Abstract
Objective
Digital health applications are digital medical products (e.g. apps and web applications) that are prescribed by healthcare service providers and reimbursed by health insurances. Before digital health applications can be prescribed, manufacturers must fulfill the requirements of the approval process. The aim of this study is to identify the challenges faced by digital health applications manufacturers in the German fast-track process.
Methods
Use of a questionnaire-based online survey with 10 digital health applications manufacturers from March to October 2023. The survey design is based on the German digital health application regulation and the digital health application guideline. Data collection via LimeSurvey and HubSpot and data visualization in bar charts.
Results
Digital health applications manufacturers see the fast-track process as a challenge, especially in terms of demonstrating positive care effects and compliance with regulatory standards. Challenges include conducting clinical trials, negotiating pricing and improving market acceptance even with innovative approaches. Views on the profitability and relevance of the digital health application model are varied, with concerns over pricing and market strategies predominating.
Conclusions
The fast-track approval of digital health applications in Germany is a significant barrier to innovation and market entry. Collaboration between stakeholders is essential to optimizing processes while ensuring safety and accessibility for patients. However, even with the limitations, the results provide insights for future research and policy refinements that are necessary to improve the implementation of digital health applications in healthcare.
Keywords
Introduction
The German healthcare system has over 73 million insured persons and aims to provide high-quality medical care for all members.
1
The German Digitalization in Healthcare Act (DVG) of 2019 aims to achieve this goal through the use of digital solutions as part of a reimbursement system.
2
This allows digital health applications (DiGA) in the form of digital medical products such as apps or web applications to be prescribed to the insured person by physicians or psychotherapists and reimbursed by the health insurances.3,4 For this purpose, the DVG introduced the DiGA fast-track process to allow manufacturers to integrate their DiGAs into the reimbursement system, whereby the manufactures negotiate the price for the DiGA with the German national association of statutory health insurance funds (
The current situation of DiGAs in Germany shows that a total of 369,526 activation codes were redeemed by DiGA users in the time between the introduction of the DiGA fast-track process in October 2020 until September 2023. 8 In terms of user demographics, of all DiGA users (68,993), approximately 66.5% (45,883) of DiGA users are women and the average age across genders is 44.9 years. 9 Most DiGAs focus on back pain, tinnitus, and migraines, but mental health DiGAs are the most popular with 30,7% of all users. 9 The usual duration of DiGA use is around 90 days. 10
However, although innovative approaches such as the DiGA fast-track process are being implemented, several challenges are identifiable. The slow growth of DiGAs is illustrated by the limited number of new publications—8 in 2020, 14 in 2021 and 12 in 2023. 10 A YouGov survey highlights the need for education, with 62.5% of respondents not knowing about DiGAs. 11 The low prescription rate of around 4% of physicians and psychotherapists as well as insolvency cases of DiGA manufacturers with high reimbursements illustrate the challenges for comprehensive integration.9,12,13 These challenges demonstrate barriers to the effective integration of DiGAs into patient care that need to be overcome to realize the full potential of DiGA in Germany.
This work aims to identify and analyze the challenges of the fast-track process from the perspective of DiGA manufacturers. Motivated by the discrepancy between the potential of DiGAs and their low acceptability, this study aims to gain new insights and serve as a basis for future studies to continuously monitor and improve the approval process. Additionally, it will be reflected to what level the German model can be seen as effective and adaptable, especially in comparison to other international approaches.
Material and methods
An exploratory survey was conducted for the study to generate hypotheses on the challenges of DiGA approval. Based on the German Digital Health Application Regulation (DiGAV) and the Guideline for the fast-track process for DiGA (DiGA-guideline), a questionnaire was designed to ask DiGA manufacturers about the challenges related to fast-track process in an online survey. The DiGAV is a regulation for DiGA approval that contains requirements that must be demonstrated by manufacturers before DiGA can be listed in the DiGA register. 6 The DiGA-guideline is a guide for manufacturers, service providers and users with a description of the application process, the requirements, the DiGA register, and the support options. 7
All 30 DiGA providers (n = 30) were contacted and 10 of them participated in the survey. The selection of participants for the survey included all DiGA manufacturers, including registered and deleted DiGA applications in the DiGA register. Suitable contacts were identified through extensive research on company websites and in professional networks. The survey was conducted anonymously for data protection reasons in order to guarantee the confidentiality of the participants. No details about the company were collected.
The survey was conducted between March and October 2023. For this purpose, structured data was collected from participants via written online surveys using an optimized standard questionnaire, which was previously pretested. The questionnaire contains predefined categories (Cat.), items and answers, including multiple-choice, single-choice, or open free-text answers (Table 1). The survey was conducted in German. The English translation of the questionnaire was carried out for the purposes of publication.
Questionaire for the online survey.

