Abstract
Objectives
Multiple endoscopic sessions may be necessary for treatment and surveillance of Barrett’s esophagus (BE)-associated neoplasia. Adherence to an endoscopic therapeutic regimen is important for longitudinal management of BE. The objective of this study was to identify the factors associated with adherence to therapy for BE-associated neoplasia.
Methods
We retrospectively identified patients with BE whom were referred to a tertiary center for endoscopic mucosal resection (EMR) or radiofrequency ablation (RFA) between 2009 and 2012. Demographic and clinical data were extracted from the medical record.
Results
We had 69 subjects meet our inclusion criteria. Referral diagnosis was low-grade dysplasia in 9 (13%) subjects, high-grade dysplasia in 33 (48%) subjects and adenocarcinoma in 26 (38%) subjects. The majority (55%) lived more than 100 miles from the treatment center. The primary third-party payer was US Medicare for 54% of the subjects and private insurance for 36% of them; 45% of the subjects were seen in the clinic by the treating endoscopist, prior to endoscopic therapy and 71% underwent EMR as the initial treatment, while 29% underwent RFA without prior EMR. We found that 72% of subjects were adherent to therapy, including: 23 (33%) completing endoscopic therapy with documented post-treatment surveillance, 18 (26%) with ongoing endoscopic therapy, and 9 (13%) whom underwent esophagectomy. Subjects seen in gastroenterology clinical consultation were significantly more likely to demonstrate adherence than those referred for open access endoscopy (Lasso OR 2.31).
Conclusions
Patients seen in a clinical consultation prior to endoscopic therapy for BE-associated neoplasia were more likely to demonstrate treatment adherence, compared to patients referred for open-access endoscopy. A clinic visit prior to therapy may define expectations regarding treatment course and increase the likelihood of patient adherence.
Keywords
Introduction
Endoscopic therapy is a first-line therapeutic option for treatment of Barrett’s esophagus (BE) and intraepithelial neoplasia. Professional practice guidelines support the use of endoscopic treatment options, including endoscopic mucosal resection (EMR) and radiofrequency ablation (RFA), for treatment of BE containing high-grade dysplasia (HGD) 1 ; and a recent international consensus statement endorsed endoscopic therapy over surgery for most patients with BE containing HGD. 2 Controlled prospective trials demonstrate the ability of both photodynamic therapy (PDT) and RFA to offer remission of dysplasia in a majority of treated patients and to reduce the rate of progression to cancer, during reported follow-up3,4; however, endoscopic therapy may require multiple treatment sessions over a number of months to achieve the desired therapeutic outcome (typically, eradication of all intestinal metaplasia). For instance, in a randomized prospective trial, subjects with BE dysplasia could receive up to four RFA treatments within the first year of the study protocol; while the majority of patients (65/119 (55%), including subjects initially treated with sham who elected a crossover to the RFA arm) received further RFA after the 1-year primary endpoint. 5
Endoscopic factors which predict response to therapy have been identified. Persistence of acid reflux, despite pharmacologic gastric acid suppression; hiatal hernia size; and initial BE segment length are associated with an incomplete response to RFA. 6 Patients with long segments of columnar epithelium and large hiatal hernias may therefore require an increased number of RFA treatment sessions to achieve desired outcomes. 7
Moreover, successful endoscopic therapy, with endoscopic and histopathologic eradication of dysplasia and/or intestinal metaplasia, does not alter predisposition to gastroesophageal reflux and does not eliminate the need for continued endoscopic surveillance. Recurrence of BE has been reported in extended follow-up after RFA.8,9 Such data led to the concept of endoscopic therapy offering ‘remission’, rather than ‘cure’, of BE. 10
In context, therefore, the decision to embark upon endoscopic therapy for BE-associated neoplasia is a process which requires commitment by both patient and physician. In studies assessing adherence to therapy in chronic disease management, there has been a link with low household income, insurance status, and higher coinsurance or copayments to prescription drug noncompliance11,12; and increases in prescription coverage and lower copayments have been associated with higher medication adherence rates for chronic diseases, including diabetes, hypertension, hyperlipidemia and congestive heart failure.13,14 Whether patient factors influence adherence to longitudinal management of BE is uncertain. The aim of this study was to identify the factors associated with adherence to therapy for BE-associated neoplasia.
