Abstract
Background
International guidelines suggest endoscopic resection for all patients with low-risk mucosal cancer. Ultimately, it is essential to treat the remaining Barrett’s esophagus as part of the treatment. Different thermal ablative therapies have been implemented to effect this treatment. They can lead to potential post-therapeutic stenosis. Furthermore, a histologic assessment of treated mucosa is not possible.
Objective
Clinical evaluation of a novel, non-thermal resection device (EndoRotor®) in the treatment of non-neoplastic Barrett’s esophagus was conducted.
Methods
Fourteen patients with early Barrett’s carcinoma were treated with endoscopic resection. Subsequently, EndoRotor® therapy was performed for resection of the remaining Barrett’s mucosa. Complications were assessed during the study. After a three-month period patients received follow-up endoscopy to evaluate post-therapeutic stenosis.
Results
On average, 674 mm2 (172 mm2 – 1600 mm2) of Barrett’s mucosa was treated with the novel device. In six (37.5%) cases, intra-procedural bleeding occurred with the need for hemostasis. All bleeding could be managed by endoscopic therapy alone. After a three-month follow-up there was no post-therapeutic stenosis registered.
Conclusion
EndoRotor® resection is a feasible non-thermal treatment of non-neoplastic Barrett’s esophagus. Larger trials have to evaluate risks and benefits of this novel device.
Key summary
Established knowledge on this subject.
After endoscopic resection of low-risk Barrett’s carcinoma, ablative therapy is essential. Thermal ablation methods always have the potential for post-therapeutic stenosis. With thermal ablation techniques histologic assessment is not possible. What are the significant and/or new findings of this study?
The EndoRotor® resection system is applicable in the esophagus. Athermal resection might decrease post-therapeutic stenosis. The use of this novel device enables assessment of resected specimen after ablation.
Introduction
Endoscopic treatment of early Barrett’s adenocarcinoma (EAC) has been established as a two-step approach. In the first step, endoscopic resection for all visible neoplasia is carried out. Usually early cancer can be identified as changes in the surface structure. In case of low-grade and high-grade intraepithelial neoplasia, it might be significantly more difficult to identify neoplasia. Major effort should be undertaken to localize the lesion as there is always a substantial risk that it is undiagnosed cancer. To avoid inappropriate therapy, all visible lesions should undergo endoscopic resection. Furthermore, there is greater inter-observer agreement in resected samples than in biopsies between pathologists. 1 In case of non-visible neoplasia, i.e. after four-quadrant biopsy screening, all intraepithelial neoplasia should be double-checked by a senior reference pathologist. Confirmed intraepithelial neoplasia has a significantly higher progression rate.
In the second step, the ablation of all residual intestinal metaplasia is necessary to avoid recurrence of neoplasia or development of metachronous lesions. Thermal ablation is used as the current standard for this indication. Photodynamic therapy (PDT) was the first technique used as a thermal therapy in Barrett’s esophagus. 2 As a result of considerable technical efforts using PDT and management of relevant side effects for patients, argon-plasma coagulation (APC) became the standard treatment for non-neoplastic intestinal metaplasia in the esophagus. 3 Using APC for ablation of the remaining Barrett’s mucosa after endoscopic resection, the recurrence of metachronous lesions can be reduced substantially. 4 Another alternative option for this indication is the use of radiofrequency ablation (RFA). RFA can be used as a balloon-based device with circumferential electrodes or as a focal catheter in different sizes. Easy handling and good Barrett’s eradication rates with acceptable complication rates have spread the use of this rapidly evolving new modality. 5 Following the current guidelines, endoscopic therapy in early adenocarcinoma of the esophagus has been established as a safe treatment option with excellent long-term results. 6 However, a major complication for all thermal treatment is the development of post-therapeutic stenosis. On the one hand, the greater the applied energy to the esophageal mucosa, the higher the risk for bleeding, perforation and post-therapeutic stenosis. On the other hand, the thickness of esophageal mucosal layers is vastly different among patients, which can result in insufficient treatment with remaining intestinal metaplasia under the neo-squamous epithelium. 7 Moreover, detection of neoplasia can be missed even by experienced endoscopists. One major disadvantage to all thermal ablation modalities is the lack of possibility for retrieving tissue samples of the mucosal and submucosal layers.
