Abstract
Background
Accurate staging of superficial esophageal squamous cell cancer (ESCC) for endoscopic therapy is challenging. Optical coherence tomography (OCT) has been shown to be superior to high-resolution endoscopic ultrasound (EUS). Volumetric Laser Endomicroscopy (VLE), a second-generation OCT, has recently become commercially available.
Objective
To assess if VLE can determine which patients with superficial ESSC can undergo endoscopic therapy.
Methods
This is a multi-center retrospective study. Patients were included if (a) they had visible ESCC, (b) they underwent VLE and EUS for staging, and c) if superficial disease was suspected then endoscopic resection had to be performed to have accurate histology to compare the VLE scan to. VLE scans were then compared to the gold standard: histology for superficial disease and EUS for disease T1b and greater.
Results
Seventeen patients were included with the following disease: squamous intraepithelial neoplasia (4 patients), T1a disease (6 patients), T1b (2 patients), T2 disease (2 patients) and T3 disease (3 patients). VLE was able to distinguish superficial disease, defined as disease limited up to the lamina propria, from non-superficial disease in all cases.
Conclusions
VLE may be able to determine which ESCC patients are candidates for endoscopic therapy. Prospective studies are needed to confirm this.
Key points
Volumetric laser endomicroscopy (VLE) is a balloon-based second-generation optical coherence tomography technology that provides high-resolution esophageal imaging. Determining the depth of involvement in superficial squamous esophageal cancer is difficult but necessary when selecting patients for endoscopic therapy. In this study of 17 patients, VLE was able to distinguish superficial disease from non-superficial disease in all cases. This has implications for selecting candidates for endoscopic therapy.
Introduction
Eighty percent of esophageal cancers occur in the non-western world; and ninety percent of these are esophageal squamous cell carcinoma (ESCC). 1 The precursor to ESCC is squamous intraepithelial neoplasia (SIN). 2 Endoscopic therapy is accepted for ESCC and SIN if the lesions are limited to the epithelium or lamina propria.2,3 Surgery with or without radiation/chemotherapy is the accepted standard if these lesions involve the deep submucosa. Lesions limited to the mucscularis mucosa or superficial submucosa are considered a gray zone for endoscopic treatment.2,3 These standards of therapy are based on the risk of lymph node metastasis (LNM).4,5
Recognized endoscopic therapies include radiofrequency ablation (RFA), and resection by endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD).2,6,7 RFA has the drawback of not obtaining histology resulting in a lack of awareness of histologic features that may predict LNM. 3 Therefore, RFA is most efficacious in patients that are low-risk for LNM; when disease is confined to the lamina propria 3 . ESD may be considered for cases of deep mucosal and early submucosal involvement, when patients are not suitable candidates for surgery and when the risk of LNM is considered low.
Determining which patients have ESCC limited to the lamina propria can be challenging. In a recent retrospective study, specimens from patients that underwent ESD for flat ESCC, were examined. 3 The study showed that one-third of patients that met clinical and endoscopic criteria for RFA had histologic criteria that were considered contraindications for RFA. The study concluded that determining which patients should receive RFA therapy is challenging. The challenge stems from inaccurate pre-therapy staging of ESCC. 8 High-frequency endoscopic ultrasound (HF-EUS) and EUS are limited in imaging superficial ESCC. 8 However, studies have shown that optical coherence tomography (OCT) can produce high-resolution images of staging ESCC. A prospective study compared OCT to HF-EUS in 123 patients with superficial ESCC and found that OCT had a higher accuracy (95% vs 81%; p < 0.05) 8 . First-generation OCT, used in that study, is not commercially available.
Recently, second-generation OCT, termed volumetric laser endomicroscopy (VLE), has been developed and is commercially available in the United States of America. VLE is a second-generation OCT technology. It is balloon-based, with the probe within a balloon that allows for centering within the esophagus. It differs from previous versions of OCT in this regard, where previous versions only used a probe. It uses infrared light to produce real time high-resolution cross-sectional imaging of the tubular esophagus. VLE can scan a 6 cm length of the esophagus in approximately 90 s, providing surface and subsurface wide-field cross-sectional imaging with an axial resolution of 7 µm to a depth of 3 mm.9,10 It has mainly been used for dysplasia detection in Barrett’s esophagus.9,11,12 At present, literature on VLE use for ESCC is limited. 13 The aim of our study was to examine the potential role of VLE for staging various SIN and ESCC lesions to determine its appropriateness for endoscopic therapy.
