Abstract
Objective
Today there are several health and medical apps (mHealth) in app stores. Germany is the world's first country that introduced apps paid by the regular health insurance service. Even though breast cancer is the most common cancer in women, mHealth for breast cancer has been largely unexplored.
Methods
A total of 33 apps from two major mobile application marketplaces (Google Play Store/Android; App Store/iOS) have been selected for analysis.
Results
The app analysis shows that there are currently only 10 mHealth apps in German, which are specifically dedicated to breast cancer patients. The features of these apps fall into two categories: improvement of health literacy and indirect intervention. These apps can be used for all phases of the patient journey starting with the diagnosis.
Conclusions
mHealth apps have the potential to support the adherence of breast cancer patients. In order to exploit this future potential, the app quality, as well as the information about the available apps, must be urgently improved. Currently, it is very difficult both for laypersons and for doctors/other therapists to identify high-quality apps. Guidance from independent or governmental institutions would be helpful to further the digitalization in health care.
Introduction
Smartphones are indispensable in today's everyday life. The legislation of the “Digitale-Verordnung-Gesetz (DVG)” on 19 December 2019 builds the foundation for a better supply of digitalization and innovation in Germany. From now on, mobile applications (apps) can be ordered and paid on prescription and consultation hours can be held by video. 1 This makes Germany the world's first country that introduced apps as a regular health insurance service. However, if one considers the Digital-Health-Index of the Bertelsmann Foundation, Germany only ranks at position 16. The leading countries are Estonia, Canada, and Denmark, although apps for health care are rarely used in these countries. 2
There are already a large number of health and medical apps (commonly called mHealth) on the German health market. In 2017, more than 300,000 health apps were available in the Apple App Store and Google Play Store. 3 Usage scenarios vary from wellness or fitness offers to complex applications for diagnosis and to support in aftercare. In the long run, the DVG will lead to even more popularity because the awareness of the German population for health topics is increasing steadily. While in 2015 only every third German smartphone user used health apps, this went up to 50% in 2017.4,5 So far primarily athletes (43%), overweight people (26%), and chronically sick (27%) are using health apps. 6
Chronic patients count as the most important target group for mHealth applications and can benefit most from it. 7 A study published by McKinsey in 2018 shows that by digitizing health care in Germany, up to €34 billion could be saved in the federal health care system. From that amount, €2 billion are lost due to the lack of digital management of chronic diseases. 8 Those also include breast cancer which is the most common type of cancer in women with almost 30% of all cancer diseases in Germany. In men, however, breast cancer ranges very low with 0.3%. 9 In 2016, 233,570 women were diagnosed with cancer in Germany of whom about 68,900 were diagnosed with breast cancer. 10 Women from age 50 onwards are affected more frequently. The highest risk is from 65 to 69 years. From the age of 70, the risk of breast cancer starts to decrease slightly. But also younger women are often diagnosed with breast cancer.
A major improvement in diagnosis was the introduction of mammography screening in 2005 for women of 50 to 69 years. Although the number of new cases rose after introduction, the mortality rate declined because of better possible therapies in the early stages. 11 Especially oral chemotherapy is becoming increasingly popular. 12 These offer patients the opportunity to be more independent during their cancer therapy but on the other side also make high demands. There is a risk of irregular intake or non-adherence.12,13 Many breast cancer patients are affected by these two factors because other treatments such as antihormone therapy, also called endocrine therapy, are long-term focused. Patients must take these medicines for at least 5 to 10 years. 14 Various studies detected that over 50% of patients were not adhering to the antihormone therapy standard after the surgical removal of the tumor. 15 Furthermore, non-adherence is also connected to non-medicinal areas of cancer therapy. Since non-adherence can result in serious consequences for patients and also affects the entire health care system, enforcing breast cancer patients’ adherence is extremely important. The annually incurred costs in Germany due to non-adherence are estimated up to €10 billion. 16 This also emphasizes the economic relevance of the topic.
