Abstract
Case summary
A 13-year-old castrated male American Shorthair cat was referred for evaluation following a 3-week history of poor balance and decreased activity. The MRI findings revealed a well-defined left caudal cerebellar mass with a diameter of 1.2 cm, consistent with a meningioma. CT and MRI scans did not reveal metastasis. After a suboccipital craniotomy, the mass was resected subtotally as a result of parenchymal invasion. A histopathological examination revealed a benign fibrous meningioma with invasion into the cerebellum at the tumour margins. Postoperatively, the cat remained asymptomatic for 25 months, but ataxia recurred, and tumour recurrence was confirmed using MRI and post-mortem examination.
Relevance and novel information
This case demonstrates that even feline meningiomas with benign features can exhibit brain invasion. Since brain invasion is an indicator of malignancy in meningioma, a detailed histopathological evaluation of the tumour margins is essential for accurate grading and prognosis.
Introduction
Meningioma is the most common type of brain tumour in cats. 1 Subtypes of meningioma in domestic animals include meningiotheliomatous, fibrous, transitional, psammomatous, angiomatous, papillary, granular cell, myxoid and anaplastic.2,3 While most subtypes of meningioma exhibit benign tumour characteristics, the anaplastic subtype is classified as malignant. Even if a meningioma is not of the anaplastic subtype, it can be classified as malignant based on features such as a high mitotic rate, hypercellularity with uninterrupted patternless growth, extensive necrosis, brain invasion and metastasis. 3 In cats, unlike in humans and dogs, most meningiomas are benign. A previous study reported that invasion into the brain parenchyma was not observed in 80 cases of feline meningioma. 1 However, another study reported multiple malignant meningiomas with intracranial metastasis in a cat. 4
Brain invasion, a malignant indicator, is not rare in meningiomas in humans and dogs.5 –7 Several studies have focused on brain invasion in meningiomas, particularly in humans. It is important to note that there are several meningiomas in humans that exhibit no malignant features other than brain invasion, referred to as brain-invasive but otherwise benign meningiomas (BIOBMs). 8 However, such cases have not been reported in cats.
Case description
A 13-year-old castrated male American Shorthair cat was referred for evaluation following a 3-week history of ataxia and decreased activity and appetite. The only neurological examination abnormalities were obtundation and absent pupil light reflexes in both eyes. Blood tests, including a complete blood count and serum biochemistry, showed values within normal ranges. Based on signs and examinations, we suspected intracranial lesions especially in or near the preopticular area, the oculomotor nerve and the cerebellum.
Imaging studies were conducted using a 1.5 Tesla MRI system (Vantage Elan; Canon Medical Systems) and a CT scanner (Aquilion Lightning; Canon Medical Systems). The MRI findings (Figure 1a–c) revealed a well-defined, left caudal cerebellar mass with a diameter of 1.2 cm. The mass appeared hypointense in T2-weighted images and isointense in T1-weighted images (T1WIs). It exhibited almost homogeneous enhancement and a dural tail sign in contrast-enhanced T1WIs. Furthermore, T2-weighted and fluid-attenuated inversion recovery sequences revealed a hyperintense signal in the surrounding cerebellar parenchyma, ie, a mass effect, implying that the mass was extra-axial. Other findings included a cerebellar herniation resulting from mass compression. The CT scan of the skull suggested microcalcification within the mass, but no specific findings in the adjacent skull region (Figure 1d). Imaging studies revealed no evidence of metastasis.