Methods
This is a multi-center retrospective study from four academic tertiary care centers in the USA (Long Island Jewish Medical Center, New Hyde Park, NY; Columbia University Medical Center, New York, NY; Mayo Clinic Arizona, Scottsdale, AZ; and Weill Cornell Medical College, New York, NY) from January 2014 to November 2017. Institutional review board approval was obtained from all centers from 13 June 2014 to 5 September 2017. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution’s human research committee.
Patients were included if they (a) had visible ESCC or SIN, (b) underwent VLE of the visible abnormalities and (c) had EUS for staging. If superficial disease was suspected/present (defined as limited up to the lamina propria) then endoscopic resection had to be performed to have an accurate histologic stage to compare the VLE scan to. This was decided given the lower accuracy of EUS for staging of superficial disease. 8 EMR and ESD histology were considered the gold standard for superficial ESCC or SIN staging. EUS was the gold standard for staging of invasive ESCC staged T1b or greater, as this has been shown to be accurate and has less inter-observer variability for invasive disease.8,14 Staging was performed using standard guidelines. 15 Non-visible lesions were not included as the VLE images are more challenging to correlate to endoscopy.
VLE (NvisionVLE, Ninepoint Medical, Bedford, MA, USA) was performed in the usual manner described in the literature
9
by experienced users in VLE (AJT, AS, NF and MK). VLE is FDA cleared for use in the gastrointestinal (GI) tract but is marketed mainly for use in the esophagus. The lesions in this study were endoscopically evident as there was a sharp contrast between normal esophageal mucosa and diseased mucosa. All patients were referred for endoscopic staging prior to deciding treatment. All patients underwent high-definition white light endoscopy with Lugol’s chromoendoscopy. VLE