Abstract
Cutaneous meningiomas are rare tumors in human beings and animals. Two canine cases of cutaneous meningiomas affecting the eyelid are described in the current study: the first from a 5-week-old female Springer Spaniel dog with an 8 cm in diameter congenital mass expanding the left upper eyelid and medial canthus; the second from a 10-year-old female spayed Maltese–Poodle mix dog with 3 firm subcutaneous nodules affecting the right upper eyelid. All masses were removed surgically. Histologically, tumors were composed of spindle-to-epithelioid cells arranged in small lobules forming solid concentric whorls. Neoplastic cells were positive for vimentin and S100 and negative for pancytokeratin, glial fibrillar acid protein, and neurofilament. Transmission electron microscopy revealed meningothelial cells with convoluted interdigitating processes, desmosomes, and hemidesmosomes, and moderate numbers of cytoplasmic microfilaments. None of the cases presented a primary neuroaxial meningioma. The first case presents clinicopathological features consistent with human type I (congenital) cutaneous meningioma. The second case is consistent with a type II (acquired ectopic) tumor, and both are hypothesized to arise from ectopic arachnoid cells displaced during development.
Keywords
Meningiomas are the most common intracranial tumors in dogs.2,16,17 The tumors arise from the neoplastic transformation of arachnoid cells (meningothelial) of the arachnoid membrane and pia mater of the nervous system. 10 Meningiomas in dogs are mostly intracranial but spinal cord, orbital, paranasal, and cutaneous meningiomas have also been reported.5,7,14,18 Extracranial cutaneous meningiomas occur occasionally in human beings but are extremely rare in dogs and other species. In human cases, cutaneous meningiomas can be congenital or acquired and usually affect the scalp and skin around facial sensory organs. 9 The histogenesis of these tumors in human beings is uncertain but the main theory is that the tumors originate from displaced meningothelial cells through the skin during embryogenesis or extension of an intracranial tumor. 8 The present report describes the clinicopathological features of 2 cases of canine periocular cutaneous meningiomas, which are directly comparable with the 2 human subtypes.
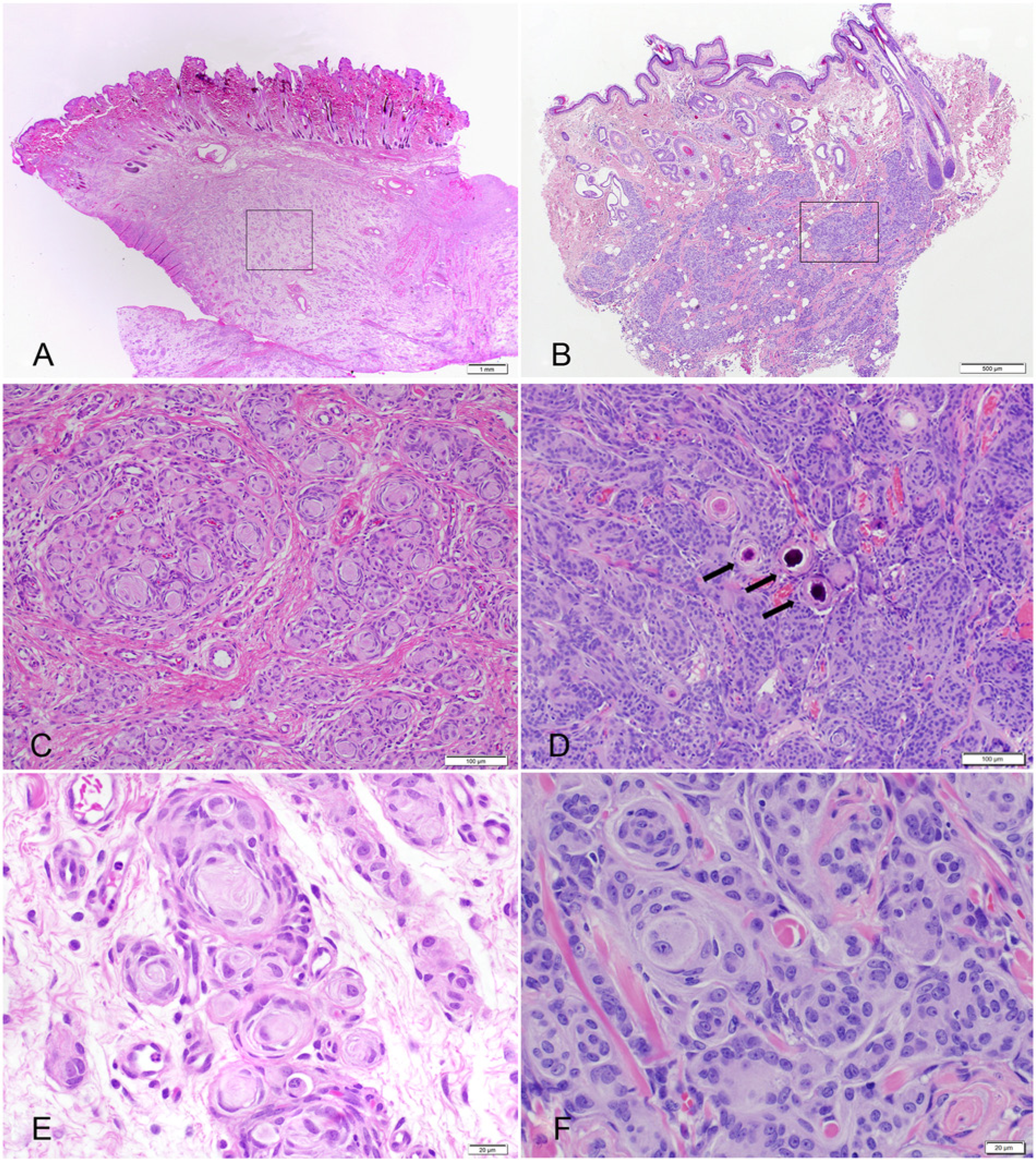
The first case, a 5-week-old female Springer Spaniel dog, presented to the University of Wisconsin–Madison Veterinary Medical Teaching Hospital (Madison, Wisconsin) with a history of a congenital, progressively growing, nonpainful, 8 cm in diameter, soft, nonulcerated, alopecic, and pedunculated mass attached to the medial canthus and expanding the left upper eyelid. Ultrasound of the mass showed diffusely hyperechoic tissue with pockets of lucency and large