Diagnostic MRI and CT images before the first surgery. (a) A horizontal T2-weighted image reveals a hypointense mass with a hyperintense signal in the surrounding cerebellar parenchyma. (b) Transverse and (c) horizontal contrast-enhanced T1-weighted images reveal almost homogeneous enhancement of the mass with a dural tail sign. (d) A transverse CT image at the level of the cerebellum suggests microcalcifications in the mass (white arrowhead)
Treatment with corticosteroid (0.9 mg/kg PO q24h) was started with anti-inflammatory intent and isosorbide (0.9 ml/kg PO q12h) was administered to reduce intracranial pressure. Consent for surgery was obtained from the owner and a suboccipital approach was performed to access the caudal cerebellar lesion. After premedication with fentanyl (3 μg/kg IV), midazolam (0.2 mg/kg IV) and medetomidine (3 μg/kg IV), general anaesthesia was induced with propofol (1 mg/kg IV) and rocuronium (0.2 mg/kg IV, to ensure suppression of the cough reflex) and maintained with sevoflurane at an end-tidal concentration of 1.0–1.5%. Fentanyl (10–15 μg/kg/h IV) and medetomidine (0.5–1.0 μg/kg/h IV) were administered for intraoperative analgesia. After the skin and muscle incision, the occipital bone was drilled using a 2.5 mm round burr to make a 1.5 × 1.0 cm oval bone window. After dural incision, a pale or off-white mass became visible and a piece with a diameter of 1.0 cm was resected from the inside of the mass to reduce in volume under an operating microscope (Leica M525 F50; Leica Microsystems) (Figure 2a). Subsequently, an attempt was made to detach the marginal part of mass from the cerebellar parenchyma, but part of the mass was found to be adherent to the cerebellum. An artificial dura was sutured to the defective dura using 5-0 nylon sutures. The muscle and skin layers were closed routinely. For the postoperative analgesia, fentanyl (3.0 μg/kg/h IV) and medetomidine (0.75 μg/kg/h IV) were administered for 10 h, followed by buprenorphine (10 μg/kg SC q12h) for 3 days. No complications related to the surgery or anaesthesia were observed after awakening from the anaesthesia. The patient had an appetite the day after surgery and was discharged 3 days postoperatively, continuing the treatment of corticosteroid (0.9 mg/kg PO q24h) and isosorbide (0.9 ml/kg PO q12h).
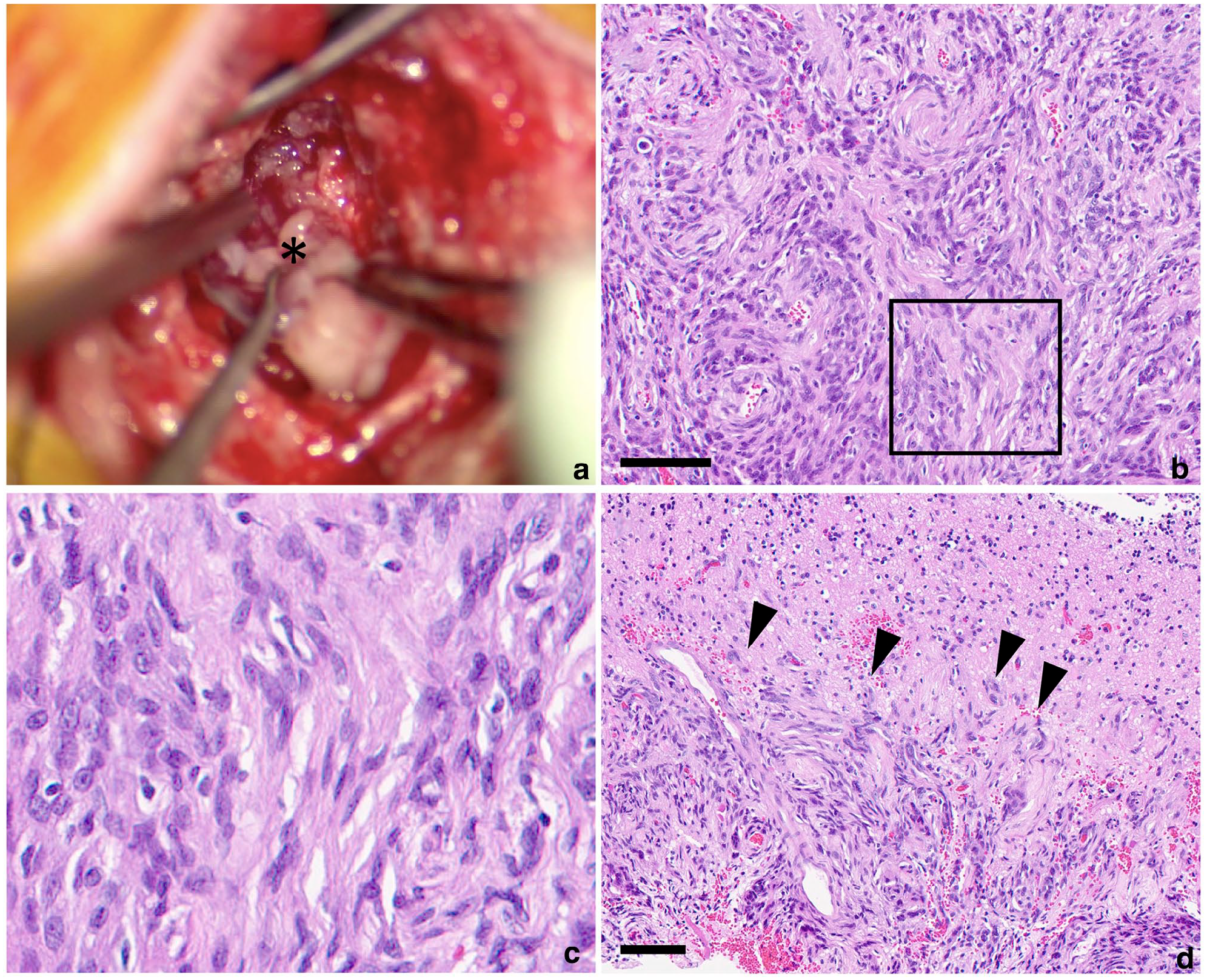
Gross and histological images of the meningioma at the first surgery. (a) The meningioma, accessed via a suboccipital approach, appears as a milky white mass (asterisk). (b) Low- and (c) high-magnification (square in panel b) images of H&E-stained sections of meningioma show wavy growths of spindle-shaped tumour cells with nuclei of mildly irregular size. (d) A low-magnification image of an H&E-stained section of meningioma shows tumour cells forming finger-like protrusions (arrowheads) invading the cerebellar parenchyma. Bars = 100 μm. H&E = haematoxylin and eosin