Parallel to the changes in possible therapies, the doctor–patient relationship has also changed. There has been a transition from paternalism to a partnership-based relationship. Today doctors and patients make the therapy decisions together.17–20 Thereby informing the patients plays a major role. However, doctors usually do not have enough time to inform and educate patients entirely. In Germany, doctors spend on average only 7.6min per patient. 21 In addition to clinical competence, the quality of care is most important for cancer patients. Since the doctor usually has little time, health care professionals (HCPs), that is, medical professionals such as nurses, play an important role. It is therefore crucial that HCPs have the knowledge and ability to communicate with the patient. 22
Moreover, an increasing amount of information for the patient is available on the internet. Unfortunately, it often lacks medical quality. 23 For this reason, to avoid misinformation, the patient usually gets information brochures from her/his doctor or the medical institution. Compared to other cancer types, these information materials are often less helpful in breast cancer and rarely serve as a decision support. 24 Due to the general use of health apps and missing reliable sources of information for breast cancer patients, apps offer a particularly great potential.
Definition of adherence
The World Health Organization (WHO) defines the term adherence as follows: “Adherence is the extent to which a person's behavior—taking medication, following a diet, and/or executing lifestyle changes, corresponds with agreed recommendations from a health care provider.” 25
Therefore, an adherent patient behaves compliant with his medical treatment specifications, such as taking a specific medication. Conversely, if a patient does not follow the therapy specifications, the term is non-adherence. Non-adherence of patients can be very diverse. Among others, this includes canceling visits to doctors, not complying with prescribed diets, and the irregular intake of prescribed medication.
26
The academic literature does not mention any standardized limit, as of which a patient is considered as adherent. Numerous studies, which have examined the adherence of cancer patients in general or of breast cancer patients in particular, often indicate a limit of 80%.27–32 Non-adherence can develop consciously or unconsciously.
33
Factors for non-adherence may be different. The WHO names five dimensions that influence adherence:
Socio-economic factors (e.g. age, education, social, and financial situation). Patient-associated factors (e.g. motivation, acceptance of illness, psychosocial status, and forgetfulness). Disease-associated factors (e.g. serious illness and comorbidities). Therapy-associated factors (e.g. treatment duration and side effects). System-associated factors (e.g. doctor/HCPs–patient relationship).
25
Socio-economic, patient-associated, and disease-associated factors are among the predisposing factors.
30
Definition of patient empowerment