The EndoRotor® device (Interscope Medical Inc, Whitinsville, MA, USA) was initially developed for endoscopic treatment of adenomatous tissue in the colon. Because of the lack of cauterization, this novel non-thermal resection device might reduce post-therapeutic stenosis and simultaneously allows for post-procedural tissue evaluation. This pilot study evaluates the feasibility of the novel EndoRotor® device for therapy of esophageal lesions.
Material and methods
Patients who presented with EAC or intraepithelial neoplasia of the esophagus at a tertiary center were considered for this feasibility study. All patients underwent systematic staging including upper gastrointestinal (GI) endoscopy with high-definition endoscopes and chromoendoscopy with 1.5% acetic acid. In accordance with current guidelines patients underwent endoscopic resection when mucosal or limited submucosal infiltration without high-risk criteria were assumed. Local therapy was continued when the final histological result revealed early mucosal adenocarcinoma pT1a or pT1b (sm1), G1/2, with no lymphatic or vascular invasion and an R0 resection margin at the base. Endoscopic therapy was accomplished when all neoplastic tissue was resected. Subsequently patients were included to undergo EndoRotor® therapy. All patients signed written consent for ablative therapy. In case of active reflux disease, which can make assessment very difficult, patients were treated by intensive anti-secretory therapy and diagnostic endoscopy was repeated before resection was performed. During all upper GI endoscopies with Barrett’s esophagus, the full extent of all Barrett’s epithelium was described using the Prague classification.8,9
Technical equipment
The EndoRotor® resection system is a catheter-based system for non-thermal ablative therapy via an endoscope. The system includes several catheter lengths for manufacturer endoscopes including a 2550 mm catheter (for esophageal disease), which was used in this study. The outer cannula can be repositioned to direct the cutting orientation to the appropriate resection trajectory and its inner rotating blade combined with a vacuum enables the actual resection. Vacuum levels can be adjusted depending on the level of tissue contact to pull tissue into the cutting orifice, while the system self-irrigates to transport the samples back to a collection trap (Figure 1). The user initiates the resection process by tapping the blue pedal to start the rotation of the inner cutter blade. The rotation stops automatically after eight seconds if resection is not started. To initiate resection, continuous pressure on the orange foot pedal activates suction to the catheter. At the tip of the catheter there is a ∼2 mm window. Direct contact of this window to the tissue when the device is active facilitates the actual resection. To stop the intervention, the tip of the probe can be lifted by using the wheels of the endoscope or the blue pedal may be pressed to cancel all functions. For better orientation, there is a solid black line at the 180-degree position and on either side of the black line at 90 degrees there are dashed lines. They indicate the cutter position is exactly 90 degrees from those lines. To control the cutting depth the operator can set the rotation speed between 1000 (low) or 1750 (high) revolutions per minute (rpm) and vary the level of suction from 75 mmHg to 400 mmHg. The size of resection tissue samples depends on the cutting position, vacuum and rotation speed. Usually the samples have a size of 2–2.5 mm, which is comparable to standardized biopsy with forceps. The applied suction sends the samples directly to the console with a disposable trap and filter (Figure 2). All treated tissue is captured for pathological assessment. Ablative procedures were performed under sedation with propofol and midazolam. The catheter-based EndoRotor® resection system was introduced through the working channel of a standard endoscope (Fujifilm, Tokyo, Japan, with at least 3.2 mm working channel). The rotation speed was set either to 1000 rpm (low) or 1750 rpm (high) for treatment. Barrett’s mucosa was injected with a mixture of saline and epinephrine (1:100,000) as well as indigo carmine for better visualization prior to EndoRotor® therapy.
Endoscope with EndoRotor® catheter. At the tip of the probe there is a small window with a cutting blade for non-thermal resection. The EndoRotor® resection system with suction device, probe and trap to collect the resection specimen.