Histologically, resected specimens were diagnosed as invasive fibrous meningioma. The tumour was characterised by intricate, wavy growths of spindle-shaped tumour cells (Figure 2b), weakly acidophilic cytoplasm, round-to-elongated nuclei of mildly irregular size and well-defined nucleoli (Figure 2c). Tumour cell atypia and proliferative activity were low. In specimens from the tumour margins, resected cerebellar tissue was found adjacent to the tumour tissue, showing invasion by tumour cells forming finger-like protrusions (Figure 2d). The border between the tumour and the cerebellum was disrupted and evaluated as not completely resected. Calcification and neovascularisation were observed within the tumour. No necrosis or occipital bone invasion adjacent to the tumour were observed.
At 10 days postoperatively, ataxia was clearly improved, and a weak pupil light reflex was recovered in both eyes. At the 1-month follow-up, ataxia was barely detectable, and corticosteroid and isosorbide were tapered by 50%. Chemotherapy with hydroxyurea was considered owing to incomplete resection and brain invasion. However, after consultation with the owner, supportive therapy with corticosteroid (0.45 mg/kg PO q24h) and isosorbide (0.5 ml/kg PO q12h) was chosen. The cat showed no signs of ataxia and was in good overall health for 25 months. However, clinical signs recurred, and tumour recurrence at the same location was confirmed by MRI. A second tumour resection surgery was performed; however, because of the tumour’s proximity to the brain stem, a complete resection was not possible. In addition, controlling bleeding from the mass was difficult, resulting in a volume reduction of less than 30%. The histopathological diagnosis was the same as that during initial surgery. Because the marginal component of the meningioma could not be removed, the brain invasion could not be evaluated. The cat’s appetite and activity gradually recovered, and he was discharged 10 days postoperatively. After discharge, the cat’s appetite and activity recovered further, but the ataxia did not. The cat was given no further treatment except for supportive therapy with corticosteroid and isosorbide, and spent his final days at home, according to his owner’s wish. Appetite and activity gradually decreased, and the cat died 3 months after the surgery.
A post-mortem examination was conducted, and the tumour was identified as a fibrous meningioma, consistent with previous biopsies. Grossly and histopathologically, no metastatic lesions were detected. The tumour caused compression of the cerebellum (Figure 3a). The tumour surface appeared irregular and milky to pale yellow in colour (Figure 3b). Grossly, the boundary between the tumour and the cerebellum appeared clear. However, a histological examination of the tumour margins revealed invasion into the cerebellum in some areas (Figure 3c). In addition, invasion into the adjacent occipital bone was observed (Figure 3d). Grossly and histologically, there was no haemorrhage or necrosis in the brain. It is presumed that the pressure from the tumour caused intracranial hypertension and loss of appetite, leading to deterioration of the cat’s general condition, which resulted in death.