There is also no commonly accepted definition for patient empowerment.34,35 Patient empowerment is often seen as a process, but sometimes as a result.34,36 The WHO describes patient empowerment as a “process through which people gain greater control over decisions and actions affecting their health.” 37 Hence, patient empowerment is a process in which patients gain the appropriate knowledge from their medical service providers and are thus enabled to actively participate in the decision-making and treatment process. 38 Patient empowerment is decisive for adherence and can be seen as a basis for this.39–43 In Germany, doctors take an average time of 7.6min consultation time per patient. 21 In this short period of time, the patient's information needs cannot be explained in detail and individual education usually misses out. Concerning breast cancer, HCPs and special breast care nurses play an important role. In addition to the patient's health care, they engage more in informing the patient, communicating with him/her, and supporting the aftercare. As shown in Figure 1, the combination of these adherence factors combined with patient empowerment result in adherence and persistence.30,44
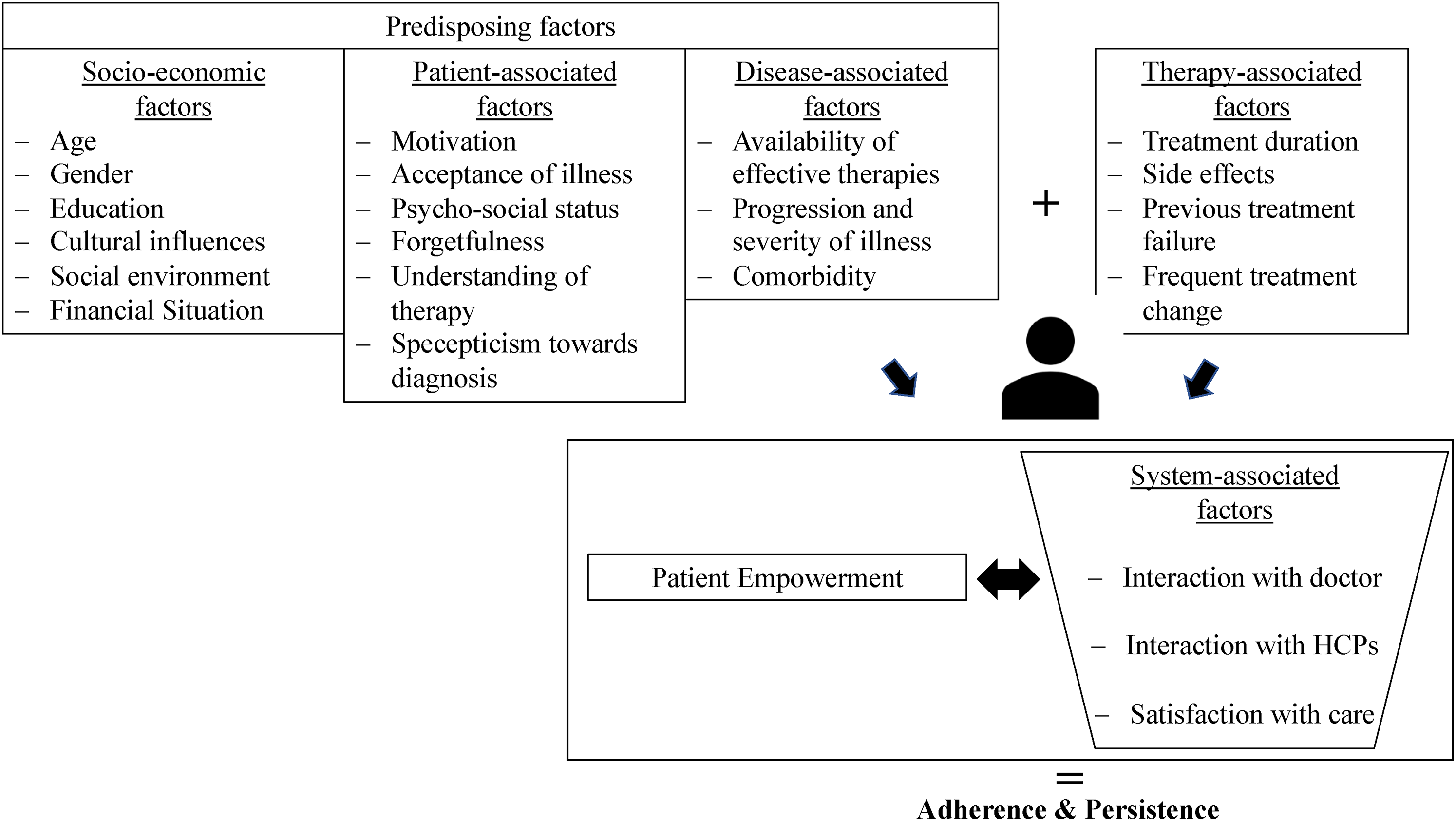
Factors of adherence. Source: Diagram by the authors based on Geynisman and Wickersham. 30
Empowerment and adherence from the patient's point of view
In the context of cancer, the majority of patients want to be informed as precisely as possible about diagnosis, treatment options, prognosis, side effects as well as being involved in all decision-making processes.19,45,46 In addition to the lack of time in the doctor's patient talk, doctors tend to withhold certain information from patients by assuming that this information is too complex or worrying for patients. In the process of patient empowerment, research using the internet plays a major role. Patients feel like experts in the treatment process. They can also compensate for information gaps or obtain additional information material. As studies show, patients benefit from both, the information offer and the exchange with other breast cancer patients.46,47 However, the almost inexhaustible possibility of obtaining information on the internet also increases the risk of overburdening, insecurity, and misinformation on the patients’ side. This often creates a potential conflict in doctor–patient communication. 23
Nevertheless, breast cancer patients who are well informed and actively participating in the treatment decision can benefit from a higher quality of life, lower fatigue, better involvement in their social environment, and better physical condition. 46
Patients who have a high level of empowerment through a well-functioning doctor–patient relationship benefits from better clinical, health, and aftercare outcomes leading to a higher quality of life.40,48 In addition to improved patient health, better disease management, higher adherence, and effective use of health services can also be attributed to patient empowerment. 40 Consequently, costs for the entire health system can be saved and at the same time, better health outcomes and higher patient satisfaction can be achieved.17,49 In its “Health 2020” program, the WHO has defined patient empowerment as a key factor in achieving better health outcomes within the EU. 50
Increasing relevance of adherence in breast cancer therapy
