Abstract
Background and objective
Despite advances in imaging, analysis of part-solid nodules (PSNs) using conventional clinical-radiological features (CRF) remains highly subjective. Therefore, we introduced a clinical-radiological transformer-attention algorithm, the Prior Clinical-Radiological Transformer-Attention Net (PCRTA-Net), and evaluated its efficacy for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as PSNs.
Methods
In this retrospective study, 277 patients with lung adenocarcinoma presenting as PSN were randomly divided into training (n = 194, 70%) and validation (n = 83, 30%) sets. Four diagnostic models were developed: (1) Transformer Attention Net (TA-Net), derived from computed tomography imaging analysis; (2) PCRTA-Net, integrating CRFs with TA-Net; (3) radiomics analysis based on feature engineering; and (4) CRF model utilizing multiple dimensionality reduction methods, including principal component analysis, least absolute shrinkage and selection operator, and recursive feature elimination with cross-validation. Model performance was evaluated using area under the curve (AUC), decision curve analysis, and net reclassification index (NRI) and integrated discrimination improvement (IDI) heatmaps.
Results
PCRTA-Net demonstrated superior performance over the other models, achieving an AUC of 0.9084 (95% CI: 0.8055–0.9779). It consistently showed a superior net benefit across nearly the entire range of clinically relevant threshold probabilities (3.7–95%) compared with the other models. It achieved the highest NRI and IDI in the heatmaps, confirming its incremental predictive value.
Conclusion
PCRTA-Net is a diagnostic algorithm integrating CRF with TA-Net that enhances the preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as PSN. It exhibited superior diagnostic performance, potentially facilitating its application in precision medicine.
Keywords
Introduction
Lung cancer is one of the most prevalent malignancies worldwide, with adenocarcinoma being the predominant histological subtype.1,2 Widespread clinical adoption of low-dose computed tomography (CT) has significantly improved small pulmonary nodule detection. 3 These nodules are classified into three distinct categories based on their radiological features: pure ground-glass nodules (pGGNs), consisting entirely of ground-glass opacities; solid nodules (SNs), which exhibit exclusively solid components; and part-solid nodules (PSNs), characterized by a combination of both ground-glass and solid elements. 4 Among these, PSNs represent a unique clinical entity, showing a reduced propensity for invasive behavior, pleural invasion, lymphatic metastasis, and airspace dissemination compared to SNs. Furthermore, patients with PSNs experience intermediate survival outcomes, with improved recurrence-free survival (RFS) and overall survival rates compared to those of patients with SNs, but lower overall survival rates than patients with pGGNs.5–7
Pathologically, PSN postoperative diagnosis corresponds to adenocarcinoma in situ (AIS), minimally invasive adenocarcinoma (MIA), or invasive adenocarcinoma (IAC), depending on the degree of invasiveness. Postoperative outcomes vary significantly among these subtypes; AIS and MIA demonstrate a 5-year RFS approaching 100% after complete resection, whereas that of IAC is considerably lower at 74.1%.8,9 Consequently, limited resections such as wedge resection or segmentectomy are preferred for AIS and MIA to preserve lung function and reduce surgical morbidity. In contrast, IAC typically requires lobectomy to lower the recurrence risk, although this approach is associated with higher rates of complications such as mediastinal fistula and hydropneumothorax.10,11 Therefore, accurate preoperative differentiation of IAC from other PSN pathological types is crucial for optimizing surgical planning and improving patient outcomes.
Despite advances in imaging, utilization of the conventional clinical-radiological features (CRF) of PSNs remains highly subjective and relies heavily on qualitative or semi-quantitative features such as lesion size, morphological characteristics, and consolidation-to-tumor ratio (CTR). This approach lacks standardized quantitative metrics, leading to interobserver variability and suboptimal accuracy in predicting invasiveness.12,13 Additionally, traditional CRF methods often fail to fully exploit PSN pixel-level CT data, which may contain critical tumor heterogeneity information.
Radiomics, an emerging computational technique, addresses these limitations by extracting high-dimensional quantitative features from medical images. Although radiomics captures subtle texture patterns and can improve predictive performance, it depends on handcrafted feature engineering, which may overlook complex spatial relationships, and is susceptible to imaging protocol and segmentation method variations.14–17
To overcome these challenges, deep learning models, particularly those using Transformer-Attention Networks (TA-Net), offer promising solutions. These models automatically learn hierarchical representations from raw imaging data, thereby eliminating the need for manual feature extraction. Attention mechanisms enable the focused analysis of diagnostically important regions by evaluating pixel intensities and spatial patterns. TA-Net enhances the characterization of tumor heterogeneity, thereby improving interpretability and predictive accuracy. 18 Therefore, we developed two deep learning models: TA-Net, a CT imaging-based model that employs self-attention mechanisms to identify discriminative imaging biomarkers, and Prior Clinical-Radiological Transformer-Attention Network (PCRTA-Net), an integrated model combining CRF with TA-Net.