Methods
Approval to conduct this retrospective study was granted by Vanderbilt University’s Institutional Review Board (IRB). We performed a database query to identify patients with BE referred to the Vanderbilt Barrett’s Esophagus Endoscopic Treatment Program (V-BEET), whom underwent EMR and/or RFA between September 2009 and November 2012. Study data were collected and managed using the Research Electronic Data Capture (REDCap) tools hosted at Vanderbilt University. REDCap is a secure, web-based application designed to support data capture for research studies. 15
Demographic and clinical data were extracted from the electronic medical record. Demographic data included age, gender, marital status, third-party insurance payor, and driving distance between the patient’s primary residence and the medical center. Clinical data included history of current or past tobacco and/or alcohol use, family history of gastrointestinal malignancy, and whether an outpatient clinic consultation with the treating endoscopist was completed prior to initiation of endoscopic therapy. Endoscopic data included BE segment length, as well as number and sequence of endoscopic procedures with modality of therapeutic intervention (EMR versus RFA).
Patients could be referred for EMR/RFA following an office consultation or a diagnostic endoscopic procedure, performed by another gastroenterologist or a thoracic surgeon within our institution, or by another gastroenterologist outside our institution. For cases in which the initial diagnostic endoscopy had been performed outside our institution, histopathology specimens were routinely requested for expert histopathological review at our institution. A clinic visit with the endoscopist (PY) performing EMR/RFA was suggested, but not required, prior to endoscopic therapy. After the EMR/RFA, a telephone follow-up was performed for all patients, with results of histopathology (in the cases of EMR) discussed with the patient at that time. Patients with BE neoplasia were routinely discussed in a multidisciplinary esophageal tumor board that included gastroenterologists, thoracic surgeons, medical oncologists and radiation oncologists, prior to commitment to a therapeutic course. Clinical follow-up after endoscopic therapy was not standardized.
For the purpose of the primary study outcome, adherence to therapy was defined as completion of endoscopic therapy with documented post-treatment endoscopic surveillance at our institution, ongoing endoscopic therapy at our institution, or surgical esophagectomy. Patients lost to local follow-up or otherwise without documented endoscopic or pathologic data within an appropriate post-treatment timeframe were considered non-adherent to therapy.
Statistical analysis was performed using the R statistics program. Univariate analysis was performed with testing of significance using the Chi square test, Wilcoxon rank sum test or Pearson test; for all analyses, a p-value <0.05 was considered significant. The number of potential predictors, relative to number of outcomes in our dataset, was large for a traditional multivariable logistic regression approach. Therefore, to control for confounding variables and generate odds ratio (OR) estimates, a generalized elastic net regression technique was employed, to construct a multivariable logistic regression model with the optimal number of predictors. 16 The elastic net allowed for fitting a multivariable model that included a penalty for the number of predictors in the model. A Lasso penalty with leave-one-out (LOO) cross-validation was used to estimate the penalty parameter and results were reported using the penalty that gave the best cross-validated fit (smallest binomial deviance).
Following the approach of Wu et al., 17 an OR was generated from a traditional multivariable logistic regression model, using the predictors selected by the elastic net. We considered 10 predictors for potential inclusion, and a model with only three predictors (family history of gastrointestinal malignancy, clinical visit with an endoscopist prior to treatment and first treatment RFA) was selected by having the best cross-validated fit. For these predictors, the ORs from unadjusted, adjusted and Lasso-penalized logistic regressions (Lasso OR) were presented. The LOO index, calculated using the likelihood ratio test to compare the model containing all predictors to a model with one predictor removed, described the significance of each predictor after controlling for the other predictors in the model. LOO indices are similar to p-values in that smaller values indicate stronger statistical significance.