In treating cancer, oral chemotherapy is a strongly growing area. While only 5% of chemotherapy drugs were available in oral form in 2003, 20% to 25% of the drugs in development were oral active agents. 12 These modern oral cancer drugs offer many benefits to patients, such as shorter treatment times, better independence, and improved quality of life, as they are administered by the patient themselves. On the other hand, there is also the risk of irregular intake and non-adhering patients. In addition, side effects management must be handled rather by the patient than by the doctor. If side effects occur by the drug, the patient must judge them and contact the doctor if necessary. Via oral medications, a paradigm shift in the field of cancer treatments is taking place. Since doctors only have a limited amount of time, the interaction with HCPs is important.12,13,51
Especially in antihormone therapy (endocrine therapy), adherence of breast cancer patients has a high priority. In endocrine therapy, three active substances are available: anti-estrogens, aromatase inhibitors, and GnRH analogs. The anti-estrogens are split between two substances: tamoxifen and fulvestrant. Both anti-estrogens and aromatase inhibitors are taken as oral pills. 14 Previous studies showed that over 50% of patients did not finish their antihormone therapies as recommended by the doctor. 15 The adherence of breast cancer patients ranged from 65% to 79% in tamoxifen and from 72% to 80% for aromatase inhibitors, but decreased to around 50% over the fourth and fifth year. Additionally, half of the patients cancel the antihormone therapy after five years. This means that a significant proportion of breast cancer patients cancel the antihormone therapy after five years and do not receive the full medical benefit of their therapy. 52
Many patients think that oral chemotherapy is less effective than an intravenous one and others also see it as their last chance. 53 For this reason, information for patients is essential. In a study, German brochures were examined by various cancer societies such as the pharmaceutical industry, statutory health insurance companies, and self-help organizations. The results indicate that the brochures for breast cancer patients are less helpful than those for other cancers and that they can rarely serve as a decision-making aid for patients. 24
Patient journey of breast cancer patients
The patient journey was modeled according to the so-called “customer journey.” This refers to a journey of the customer (or even the patient) across various points of contact to achieve a specific goal. Not every stage is always pleasant for the customer because it can include aspects or even results that are not desired by Lindner 54 and Halvorsrud et al. 55 The patient journey is therefore a series of events along the course of a disease. It starts with perception about diagnosis, goes via treatment, and recovery up to aftercare. In German literature, a uniform definition has so far been lacking. 54 However, a generally valid definition is essential for patients, doctors, patient organizations, and other stakeholder groups to work together on a common basis. 55 The basis for this article is the patient journey in Figure 2.