blood vessels at base of the mass. Fine-needle aspiration and cytological evaluation of the mass revealed a mildly cellular sample with 2 neoplastic populations. The first was composed of elongated fusiform cells with increased nuclear-to-cytoplasmic (N:C) ratio, wispy basophilic cytoplasm, and oval nuclei with stippled chromatin. The second was composed of epithelioid cells with low N:C ratio, and deeply eosinophilic and granular cytoplasm with oval nuclei that formed rare small clusters. The mass was surgically removed and fixed in formalin. On cut section, the mass was gray, slightly translucent, and gelatinous to rubbery. The tissue was routinely processed and stained with hematoxylin and eosin (HE), Bielschowsky, and Luxol fast blue. Histologically, the dermis and subcutis contained a poorly demarcated, unencapsulated, and invasive mass composed of spindle cells forming small lobules (Fig. 1A). Within the lobules, spindle cells frequently formed small whorls, usually with a small amount of concentric laminated fibrillar eosinophilic material centrally creating an “onion-bulb” appearance (Fig. 1C, 1E). Between the lobules, cells formed loose to dense streams that multifocally dissect preexisting tissue elements. The cells presented indistinct borders, small amounts of fibrillar to wispy eosinophilic cytoplasm, and an oval to crescentic nucleus with finely stippled chromatin and inconspicuous nucleoli. Mitotic figures were uncommon at 3 per ten 400× fields. Anisocytosis and anisokaryosis were mild. Bielschowsky and Luxol fast blue histochemical stains failed to demonstrate any axons or myelin within whorls, while preexisting peripheral nerves in the sections (internal control) stained properly. Sections were immunohistochemically stained with vimentin, a cytokeratins AE1 and AE3, a glial fibrillar acid protein (GFAP), a S100, a and neurofilament a (Table 1). All neoplastic cells and extracellular matrix in the center of the whorls were vimentin positive and presented scattered positivity for S100 (Fig. 2). The neoplastic tissue failed to stain with cytokeratin, GFAP, and neurofilament. Formalin-fixed saved tissue was postfixed with 2.5% glutaraldehyde (pH 7.2) for 12 hr and processed for transmission electron microscopy (TEM). TEM revealed that the mass was composed of aggregated clusters of swollen and stellate meningothelial neoplastic cells with oval nuclei, moderate numbers of cytoplasmic microfilaments, and cellular membranes presenting remarkably complicated interdigitating processes, desmosomes, and hemidesmosomes creating a jigsaw pattern diagnostic of meningioma (Fig. 3).
Antibodies and immunohistochemical staining procedures.*

GFAP = glial fibrillary acid protein. Sources: aDako North America Inc., Carpinteria, CA; bThermo Fisher Scientific Inc., Pittsburg, PA.
