Abstract
Background
Germany has one of the oldest social security systems in the world. Population coverage has subsequently increased, reaching coverage of approximately 90% of the population in the statutory health insurance (SHI) system today. Before this background, Germany has been pioneering the integration of digital therapeutics (DTx) into its SHI system by the introduction of the Digital Healthcare Law (Digitale-Versorgung-Gesetz, DVG) in 2019. Thereby, patients became eligible for digital health applications (Digitale Gesundheitsanwendungen, DiGA), which are available upon prescription by qualified healthcare professionals.
Challenge
As conventional healthcare delivery often lacks direct outcome measures as and is mostly still reimbursed on a fee-for-service basis, DiGA offer the opportunity to continuously provide individual outcome and performance data. They are, therefore, well-suited for a performance-based payment framework. While the DVG introduced the option for performance-based reimbursement components in 2019 already, the ongoing debate about the value of DiGA and to what extent they can contribute to the healthcare system has now been reflected in a 2023 health policy bill by the German Federal Ministry of Health, which aims to introduce a mandatory performance-based reimbursement component for DiGA.
Proposal
In this light, we propose a framework for performance-based reimbursement of DiGA, involving an intervention-specific, performance-linked reimbursement framework with shared accountability between manufacturers and payers. The approach aims to align the often contradicting interests of the involved stakeholders to incentivize the delivery of high-value digital health care. Yet, the proposal also acknowledges the need for further research to establish a robust foundation for implementing such a framework.
Introduction
Digital therapeutics (DTx) are broadly defined as ‘evidence-based therapeutic interventions that are driven by high-quality software programs to treat, manage or prevent a disease or disorder’ 1 and provide a new branch of therapeutic means. Yet, to make DTx available to broader patient populations, the integration of DTx into existing and often highly complex healthcare systems poses a challenge. The introduction of the Digital Healthcare Law (Digitale-Versorgung-Gesetz, DVG) in 2019 and the resulting implementation of DTx as so-called digital health applications (Digitale Gesundheitsanwendungen, DiGA) in 2020 put Germany at the global forefront of the integration of such novel digital tools into a highly developed healthcare system. The introduction of DiGA included an accelerated market approval process and the eligibility for reimbursement through the collectively funded statutory health insurance (SHI) system. Both features have since fostered an ongoing discussion about applicable evidence for market approval decisions, the role of DTx in conventional care pathways and the relationship between patients, providers, payers and respective DTx elements.
Payers have since criticized the accelerated market approval process, the early eligibility of DiGA for reimbursement and the risk of a significant budget impact resulting from DiGA. While a comprehensive overview of the German healthcare system can be found elsewhere,2–4 this article only introduces core principles of the SHI system before focusing on the current market approval process and reimbursement framework for DiGA. It then discusses the potential role of performance-based reimbursement in the light of the recently passed Law to Accelerate the Digitization of Healthcare (Gesetz zur Beschleunigung der Digitalisierung des Gesundheitswesen, DigiG) from early 2024 which now aims to establish mandatory performance-based reimbursement principles for DiGA. The article then provides a suggestion of how to incentivize both payers and manufacturers to enable maximum benefit from the prescription and use of DiGA.
Background
Principles of the statutory health insurance system in Germany
The German social security system dates back to 1883, when the society faced the challenges of rapid industrialization with rising social unrest among factory workers over dire working conditions. Its foundation relied on two basic principles. Firstly, employers and employees were given the obligation to contribute to so-called sickness funds that would provide payments to employees if they developed an illness leading to sick leaves. Secondly, payers, healthcare providers and other stakeholders in the healthcare system received the mandate for self-governance. This limited the direct political influence to changes in the German social laws and allowed healthcare stakeholders to independently govern the healthcare system. While these principles of solidarity and self-governance still guide the SHI system today, the system has evolved significantly ever since. The SHI system—consisting of 95 individual SHI in 2024—has become the default health insurance for the vast majority of the German population. Instead of payments, it now mostly provides services from a comprehensive benefits package that is defined by the Joint Federal Committee (Gemeinsamer Bundesausschuss, G-BA), a governing body consisting of SHI, providers and hospital representatives. Through the DVG—a change to the German social laws—DiGA were established as a new branch of therapeutic means that were included in the benefits package of the SHI in 2019.