This study aimed to validate the superiority of PCRTA-Net over standalone TA-Net, conventional CRF, and radiomics analysis, and to further demonstrate its diagnostic clinical utility. This approach seeks to guide personalized surgical decision-making in patients with lung adenocarcinoma presenting as PSNs, ultimately optimizing patient outcomes.
Methods
Study population
This retrospective study included consecutive patients who underwent surgical resection of pulmonary nodules at Hunan Cancer Hospital, The Affiliated Cancer Hospital of Xiangya School of Medicine, Central South University between January 2021 and June 2023. It was performed in accordance with the Declaration of Helsinki and approved by the institutional review board (no: SBQLL-2022-134). The requirement for individual informed consent was waived in accordance with national regulations governing retrospective biomedical research using anonymized clinical data.
The initial inclusion criteria were: (1) radiologically confirmed PSNs measuring 5–30 mm on high-resolution CT scans; (2) clinical T1N0M0 staging according to the 8th edition of the TNM classification; and (3) available complete preoperative imaging data. Patients were excluded based on the following criteria: (1) diagnosis of any concurrent or prior malignancy within the past 5 years; (2) recipient of neoadjuvant therapy, including radiation, chemotherapy, or combined modality treatment; and (3) final histopathological diagnosis indicating atypical adenomatous hyperplasia rather than adenocarcinoma spectrum disease. Patient characteristics are shown in Supplemental Figure 1.
Imaging acquisition and radiological evaluation
All preoperative thoracic CT examinations were performed using multidetector CT systems from SOMATOM Definition AS + (Siemens Healthineers, Forchheim, Germany) and IQon Spectral CT (Philips Healthcare, Best, the Netherlands). Detailed technical parameters for image acquisition are provided in Supplemental Material S1.
Standardized Digital Imaging and Communications in Medicine files were spatially registered and subsequently archived in our institutional picture archiving and communication system for centralized analysis. This single-center approach ensured consistent image processing protocols and minimized variability introduced by multi-site data acquisition or scanner heterogeneity. Two board-certified thoracic radiologists (W.C. and H.Z.; each with over 10 years of experience) were blinded to all histopathological results and independently evaluated all eligible PSNs using lung window settings (width: 1500 HU; level: −700 HU). Detailed technical parameters for lesion segmentation are provided in Supplemental Material S2. To assess the reliability of the quantitative parameters, we used the intraclass correlation coefficient (ICC). Meanwhile, Cohen's kappa was employed to evaluate the consistency of the non-quantitative parameters.
Quantitative assessments were performed according to the protocols outlined in the 8th edition of the TNM staging system for pulmonary malignancies. 19 PSNs coexisted with ground-glass opacities and solid components. Ground-glass opacity was defined as a region with a mild, uniform increase in density that did not obscure the vascular markings beneath. The dominant solid component was characterized by an area exhibiting complete vascular obscuration and uniform attenuation. The longest orthogonal axis of the solid component in the axial plane was measured to determine lesion size. For nodules with multifocal or irregular solid parts, multiplanar reconstruction was used to identify the largest contiguous solid region. CTR was calculated by dividing the maximum diameter of the solid component by the total tumor diameter, a method that has been previously validated.20–22 A schematic panel demonstrating the calculation of CTR is included in Figure 1.

Overall study design illustrating the framework and approach utilized in this research.
Study design conceptual framework
We employed a binary classification task to discriminate between IAC and AIS/MIA. Figure 1 illustrates the overall study design. Four diagnostic models were developed: (1) TA-Net, based on CT imaging analysis; (2) PCRTA-Net, integrating CRF with TA-Net; (3) radiomics analysis utilizing feature engineering; and (4) a CRF model employing multiple dimensionality reduction methods, including principal component analysis (PCA), least absolute shrinkage and selection operator (LASSO), and recursive feature elimination with cross-validation (RFECV).
Transformer-based multi-head attention model development
An effective cross-modal fusion technique is essential for integrating diverse sources of information such as medical images and clinical data.18,23 Therefore, we introduced PCRTA-Net, an end-to-end deep learning architecture designed to seamlessly fuse prior CRF with CT image volume.
Because the lesion volumes and mask regions were relatively small, we extracted a 32 × 32 × 32 mm³ cubic patch from the volume of interest (VOI), with the patch center aligned to the centroid of the 3D mask within the CT image. The extracted lesion patch was then normalized within the lung window range (−1150, 350). PCRTA-Net accepts VOI cubic patch as the input, along with feature maps derived from qualitative or semi-quantitative features such as lesion size, morphological characteristics, and CTR.12–13 To extract meaningful representations, our backbone encoder integrates 3D residual learning24,25 and transformer-based multi-head attention mechanisms.26,27 The overall architecture of PCRTA-Net is depicted in Figure 2. Additionally, we designed TA-Net, which is derived solely from CT imaging analysis and does not incorporate the CRF.