Results
Demographic data
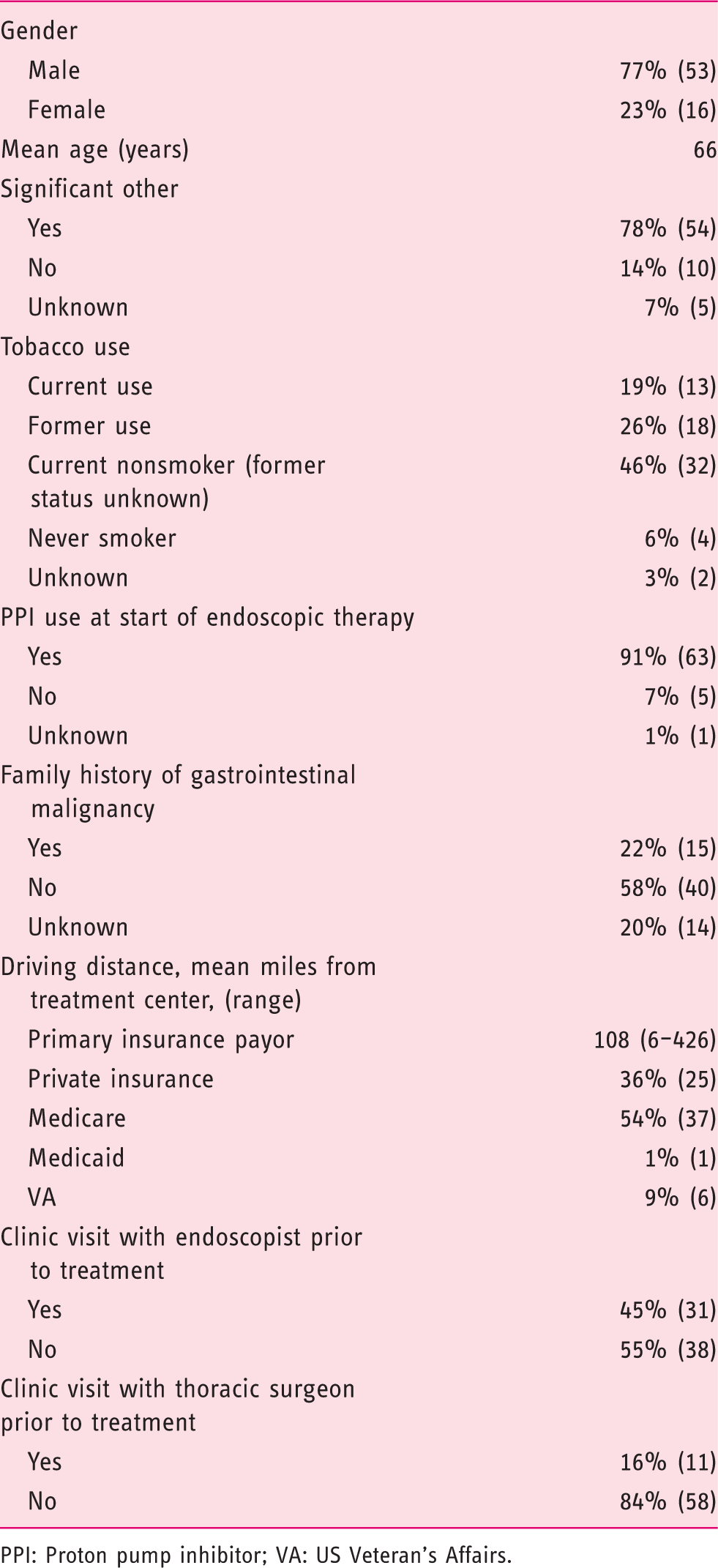
PPI: Proton pump inhibitor; VA: US Veteran’s Affairs.