was performed prior to Lugol’s staining. Locating these lesions on VLE was straightforward as there was a sharp contrast between normal tissue and lesions with ESCC or SIN. All the lesions were surrounded by normal esophageal tissue both on high-definition white light endoscopy with Lugol’s chromoendoscopy and VLE. Although VLE with laser marking was available for over half of these cases, it was not performed as the lesions in question were evident on endoscopy. In addition to the sharp contrast seen of normal esophagus versus abnormal ESCC or SIN (both on endoscopy and VLE), the location of the lesions on VLE compared to endoscopy were confirmed by using the registration lines on the VLE balloon and images. Figure 1 depicts normal esophageal layers on VLE. Figures 2 and 3 depict images with superficial and invasive disease, respectively. All procedures utilized a 20 mm balloon.
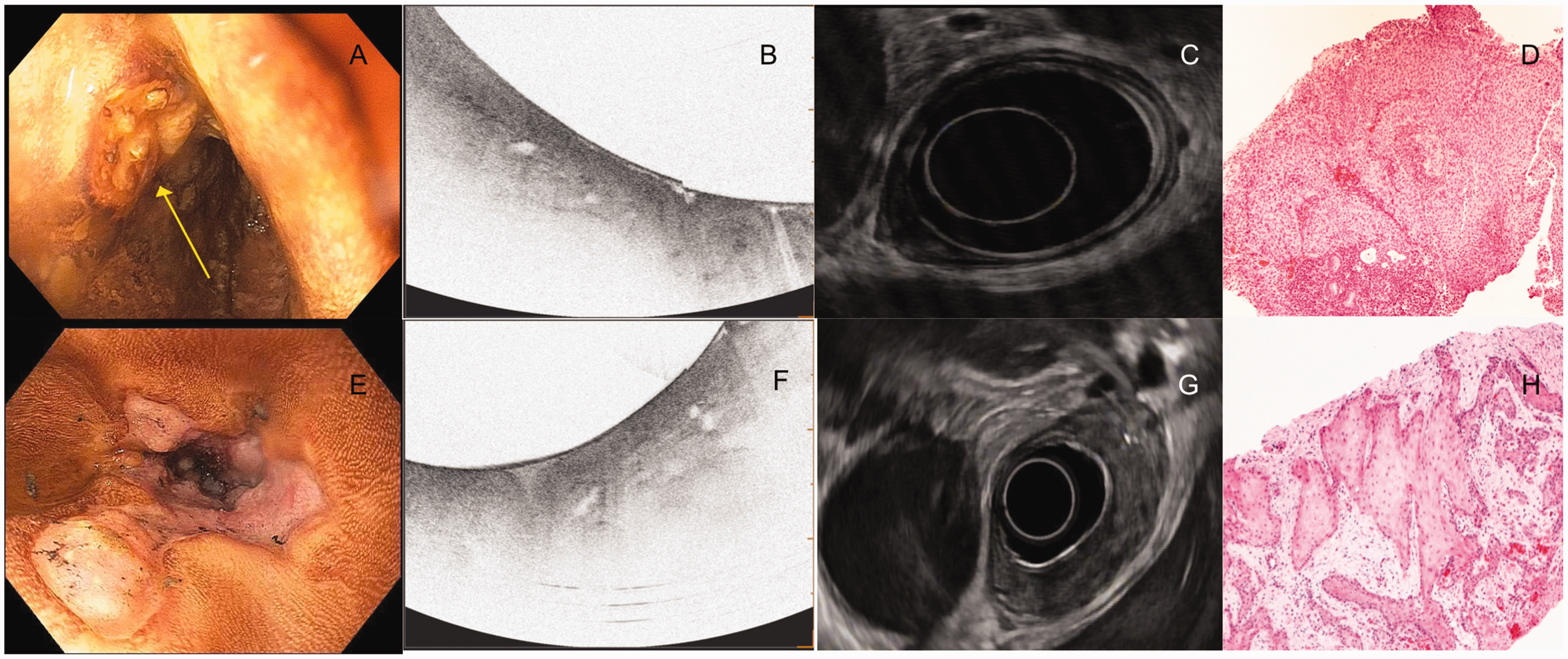
An example of normal esophageal layers and corresponding histology (not from the same patient). Image courtesy of Ninepoint Medical. Images from patients with T1a squamous cancer. The first column from (a)–(c) shows the endoscopic, VLE and histology images of a patient with high-grade squamous intraepithelial neoplasia. The yellow arrow points to the lesion on endoscopy (a). The VLE image (b) shows surface hyperreflectivity constrained to the epithelium (red arrows) with preservation of layers below. The histology (c) shows disease limited to the to the epithelium. The second column from (d)–(f) shows the endoscopy with Lugol’s chromoendoscopy with surrounding cautery marks outlining the lesion for resection (d), VLE and histology images of a patient with squamous cell cancer invading the lamina propria. The VLE image (e) shows surface hyperreflectivity (red arrows) extending to the lamina propria but preservation of the layers below it. The corresponding histology shows disease limited to the lamina propria. The