Comprehensive architecture of the PCRTA-Net, highlighting its structural components and functional pathways. PCRTA-Net: prior clinical-radiological transformer-attention net.
Given the limited size of medical datasets, the backbone network must be compact with few parameters to facilitate training. Both residual learning and multi-head attention meet these requirements and have shown outstanding performance in image classification tasks.24–27 The architecture comprises four transformer-based multi-head attention blocks, 27 each incorporating convolution layers, rectified linear activation function (ReLU) activations, multi-head attention, fully connected layers, layer normalization, and skip connections. Multi-head attention provides several advantages in modeling complex feature dependencies. Specifically, each attention head learns to focus on different subspaces of the input features, thereby capturing diverse and complementary relationships within the data. This mechanism allows the model to attend simultaneously to both local and global contextual information, enhances the representation of subtle feature interactions, and reduces the risk of overlooking critical details. Moreover, by aggregating the outputs of multiple heads, the model achieves improved robustness, richer feature expressiveness, and greater generalization capability. The backbone network includes head and tail blocks. The head block consists of a convolutional layer, batch normalization (BN), ReLU, and MaxPool layer, which transform input volumes into feature maps, while increasing the channels from 13 to 32 and reducing spatial dimensions. The BN mitigates the internal covariate shift, and ReLU prevents vanishing gradients. 28 MaxPool further compresses features to prevent overfitting. The tail block features a convolution layer that reduces hierarchical feature dimensions by merging spatial information. Finally, a softmax activation function is used to predict the risk of IAC.
Model training involved data augmentation through random flipping along three spatial directions. We trained PCRTA-Net without transfer learning or pretrained models. A cross-entropy loss function was employed with adjusted loss coefficients to address class imbalance: two for IAC samples and one for AIS/MIA samples. Implementation was performed using PyTorch 29 with the ADAM optimizer, 30 set at a learning rate of 1 × 10−4, using default hyperparameters. Training was performed for 50 epochs with a batch size of four. Experiments were conducted on a workstation equipped with an Intel® Core™ i9-14900 K processor and an NVIDIA GeForce RTX 4090 GPU.
CRF and radiomic workflow
The CRF model employs multiple dimensionality reduction techniques, including PCA, 31 LASSO, and RFECV. 32 The technical parameters for CRF model construction are provided in Supplemental Material S3. Radiomics analysis is based on feature engineering and follows a structured pipeline comprising three main phases: image normalization, feature extraction, dimensionality optimization, and predictive modeling. 33 Computational image processing technical parameters and the radiomics pipeline are presented in Supplemental Material S4.
Model comparison statistical methods
A comprehensive statistical evaluation framework was employed to assess the diagnostic performance of the four models (TA-Net, PCRTA-Net, radiomics analysis, and CRF model) in discrimination of IAC from AIS/MIA. Model performance was quantified using receiver operating characteristic (ROC) curve analysis, with the area under the curve (AUC) and its 95% confidence interval (CI) serving as the primary metric. 34 The model that demonstrated the highest AUC robustness during validation was recognized as the optimal configuration. Additionally, we calculated other classification metrics, including accuracy, F1-score, negative predictive value (NPV), positive predictive value (PPV), sensitivity, and specificity—all with their associated 95% CI—derived from confusion matrices to provide a more comprehensive evaluation.
To assess the model reclassification performance, we further computed the net reclassification improvement (NRI) and integrated discrimination improvement (IDI) indices, which were visualized using heatmaps. Finally, we evaluated the clinical utility of the different models through decision curve analysis (DCA) to determine their clinical net benefit across the full range of incidence thresholds, from 0% to 100%.
Results
Study population characteristics
This retrospective study included 277 consecutive patients with pathologically confirmed lung adenocarcinoma who presented with PSNs on preoperative imaging. The cohort was randomly divided into a training set (n = 194, 70%) and a validation set (n = 83, 30%) using computer-generated randomization. Histopathological evaluation revealed IAC in 183 patients (66.1% prevalence). The IAC distribution was similar between the two groups, with 66% in the development set (n = 128) and 66.3% in the validation set (n = 55). Furthermore, a comprehensive assessment of demographic, clinical, and radiological parameters confirmed that the baseline characteristics were well-balanced across both datasets, as all intergroup comparisons showed no significant differences (p > .05). This finding ensures an equitable distribution of potential confounding variables. Additionally, the ICC and Cohen's kappa for CT radiological features—which ranged from 0.906 to 0.929—demonstrated excellent agreement (Table 1 and Supplemental Figure 2).
Comparative analysis of clinical-radiological features between the training and validation sets.