Endoscopic data
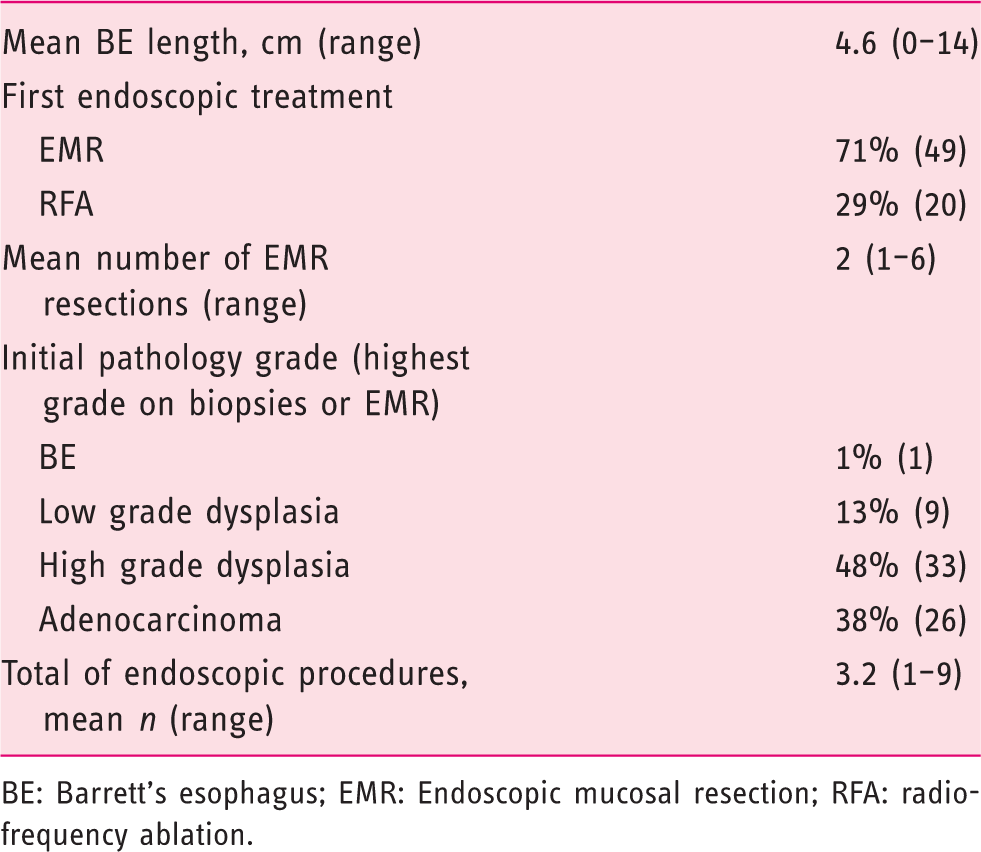
BE: Barrett’s esophagus; EMR: Endoscopic mucosal resection; RFA: radiofrequency ablation.
We found that 72% (50/69) of subjects were adherent to therapy, including 33% (23/69) whom completed endoscopic therapy with documented post-treatment surveillance, 26% (18/69) whom were enrolled in ongoing endoscopic therapy and 13% (9/69) whom underwent surgical esophagectomy.