DiGA approval process
To determine whether a DTx can become a DiGA—and hence can become eligible for reimbursement through the SHI system—the Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, BfArM), a regulatory authority, assesses the formal applications from DTx manufacturers in a market approval process. Through a unique provision of DiGA regulation, manufacturers can pursue a so-called fast-track market approval process, which allows a provisional market approval based on a clinical evaluation plan and sufficient preliminary data on its effectiveness (positiver Versorgungseffekt, pVE). If, on this basis, BfArM deems a successful evaluation likely, provisional market approval is granted once mandatory safety, data protection, quality, functionality and data interoperability standards are met. Safety is considered given if the DTx carries a Conformité Européenne (CE) mark, the formal confirmation of a medical device complying with the European regulations for medical devices. Sufficient data protection standards are assumed to be given once the General Data Protection Regulations (GDPR) of the European Union are met. The use of patient data is further limited by Article 4 of the DiGA ordinance (DiGA-Verordnung, DiGAV) to the very specific reasons of ensuring the intended use of the DiGA, allowing the collection of outcome data during the provisional market approval period, collecting outcome data for performance-based reimbursement components and—with the requirement of an additional informed consent by patients—improving the functionality of the DiGA. The quality of the DTx is assessed through a catalogue of criteria including the avoidance of any commercial advertising, providing only medical information that is backed by generally accepted medical standards and guidelines and the documented minimization of barriers to access for disabled persons. The quality criterion extends to the requirement to provide information material to healthcare professionals who can prescribe the respective DTx. Data interoperability requires, as of August 2024, compatibility with internationally recognized interface and semantic standards (e.g. HL7 FHIR, SNOMED CT and ISO norms). Further details on the latter aspect can be found in the work by Weber and Heitmann. 5 If the provisional market approval is granted, the manufacturer is obliged to present comprehensive and confirmatory data supporting the pVE, generally within 12 months, before BfArM issues a definitive decision on the permanent market approval. If comprehensive evidence can be presented at the time of the initial application for market approval already, the direct and permanent market approval is granted. However, this conventional market approval process is currently less attractive for manufacturers—mostly start-ups—as the provisional market approval allows revenues to be generated throughout the provisional market approval period. As of September 2023, the latest comprehensive overview available, 81.8% of all DiGA (45/55) had pursued a fast-track market approval, while only 18.2% (10/55) pursued a conventional process with a permanent market approval. 6 Additional details on the fast-track approval process have been discussed and can be found elsewhere. 7
DiGA reimbursement system
All SHIs must reimburse DiGA with both provisional and permanent market approval. Since the introduction of the DVG in 2019, the SHIs, mainly through their National Association of Statutory Health Insurance Funds (GKV-Spitzenverband, GKV-SV), are advocating to revise the fast-track market approval process. Their primary argument is that the evidence threshold required for provisional market approval is inadequate. Moreover, they question the pricing approaches pursued by manufacturers during the time of provisional market approval—during which reimbursement rates can be freely set by the manufacturer up to a specific cap-price—to be unduly high.
After permanent market approval of a DiGA, the GKV-SV and the manufacturers are mandated to negotiate permanent reimbursement rates. However, since the introduction of the DVG in 2019, a significant number of these negotiations failed, requiring the involvement of a legally appointed arbitration board. This impasse reflects the fundamental differences between the manufacturers and the GKV-SV in their perception of the value provided by DiGAs. This controversy culminated in the bankruptcy of a leading DiGA manufacturer over an arbitration board decision on its reimbursement rate in 2023 8 and highlights the need for a comprehensive discussion on reimbursement agreements in DTx.
Status quo of performance-based reimbursement
DTx in general, and DiGA in particular, provide an ideal use case for innovative reimbursement agreements, as the use of the DiGA itself generates outcome data that can be used for performance assessment. This data includes usage patterns and adherence information, the development or subsidence of symptoms and the acquisition and use of condition-specific knowledge, patient satisfaction and more. Per definition, this data falls under strictly regulated medical data, and its use for scientific analysis is highly controlled. Yet, the German social laws explicitly suggest outcome-based reimbursement agreements for DiGA (‘[The agreements are also to cover performance-related price components]’, Article 134 German Social Law Book V, SGB V), and the otherwise strict data protection requirements provide an exclusion for the collection of user data for the determination of outcome-based reimbursement components (Article 4, DiGAV). Surprisingly, however, no such reimbursement agreement has been negotiated or arbitrated as of August 2024.
