Abstract
Background
Little is known about the outcomes of endoscopic submucosal dissection in elderly patients with superficial squamous esophageal neoplasms.
Objective
To assess the efficacy and safety of endoscopic submucosal dissection for superficial squamous esophageal neoplasms in elderly patients (≥65 years) compared with non-elderly patients.
Methods
All patients with superficial squamous esophageal neoplasms receiving endoscopic submucosal dissection were retrospectively analyzed. Among them, 130 were aged 65 or older (group A), and 201 were aged younger than 65 years (group B). Therapeutic efficacy, adverse events, and follow-up data were evaluated.
Results
Group A had a higher prevalence of concomitant diseases than group B (52.3% vs. 14.9%, respectively). R0 resection rate was 82.3% in group A and 84.6 % in group B (P = 0.717). The curative resection rate was 80.8% in group A and 83.6% in group B (P = 0.653). The rate of procedure-related non-cardiopulmonary adverse events was 20.8% in group A and 16.9% in group B (P = 0.377). The incidence of cardiopulmonary adverse events during or after the procedure was 6.2% in group A and 2.5% in group B (P = 0.094). No procedure-related mortality was reported in either group.
Conclusion
Endoscopic submucosal dissection is effective and safe for treating superficial squamous esophageal neoplasms in elderly patients.
Keywords
Introduction
Esophageal squamous cell carcinoma (ESCC) is currently the dominant histological type in Asian countries, and esophageal squamous intraepithelial neoplasia (ESIN) represents the strongest risk factor for the development of ESCC.1,2 Radical esophagectomy has traditionally been the preferred treatment for early-stage ESCC and ESIN, but it carries relatively high morbidity and mortality rates, especially in patients at high risk of surgery.3,4
Endoscopic submucosal dissection (ESD) is considered a promising alternative for patients with superficial squamous esophageal neoplasms (SSENs) including ESIN and early ESCC because it can achieve an en bloc resection regardless of lesion size and offers the advantage of histopathologic verification.5,6 A recent meta-analysis reported favorable outcomes with ESD in endoscopic eradication of SSENs with low risk of lymph node metastasis. 7 Elderly patients, considered to be medically vulnerable, have multiple chronic diseases, and their physical conditions are generally worse than those of non-elderly patients. While usually safe, ESD can pose a risk of serious complications, especially for elderly patients with underlying co-morbidities. In recent years, ESD for the elderly has been reported to be a reliable procedure in the stomach and colon.8,9 However, esophageal ESD seems to be technically more difficult than gastric ESD due to the narrower space. Until now, only limited data have been available regarding the results of ESD for SSENs in elderly patients.10–11 To evaluate the short-term and long-term outcomes of ESD in elderly patients (≥65 years) with SSENs, we reviewed our experience with ESD in elderly versus non-elderly patients (<65 years).
Materials and methods
Study cohort and variables
Between January 2010 and December 2013, we retrospectively analyzed the clinical data of patients with initial-onset SSENs who underwent ESD. Among these patients, 130 were aged 65 or older (group A), and 201 were younger than aged 65 years (group B). The data were obtained from a prospectively collected database of patients who underwent ESD at a single tertiary academic hospital (the Nanjing Drum Tower Hospital, China). Esophageal ESD was considered when the lesions were clinically diagnosed as follows: (1) endoscopic signs of early lesions or endoscopic ultrasonography (EUS) and endoscopic examinations confirming tumor limited to the mucosa; (2) histological confirmation of ESCC restricted to the mucosa (m1 for intraepithelial carcinoma and m2 for carcinoma invading the lamina propria), or high-grade intraepithelial neoplasia (HGIN), or low-grade intraepithelial neoplasia (LGIN); (3) lesions with m3 (muscularis mucosa) invasion without lymphatic and vascular involvement; (4) no signs of lymph node or distant metastases.
For evaluation of staging, all patients underwent white light imaging, iodine staining, narrow band imaging with magnification, EUS, and chest and abdominal computed tomography (CT). Data were collected on gender, age, and comorbidities including hypertension, diabetes, cardiovascular disease, chronic kidney disease, any previous cerebrovascular event, physical status, endoscopic characteristics, histological findings, procedural details and outcomes, duration of hospitalization, procedure-related adverse events, and mortality. Each patient’s status was classified according to the physical status classification of the American Society of Anesthesiologists (ASA). 12 The study was approved by the institutional review board of our hospital on February 24, 2014, and written informed consent was obtained from all patients.