MC: multiple choice; SC: single choice.
The digital implementation of the questionnaire was realized via LimeSurvey (by LimeSurvey GmbH. Germany), 14 supported by the Customer Relationship Management platform HubSpot (by HubSpot, Inc., USA), 15 which ensures automated mailing, success measurement and tracking as well as compliance with all data protection requirements in accordance with the European General Data Protection Regulation. 16 An ethics vote was obtained for the survey from the Ethics Committee of the Faculty of Medicine at Justus Liebig University Giessen (Germany) (file number: AZ 199/23). Before participating in the online survey, participants were informed about the purpose and procedure of the study. A technical requirement for starting the survey was that the participants had to expressly consent to anonymized data processing and possible publications in advance. Meta-data of the participants was used to prevent the possibility of multiple participation. After the online survey was successfully completed, the metadata was deleted for data protection reasons and to ensure anonymity.
For this study no copyrighted tools or questionnaires were used that need additional permission. The questionnaire is based on the publicly available guidelines of the German Digital Health Application Regulation (DiGAV) and the DiGA guideline. The digital implementation of the questionnaire was realized via the free and open source platform LimeSurvey as well as the free version of the HubSpot platform.
After the survey results were collected and analyzed, the data was filtered by separating relevant and irrelevant data for the research question. 17 To analyze the data, the absolute numbers of responses were plotted in a bar chart to determine how often each response was selected.
Results
In total 10 DiGA manufacturers participated in the survey and all of them rated the requirements of the fast-track process as at least challenging (Figure 1). The biggest challenges of the fast-track process were identified by all participants as the positive care effect and by the majority as the security, functionality, data protection and interoperability as well as the time schedule of the BfArM (Figure 2).

Result for the question “How challenging was the entire BfArM fast-track process for you?”.