third column from (g)–(i) shows the endoscopy with Lugol’s chromoendoscopy with surrounding cautery marks outlining the lesion for resection (g), VLE and histology images of a patient with squamous cell cancer invading the muscularis mucosae. The VLE image shows diffuse surface hyperreflectivity and effacement of all layered architecture below it. The corresponding histology shows disease limited to the muscularis mucosae. Images from patients with T2 and T3 invasive squamous cell cancer. The first column from (a)–(d) shows the endoscopy with Lugol’s chromoendoscopy, VLE, EUS and histology images from a pinch biopsy from a patient with T2 ESCC. The VLE image (b) shows surface hyperreflectivity and effacement of all layered architecture. The corresponding EUS shows a hypoechoic lesion involving the muscularis propria but not invading beyond it. The second column (e)–(h) shows the endoscopy with Lugol’s chromoendoscopy, VLE, EUS and histology of a pinch biopsy from a patient with T3 ESCC. The VLE image (f) shows surface hyperreflectivity and effacement of all layered architecture. The corresponding EUS shows a hypoechoic lesion invading beyond the muscularis propria.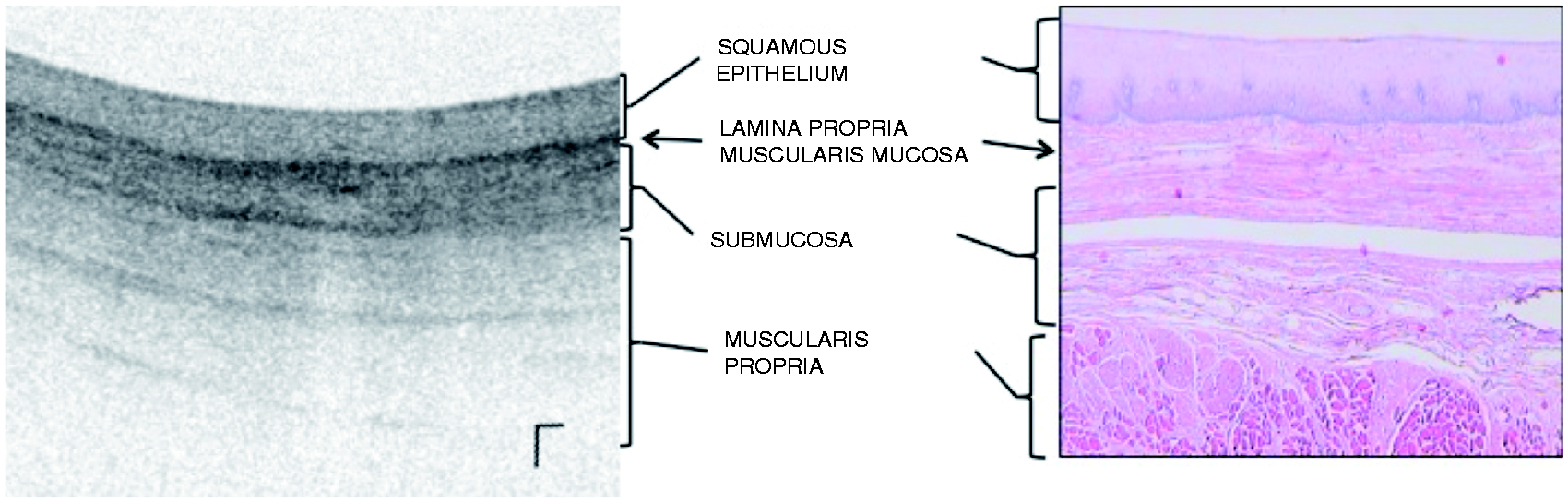
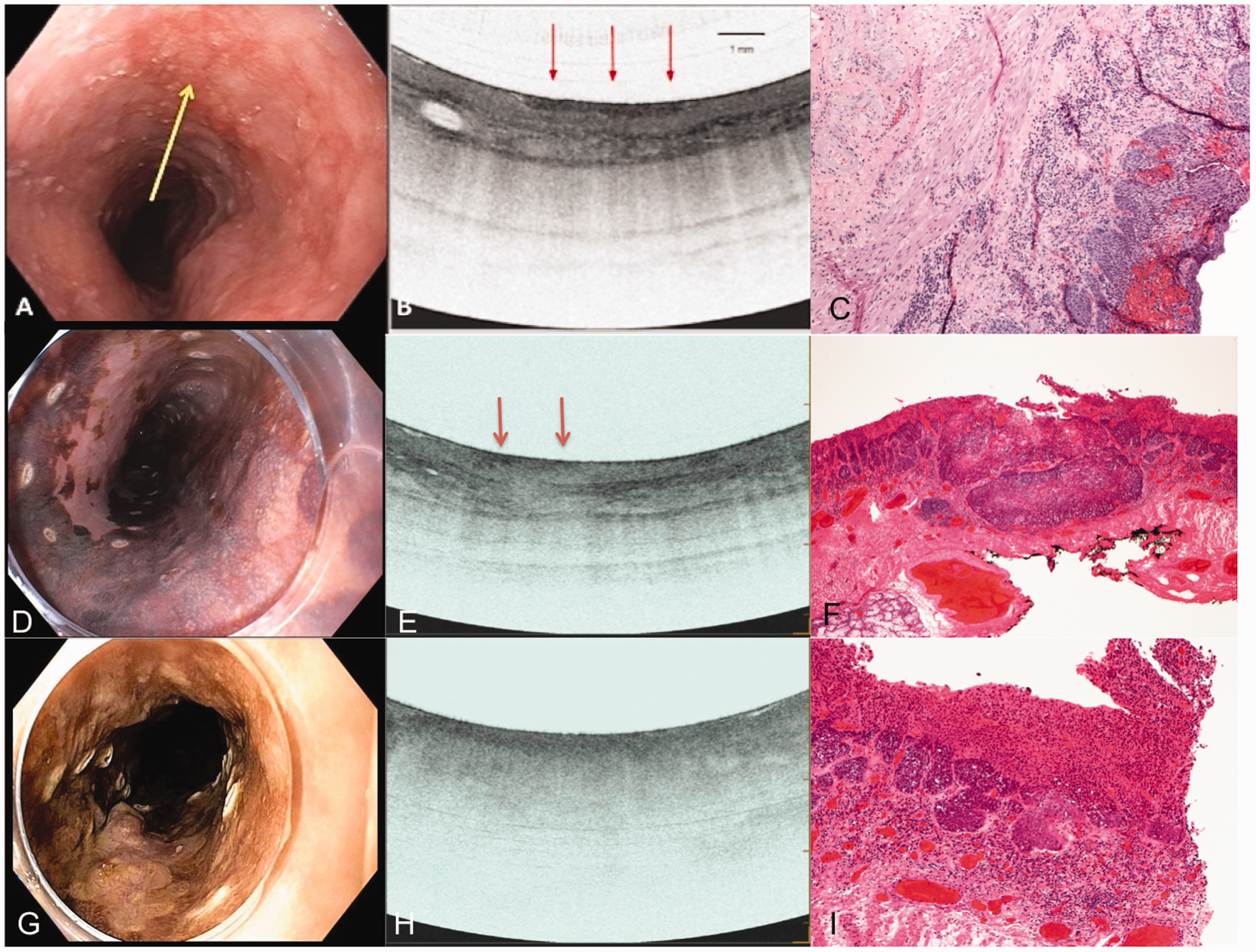
This study incorporated previously recognized OCT criteria to stage SIN and ESCC (Hatta criteria based on probe-based OCT). 16 This staging is based on OCT signal penetration into a layer with preservation of the layers below it. In our experience, we have noticed that this staging is similar for VLE with SIN disease; a surface hyperreflective (darker) signal from the neoplasm is visualized only involving the epithelium with preservation of the layers below it. However, with disease involving deeper layers, a surface hyperreflectivity is seen with effacement (loss of the traditional layering) below the darkened surface. The extent or depth of disease involvement is determined by which layers