Patient journey of breast cancer patients. Source: Own representation based on Thrift-Perry et al. 56
Therefore, breast cancer patients undergo a patient journey that can be divided into five phases: disease perception, diagnosis, coordinated care, treatment, and aftercare. 56
Depending on the phase of the patient journey, different stakeholder groups are involved in the process: patient organizations, health insurance companies, doctors, HCPs, publishers of information brochures for patients, psycho-oncological services, and self-help groups. In Germany, health insurance companies take over the annual breast cancer examination for women starting with 30 years of age. 57
Definition of mHealth
There is not yet a uniform definition for the term Mobile Health, also called mHealth. The WHO defines mHealth as a “medical and public health practice supported by mobile devices, such as mobile phones, patient monitoring devices, personal digital assistants (PDAs), and other wireless devices.” 58 Therefore, mHealth supports medical treatments to leverage public health and health care using various mobile technologies. This definition was also adopted by the European Commission. 59 mHealth is a sub-area of Electronic Health or eHealth. 58
eHealth covers all applications based on information and communication technologies (ICT). Information processing and distribution take place electronically in order to promote and support patients’ care and treatment in general. The use of ICT should be safe and cost-effective. In addition to mobile applications, eHealth also includes telemedical applications and “elektronische Patientenakte” = “electronic health record.”60,61
eHealth is currently thought of as a part of the larger concept of Digital Health. A definition attempt for Digital Health is: “The cultural transformation of how disruptive technologies provide digital and objective data accessible to both caregivers and patients leads to equal level doctor–patient relationship with shared decision-making and the democratization of care.” 20 Digital Health is therefore the use of different technologies that provide digital data to both patients and HCPs in order to enable a participatory therapy decision-making for patient and physician. 20 Digital Health supports people in promoting and maintaining their health on their own authority. It supports the goal of individually adapting medical care with quality, effectiveness, and efficiency, in order to improve the health of the entire population.62,63 Having no uniform definition for digital health, it can be seen as an umbrella term for all digital applications in health care.
This also includes mHealth applications. These differ fundamentally from eHealth or Digital Health since they are significantly more relevant for the patient. They not only serve the mere provision of information or organization, but also offer the patient the opportunity to actively participate in the treatment by using apps and without having to involve doctors or HCPs. 64
However, so far it is still very difficult for patients as well as for doctors to find and assess high-quality apps across the different app stores. No uniform quality standard exists yet for DiGA (i.e. “Digitale Gesundheitsanwendung” = “Digital Health Application”). 65 Quality labels such as CE marking, or even better labels such as “GS” (“Geprüfte Sicherheit” = “Proven Security”) and TÜV could help. Nevertheless, these are not yet widespread. In 2018, out of 8.767 German-language health & medicine apps, only 41 apps were marked in the app stores with a seal of approval. Only 34 apps had a CE marking and only a handful others had a non-governmental seal of quality. The CE marking is hardly used until now. 66 However, a CE marking is merely evidence that regulatory standards have been met with the aim of authorization as a medical device. 66
Another guide for patients could be the White List (“Weisse Liste”). So far, the Bertelsmann Stiftung project offers patients an online orientation in the search for doctors and hospitals. This offer will be expanded to include information on DiGA in the future. First, the patient will be provided with detailed supplier information of the respective DiGA, which will be gradually extended with quality characteristics. 65 So far, this comprehensive information for health & medicine apps is available in an early beta version. 67
Classification procedure for digital health applications
In 2016, the Bertelsmann Stiftung developed a classification procedure for digital health applications specifying seven application types:
Type 1: Improvement of health literacy Type 2: Analysis & realization Type 3: Indirect intervention: Promoting self-efficacy, adherence & safety Type 4: Direct intervention: Change of skills, behavior & states Type 5: Documentation of health and disease record Type 6: Organization & administration Type 7: Procurement & supply
63
It has been realized that DiGA has the potential to improve the patient's care and independence. However, the necessary requirements in prevention and supply have not yet been implemented in the market. In addition, there are no meaningful evaluation procedures for digital applications that allow transparency for the user.