Result for the question “What were the three biggest challenges of the DiGA fast-track process?”.
All DiGA manufacturers found demonstrating the positive care effect at least challenging. The majority found it very challenging (Figure 3). All except two participants cited the realization of the clinical trial as the biggest challenge. One participant pointed out that the increase or change in evidence requirements over time is difficult to plan, especially for registered and established study designs (Figure 4). In this context, all participants except one named the coordination and logistics as well as the recruitment of study participants as one of the biggest challenges (Figure 5). Participants used innovative approaches and technologies such as remote virtual trials, eCRF systems, real-world data, and digital biomarkers to demonstrate the positive effect of care (Figure 6).
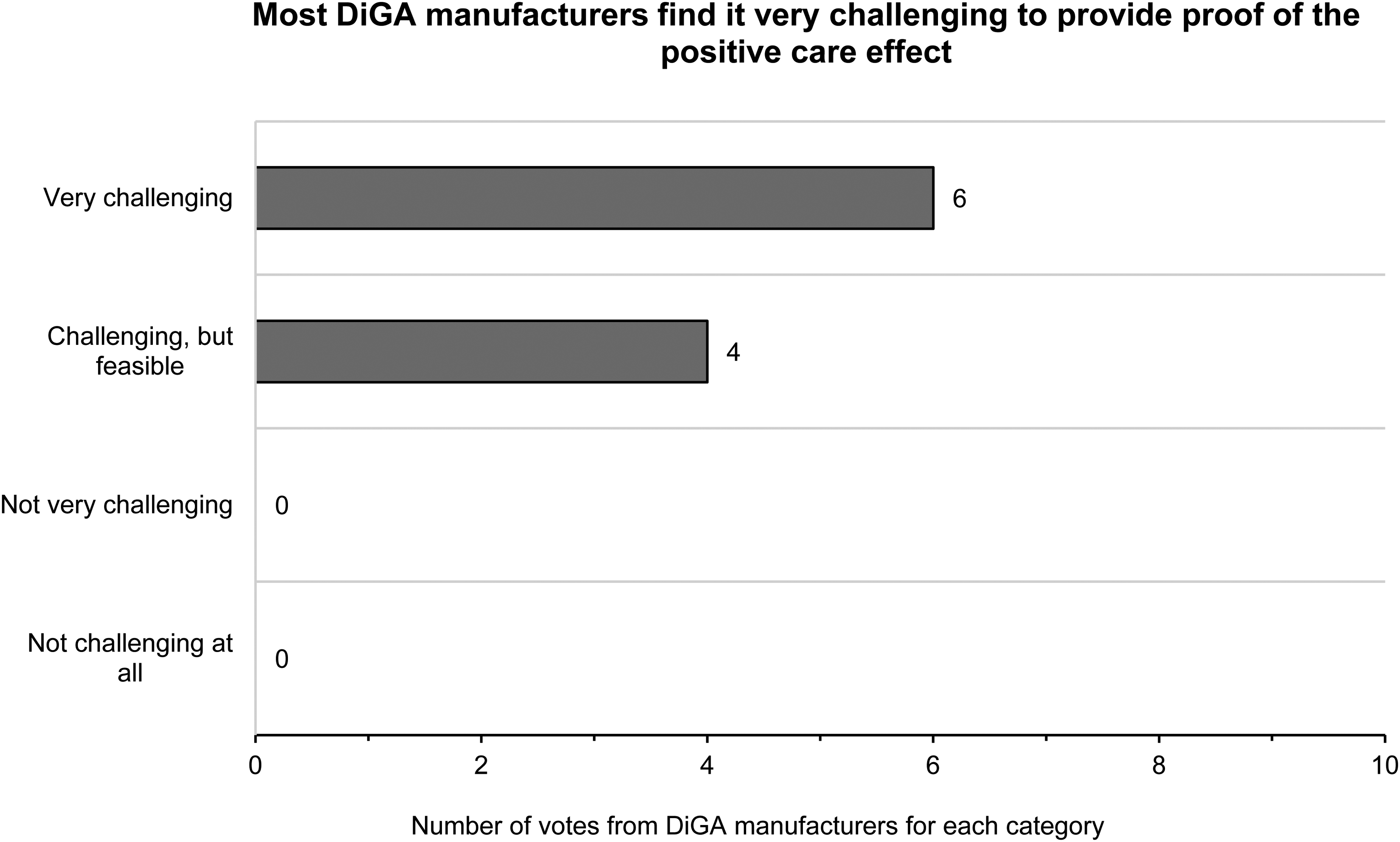
Result for the question “How challenging was it for you to prove the positive care effect?”.

Result for the question “What was the biggest challenge for you in proving the positive care effect?”.

Result for the question “what were the three biggest challenges in planning and conducting your study?”.

Result for the question “Have you used any innovative approaches and/or technologies to demonstrate the positive care effect?”
In terms of reimbursement, DiGA manufacturers found price negotiation and setting the price challenging (Figure 7). The majority of DiGA manufacturers would choose the DiGA model again if they were to release a new health app (Figure 8).

Result for the question “In what aspects were the pricing and price negotiations a challenge?”.