IAC: invasive adenocarcinoma; CTR: consolidation-to-tumor ratio; RUL: right upper lobe; RLL: right lower lobe; RML: right middle lobe; LUL: left upper lobe; LLL: left lower lobe; ICC: intraclass correlation coefficient.
CRF model performance
The CRF model was developed through a rigorous multistep feature selection process that integrates three complementary dimensionality reduction techniques (Supplemental Figure 3). First, the RFECV identified three key predictors: CTR, age, and lesion size. Concurrently, LASSO regression selected CTR and the vascular convergence sign as the most discriminative features. Furthermore, PCA generated eight principal components from a range of clinical and radiological variables including age, CTR, lesion size, lobulation sign, location, margin, pleural indentation sign, sex, shape, spiculation sign, vacuole sign, and vascular convergence sign. Intersection analysis using an UpSet plot demonstrated that CTR was the only feature consistently selected across all three methods (Figure 3), underscoring its critical role in distinguishing IAC in lung adenocarcinomas presenting as PSNs. The final CRF model, which incorporated CTR as the primary predictor, demonstrated robust diagnostic performance, with an AUC of 0.7864.

Intersection analysis employing an UpSet plot to delineate three complementary dimensionality reduction techniques: RFECV, LASSO, and PCA. RFECV: recursive feature elimination with cross-validation; LASSO: least absolute shrinkage and selection operator; PCA: principal component analysis.
Radiomics analysis performance
The radiomics analysis pipeline initially extracted 1239 quantitative imaging features from each PSN. After applying two-tailed t-tests (α = 0.05), 539 features showing statistical significance were retained for further analysis. Next, a pairwise correlation analysis (Pearson's |r| > 0.9) was used to remove redundant features, resulting in a set of 532 non-collinear candidates. These features were then subjected to