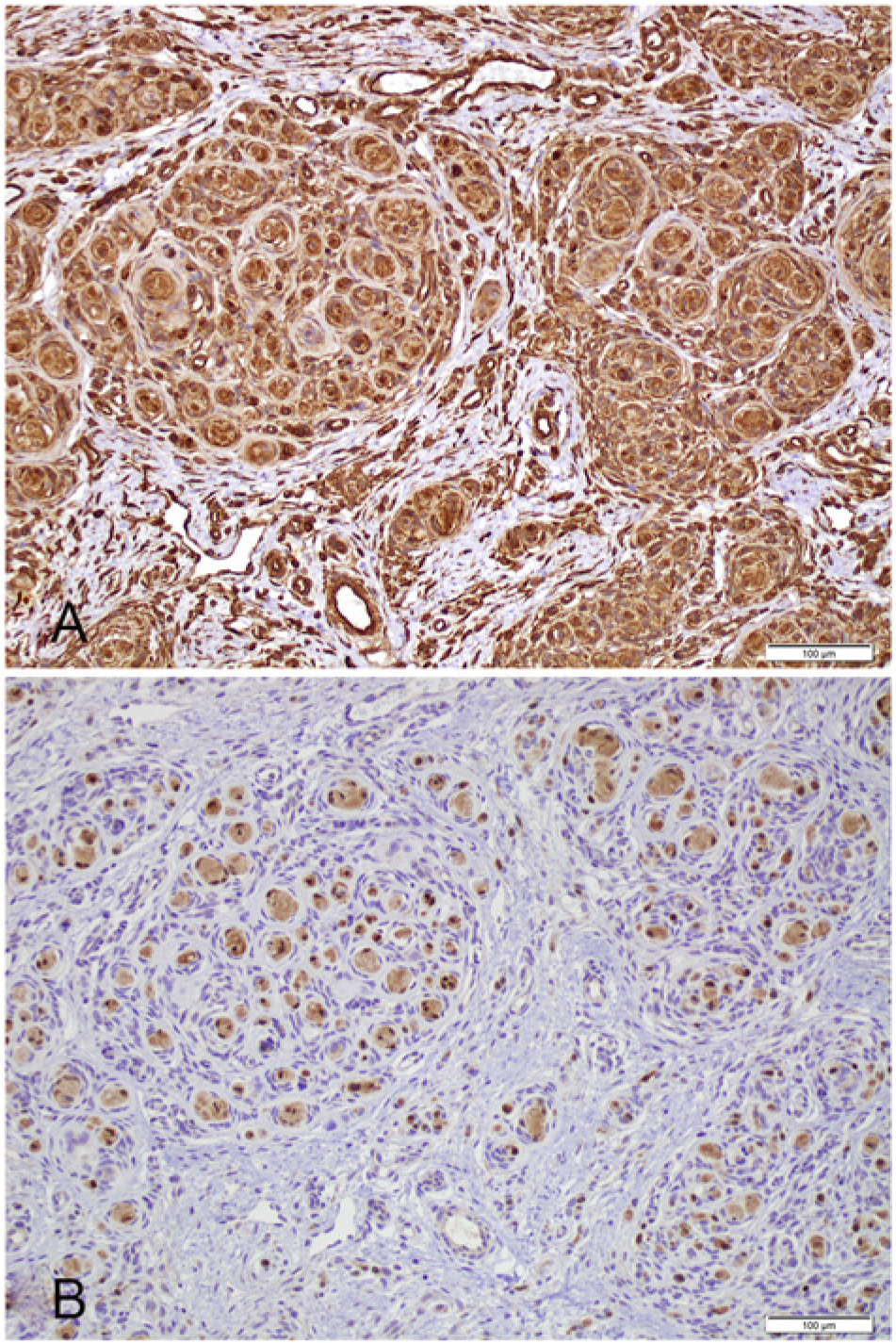
Neoplastic cells were vimentin positive (
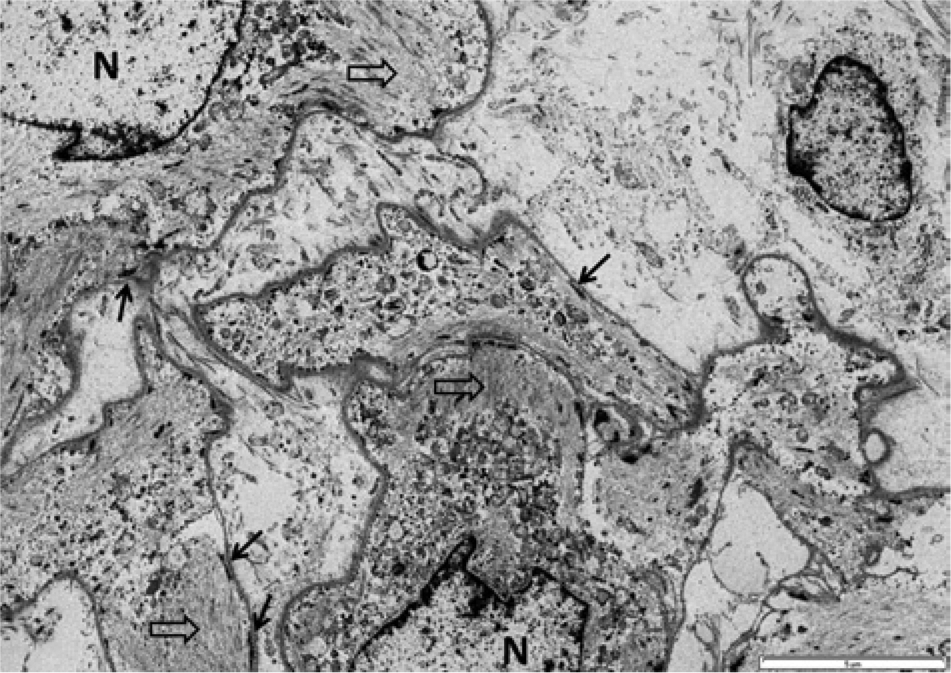
Transmission electron microscopy. Note intimately associated swollen and stellate meningothelial neoplastic cells with oval nuclei (N), cytoplasmic microfilaments (open arrows), and cellular membranes presenting remarkably complicated interdigitating processes and eventual desmosomes and hemidesmosomes (arrows) creating a jigsaw pattern diagnostic of meningioma. Bar = 5 μm.
The second case, a 10-year-old female spayed Maltese–Poodle mix dog, presented to a veterinary ophthalmologist with a 2-year history of 3 firm subcutaneous periocular nodules affecting the upper eyelid of the right globe. All masses were well delineated, firm, nonulcerated, and were located near the lateral canthus, mid-lateral eyelid, and central eyelid, measuring 0.9 × 0.7 × 0.5 cm, 0.4 × 0.3 × 0.4 cm, and 0.5 × 0.5 × 0.4 cm, respectively. The patient was otherwise healthy. The masses were surgically removed, fixed in formalin, and submitted to the Comparative Ocular Pathology Laboratory of Wisconsin (Madison, Wisconsin). At cut sections, the fixed specimens were firm and gritty and presented a white-to-tan irregular cut surface. Samples were submitted for routine histologic processing and stained with HE, Masson trichrome, and alcian blue–periodic acid–Schiff. Histologically, the 3 masses presented