From our experience, any proposed reimbursement agreement departing from conventional fee-for-service agreements was opposed by different system stakeholders, primarily due to concerns related to the expected and disproportional resulting administrative burden. In this context, only volume-based rebates were proposed as an instrument to minimize the potential budget impact of a scalable digital therapeutic. Yet, publicly, the GKV-SV advocates strongly for the introduction of innovative reimbursement frameworks that rely on use and outcome. This has been illustrated by a recent statement by a GKV-SV chairperson saying ‘[(…) information on the actual usage of DiGA should be made transparent by manufacturers and be taken into account in reimbursement. After all, a medical service is only reimbursed if it is actually used by the insured person and not already when the appointment is made. Consequently, we want to pay for therapeutic benefits, not downloads]’. 9
Along similar lines, different authors have proposed to use therapeutic adherence as a unidimensional indicator for quality and expected therapeutic benefit of DiGA.10,11 This has been put very explicitly by, for example, Kolominsky-Rabas et al. who argue that ‘[(…) reasons for non-adherence to digital interventions are lack of time, personal dissatisfaction with the intervention content, and intervention content that users perceive as impersonal.]’. 11 Both accounts represent a payer perspective and reflect a unidimensional therapy-centered understanding of adherence, which is not in line with long-standing guidance from the World Health Organization (WHO) on adherence, highlighting the equal importance of therapy, condition, patient, socioeconomic and also healthcare system-related factors influencing adherence. 12 Yet, the perceptions put forward by the stakeholders above seem to have influenced the recent policy-making efforts.
Recent policy-making efforts
Before the background of this ongoing discussion, the Federal Ministry of Health introduced a Law to Accelerate the Digitization of Healthcare (Gesetz zur Beschleunigung der Digitalisierung des Gesundheitswesen, DigiG) in early 2024, which aims to further advance the integration of digital health into the German healthcare system by building on the Digital Health Care Act (Digitale-Versorgung-Gesetz, DVG) from 2019. While the DigiG introduces numerous changes to, e.g. digital prescriptions, telehealth coverage, data interoperability standards and data protection measures, it also proposes a revision of the reimbursement principles for DiGA, of which one is of particular interest. Specifically, a mandatory performance-based reimbursement component that accounts for a minimum of 20% of the reimbursement rate is proposed and will come into effect by January 1, 2026. The DigiG suggests to assess performance preferably by ‘[…] the duration and frequency of use of the digital health application […] patient satisfaction with the quality of the digital health application, and […] the patient-reported health status during the use of the digital health application’ (Article 139 German Social Law Book V, SGB V).
While the DigiG lacks a more specific elaboration on what a DiGA and its respective manufacturer should be accountable for and does not specify suitable data sources and outcomes, it demonstrates a clear inclination to pilot performance-based reimbursement in the field of DiGA.
In light of the debate leading up to the passing of the DigiG about the applicability of DiGA evidence to a real-world care setting, the perceived limitations of the fast-track market approval process and an initially proposed yet eventually abolished change to introduce a mandatory free testing period for DiGA, we consider the recent introduction of the DigiG a starting point to propose the implementation of a performance-based reimbursement model that allows the introduction of performance-based reimbursement principle for DiGA while accounting for the novelty of DTx in the German healthcare system and their dependency on system factors to provide a maximum benefit.
Proposed performance-based reimbursement framework
To capture the potential of innovative reimbursement agreements and maximize their effect on incentivizing highly effective DTx in the German healthcare system, a constructive perspective toward the different approaches to structuring reimbursement as well as their potentials and limitations is required. The following approach explores different dimensions of a performance-based reimbursement framework. It considers their applicability to the German healthcare system, the ease of implementation and the reflection of the patient, public (i.e. GKV-SV, individual SHI and physicians’ chambers) and private (i.e. providers and manufacturers) stakeholders’ interests in an evolving policy environment.