are preserved below it. This is different from OCT staging without a balloon, as the balloon compresses the mucosa and creates this effect. Another difference is that the Hatta criteria define hyperreflective surface signals as white and hyporeflective signals as black due to how they process the image. 16 The VLE literature describes darker surface signals as hyperreflective,9,10,17 and thus to be consistent with the VLE literature, we did the same. After the VLE was performed, the endoscopist interpreted the VLE images and the findings were recorded in the endoscopy report. The endoscopist had no knowledge of the histology findings from EMR/ESD specimens. For the purposes of this study, VLE images were re-reviewed by another experienced VLE user (SI), to ensure reliability of the VLE findings among all sites. This user was blinded to the histology and VLE findings during his review.
If lesions were suspected of being superficial disease, then EMR/ESD was performed of the lesion as it provides histopathology staging and information regarding lymphovascular invasion, which is considered important for determining the depth of invasion, accurate staging and prognosis.18,19 The histology images were interpreted by an expert GI pathologist and confirmed by a second GI pathologist. The pathology findings were read independent of the EUS and VLE findings and were read after the endoscopy was performed.
Endoscopic ultrasound of invasive disease was performed by the same user using a radial echoendoscope with an advanced processor (GF-UE160-AL5 and Prosound F75, Olympus America, Center Valley, PA, USA) using established EUS staging criteria for ESCC. 14 EUS findings were recorded on the procedure report. VLE scans were then compared to the gold standard to determine if a VLE pattern correlated to the histologic stage SIN or ESCC.
Results
Volumetric laser endomicroscopy and endoscopic ultrasound features of patients included in this study.