LASSO-regularized regression with embedded five-fold cross-validation, which determined an optimal regularization parameter (λ = 0.026) and ultimately selected 11 discriminative features (see Supplemental Figure 4). The final radiomic model exhibited strong diagnostic performance, achieving an AUC of 0.8305 in the validation set.
Comparison of model diagnostic performance
The four evaluated models demonstrated distinct performance profiles in differentiating IAC from AIS/MIA within the PSNs. ROC analysis (Supplemental Figure 5) identified PCRTA-Net as the top-performing model, with an AUC of 0.9084 (95% CI: 0.8055–0.9779), significantly outperforming TA-Net (AUC = 0.8552, 95% CI: 0.7717–0.9325), radiomics analysis (AUC = 0.8305, 95% CI: 0.7081–0.9262), and the CRF model (AUC = 0.7864, 95% CI: 0.6976–0.8636). DeLong's test confirmed statistically significant differences in AUC performance when comparing PCRTA-Net with TA-Net (p = .0339), radiomics analysis (p = .0315), and the CRF model (p = .0018) (Supplemental Table 1). Furthermore, this performance hierarchy was consistently observed across multiple classification metrics (Table 2 and Figure 4), with PCRTA-Net achieving superior balanced accuracy (0.8313, 95% CI: 0.7470–0.9036) and F1-score (0.8627, 95% CI: 0.7907–0.9245).
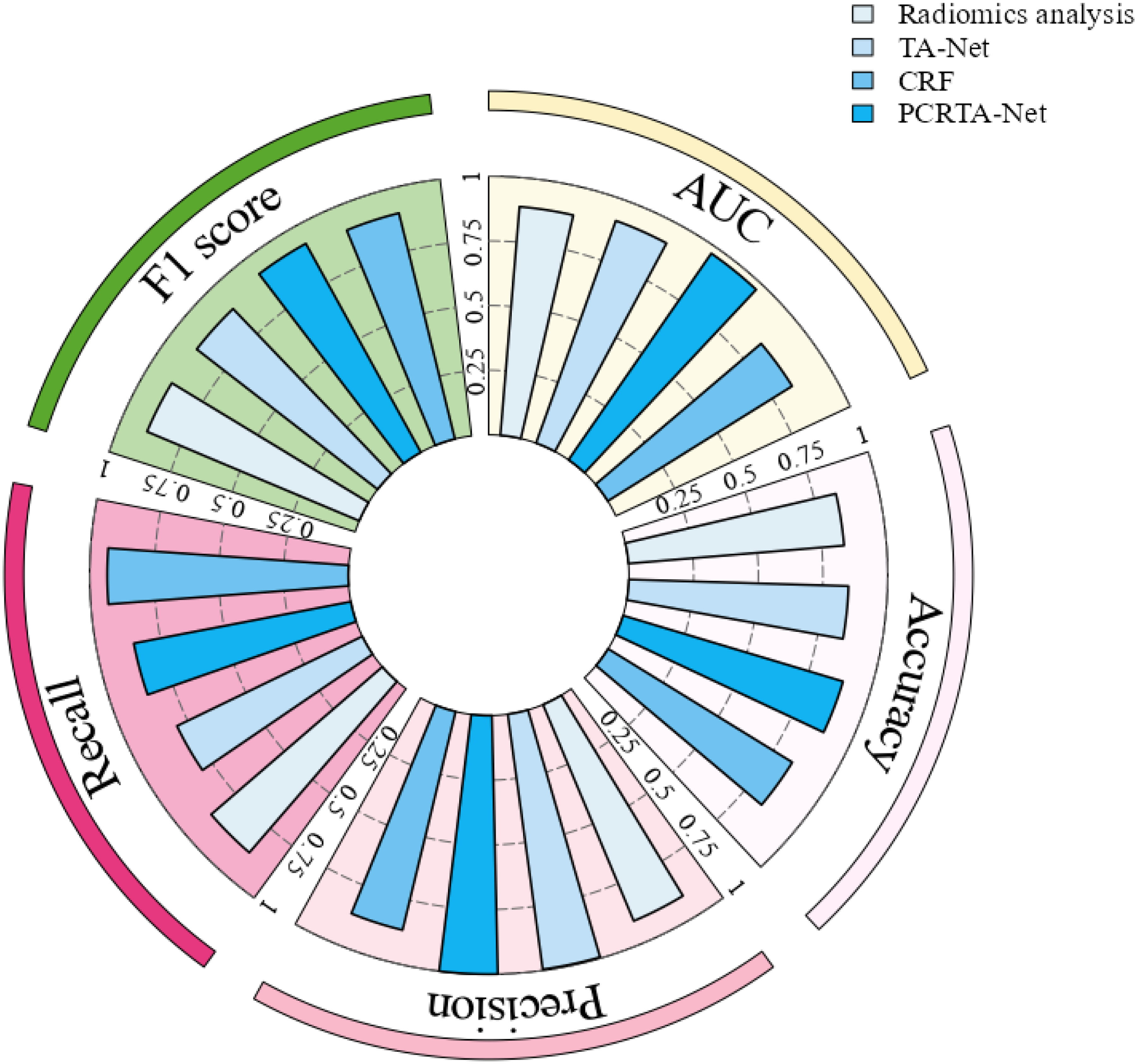
Visualization of the diagnostic performance of four models (TA-Net, PCRTA-Net, radiomics analysis, and CRF model) through a polar coordinate graph representation. TA-Net: transformer attention net; PCRTA-Net: prior clinical-radiological transformer-attention net; CRF: clinical radiological features.
Comparative diagnostic efficacy of PCRTA-Net, TA-Net, CRF model, and Radiomics analysis in Assessing Invasiveness of Part-Solid Nodule Lung Adenocarcinoma.
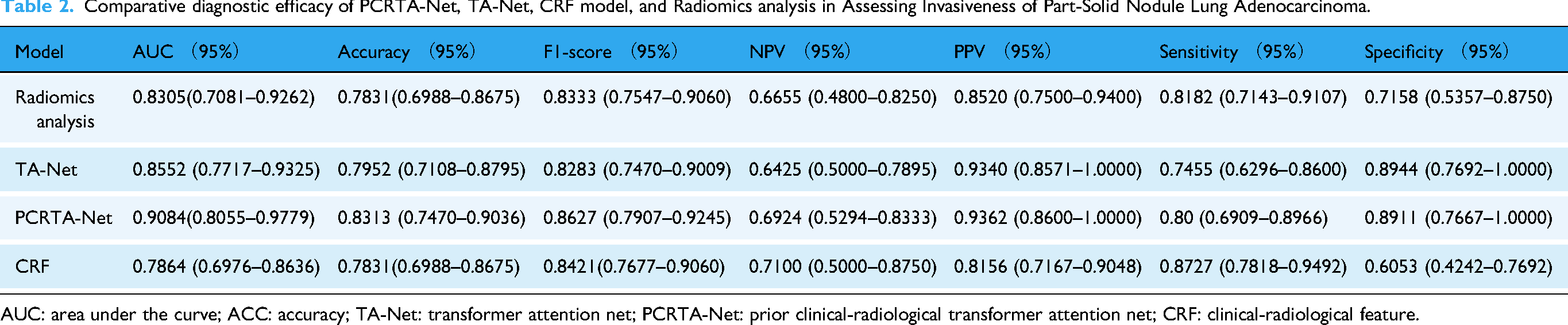
AUC: area under the curve; ACC: accuracy; TA-Net: transformer attention net; PCRTA-Net: prior clinical-radiological transformer attention net; CRF: clinical-radiological feature.