Scope
The level of implementation reflects the scope of the reimbursement framework and can, in principle, range from a population-based to an individual perspective. Population-based payment systems aim to shift volume-based incentives to quality-based incentives and are typically not connected to individual services, but to a period over which a patient is under the care of a specific provider or a specific health system. Payments are typically made on a per-member-per-time basis for the ‘population under care’ and provide the incentive to shift care from reactive to preventive care, but often fail to include explicit quality measures for the services provided.14,15 This approach remains a core principle for determining provider reimbursement in the German ambulatory care context and has also been, for example, applied in certain Medicaid programs in the United States. 16 Yet, it has not found wide applicability for the reimbursement of specific therapeutic measures. Therefore, and since the population-wide effects of DiGA are difficult to detect due to their recent introduction and still low system penetration, this level of implementation seems premature.
A narrower level of implementation is based on ‘episodes of care’ and is at the center of well-established performance-based reimbursement frameworks.17,18 Episodes of care typically include services from different providers for a variable length of an episode. Although episode-of-care-based reimbursement could support the efforts to integrate DiGA into conventional healthcare delivery pathways, we consider the ‘episode of care’ as an inadequate reflection of the performance of any specific DiGA, as it would be challenging to isolate the performance of a specific DiGA within this framework. While efforts for the integration of DiGA into structured disease management programs for diabetes mellitus, obesity and other chronic conditions are ongoing, the system-wide implementation of DiGA in such programs is not yet foreseeable. In addition, the German system for diagnosis-related groups (G-DRG), which is the core framework for determining reimbursement for hospital-based treatments, has increasingly been viewed critically by the public and the current legislators, as it has been perceived as a key driver of the cost pressure in the German healthcare system and is undergoing reform itself. 19
Lastly, the implementation of a performance-based reimbursement framework could be at the level of one specific intervention. An intervention-based reimbursement is feasible and can be designed to align accountability with the specific outcomes attributed to the DiGA. The latter appears, for now, to be the most applicable level of implementation, as DiGA cannot yet be expected to have a relevant population-wide effect and are not yet widely established in conventional healthcare delivery pathways. At the same time, an intervention-based focus considers the payer perspective. It can help to distinguish low-quality from high-quality DiGA while limiting the effect of healthcare system influences.
Measurement
A precondition to the effectiveness of a performance-based reimbursement framework is capturing the performance of the DiGA. Although performance measurement of healthcare interventions has been at the center of health economic research for years, its applicability to DTx is subject to discussion. In general terms, Porter coined the overarching definition that value-based healthcare should aim to maximize the ‘health outcomes achieved per dollar spent’. 17 At the same time, other initiatives have focused on identifying specific health outcomes that are meaningful to patients (e.g. ICHOM 20 or COMET 21 ). A more generic approach includes utility measures, such as quality-adjusted life years (QALY) or disability-adjusted life years (DALY). Each standardized outcome set for given diagnoses and utility measures has potential and has been employed successfully to inform cost-effectiveness analyses for pharmaceuticals. Yet, they are not ideal for a DiGA reimbursement framework in the German system, as the continuous collection of such outcome data could impair the usability of the DiGA and, hence, interfere with the therapeutic mechanisms of the respective DiGA. This is particularly relevant, as many DiGA rely on behavioral or educational means to convey their medical benefit. Furthermore, the concept of utility measures has historically been viewed skeptically in reimbursement decisions in the German healthcare system, as assigning a monetary value to health status is perceived as morally challenging.22,23 While the definition of any generic outcome set (i.e. independent of the spectrum of indications the respective DiGA addresses) would facilitate the applicability of a generic reimbursement principle, it would not account for the specificities of the medical condition addressed. This, too, applies to measures of engagement, adherence or retention. Although high levels of engagement have demonstrated positive effects on medical effectiveness in certain conditions, the same relationship cannot be assumed for all interventions and indications. Hence, these measures warrant careful consideration before designating them as a reimbursement target. On the contrary, the required effort to collect meaningful and specific medical performance data requires balancing so that no condition-specific barriers to assessing performance arise (e.g. the collection of multi-item questionnaire-based outcomes for mental health conditions such as depression might be more difficult than the collection of self-reported pain intensities on a unidimensional scale). Similar limitations apply to adherence measures or patient satisfaction scores, as these do not necessarily reflect the performance of the respective DiGA, but are influenced by a multitude of context factors. Before this background, the advantages and disadvantages of different concepts capturing the potential value of a DiGA become clear. While a homogeneous assessment of performance might be a promising perspective, we deem the use of condition-specific performance-based reimbursement agreements as most applicable in the current context.