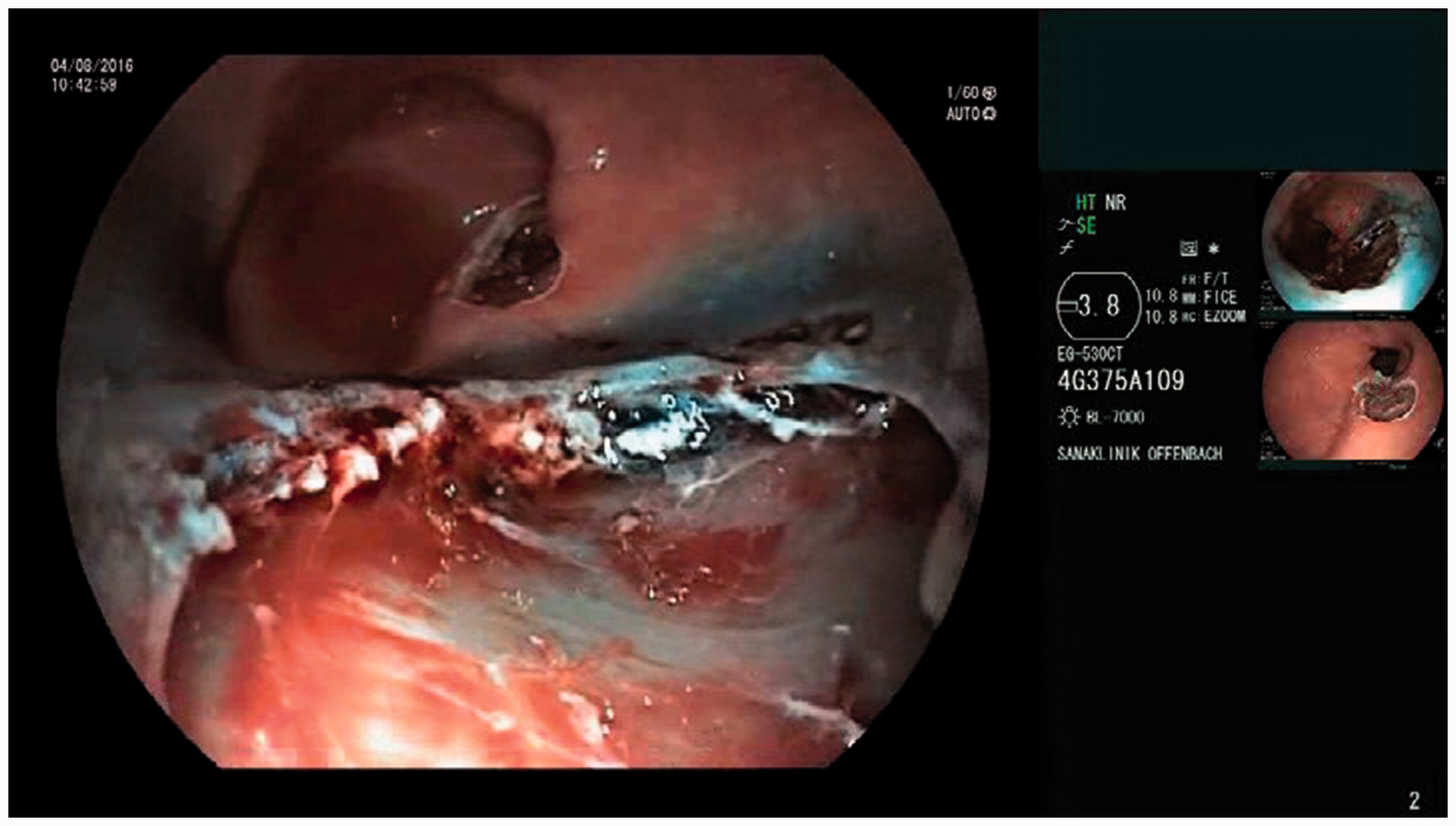
Under direct visual control vacuum (75 mmHg to 300 mmHg) was applied to the catheter and blade rotation was started. Applying gentle pressure to the catheter tip against the mucosa and consequent movement over the esophageal mucosal started the EndoRotor® resection. Continual movement of the EndoRotor® catheter contributes to depth control and is crucial to avoid perforation as the catheter serially resects tissue layers (Figures 3 and 4). To analyze the effectiveness of the EndoRotor® device, all treated areas were photo-documented. For measuring the surface, it was assumed that the full circumferential (C) length of the esophagus was 4 cm. The longitudinal length was measured by retracting the endoscope. Complete eradication was not the goal of treatment in this study protocol. Therefore, recurrence for Barrett’s esophagus could be given only for the treated area.