Univariate analysis: Factors associated with adherence
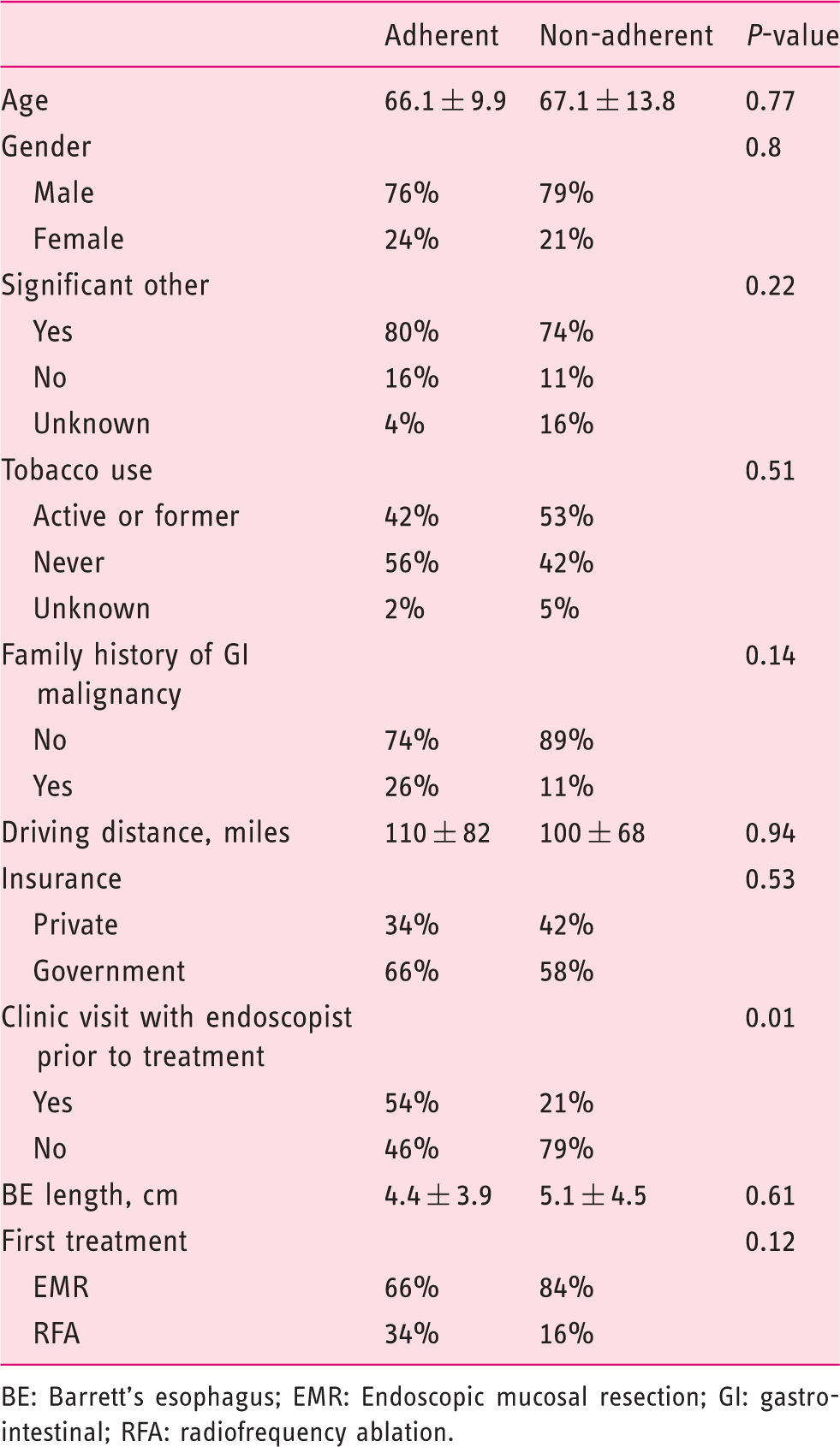
BE: Barrett’s esophagus; EMR: Endoscopic mucosal resection; GI: gastrointestinal; RFA: radiofrequency ablation.
Multivariable analysis: Factors associated with adherence
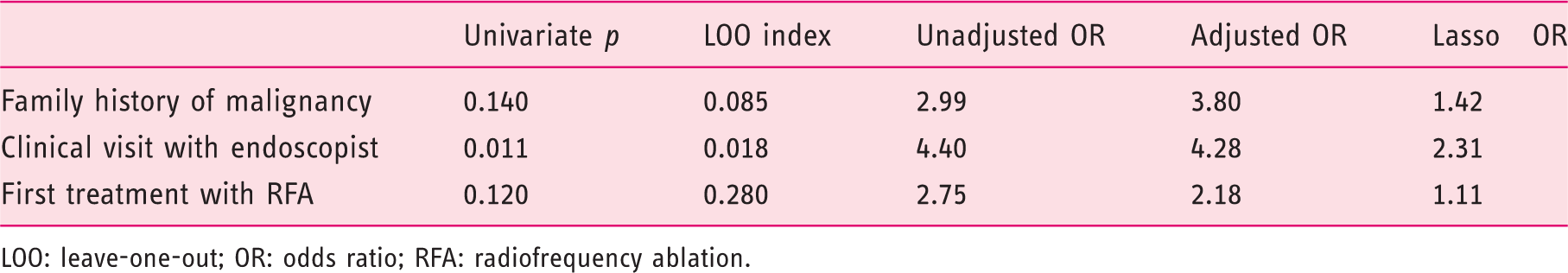
LOO: leave-one-out; OR: odds ratio; RFA: radiofrequency ablation.