A performance-based reimbursement model determines the effective reimbursement rate based on achieving an agreed-upon outcome, which should become the subject of the already mandatory reimbursement negotiations. A benefit of an outcomes-based reimbursement model is that accountability is tied to real-world performance. The definition of, for example, clinically relevant improvements is feasible for a broad range of conditions and could therefore also become the subject of reimbursement negotiations. Hence, a reimbursement framework based on such clinically relevant improvements could be easily implemented. By following this approach, the legal provision for payers and manufacturers to negotiate reimbursement rates would even be strengthened and not undermined by preempted regulations that might not reflect the use case of the specific intervention.
Risk
Performance-based risk-sharing arrangements (PBRSA) have been well established in the field of innovative pharmaceutical therapies for which payers aim to minimize the risk for high-budget impacts from novel drugs with unclear real-world benefits and are increasingly implemented in the United States and the European Union.24–26 Outcome-based PBRSAs often distinguish conditional coverage (e.g. coverage is only granted under the condition of generating further evidence or if certain short-term therapy goals are met) and performance-linked reimbursement (e.g. outcome guarantees or improvements in pattern or processes of care). As the provisional market approval under the fast-track process is an example of conditional coverage and since the DigiG proposes rebates for DiGA not yielding a guaranteed outcome, we deem this an appropriate reference framework to discuss a DiGA-specific PBRSA. Yet, we see several required adjustments to account for the specific prescription and use patterns of DiGA.
Specifically, we deem a risk-sharing mechanism necessary, since large parts of the administrative activation process for DiGA are under the control of the SHI of the respective patients. At the same time, however, manufacturers should be fully accountable for the outcomes that can be attributed to the use of the respective DiGA. By implementing a shared-risk model, performance-based reimbursement can become an instrument to align the otherwise opposing interests between payers and manufacturers on maximizing the health benefit generated through a DiGA.
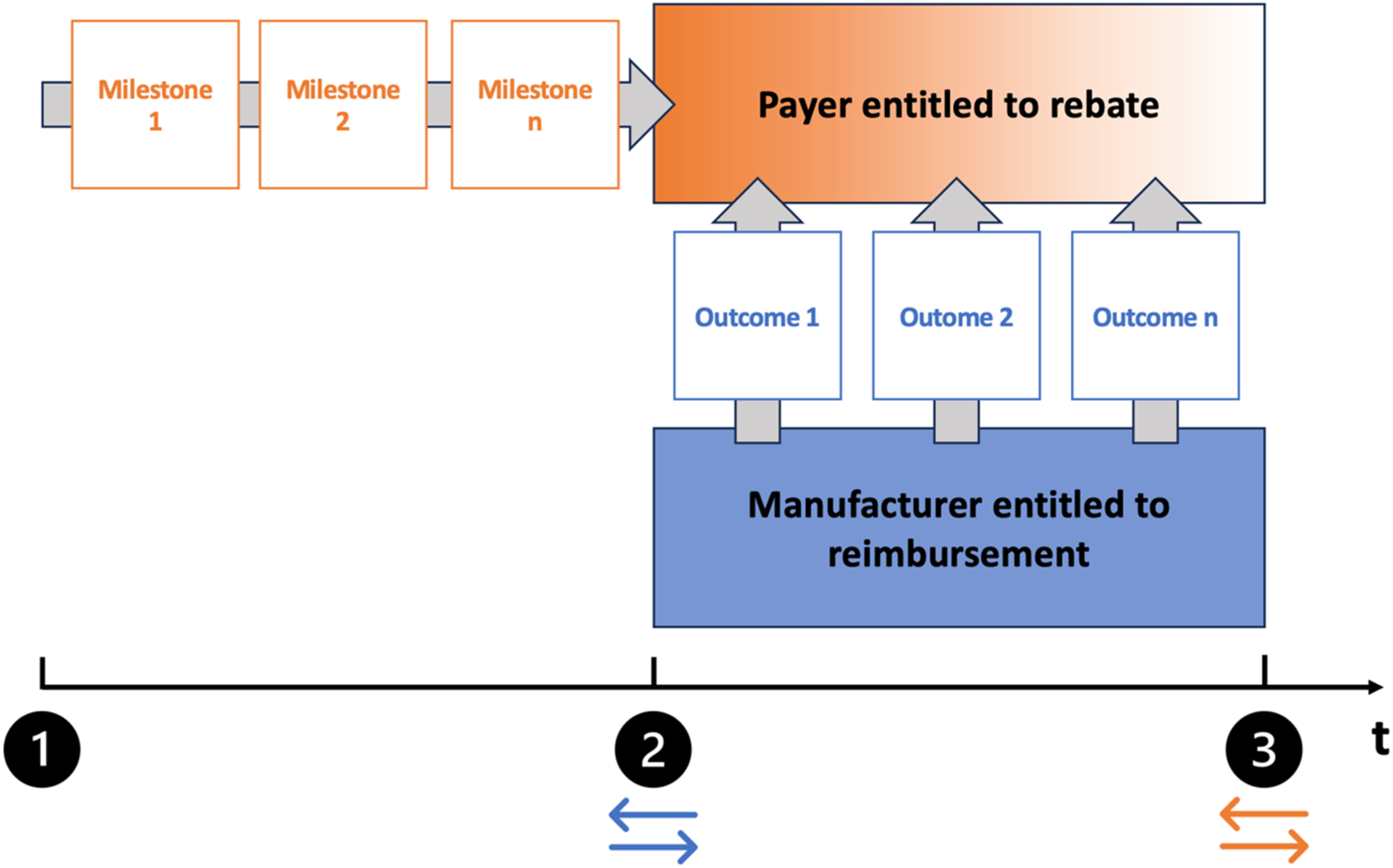