Endoscopic view after saline injection and first cut. The EndoRotor® probe is clearly visible on the left-hand side. Esophageal wound after ablation with the EndoRotor® system. At the base of the mucosal defect the intact circular muscle can be clearly seen with no clinically relevant bleeding.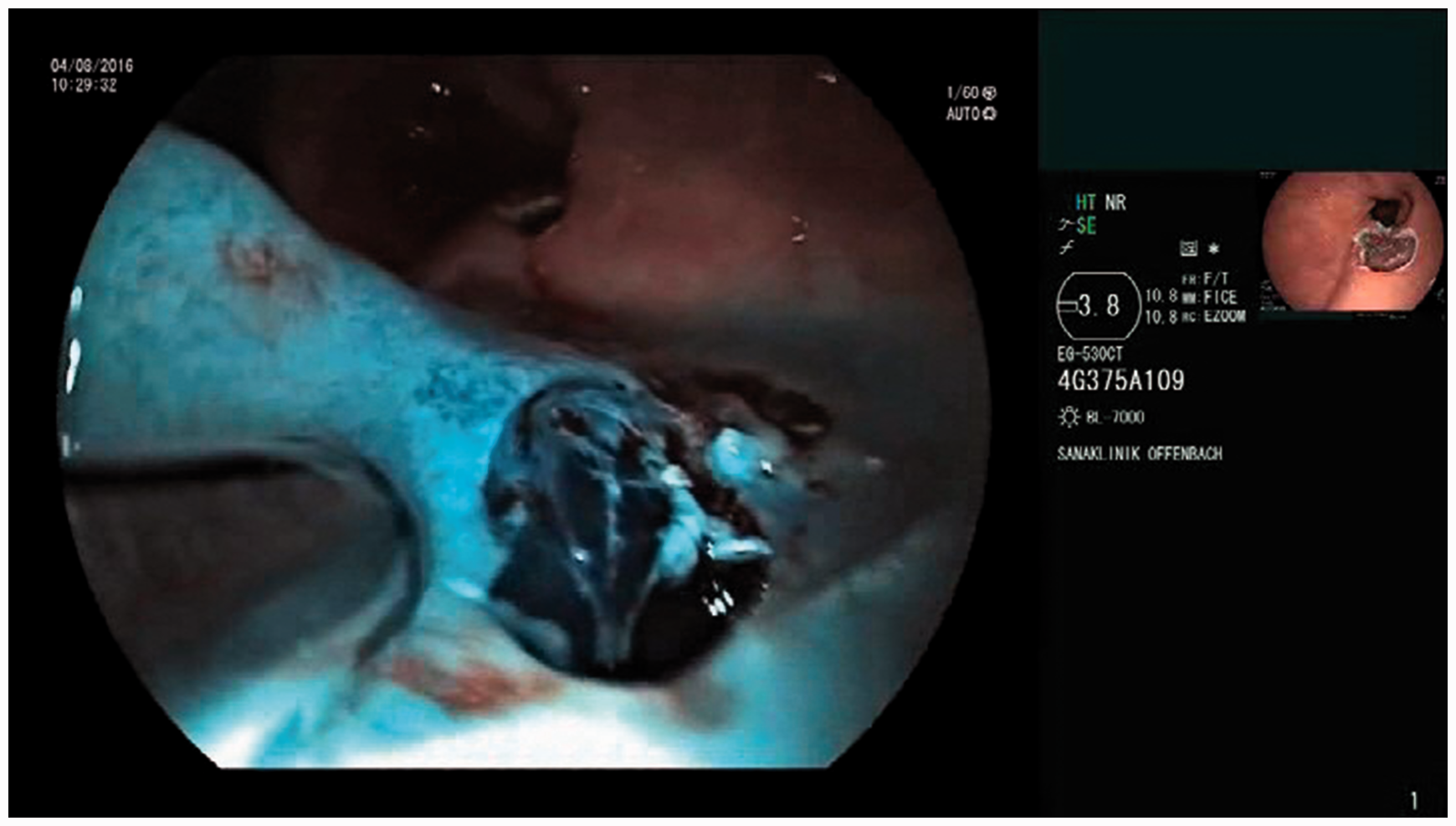
Minor complications were defined as post-procedural bleeding with a decrease of hemoglobin <2 g/dl. To analyze bleeding complication rate, every patient had gastroscopy the day after EndoRotor® therapy.