65
For this reason, the AppQ project was launched by the Bertelsmann Stiftung and funded by the Federal Ministry of Health. Its aim was to develop a quality criteria core set for users to facilitate the evaluation of DiGA. Another concern was to use the quality criteria core set to let providers fill in a self-information for publishing the information on the quality of the digital health applications in the White List (oder “Weisse Liste”?). AppQ looked at DiGA from a patient perspective and also took the communication with HCPs into account. Accordingly, the focus was set on the microlevel and not the system level. The hypotheses examined were assigned to the categories “economic,” “social/ethical,” and “organizational” based on the health technology assessment (HTA). 65
The HTA is defined by the WHO as: “Health technology assessment (HTA) refers to the systematic evaluation of properties, effects, and/or impacts of health technology. It is a multidisciplinary process to evaluate the social, economic, organizational, and ethical issues of a health intervention or health technology. The main purpose of conducting an assessment is to inform a policy decision making.” 68 Therefore, it is an instrument for the systematic evaluation of medical technologies, taking into account socio-cultural, ethical, organizational, and economic aspects. 68 The most important component of the HTA is the economic analysis, which determines the cost and benefits of medical technology for health care systems. 69 In practice, however, HTA is usually only used to evaluate medical aspects. 70
Analysis of apps for breast cancer patients
Mobile app search
On 13 February 2020, searches in the Google Play Store (Android) and the App Store (iOS) with the terms “breast cancer” and “cancer” were conducted. Apps were downloaded if the title, description, or reviews showed that:
targeted apps for breast cancer patients or cancer patients in general with a possible application to breast cancer patients does not apply to doctors or medical professionals is available in German is available both for the Android and iOS operating systems. These operating systems were selected as around 90% of apps for cancer patients are available for Android and IOS.
71
Findings
In total, 44 apps were found for cancer and breast cancer patients in Germany. Of these, only 33 were available for both Android and iOS. Eleven apps were only available for one operating system and contained numerous spelling and grammar errors in the description. They were therefore not included in the analysis. Two of the 33 downloaded apps could not be opened in the practical test, neither on iOS nor on Android. A total of 31 apps were analyzed (Figure 3). Thirty apps aim at adults and one app is focused on children.

Selection process of the examined apps. Source: Own presentation.
In 2016, the Bertelsmann Stiftung developed a classification procedure for digital health applications specifying seven application types.
For the apps included in the analysis, the classification procedure of the Bertelsmann Stiftung for digital health applications in seven application types was carried out.
63
In addition, the following data was collected:
number of downloads in the Google Play Store end price for the user need for access after the download of the app application for cancer types and matching phases of the patient journey participation of doctors identification with labels motivation or emotional focus of the app available languages.
Among the apps examined, two already need to be paid for the download and three more charges for using the full functionality. For seven apps, a user account must be created and two leave it up to the user to create an account to save the data. Only 16 of 31 apps specifically target breast cancer and six of them generally target breast change. The app stores currently only offer 10 apps that explicitly address breast cancer patients.
In Brouard et al. 72 study, 117 apps for oncological patients were examined with respect to information and therapy monitoring. Only 27.4% of these apps were scientifically validated. 72 Another study by Collado-Borrell et al. 73 investigated 166 apps for cancer patients. They also lacked the participation of medical professionals and the scientific basis of information in the apps. 73
This analysis also provides confirmation: only one of 31 apps uses the non-governmental quality seal of TÜV Saarland for advertising. On the other hand, two apps that are already certified as class 1 medical devices do not state this in their app stores description. This means that breast cancer patients cannot even recognize the quality of these certified apps. The same is true for the participation of doctors.
The following fact can be extracted from the descriptions of the apps:
The editors of eight apps are medical facilities, doctors, a cancer society, or cancer aid. Two other apps have been created in collaboration with medical institutions. One allows a connection with the doctor. One refers to quality-assured information. Another one to the summary of medical studies.
In summary, 13 of 31 apps point to tested knowledge. The apps studied are usually available in several languages.
The classification of the examined apps into different application types gave the following results (Table 1).
Number of apps per application type.

The majority, that is, 27 apps, focus on “improvement of health literacy” (type 1). Apps with functions according to type 1 should provide the patient with knowledge before a potential illness. Among other things, for breast cancer, these are apps that provide information about the disease, the preventative palpating of the breast, help in finding experts, or in making decisions. With this information, patients are given the opportunity to act independently within and outside the health system.
In the present investigation, there is no app intended for “Analysis & Recognition” (type 2). Applications according to type 2 should help especially when evaluating information to make an assessment or diagnosis.
With 13 apps, the “indirect intervention” (type 3) is most frequently represented after type 1. This type of application allows the patient to also collect and evaluate health-related data and information. This should contribute to the promotion of patient adherence, safety, and self-efficacy. Functions for memorization of revenue, exchange between patients, and motivation or control are also included in these apps.