identical features and were composed of a highly cellular, poorly delineated, and infiltrative neoplastic tissue expanding and replacing the subcutaneous tissue and deep substantia propria and dermis of the eyelid (Fig. 1B). The tissue presented epithelioid neoplastic cells mainly arranged in discrete concentric whorls that coalesced and were connected by thin bundles of spindle-shaped cells embedded in a loose alcian blue–positive and Masson trichrome–negative extracellular matrix. Multifocally, epithelioid neoplastic cells concentrically surrounded multilaminated basophilic mineral concretions (psammoma bodies; Fig. 1D) and surrounded and compressed peripheral nerves in the deep dermis. These cells presented indistinct borders, small amounts of fibrillar eosinophilic cytoplasm, and an oval nucleus with finely stippled chromatin and indistinct nucleoli (Fig. 1F). Cellular pleomorphism was moderate, and there were 5 mitotic figures per ten 400× fields. Neoplastic cells were immunohistochemically positive for vimentin, regionally positive for S100, and negative for cytokeratins AE1 and AE3. Formalin-fixed tissue was also submitted for TEM, and neoplastic cells presented similar ultrastructural features as described for the first mass. The histologic, immunohistochemical, and ultrastructural characteristics of both tumors are consistent with a meningotheliomatous extracranial cutaneous meningioma.