Gross and histological images from the post-mortem examination. (a) The cerebellum shows a depression (arrowhead) caused by tumour pressure. (b) A pale yellow tumour (asterisk) with an irregular surface is attached to the occipital bone. (c) A low-magnification image of an H&E-stained section of meningioma reveals tumour invasion into the cerebellum (arrowheads indicate finger-like protrusions). (d) A low-magnification image of an H&E-stained section of the occipital bone reveals a cluster of tumour cells within the occipital bone. Bars = 250 μm. H&E = haematoxylin and eosin
The histopathological examination revealed that the meningioma displayed benign characteristics, except for the consistent invasion of the brain. Brain invasion was evident in specimens from the first surgery and the post-mortem examination. The number of mitotic figures of the meningioma during the first surgery, second surgery and post-mortem examination were 0–1 per 10 high-power fields (2.37 mm2) at all time points.
Discussion
This report presents histopathological evidence of brain invasion in a cat with a meningioma showing benign features. Brain invasion is an important criterion in meningioma grading, and its presence alone is sufficient to classify the meningioma as a World Health Organization (WHO) grade II atypical meningioma.3,7,8 In human grade II atypical meningioma, brain-invasive meningiomas without additional malignant features are referred to as BIOBMs. 8 The current case is consistent with the features of human BIOBMs. Studies suggest that human BIOBMs progress similarly to WHO grade I benign meningiomas and have a longer period without tumour progression than other WHO grade II atypical meningiomas.9 –11 These reports suggest that the prognosis is relatively good for human BIOBMs, even in the presence of brain invasion. In the present case of a BIOBM in a cat, the postoperative survival time and the interval between the first and second surgeries were 28 and 25 months, respectively, which are similar to the prognosis for WHO grade I benign meningiomas.12 –14 A large-scale study is required to clarify how brain invasion affects the prognosis for cat meningiomas.
The MRI findings in this case revealed a typical, well-defined solitary meningioma with a dural tail sign, and no findings implying brain invasion were noted. However, during surgery, the tumour was found to be attached to the cerebellum, indicating less distinct boundaries than typical benign meningiomas. This raised concerns about potential brain invasion, prompting a thorough histopathological examination of the specimen margins. To detect microscopic brain invasion, tumour tissue that is adherent to the brain should be closely examined histopathologically.
In humans, it has been shown statistically that brain invasion by meningiomas is more common in men. 9 Furthermore, histological invasion typically presents as a finger-like pattern in men and a cluster-like pattern in women. The present case in a cat followed these trends. In humans, studies have been conducted on gonadal steroid hormone receptor expression in meningioma, 15 but the mechanisms that cause brain invasion are unclear. The acquisition of brain invasiveness is thought to require extracellular matrix degradation, acquisition of cell migration using adhesion molecules (eg, integrin, E-cadherin and selectin), neovascularisation and signalling by growth factors (eg, hepatocyte growth factor, endothelial growth factor, and vascular endothelial growth factor). 16 Hormone receptor signalling may be involved in these complex processes.
In the present case, post-mortem examination revealed invasion of the occipital bone. Histological examination and imaging at the first surgery showed no tumour invasion of the bone, indicating a gradual infiltration over time. Unlike brain invasion, bone invasion is common in cat meningiomas and does not affect prognosis. 17 Other characteristic findings in this case included calcification within the tumour. Little is known about the association between calcification and prognosis in domestic animals. In humans, meningiomas with calcification have been reported to have slow growth. 18 For a detailed prognosis, the characteristics of the meningiomas need to be studied carefully.
Conclusions
In cats, a meningioma with benign features can exhibit brain invasion. Large-scale clinical studies are needed to clarify whether brain invasion in benign meningioma affects prognosis.
Footnotes
Acknowledgements
We would like to thank the owner of the cat for allowing us to carry out the post-mortem examination and gain further understanding of the disease, and ultimately advance veterinary medicine. We also thank staff who assisted with the surgery and post-mortem examination.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