Result for the question “Which reimbursement path would you choose in the future if you were to launch another health app on the market?”.
On the profitability and importance of the DiGA model, participants answered with different perspectives. Some expressed worries about training and incentives for physicians, highlighting the need for better education to make the current model more cost-effective. On the other hand, there was optimism about future integration into regular care, with the expectation that DiGAs would become established, and prices could fall. A further point related to pricing, with concerns raised about the current pricing structure for companies that only offer a single DiGA. The viability assessment found that while the model is profitable, small companies face challenges, while economies of scale could encourage the production of multiple DiGAs. The need for closer integration of service providers and services was cited as a pre-requisite for profitability. Although the DiGA model is viewed positively as a reimbursement model with increasing importance, some participants expressed reservations, particularly with regard to the increasing difficulties in the fast-track process and the high data protection requirements, which could have a negative impact on user-friendliness.
The fast-track process was a large incentive for the majority of participants to launch their medical devices as DiGAs on the healthcare market (Figure 9). The DiGA guide was an important tool for manufacturers, even if opinions on its usefulness were mixed. The majority of participants found the guide at least helpful, while two other participants found it very helpful (Figure 10).

Result for the question “To which level was the fast-track process an incentive for you to establish the medical device on the healthcare market as a DiGA?”.

Result for the question “How helpful was the DiGA guideline for you?”.
The biggest challenge for the acceptance and marketing of DiGA was identified by all participants except one as promoting acceptance among service providers (Figure 11). As methods with which the manufacturers wanted to increase acceptance by physicians and the prescription rate, all manufacturers except one also used training courses/training documents for physicians and healthcare professionals. In addition, targeted marketing and advertising activities and intensive communication with medical facilities and clinics were used (Figure 12). In terms of the effectiveness of DiGA manufacturers’ activities to increase acceptance and prescription rates, participants felt that their activities were moderately effective (Figure 13).

Result for the question “What were the two biggest challenges in marketing and distribution of your DiGA?”.