The confusion matrix analysis (Figure 5) further revealed that PCRTA-Net maintained the best balance in classifying both positive (IAC) and negative (AIS/MIA) cases. Specifically, it correctly identified 30.1% (25) of the AIS/MIA cases as true positives and 53.0% (44) of the IAC cases as true negatives, with relatively low false-positive (3.6%) and false-negative rates (13.3%). In contrast, radiomics analysis showed a higher false-negative rate of 12.0% and a lower true-positive rate of 24.1%. TA-Net (Figure 5(b)) demonstrated a moderate performance but still had a higher false-negative rate (16.9%) than PCRTA-Net. The CRF model (Figure 5(d)) exhibited the poorest balance, with false positives at 13.3% and false negatives at 8.4%, tending toward the overprediction of IAC.
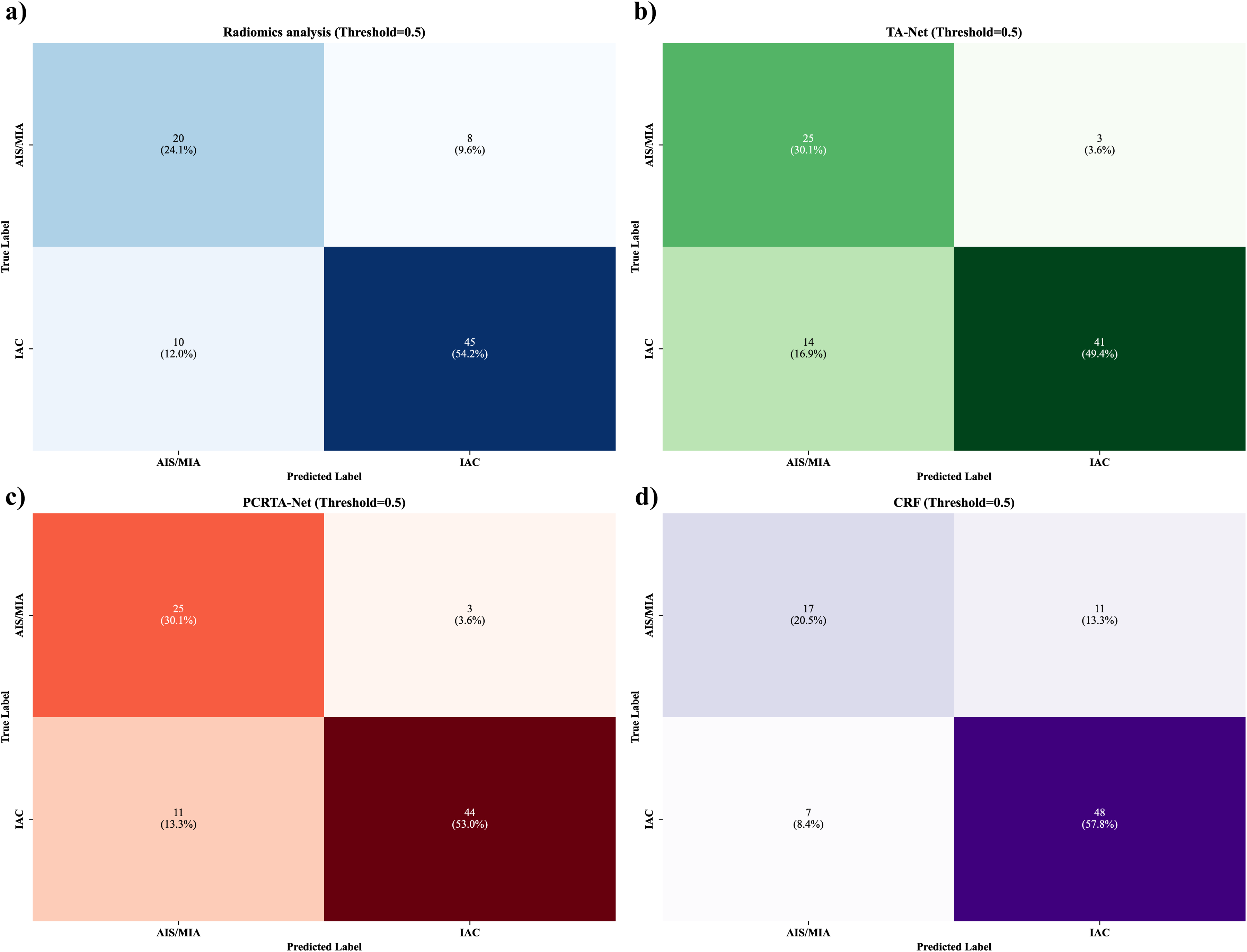
Confusion matrix analysis of the four models (TA-Net, PCRTA-Net, radiomics analysis, and CRF model), showcasing the classification accuracy and errors. TA-Net: transformer attention net; PCRTA-Net: prior clinical-radiological transformer-attention net; CRF: clinical radiological features.