Apps related to “direct intervention” (type 4) were not found. Applications according to type 4 would primarily offer support in case of behavioral or health problems, such as a magnifying glass function.
Applications of type 5 support the patient in “documentation of health and disease history,” comparable to a patient record. For example, this supports storing of findings. Three apps for breast cancer patients were found in this category.
Type 6 helps the patient with the “Organization & Administration” of, for example, appointments or invoices. Three apps were identified for planning doctor's appointments.
There is no app for breast cancer patients concerning type 7. This application type helps with “Procurement & Supply” of, for example, medicines. 63
This distribution shows that apps are dominated by functions of the types “improvement of health literacy” (type 1) and “indirect intervention” (type 3). For type 1 and type 3, a great potential is seen in breast cancer. 63
The emotional support of cancer patients improving their own motivation is very important. Table 2 shows how the analyzed health & medicine apps motivate.
Number of apps for motivation.

Therefore, the way of motivation or influence the emotional response of the patients is very different in each app: Three apps use experience reports, one sports award, one weekly goal, two by connecting with other patients. One app allows the sharing of emotional states with persons of trust and another offers the possibility to create a bead chain for oneself or their cancer-stricken children with each treatment step. The app for children addresses young patients emotionally by using gamification.
The aim of these features is to motivate patients and also prove theories about motivation and behavioral change. These features are referred to as behavioral change techniques (BCTs) and represent the smallest components of behavioral intervention. These can be used alone or in combination with other BCTs to influence the behavior of patients. 74
Categories for BCTs include objectives and planning, observation of behavior and feedback, social support, or promotion of self-confidence. Also, the formation of knowledge, for example, the information on health effects, can lead to behavioral changes. 75 Finally, BCTs should motivate the patient and increase adherence.
The apps examined in this work rarely integrate one or more BCTs. Out of the 31 apps examined only 10 include BCTs for motivation and from these only five apps are designed for breast cancer patients and one for children. In an examination of fitness apps, Direito et al. 76 also found unused potential for behavioral changes. Although apps will very likely be an important tool for patients’ behavioral change in the future, this has not yet been properly implemented by the providers of breast cancer apps. 77
However, there are already very good examples in the area of fitness and weight loss apps. Apps such as Weight Watchers, Gymondo, or Runtastic offer digital reward systems that motivate the participants to use an app in the long term to achieve their goals.
After the classification of apps into different application types, in the next step, we analyze how patients can really use the apps throughout the different phases of the patient's journey (Figure 4):

Number of apps per phase of the patient journey. Source: Own presentation.
The patient journey was extended by the pre-phase “prevention.” One app was found for pure prevention. It offers a training program to prevent cancer.
Six apps were found for the prevention and perception of breast cancer. They are all intended for self-examination of the breast and sometimes provide additional assistance when abnormalities are discovered at the chest.
The majority of apps (19) supports patients in different phases from diagnosis to aftercare. These apps offer a wide range of functions, including information, expert search, exchange with other patients, patient care diaries, drug reminder features as well as tips for sports and nutrition.
Only one app was found for phases 4 and 5 that does not specifically aim at cancer patients. It generally serves the management of different diseases in order to remember the medication intake or doctor's appointments.
Last but not least, two apps for aftercare only were identified. One of them targeted the participants of a particular cure.
At this point, it seems that so far app companies assign the greatest potential to the diagnosis phase. Although there is a great need for aftercare, just a few apps are designed for this phase although the aftercare phase is particularly important for breast cancer. Bender et al. 71 studied 295 English-language apps related to cancer. Of these, 138 apps (46.8%) focused on breast cancer. Among these 138 apps, four apps were designed for prevention and 17 apps for disease detection (phase 1) during the patient journey. Only three apps were intended for the management of the disease (phase 2). Only three of the 295 apps were designed for the communication of cancer patients within patients’ community or with their HCPs. 71
Additionally, the study of Jupp et al. 13 investigated apps for cancer patients, and the majority of these apps provided functions for the prevention of and information about cancer. Features to communicate with other cancer patients or self-help groups were also not present very often. 13
According to the results of this analysis, a need for action was detected to develop apps that support diagnosis (phase 2) and the subsequent phases. Today, 10 German-language apps exist that explicitly address breast cancer patients. Merely two apps enable patients to communicate with each other. However, the offer is still expandable, especially with regard to the quality of information as well as patients’ motivation and emotional address.