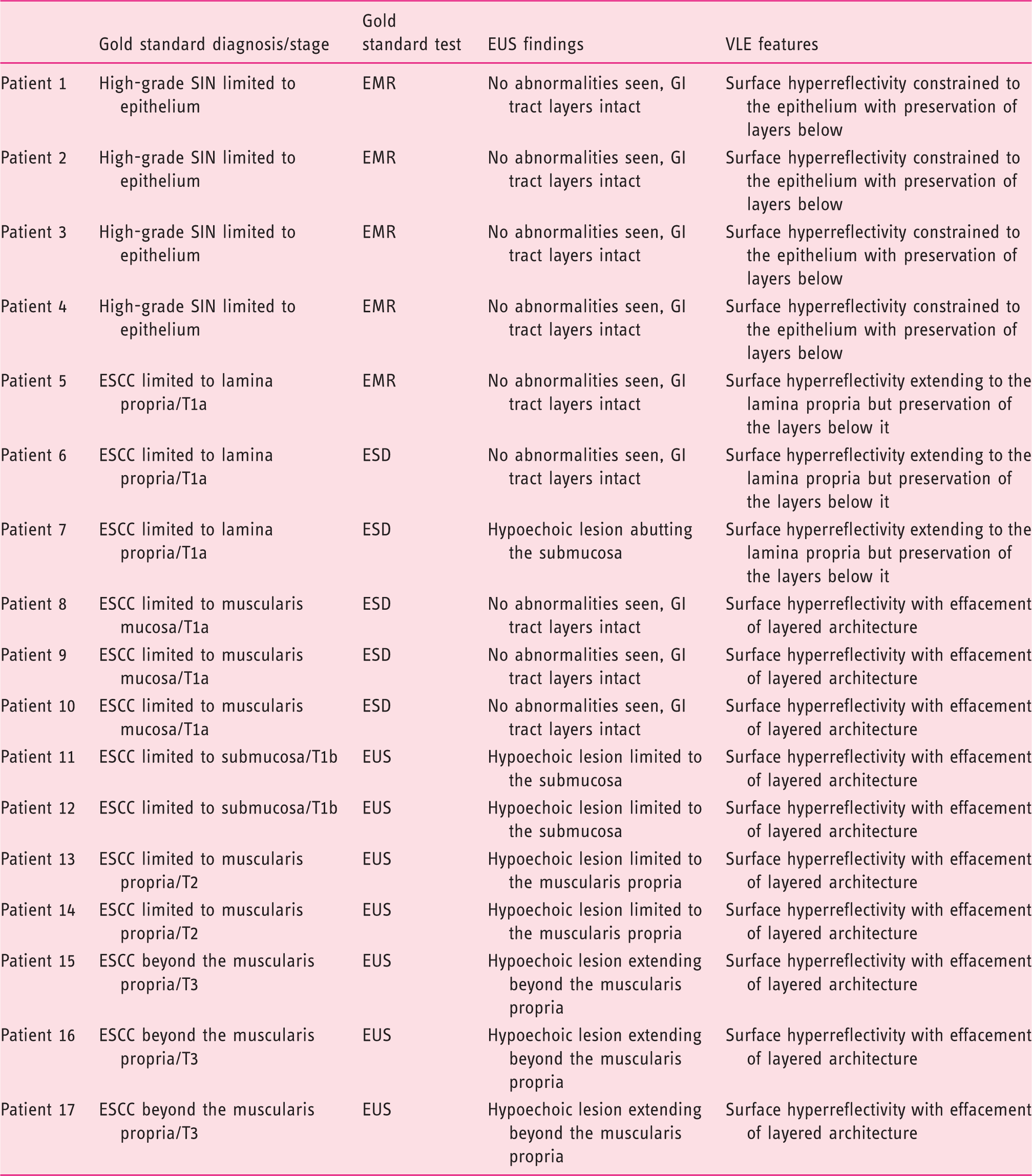
EMR: endoscopic mucosal resection; ESCC: esophageal squamous cell carcinoma; ESD: endoscopic submucosal dissection; EUS: endoscopic ultrasound; GI: gastrointestinal; SIN: squamous intraepithelial neoplasia; VLE: volumetric laser endomicroscopy.
With regard to the VLE findings, patients 1–4 had a surface hyperreflectivity in the epithelium with preservation of the layers below it (Figure 2). For patients 5–7, there was surface hyperreflectivity extending to the lamina propria but with preservation of the layers below it; the submucosa and layers below were intact (Figure 2). For patients 8–17, surface hyperreflectivity was seen with effacement of all layered architecture below it (Figures 2 and 3).