ESD procedures
In patients who were receiving anticoagulants daily, anticoagulants or antiplatelets were discontinued for 1 week before ESD, if possible. During ESD, all patients were under general anesthesia and tracheal intubation. All ESD procedures were performed in a standardized way, 6 and performed by four experienced endoscopists (Zhang XQ, Lv Y, Ling TS, and Wang L).
Adverse events
Adverse events were classified according to whether they occurred during or after ESD. Cardiopulmonary adverse events were defined as any adverse event occurring during or within 1 day after ESD, as follows: (1) hypoxia, (2) ST depression in ECG at any time during ESD, (3) overt bradycardia, (4) hypotension, or (5) post-ESD myocardial or cerebral infarction within 1 day. 13 Other minor cardiopulmonary adverse events during or after ESD included tachycardia and hypertension. 13 Concerning ESD-related non-cardiopulmonary adverse events, delayed bleeding was defined when clinical bleeding signs were observed after ESD (hematemesis and/or melena or hemoglobin drop >2 g). 14 Perforation was diagnosed endoscopically just after resection or by the presence of free air on a chest x-ray or CT scan. Stricture was regarded as a complication when it was symptomatic and required endoscopic intervention. Infection was defined as body temperature >38.5 ℃ at 24–48 h after ESD and the evaluation of white blood cells.
Histological evaluation after ESD and outcome definition
Histological evaluation was performed by two experienced pathologists according to World Health Organization criteria. En bloc resection was defined as removal of the lesion in a single piece for which the margin appeared endoscopically tumor free. R0 resection was defined as en bloc resection with histopathological demonstration of horizontal and vertical margins free of both cancer and dysplasia. R1 resection was described as incomplete resection with tumor cells or dysplasia extending into the vertical or lateral margins. Curative resection in ESCC was defined as R0 resection without lymph-vascular invasion on histology, when the lesions fulfilled the indications described above. If the treatment was not curative, patients with ESCC were recommended for further treatment, such as esophagectomy with lymph node dissection or radiation therapy.
Follow-up algorithm
After ESD, all patients underwent endoscopic examination at 3, 6, and 12 months postoperatively and every 12 months thereafter. CT was performed in patients with non-curative resection at 6–12 month intervals to detect recurrence or other metastasis. Local recurrence was defined as a neoplasia detected in close proximity to the scar more than 6 months after ESD. A new lesion detected within 1 year of the initial ESD in a different area from the first lesion was considered a synchronous lesion, as opposed to a metachronous lesion, which was defined as a lesion that was detected >1 year after ESD.
Statistics
Categorical variables were compared with the chi-square test, and continuous values were compared using the t test or the Mann–Whitney rank sum test. Kaplan–Meier curves were constructed to analyze rates of metachronous lesions. The log-rank test was used to evaluate the significance of differences between curves. All tests were two-sided, and p values <0.05 were considered statistically significant (SAS v9.2, SAS Institute Inc, Cary, NC).
Results
Patient and lesions characteristics
Patients and lesion characteristics

Fisher exact test.
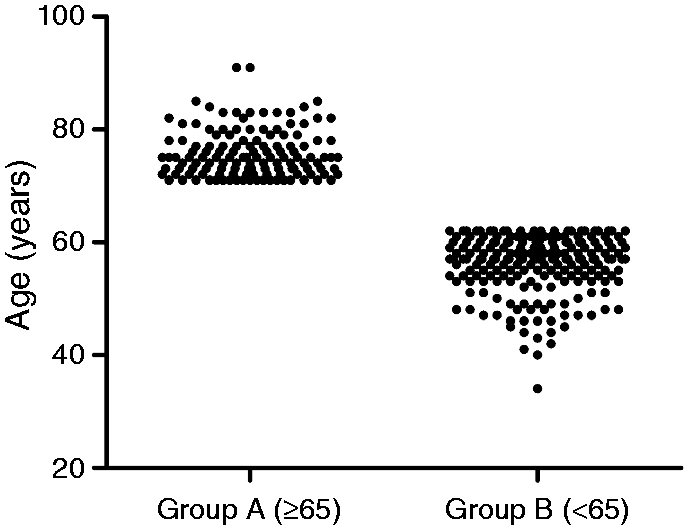
The age distributions in group A and group B.