Discussion
This study showed that 72% (50/69) of patients referred to a tertiary center for endoscopic treatment of BE-associated neoplasia were adherent to therapy. This definition of adherence included patients who had completed therapy and remained in surveillance at our institution, patients continuing in an endoscopic therapy protocol at our institution and patients who underwent surgical esophagectomy. We deliberately selected a strict threshold in defining adherence; hence, this definition may underestimate true adherence. rates as some patients may have returned to their local gastroenterologists for post-treatment surveillance. Among patients not completing endoscopic therapy as initially intended, various explanations were identified, specifically including the patient’s desire to forego further endoscopic treatment, often in the setting of progressive medical comorbidities. Therefore, lack of adherence should not in all cases be equated with being lost to follow-up.
In determining the factors associated with adherence to therapy, patients seen in gastroenterology clinical consultation with the treating endoscopist prior to initiation of a therapeutic regimen were more likely to adhere to their treatment than patients referred directly for open access endoscopy. This association remained significant, after controlling for potential confounders.
This study is unable to assess the specific features of a clinical consultation that were responsible for the increased likelihood of adherence, but clinic visits for these subjects did not follow a standardized template nor protocol; however, their clinic visits likely included discussions regarding the natural history of BE and risk of progression to invasive malignancy, with and without therapy; the technical aspects of endoscopic interventions, including the expected effectiveness and potential adverse outcomes; the potential need for multiple endoscopic interventions, followed by the need for post-treatment endoscopic surveillance; and the option of surgical esophagectomy as an alternative to endoscopic therapy and surveillance. Although the practice at our institution had previously been to offer and strongly encourage a gastroenterology clinical consultation prior to endoscopy among patients with BE whom were referred for open access for EMR or RFA; based on the results of this study, a clinical consultation is now mandatory.
Although in practice, most physicians would agree that a therapeutic relationship begins in a clinical setting, open access endoscopy has become widely prevalent in the current delivery of outpatient gastroenterology services in the USA. Potential benefits of open access endoscopy from the patient’s perspective may include lower direct and indirect costs (the latter by avoiding duplication of travel expenses and missed time from work, for an additional clinic visit), as well as shorter wait times; however, if one subscribes to the concept that endoscopic therapy offers a ‘remission’, rather than a ‘cure’ of BE, then the finding that a clinical consultation results in increased adherence to treatment should not be surprising.
In outlining a treatment plan and setting appropriate expectations, the patient and physician may recognize together that they are embarking on a chronic disease management program that requires longitudinal follow-up. This concept has been demonstrated in other gastrointestinal diseases requiring surveillance or ongoing follow-up. For example, increased adherence to surveillance protocols after colorectal cancer treatment is higher in patients with higher baseline patient-clinician information engagement scores 18 ; and a study of an outpatient inflammatory bowel disease clinic shows that physician trust is correlated with increased overall adherence to therapy and increased adherence to medications. 19
Other social and demographic factors that we examined did not appear to be associated with treatment adherence, including age, gender, marital status, insurance payor and driving distance from the treatment center. This last variable was of particular interest to us, from the perspective of a referral center to which many patients travel from out-of-state for medical care, with our expectation being that patients traveling long distances would be at increased risk of non-adherence or loss to follow-up.