VLE was able to distinguish superficial disease (disease limited to the lamina propria) from non-superficial or invasive disease in all 17 cases. In invasive disease, defined as disease extending beyond the lamina propria, VLE had a distinct signature of surface hyperreflectivity with effacement of all layered architecture. With regard to tumor (T) staging, VLE could not provide a specific T stage in non-superficial or invasive disease. VLE was able to provide a T stage in cases of superficial cancer. EUS was only able to stage disease that was T1b or higher (Table 1). In all 17 cases, VLE was able to determine which patients were candidates for endoscopic therapy by radiofrequency ablation compared to the gold standard.
Discussion
This study demonstrates that VLE may accurately determine which patients with SIN and superficial ESCC should undergo endoscopic therapy. RFA is indicted for disease limited to the lamina propria and endoscopic resection for deep mucosal disease. In this study, VLE had a specific signature for SIN, ESCC limited to the lamina propria and ESCC invading beyond the lamina propria. Thus, VLE is able to distinguish superficial from non-superficial disease in this case series. As a result, VLE and EUS are complementary. This data suggests that patients undergoing pre-therapy staging for ESCC should undergo an EUS. If the EUS is inconclusive due to superficial disease, then a VLE can define the stage to determine if endoscopic therapy is possible.
VLE can help define candidates for RFA or endoscopic resection. For example, VLE staged superficial disease in patients 1–7, who were candidates for RFA. The later patients (patients 8–17) were not candidates for RFA, as VLE staged more invasive disease. Disease limited to the muscularis mucosa (patients 8–10) is a gray zone for endoscopic treatment. 2 Although RFA cannot treat this, ESD is an option. Patients 8–10 had disease limited to the muscularis mucosa and underwent ESD that was curative. The EUS in these patients was non-diagnostic but the VLE was consistent with disease beyond the lamina propria. A VLE showing non-superficial disease with a negative EUS (not showing T1b disease or greater) indicated disease limited to the muscularis mucosa and thus indicated candidates for ESD. None of the patients underwent RFA in this study as EMR/ESD was performed for staging purposes. However, RFA could be useful in multi-focal disease or when EMR/ESD were not available.
It should also be noted that our VLE criteria are different from the original Hatta criteria, given the balloon-based nature of the VLE system. The Hatta group utilized an OCT probe that images (at a depth of 1.5 mm and an axial resolution of 11 µm) the tissue in contact with the probe. VLE utilizes a rotating probe within a balloon (that images at a depth of 3 mm and an axial resolution of 7 µm) that performs 360° wide-field imaging of a 6 cm segment of esophagus in 90 s. The balloon helps center the probe within the esophagus for better imaging. It is likely that the inflated balloon exerts a tension on the mucosa that causes effacement of layers, which is not seen in probe-based OCT.
Our study does have limitations. The relatively small cohort and retrospective observational study design limit this study. However, the observations noted in this study are very compelling for a role of VLE in the staging of superficial esophageal ESCC. ESCC is not common in the USA, and thus studying VLE for staging ESCC is difficult. On the other hand, VLE is not available in areas with a high prevalence of ESCC. Thus, this series provides a unique perspective on staging superficial ESCC that will be hard to replicate. We were fortunate to have a cohort with diverse stages from SIN, T1a, T1b, T2 and T3 disease, and thus this cohort does allow insight into VLE staging of ESCC.
Although this study is promising for the use of VLE in the staging of superficial ESCC, future studies are needed. Such studies need to examine the degree of inflation of the balloon and the psi of the balloon in relation to effacement of the VLE image. In addition, a larger prospective study will be needed. It will likely require a site outside the USA, given the rarity of superficial ESCC in the USA. In conclusion, this study shows that VLE may be useful to stage superficial ESCC. VLE may be able to decide which patients are suitable candidates for endoscopic therapy.
Footnotes
Acknowledgements
The authors wish to thank Dan Aupi for his assistance during volumetric laser endomicroscopy image collection. In addition, we thank Dr Calvin Lee for his assistance with manuscript preparation. Author contributions: conception and design (AJT); analysis and interpretation of the data (AJT, PCB and DVS); drafting of the article (AJT, PCB, SI and AR); critical revision of the article for important intellectual content (AJT, PCB, SI, DVS, CF, AR, AS, MK, IA, NF and AK); and final approval of the article (AJT, PCB, SI, DVS, CF, AR, AS, MK, IA, NF, AK).
Declaration of conflicting Interests
None
Funding
None
Ethics approval
Institutional review board approval was obtained from all centers. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institutions' human research committees.
Informed consent
Not required for this article as it is a retrospective study.