Major complications were defined as bleeding that required blood transfusion or a decrease in hemoglobin >2g/dl, the need for re-endoscopy or perforation.
Bleeding complications were managed by endoscopic clipping (Resolution Clip, Boston Scientific, MA, USA), mucosal injection with saline (Medwork, Höchstadt, Germany, injection needle 0.7 mm) with epinephrine mixture (1:100,000) or soft coagulation (Hot Biopsy Forceps 230 mm, Cook Medical, Bloomington, IN, USA). Two senior endoscopists with more than 100 endoscopic resections per year performed all procedures.
Patient data, procedural times and complications were collected.
All patients had scheduled endoscopic follow-up after five to eight weeks for assessment of late complications and evaluation of post-interventional stenosis.
Statistical calculations were performed with PASW 18 (SPSS, IBM Corporation, NY, USA).
This study was conducted in accordance with Good Clinical Practice standards. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee. It was registered in DRKS (German trial register, Freiburg) DRKS00012925 and was approved by the local ethics committee (Landesärztekammer Hessen), date of approval 9 July 2017, number: FF168/2016.
Results
All data in the feasibility study were recorded and evaluated in a meticulous assessment. However, all results in percentages should be interpreted with caution as the sample size in this study is too small for statistic evaluation.
Within a two-month period 16 endoscopic EndoRotor® interventions of non-neoplastic Barrett’s mucosa on 14 patients (12 men, two women (age minimum 45 years, maximum 79 years, mean age 64) were performed. The mean initial Barrett size was C 2.5 cm (SD 2.4) and maximum (M) 7 cm (SD 3.1) according to the Prague classification. EndoRotor® therapy could be performed in all patients. On average 674 mm2 (minimum 172 mm2, maximum 1600 mm2, SD 438 mm2) of Barrett’s mucosa was treated with the novel device.
Re-epithelialization with squamous epithelium was seen in all patients who underwent follow-up. In one case full acid suppression with daily dose of 120 mg pantoprazole was necessary.
Histologic assessment of EndoRotor®-collected specimens showed specialized (Barrett’s mucosa) or non-specialized columnar epithelium in 87.4%. In 6.3% low-grade intraepithelial neoplasia (LGIN) was diagnosed. In one case (6.3%) histologic evaluation was not possible because of technical difficulties.
Complications
After the endoscopic procedure, 9/14 patients complained of moderate esophageal pain with indication for oral or parenteral analgesia. In six (37.5%) cases, intra-procedural bleeding occurred with the need for hemostasis. One of these patients had major bleeding with the need for re-endoscopy and the need for observation in intensive care. There was one minor bleeding vessel controlled endoscopically the next day. All bleeding with indication for re-endoscopy could be managed by endoscopic hemostasis alone. There were no perforations or intervention-related deaths. In total 12.5% of post-procedural bleeding complications were recorded.
Twelve of 14 patients underwent follow-up after a three-month interval. None complained about dysphagia or had relevant stenosis visible during upper GI endoscopy.
Discussion
Comparing different ablative techniques for Barrett’s esophagus after endoscopic resection of neoplasia.
