felicia Onoviran, Sibhi Raja, Madalena Rosario and Ally Speight for your valuable contribution to the study.
ORCID iDs
Ethical considerations
This study received ethical approval from the Ethics Committee at the sponsor's site (Medical Faculty of Kiel University) on 26 April 2022 (D 616/21).
Consent to participate
All study partipicants are informed of the objective of this research by the investigator or his or her representative, how it is carried out and what a participation means. They provide written consent to voluntary participate in the study and to the use of their data for scientific work.
Author contributions
WM, APi, CJvW, RRei, MJ, DW, AY, FB, NVM and WFN designed and directed the project, and raised funding. SAv, CAL, TA, NA, LAv, NB, SBe, STdB, DB, IB, CNB, NC, MC, HCh, FC, LCG, DdS, SdD, CBD, JJF, UF, TG, THH, CHan, SHar, AH, CHin, HK, JK, NKen, NoKen, JMK, MBL, JKL, DM, AP, MP, ClPi, CoPi, APr, RZUR, GR, MWR, LR, RRo, StSch, CS, KSe, LS, SrSu, KSu, NT, EBvV, LW, GW and GvG helped designing the project. SAuf, SBo, JC, HCo, CC, MoDe, ID, MaDo, AD, KF, KG, JG, MGG, HG, SHi, MAH, LJ, NKel, LCMK, SL, YL, TL, ELS, VM, CM, MM, TN, EP, ZP, AR, KS, GT and KFvdZ carried out the project. All authors discussed the results and contributed to the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Innovative Medicines Initiative (Grant No. 853981).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
A study protocol is described here. Data collection of this study has already started and results will be presented in future publications.
Supplemental material
Supplemental material for this article is available online.