The DCA (Supplemental Figure 6) indicated that PCRTA-Net consistently provided a superior net benefit across nearly the entire range of clinically relevant threshold probabilities (3.7–95%). This broad range aligns with the observed PSN invasive prevalence in our cohort (about 10–60% as reported in prior literature5–7) and ensures robustness across potential real-world scenarios. Furthermore, a comprehensive reclassification analysis using the NRI and IDI heatmaps (Figure 6) confirmed the advantages of PCRTA-Net. It revealed NRI (Figure 6(a)) improvements of 0.055 (vs. TA-Net), 0.16 (vs. radiomics), and 0.213 (vs. CRF), and corresponding IDI values (Figure 6(b)) of 0.177, 0.282, and 0.372, respectively (all p < .005). Additionally, as shown in Supplemental Figure 7, the attention overlay maps display heat spots that are distributed not only over the tumor core but also across its boundaries and surrounding lung parenchyma. This spatially dispersed pattern reflects the global self-attention mechanism of the Transformer, which captures long-range dependencies and integrates contextual information from multiple anatomical regions. Such global attention allows the model to focus simultaneously on the lesion and its clinically relevant context, highlighting its interpretability and clinical plausibility. Collectively, these results establish PCRTA-Net as the most robust and clinically applicable model for the preoperative prediction of IAC in PSNs.

Comprehensive reclassification analysis utilizing net reclassification improvement (NRI) and integrated discrimination improvement (IDI) heat maps to compare the four models (TA-Net, PCRTA-Net, radiomics analysis, and CRF model). TA-Net: transformer attention net; PCRTA-Net: prior clinical-radiological transformer-attention net; CRF: clinical radiological features.
Subgroup analysis
The lesion size subgroups (≤20 and > 20 mm) revealed distinct diagnostic profiles across models in assessing PSN lung adenocarcinoma invasiveness (Supplemental Table 2). In the < 20 mm subgroup, PCRTA-Net achieved the highest AUC of 0.9436 (95% CI: 0.8344–1.0000), significantly outperforming the CRF model (AUC = 0.7603, 95% CI: 0.6261–0.8868). TA-Net (AUC = 0.8378, 95% CI: 0.6936–0.9448) and radiomics analysis (AUC = 0.8355, 95% CI: 0.6853–0.9627) showed comparable performance but were outperformed by PCRTA-Net. In the ≥ 20 mm subgroup, PCRTA-Net maintained superiority with an AUC of 0.8138 (95% CI: 0.6071–0.9747), though statistical significance compared with TA-Net (AUC = 0.7983, 95% CI: 0.5804–0.9588) and the CRF model (AUC = 0.7971, 95% CI: 0.6049–0.9706) was not observed. Radiomics analysis (AUC = 0.7714, 95% CI: 0.5882–0.9276) demonstrated the poorest performance in this subgroup.
The CTR subgroups (CTR ≤ 1/2 and CTR > 1/2) also revealed distinct diagnostic profiles across models in assessing PSN lung adenocarcinoma invasiveness (Supplemental Table 3). In the CTR ≤ 1/2 subgroup, PCRTA-Net demonstrated superior discriminative power with an AUC of 0.8992 (95% CI: 0.7785–0.9784), outperforming TA-Net (AUC = 0.7526, 95% CI: 0.5974–0.8982), radiomics analysis (AUC = 0.7889, 95% CI: 0.6360–0.9156), and the CRF model (AUC = 0.7218, 95% CI: 0.5855–0.8586). In the CTR > 1/2 subgroup, PCRTA-Net maintained dominance with an AUC of 0.9612 (95% CI: 0.8819–1.0000). TA-Net achieved an AUC of 0.8279 (95% CI: 0.5000–1.0000), while radiomics analysis (AUC = 0.8221, 95% CI: 0.4868–1.0000) and the CRF model (AUC = 0.6915, 95% CI: 0.6129–0.7794) displayed weaker overall performance.
Discussion
Whilst the emerging computational technique of radiomics addresses the predictive performance limitations associated with conventional CRF of PSNs, it can overlook complex spatial relationships, and is susceptible to variations in imaging protocols and segmentation methods. To address this, we developed PCRTA-Net, an innovative artificial intelligence (AI) model integrating CRF with TA-Net. In this study, we demonstrate that PCRTA-Net excels in the preoperative prediction of PSN invasiveness. It achieved the highest AUC, along with superior balanced accuracy, precision, and F1-score compared with the three other diagnostic models. Subgroup analyses based on lesion size and CTR showed that PCRTA-Net consistently exhibited superior diagnostic performance in assessing PSN lung adenocarcinoma invasiveness across different subgroups, outperforming the other models. Furthermore, its robust performance across various decision thresholds and significant reclassification improvements highlight its potential to enhance clinical decision-making and personalize surgical strategies for PSNs.