Result for the question “Which of the following methods do you use to increase physician acceptance and prescription rates?”
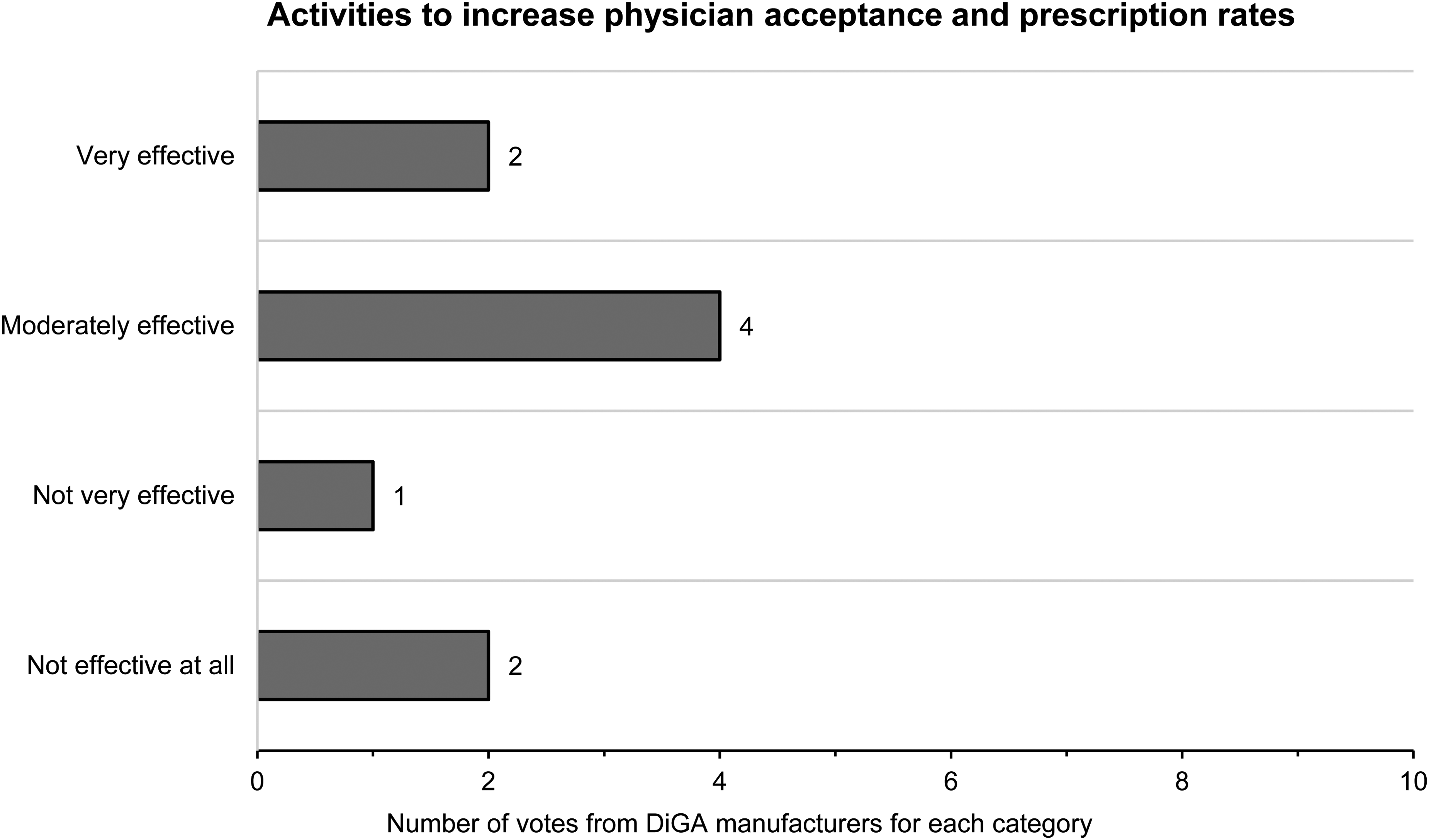
Result for the question “How effective would you rate your activities to increase physician acceptance and prescription rates?”.
Discussion
Although most participants saw fast-track procedures as an incentive to bring their DiGA to market, the majority of participants saw major challenges in the context of the fast-track process, especially in terms of demonstrating positive supply effects and the requirements for safety, functionality, data protection, and interoperability. These challenges illustrate the complexity of the legal requirements and the high standards set by the BfArM. Such challenges can hinder innovation and slow down the market launch of DiGAs. This highlights the importance to rationalize regulatory processes without affecting safety and efficacy.
Another challenge highlighted by participants relates to the realization of clinical trials, with coordination and participant recruitment mentioned as main challenges. The introduction of innovative approaches such as remote virtual trials and digital biomarkers are indicative of efforts to overcome these challenges. However, additional support and guidance may be needed to support the efficient realization of clinical trials to ensure a robust evidence base for DiGAs.
The complexity of negotiating prices and setting free prices highlights the commercial challenges faced by manufacturers. While reimbursement through the DiGA model offers opportunities for market entry, there are still doubts about profitability and pricing structures. To address these fears, a balanced approach is needed that promotes fair reimbursement mechanisms while providing incentives for innovation and affordability.
The participants gave different views on the profitability and importance of the DiGA model. While some expressed worries about training, incentives for healthcare professionals, and the current pricing structure, others were optimistic about future integration into mainstream care. The evaluation of profitability highlights the need for economies of scale and closer integration of providers to improve sustainability and long-term viability.
Promoting acceptance among service providers proved to be a major challenge for manufacturers. Strategies such as training, targeted marketing, and communication with medical institutions are being used to increase acceptance and prescription rates. However, the effectiveness of these initiatives is still moderate, highlighting the need for continuous evaluation and refinement of marketing strategies to improve the acceptance and use of DiGAs.
Limitation
The representativeness of the survey results may be affected by the limited number of participants—10 out of 30 DiGA manufacturers contacted. Although this represents a response rate of 33%, it should be noted that this was a complete survey of all DiGA manufacturers listed at the time of the survey. Even if the results are not statistically significant or fully representative, they do provide a valuable insight into the general mood within the industry. Potential reasons for nonparticipation include lack of interest, competing priorities, and time constraints. This could question the applicability of the results to the overall population of DiGA manufacturers. In addition, we did not collect detailed data on factors such as company size, annual revenue or share of total apps downloaded, which could provide further insight into representativeness and market structure. These limitations highlight the need for careful interpretation of the results and that the results may not represent the full diversity of DiGA providers.
The validity of the survey could be affected by potential interpretation bias or inaccurate responses from participants. Although the questionnaire was pretested, some participants may have had difficulty understanding the questions correctly or providing accurate information. A formally validated questionnaire was not used, which could also lead to bias in the results and affect the validity of the conclusions. The study is seen as a pilot study to provide initial insights and should be evaluated in terms of its validity.