Discussion/conclusion
In conclusion, the potential of health and medicine apps for breast cancer patients has been recognized. Nevertheless, the number of breast cancer apps can still be increased. In addition, the already available apps lack meaningful sources and references. Medical guidelines are rarely considered.78,79
Therefore, it is recommended to introduce a uniform process for quality assurance of health & medical apps as patients need more transparency in mHealth applications. 72 As early as 2014, the first study which explicitly examined apps in the context of breast cancer revealed major deficiencies regarding the app's information and the absence of HCPs. 80 Giunti et al. repeatedly pointed out these existing defects. 79 This analysis also showed that the quality of the individual apps is neither apparent from descriptions in the app stores nor from the usage after download. That is why it is currently difficult for patients, laypersons, doctors, and other therapists to identify high-quality apps. Guidance from independent or governmental institutions would be important to further improve the digitalization of the health care system.
Considering the factors of adherence, especially the patient- and system-associated factors, these are still insufficiently addressed by apps. In the case of patient-associated factors, forgetfulness is mainly addressed by memory functions, understanding of therapy specifications, and skepticism toward the diagnosis through information in the app. The remaining characteristics—motivation, acceptance of the disease, and psychosocial status—are treated insufficiently only. Based on the current study, only 13 apps use the “indirect intervention” application type and just 10 apps address the patient on an emotional basis. A significantly greater lack arises in the case of systemic factors. There is no app that allows interaction with the doctor or the HCPs. As pointed out by Giunti et al., 79 HCPs are not yet integrated into apps for breast cancer patients. While HCPs have great importance, apps must further improve to integrate them.
Limitations and further directions
Keyword searches for “breast cancer” and “cancer” were used to identify relevant apps for including them in the study. It is possible that other apps may also be useful to cancer patients but were not detected by these search terms. Furthermore, the analysis was restricted to apps available in Germany, provided in German language, and offered in the Google Play Store and Apple App Store. This might have limited the number of available and evaluated apps. Furthermore, the criteria selected were not evaluated based on patients’ opinions and experiences, but in a double-checked evaluation process by two raters. Patients’ and HCPs’ ratings would be very useful and could be included in further research.
This study provides foundations for future research. Further research should focus on app quality in combination with possibilities of interactions between patients and health experts (e.g. integrating Apps such as “WeChat,” “WhatsApp,” or similar).
So far, no study has yet been found that addresses changing needs of breast cancer patients along the patient journey, further research is recommended here.
The potential to positively influence adherence has already been recognized by app providers. Thus, individual factors of adherence are already addressed by app functions. This is an undiscovered opportunity for increasing adherence through health and medicine apps and another challenge for future app developments. Existing fitness apps (e.g. Weight Watchers or Gymondo) that offer digital reward systems, connections to real trainers, or even access to the online communities can serve as successful examples for app providers.
Health and medical apps offer several benefits for patients, physicians, and other HCPs as well as for health insurers and the German health care system. However, in order for these potentials to be fully exploited in the future, the quality of the apps needs to be improved. So far, there is no standard process for quality assurance of health and medical apps. It is hardly recognizable if Apps are developed by business companies or public institutions (e.g. public hospitals, foundations, and universities), which can affect the motivation, commercial independence, and aims of medical apps.
Footnotes
We would like to thank CW for his assistance and guidance in this research.
Contributorship
LT and SS researched literature and conceived the study. LT wrote the first draft of the manuscript. SS reviewed and edited the manuscript and approved the final version of the manuscript
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Meanwhile, LT is an employee of Sanacorp, a pharmaceutical wholesaler, there are no conflicting interests.
Ethical approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
SS.
Informed consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial registration
Not applicable, because this article does not contain any clinical trials.