Meningiomas are the most common mesodermal tumor of the central nervous system in dogs and present highly diverse morphologic patterns.7,11 According to the World Health Organization (WHO), meningiomas in domestic animals are classified into 9 different subsets: meningotheliomatous, fibroblastic, transitional, psammomatous, angiomatous, papillary, granular cell, myxoid, and anaplastic. 6 Canine meningiomas presenting features resembling the microcystic, gemistocytic-like, rhabdoid, and secretory human subtypes, as classified according to the WHO human classification of meningiomas, have been reported.10,15 Approximately 82% of all canine meningiomas are intracranial, 15% intraspinal, and 3% orbital. 7 Extracranial paranasal meningiomas have also been described. 12 Extracranial cutaneous meningiomas are extremely rare in dogs with only 2 reports in the literature describing 1 case each of a canine tumor affecting the subcutaneous tissue of the thoracic and pelvic limbs.5,18 Cutaneous meningiomas in human beings are classified into 3 types. 8 Type I is congenital, generally occurs in the scalp and paravertebral regions, and develops from ectopic arachnoid cells that become trapped in the dermis and subcutis during development. Type II tumors occur in adults and involve sensory organs of the head, including periocular, aural, and paranasal regions and cranial nerves, and are presumed to derive from nests of arachnoid cells found along the course of nerves supplying these regions that are displaced after the nerves penetrate the dura. Type III tumors occur after direct extension or metastasis from intracranial neoplasms.
Based on magnetic resonance imaging of the skull, none of the animals in the current report presented a primary intracranial tumor, thus ruling out an extracranial extension of an intracranial meningioma. The first case presents marked similarities to human cutaneous meningiomas type I (congenital). The tumor affected a 5-week-old animal, was present at birth, and was located at the upper eyelid near the medial canthus. Based on the pathophysiology of the human tumor, it can be extrapolated that this tumor most likely originated from displaced arachnoid cells during embryogenesis, explaining its early onset. Also, the medial canthus is located adjacent to multiple cranial bone sutures (i.e., nasomaxillary, frontomaxillary, lacrimomaxillary, and zygomaticomaxillary sutures 3 ) furthering even more the possibility of subcutaneous displacement of meningothelial cells. In the second case, the tumors were also in the eyelid, but in contrast to the first case, affected an adult dog. The clinical presentation and the fact that the tumor encroached multiple peripheral nerves in the dermis and substantia propria of the eyelid suggests a similarity to human cutaneous meningiomas type II (acquired ectopic) and the possibility that it originated from meningothelial cells tracking these nerves.
The histologic pattern of discrete nodules of spindle-to-epithelioid cells arranged in whorls and short fascicles present in each case is consistent with, but not specific of, meningiomas. Associate that with the atypical location of these tumors and a diagnosis solely based on morphology becomes very challenging. Possible differential diagnoses for the present cases are peripheral nerve sheath tumors, hemangiopericytomas, and anaplastic carcinomas.
The absence of immunohistochemical reaction for cytokeratin AE1/AE3, as broad spectrum pancytokeratin that stains most epithelial tissues, ruled out tumors of epithelial origin. The absence of axons and myelin in the center of the cellular whorls under Bielschowsky and Luxol fast blue stains, the immunohistochemical negativity for GFAP and neurofilament, and the weak and focal reactivity to S100 rendered the diagnosis of peripheral nerve sheath tumor unlikely. Meningiomas do not present a specific immunohistochemical staining pattern but canine tumors are usually vimentin and S100 positive, GFAP negative, and stain variably with pancytokeratins.1,4,10,13 The immunohistochemical staining pattern of tumors presented herein (vimentin and focal S100 positivity and pancytokeratin and GFAP negativity) are consistent with meningiomas. Epithelial membrane antigen has been found very useful in distinguishing human meningiomas from other spindle cell tumors, but no reagents are available for routine veterinary samples. 4 Cluster of differentiation (CD)-34 and E-cadherin have been pointed out as useful antibodies on the diagnosis of meningiomas, 13 but because of lack of availability, tumors were not stained with these antibodies. Nevertheless, the definitive diagnosis of cutaneous meningioma was based on the ultrastructural features of the tumor. The presence of convoluted interdigitating cellular processes with multiple desmosomes, hemidesmosomes, and abundant cytoplasmic microfilaments are diagnostic features of meningiomas.5,7,18
In conclusion, 2 cases of periocular cutaneous meningiomas in dogs are described in the current study. Both cases presented a meningotheliomatous histologic pattern and immunohistochemical and ultrastructural features similar to intracranial meningiomas. One tumor was congenital and presented clinicopathological similarities to human type I (congenital) cutaneous meningioma. The other occurred in an adult dog and was similar to human cutaneous meningioma type II. Although rare, cutaneous meningiomas should be kept as a differential diagnosis for periocular epithelioid tumors.
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
Thermo Fisher Scientific Inc., Pittsburg, PA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