Previous clinical models have predominantly depended on CRF to evaluate the invasiveness of PSNs.12,13 Although these features showed predictive value when considered independently, our comprehensive feature selection methods, including RFECV, LASSO, and PCA, identified CTR as the only consistent predictor across all dimensionality reduction techniques. CTR acts as a surrogate marker indicative of an infiltrative invasive growth pattern in lung adenocarcinoma manifesting as PSNs,6,2235–37 with elevated CTR values correlating with aggressive pathological characteristics. Moreover, the semi-quantitative assessment of CTR, segmented into four equal parts, is considered a valuable tool for the prognostic evaluation of PSNs. 38
Comparative analysis of the four predictive models revealed a clear performance hierarchy, illustrating the progressive advantages of advanced computational approaches in the preoperative assessment of PSN invasiveness. Radiomics analysis demonstrated superior discriminative ability compared to the conventional CRF model. This advantage stems from the ability to extract and quantify subvisual tumor heterogeneity through high-throughput feature analysis such as texture and wavelet transformations, which are linked to histopathological markers of biological aggressiveness. However, the limitations of handcrafted feature engineering become apparent when comparing with TA-Net. TA-Net utilizes a self-attention mechanism that dynamically captures long-range spatial relationships and identifies complex nonlinear patterns that extend beyond predefined radiomic features. Building on this, PCRTA-Net emerged as the most effective approach, synergistically combining the advanced imaging analysis of TA-Net with clinically validated predictors. This integration combines the pattern-recognition strength of deep learning with established pathological correlates, resulting in a robust and clinically interpretable model that achieves superior diagnostic accuracy.18,23 By overcoming the limitations of traditional radiomics through attention-based feature learning, PCRTA-Net offers a comprehensive solution for the preoperative risk stratification of PSNs, effectively bridging the gap between sophisticated computational analysis and practical clinical decision-making.
This study has several limitations. First, as this was a single-center retrospective analysis with a relatively small sample size. Therefore, our findings may have been affected by selection bias and have limited generalizability. Second, although TA-Net and PCRTA-Net demonstrated superior performance, their “black-box” nature remains a challenge; the specific imaging features emphasized by the attention mechanism are not fully interpretable. Third, the lack of long-term follow-up data limited our ability to evaluate the impact of the models on treatment outcomes such as recurrence rates or survival. Fourth, our study was confined to PSNs, leaving the applicability of the models to pGGNs or SNs untested.
In conclusion, this study established PCRTA-Net as a cutting-edge AI tool for the preoperative prediction of invasiveness in lung adenocarcinoma presenting as PSNs. By integrating TA-Net with CRF, PCRTA-Net demonstrated superior diagnostic performance compared with traditional methods. This study marks a significant advancement in the incorporation of AI-driven precision diagnostics into clinical practice, enhancing early-stage lung cancer management. It motivates researchers to explore implementation outcomes beyond technical performance, such as healthcare access, patient outcomes, and socioeconomic impact. Centering equity in AI development and deployment allows us to harness technology to reduce, rather than amplify, global health inequities. 39 This study underscores the importance of ensuring that AI serves as a true catalyst for inclusive healthcare advancement.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251393390 - Supplemental material for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules
Supplemental material, sj-docx-1-dhj-10.1177_20552076251393390 for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules by Wei Chen, Yalin Li, Lian Jian and Hong Zheng in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251393390 - Supplemental material for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules
Supplemental material, sj-docx-2-dhj-10.1177_20552076251393390 for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules by Wei Chen, Yalin Li, Lian Jian and Hong Zheng in DIGITAL HEALTH
Supplemental Material
sj-docx-3-dhj-10.1177_20552076251393390 - Supplemental material for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules
Supplemental material, sj-docx-3-dhj-10.1177_20552076251393390 for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules by Wei Chen, Yalin Li, Lian Jian and Hong Zheng in DIGITAL HEALTH
Supplemental Material
sj-docx-4-dhj-10.1177_20552076251393390 - Supplemental material for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules
Supplemental material, sj-docx-4-dhj-10.1177_20552076251393390 for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules by Wei Chen, Yalin Li, Lian Jian and Hong Zheng in DIGITAL HEALTH
Supplemental Material
sj-doc-5-dhj-10.1177_20552076251393390 - Supplemental material for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules
Supplemental material, sj-doc-5-dhj-10.1177_20552076251393390 for PCRTA-Net: A clinical-radiological transformer-attention algorithm for preoperative prediction of pathological invasiveness in lung adenocarcinoma presenting as part-solid nodules by Wei Chen, Yalin Li, Lian Jian and Hong Zheng in DIGITAL HEALTH
Footnotes
Ethics approval and consent to participate
The study protocol was approved by the institutional review board and due to the retrospective nature of the study. The informed consent requirement was abandoned.
Consent for publication
Our institution has given consent for publication.
Authors’ contributions
Wei Chen contributed to manuscript preparation, literature research, and data analysis. Yalin Li and Lian Jian contributed to data collection. Wei Chen contributed to data analysis and interpretation. Hong Zheng contributed to study conception and design, manuscript review and guarantor of integrity of the entire study. All authors have read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data sharing statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