While our study did not detect an association between driving distance from the treatment center and adherence, the ability to adequately assess this may have been hindered by a skewed distribution and the fact that the majority of patients (55%) lived >100 miles from the treatment center. The burden and cost of repeated travel could present an obstacle to adherence, for some patients. The extent to which endoscopic management of BE neoplasia will or should disseminate into community practice is unclear. Current professional practice guidelines indicate that patients with BE HGD should be evaluated in centers specializing in foregut cancer and HGD, prior to proceeding with surgery 1 ; and a recent international panel offers a strong consensus opinion for the statement that endoscopic therapy for HGD/T1m cancer should be performed in referral centers with appropriately-trained endoscopists and pathologists. 2 In a recent survey of practicing gastroenterologists, 85% of whom identified themselves as community gastroenterologists, the majority report that they refer patients with BE HGD to academic centers with BE expertise. 20 Any potential implications as to whether local availability of endoscopic BE therapy, as opposed to travel to a tertiary center, might influence patient adherence cannot be addressed by the current study. Intensive follow-up protocols, with collaboration between referring gastroenterologists and expert treatment centers, have been employed successfully in Europe. 21 Further efforts should be focused on defining the end-points for patients to transition back to local surveillance, following endoscopic therapy, with clear expectations for communication and joint patient management between the local and tertiary gastroenterologists.
This study has additional potential limitations. This was a single-center retrospective experience, in which one physician served both as the clinical consultant and the treating endoscopist. While this factor may reduce practice variability within the study, it may also lead to a lack of generalizability and external validity. The size of the cohort limits the power of the statistical analysis, and it is possible that additional factors that are either unknown or were not included in the current model could influence study findings. Future prospective studies with multiple participating centers and larger numbers of patients would be desirable, to further investigate and validate this study’s findings. An appropriate surrogate for socioeconomic status was not available in the database; therefore, the influence of socioeconomic status on adherence to BE therapy was not assessed. Additionally, it is possible that treatment adherence diminishes over time, due to either patient or physician fatigue; and hence, the duration of the patient follow-up period may not accurately capture long-term adherence rates.
By design, the initial statistical model did not include endoscopic/disease variables in assessing the factors associated with adherence. A reasonable assumption would be that patients with T1 esophageal cancer would be more likely to be adherent to therapy than patients with less advanced pathology (dysplastic or nondysplastic BE). In fact, in the post hoc analysis, patients with a pre-treatment diagnosis of T1 cancer were not more likely to be adherent than patients without cancer in this study; nor did inclusion of the presence/absence of cancer in the Lasso-penalized logistic regression model alter the results of the model: A prior clinic visit with the treating endoscopist remained the strongest predictor of adherence. These findings underscore that patient motivation(s) for maintaining adherence may not be driven solely by patient fear of or reaction to a diagnosis of cancer.
Also by study design, we elected to include surgical esophagectomy as a measure of adherence to therapy, because a decision to proceed with surgical esophagectomy is a commitment to definitive therapy for BE-associated neoplasia. Selecting between surgical and endoscopic therapy for BE HGD or intramucosal adenocarcinoma may be a complex decision involving factors including not only disease stage, but also patient age and medical comorbidities. One prior study suggests that the decision to proceed with surgical versus endoscopic therapy is influenced by whether a surgeon or gastroenterologist serves as the initial consultant 22 ; therefore, multidisciplinary consultation and collaboration may be advisable. Certainly, a surgical approach may be favored for patients with HGD or intramucosal adenocarcinoma who are unable or unwilling to demonstrate adherence to a longitudinal endoscopic therapy and surveillance program that would be necessary to confirm disease eradication and sustained remission. In our post hoc analysis, exclusion of the nine patients who underwent surgical esophagectomy did not alter the study results.
In summary, patients seen in clinical consultation prior to therapy for BE-associated neoplasia were more likely to demonstrate treatment adherence, compared to patients referred for open-access endoscopy. A clinic visit with the treating endoscopist may define expectations regarding treatment course, increase the likelihood of patient adherence; and so it merits consideration as the standard practice prior to embarking upon an endoscopic therapy and surveillance program. The study results may bear implications for other practice settings that plan to develop or have newly-developed BE endotherapy programs.
Footnotes
Author contributions
Study concept and design: LC and PY; data acquisition: LC; interpretation of data: LC, JCS and PY; statistical analysis: JCS; drafting of manuscript: LC and PY; review of final manuscript for important intellectual content: LC, JCS and PY.
Funding statement
This work (specifically Research Electronic Data Capture (REDCap)) was supported by the US National Institute of Health (NCATS/NIH grant number UL1 TR000445).
Conflict of interest
The authors declare there are no conflicts of interest.