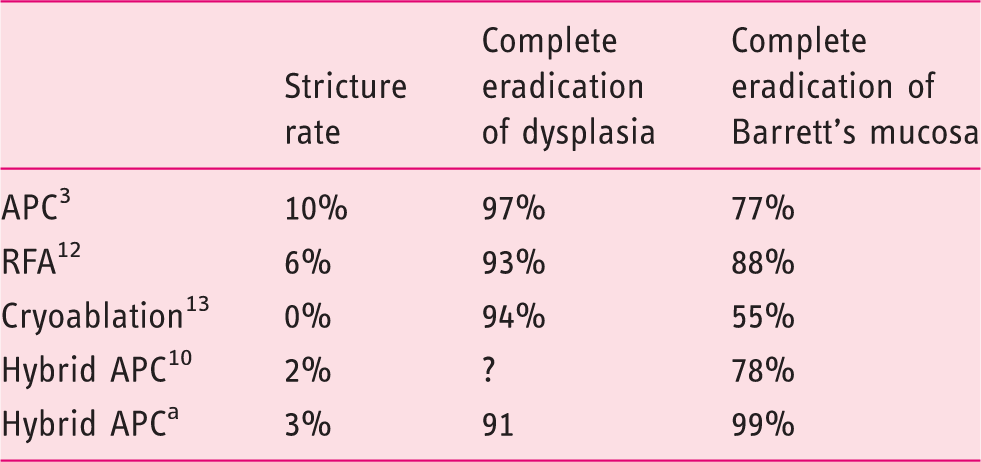
Publication pending.
APC: argon-plasma coagulation; RFA: radiofrequency ablation.
The EndoRotor® resection system was initially developed for resection of flat lesions and persistent/recurrent adenoma in the colon. Particularly after piecemeal resection of large non-pedunculated colorectal adenomas, there is a relevant rate of recurrence.14,15 Additional endoscopic resection in post-therapeutic scars is challenging. Design and construction of this novel device offers a potential solution. Differences in elasticity between scar and adenomatous tissue helps to resect only the mucosa while the stiffer muscular layer is kept unharmed. A first experience in an animal model showed promising results. 16 The aim of this pilot study was to evaluate the novel device in the esophagus.
The present feasibility study shows the first experiences using the EndoRotor® resection system for therapy in non-neoplastic Barrett’s esophagus. It could show that application is safe and practicable in clinical use. However, approximately one-third of patients had intra-procedural bleeding that could be managed by endoscopic therapy alone. Time for intervention was not systematically recorded as we expected a learning curve for endoscopists and endoscopic assistants in handling the new endoscopic probes, which were initially developed for colonoscopies. Future multicenter studies will have to define the real technical effort.
In this first pilot study, EndoRotor® therapy, being non-thermal, in Barrett’s esophagus seems to have higher bleeding complications (one minor bleeding and one major bleeding) compared to other established procedures. Using RFA for therapy of Barrett’s esophagus is well investigated with very few bleeding complications <1%.10,12,17 However, a recently published large multicenter study showed an overall rate of mild and moderate adverse events in 19% using RFA. 18 This is still significantly lower than in the present study. Future prospective studies will have to demonstrate if complication rates remain relevant using EndoRotor® in Barrett’s esophagus, especially as it remains unclear what technical setting (rpm high versus low) and level of suction is the optimal setting for use in the esophagus. In our experience it is best to start the ablation with a high speed (1750 rpm) at 150–200 mmHg suction. Evaluation of other variables like routine application of hemostatic agents should also be considered in future interventions as they might lower bleeding complications.
The major advantage of the EndoRotor® is the non-thermal therapy in combination with deep resection down to the muscle layer under direct visual control.
In particular the possibility of tissue collection during use of the EndoRotor® is a highly innovative advantage compared to all other ablation devices. During intervention, tissue is collected in a trap and can be subsequently analyzed by a pathologist. In this small pilot series, at least one low-grade intraepithelial neoplasia was found. The use of non-thermal resection might lead to less thermal damage to the deeper layers, which might subsequently decrease the stricture rate. There was no reported dysphagia or stenosis in this cohort.
In conclusion, the new EndoRotor® resection system is a potential alternative to the established ablative therapies. Larger, prospective trials are needed to highlight the statistically relevant advantages and disadvantages to establish the system in everyday clinical use. Further pending technical improvements are necessary to optimize handling in the upper GI tract.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This study was conducted in accordance with Good Clinical Practice standards. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee. It was registered in DRKS (German trial register, Freiburg) DRKS00012925 and was approved by the local ethics committee (Landesärztekammer Hessen), date of approval 9 July 2017, number: FF168/2016.
Informed consent
All patients have written informed consent for ablative therapy as well as endorotor therapy.