Reliability of survey results relates to the consistency and reliability of the survey. Repeat surveys could show if similar results can be achieved and confirm the reliability of the data. However, as the survey was not repeated, the reliability of the results remains unconfirmed.
The objectivity of the survey is supported by the use of standardized questionnaires and the clear definition of the measurement variables. Conducting the survey digitally using LimeSurvey and HubSpot could also promote objectivity as it minimizes human error in data collection. However, subjective influences in the interpretation of the results cannot be avoided, especially when it comes to categorizing or analyzing the responses.
Conclusion
In summary, the results of this study show that the fast-track process presents serious challenges for DiGA manufacturers. The fulfillment of requirements relating to positive supply effects, safety, functionality, data protection, and interoperability proved to be problematic. The complexity of the regulatory requirements and the high standards of the BfArM can slow down the market launch of DiGAs. Another challenge was the realization of clinical trials, with the coordination and recruitment of trial participants cited as the major problems. The limited participation of only 10 DiGA manufacturers affects the representativeness of the results and therefore there is no statistical significance.
The complexity of negotiating prices and setting free prices also highlights the commercial challenges faced by manufacturers. Although the DiGA model offers opportunities for market entry, there are significant doubts about profitability and pricing. Strategies such as training, targeted marketing and communication with medical organizations are being used to increase acceptance, but the effectiveness of these initiatives is still moderate.
In terms of the suitability of the German DiGA model for other countries, it shows that its specific legal and regulatory requirements may not be directly transferable. However, the general principles of the DiGA model, especially in terms of security, functionality, and interoperability, could provide inspiring guidelines for other countries. It should be a goal for manufacturers of CE marked medical devices to achieve reimbursement in as many member states as possible, harmonized rules could help. 18 The DiGA register already shows that foreign manufacturers are interested in the DiGA reimbursement system. A number of overseas companies have already received DiGA authorization: Edupression.com (Austria), 19 My Dose Coach (Italy), 20 Untire (Netherlands), 21 and Vitadio (Czech Republic). 22
The quality of the survey results highlights the importance of a critical reflection on the methodology of the survey. Future research should improve the representativeness and quality of the results, promoting co-operation between all stakeholders. It is important to use robust and methodologically solid approaches for future studies.
Footnotes
Contributorship
MS, KS, and VG were involved in study design, initial idea, and supervision; FS in statistical analysis and estimation of regulatory aspects; MB and OA in experimental execution, writing and editing. All authors have defined the execution and evaluation strategy as well as reviewed and approved the submitted version.
Consent to participate
Participant consent was given before the start of the online survey, nevertheless the ethics committee does not require participant consent.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The ethics vote of the Ethics Committee of the Faculty of Medicine of the Justus Liebig University Giessen (Germany) contains that no ethics vote is required for this study (file number: AZ 199/23).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
MS