Illustration of the proposed reimbursement framework on a time scale. Upon prescription of the DiGA (1), the payer has the obligation to meet process milestones that are defined in the obligatory reimbursement negotiations between manufacturers and payers (e.g. enabling the activation of the DiGA within a specific number of days). If the payer fails to meet the agreed milestones, the eligibility for a rebate is waived. Once the DiGA is activated (2), manufacturers are entitled to claim the negotiated reimbursement rate (blue arrows), but must deliver outcomes that are likewise subject to the obligatory reimbursement negotiations (e.g. providing a clinically relevant improvement of a specific symptom). If the manufacturer fails to deliver the agreed outcomes, the payer is entitled to claim a rebate. The extent of the rebate can vary between outcomes (shaded orange area). Once the use period of the DiGA expires (3), the accrued rebates are transferred back to the payer (orange arrows). t = time.
The proposal in practice
Based on the above, we propose an intervention-specific, performance-linked reimbursement framework that incorporates a risk-sharing model between payers and manufacturers to account for the significant role both parties play in enabling the greatest possible benefit from using a DiGA (Figure 1).
An intervention-specific outcome measure to report on a patient individual level as well as applicable thresholds to determine a clinically relevant improvement can become part of the obligatory price determination process following the permanent market approval of a DiGA. Condition-specific barriers due to differences in the complexity of collecting such measures should be taken into consideration. By doing so, payers and manufacturers are jointly responsible for selecting meaningful outcomes to assess real patient benefit.
Yet, the need to establish a joint responsibility reaches further. The current complexity of the prescription process of a DiGA (i.e. prescription by a qualified healthcare provider, submission of the prescription to the payer, the administrative process of releasing an activation code and returning the respective activation code to the patient, submission of the activation code by the patient to the manufacturer and submission of a reimbursement claim by the manufacturer to the payer) warrants an incentivization of providers and payers to enable a quick activation of the DiGA upon prescription. The current market experience reflects relevant delays in releasing activation codes, leading to delayed access to the respective DiGA, as discussed above.
Similarly, two complaints issued by the Federal Office for Social Security (Bundesamt für soziale Sicherung, BAS), a supervisory body of the SHI, regarding the interference of individual SHI with the prescription process (i.e. unjustified queries to patients and providers regarding the applicability of a DiGA prescription and diverting issued prescriptions toward less regulated non-DiGA DTx) in 2023 highlight the need for shared accountability on DiGA activation and use. 27 These systemic challenges not only pose a risk for primary adherence but also limit the opportunity of DiGA to provide timely access to therapy, which is, by its digital nature, not limited by the capacity constraints of conventional therapeutic means. While manufacturers should be accountable for providing information to providers and patients regarding the correct prescription and activation process and early access to the respective DiGA, payers should be disincentivized to delay the activation process and to avoid the submission of reimbursement claims. While the DigiG suggests ‘[…] the duration and frequency of use of the digital health application […] patient satisfaction with the quality of the digital health application, and […]the patient-reported health status during the use of the digital health application’ (Article 139 German Social Law Book V, SGB V) as appropriate outcome categories, we propose an additional set of process milestones that are in the responsibility of the payers. Process milestones could comprise the release of an activation code within a specified time, the approval of manufacturer-issued assistance services to support providers and patients in the administrative process of activating a DiGA prescription and full compliance with the treatment sovereignty of the provider (i.e. fulfillment of the prescription as issued). Manufacturers, in return, should be fully responsible for outcome metrics. Naturally, implementing such a framework would require sufficient transparency of the entire prescription and activation process of a DiGA. Ongoing policy making efforts, e.g. implementing a fully digital prescription of a DiGA, could support this transparency.
Conclusion
Performance-based reimbursement agreements offer the opportunity to maximize the benefit generated by DiGA in the German healthcare system. However, attempts to implement such a reimbursement agreement have been unsuccessful and face opposition from different healthcare system stakeholders. In the context of the recent introduction of the DigiG and its provision for mandatory performance-based reimbursement elements, we outline a framework for performance-linked reimbursement agreements and propose a risk-sharing model for the stakeholders involved to incentivize the delivery of high-value care. Key elements of the proposed framework are (a) expanding of the scope of the mandatory price negotiations to include (b) process milestones in the responsibility of the SHI and (c) outcome metrics in the responsibility of the manufacturer with unambiguous criteria for therapy success are defined. (d) Meeting the process milestones entitles the respective SHI to rebates if the manufacturer fails to provide the agreed outcomes. Yet, this article can only provide a starting point for introducing value-based reimbursement principles into the German DiGA landscape and further research is required to establish a robust ground for its implementation. This should include a systematic cross-country analysis of emerging and similar prescription DTx frameworks throughout Europe and established performance-based reimbursement agreements alike, a stakeholder panel to identify reasonable responsibilities in the process and outcome domains and a thorough validation process based on real-world prescription and claims data.
Footnotes
Acknowledgements
We acknowledge the valuable input from Aishwarya Kanneganti, Sarah Fairbrook, Vineet Pandharpurkar, Ankita Jain and Harsha Gutta on value-based reimbursement principles in the U.S. healthcare market.
Contributorship
LB developed the concept of the work. LB and FPH contributed to writing the initial draft of this manuscript. LB provided the figure. J-SP provided feedback on the initial draft and figure and revised the work thoroughly. LB and GWT revised the manuscript. GWT revised the figure and structure of the final draft. All authors contributed to reviewing and editing the final draft.
Declaration of conflicting interests
LB receives a salary from Vivira Health Lab GmbH, a DiGA manufacturer. Yet, Vivira Health Lab GmbH did not have any influence on this article and did neither provide content, nor financial support to this work.
Ethical approval
Not applicable as no patient data were included in this study.
Availability of data and materials
All data, including the referenced personal communication, can be made available upon reasonable request.
Funding
The work presented did not require external funding. Yet, LB received funding through the Berta-Ottenstein-Programme for Clinician Scientists from the Faculty of Medicine, University of Freiburg.
Guarantor
LB is the corresponding author and guarantor for this work and takes full responsibility for this article, including for the accuracy and appropriateness of the reference list.
Informed consent
Not applicable.