The macroscopic type of lesion, location, tumor size, pre-ESD and post-ESD histological types, depth of invasion, and lymphovascular invasion were similar in both groups (Table 1). When comparing the pre-ESD pathology with initial biopsy specimens, 185 of these lesions (55.9%) showed histologic concordance, 97 (29.3 %) showed an upgraded histology (58 HGID, 17.5 %; 39 ESCCs, 11.8 %), and 49 (14.8 %) showed a downgraded histology. The histologic discrepancies between the biopsy and the ESD diagnosis did not differ between the two groups (p = 0.562) (Table 1).
Short-term outcomes and adverse events of ESD
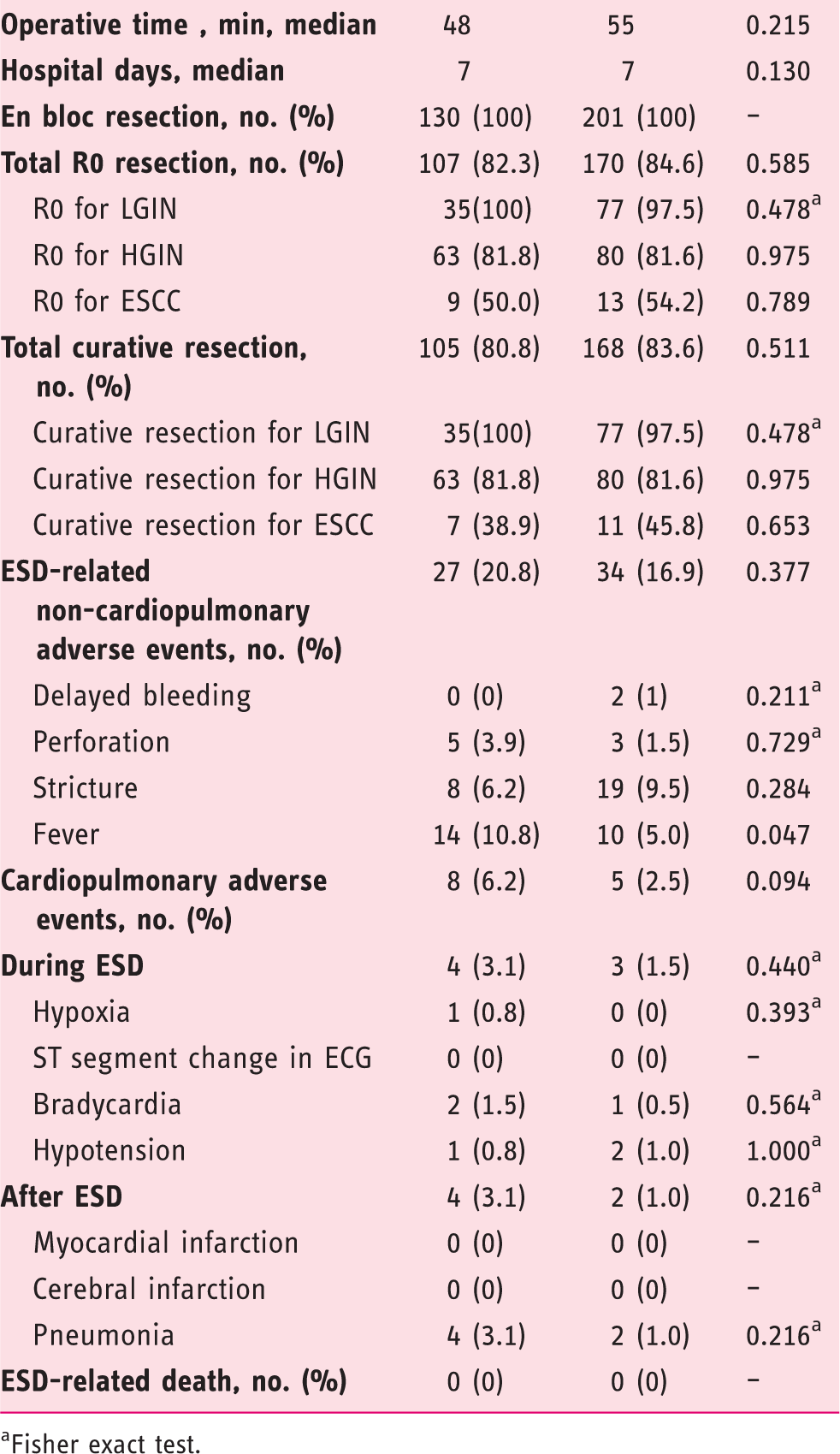
Outcomes and adverse events of endoscopic submucosal dissection in both groups
Fisher exact test.
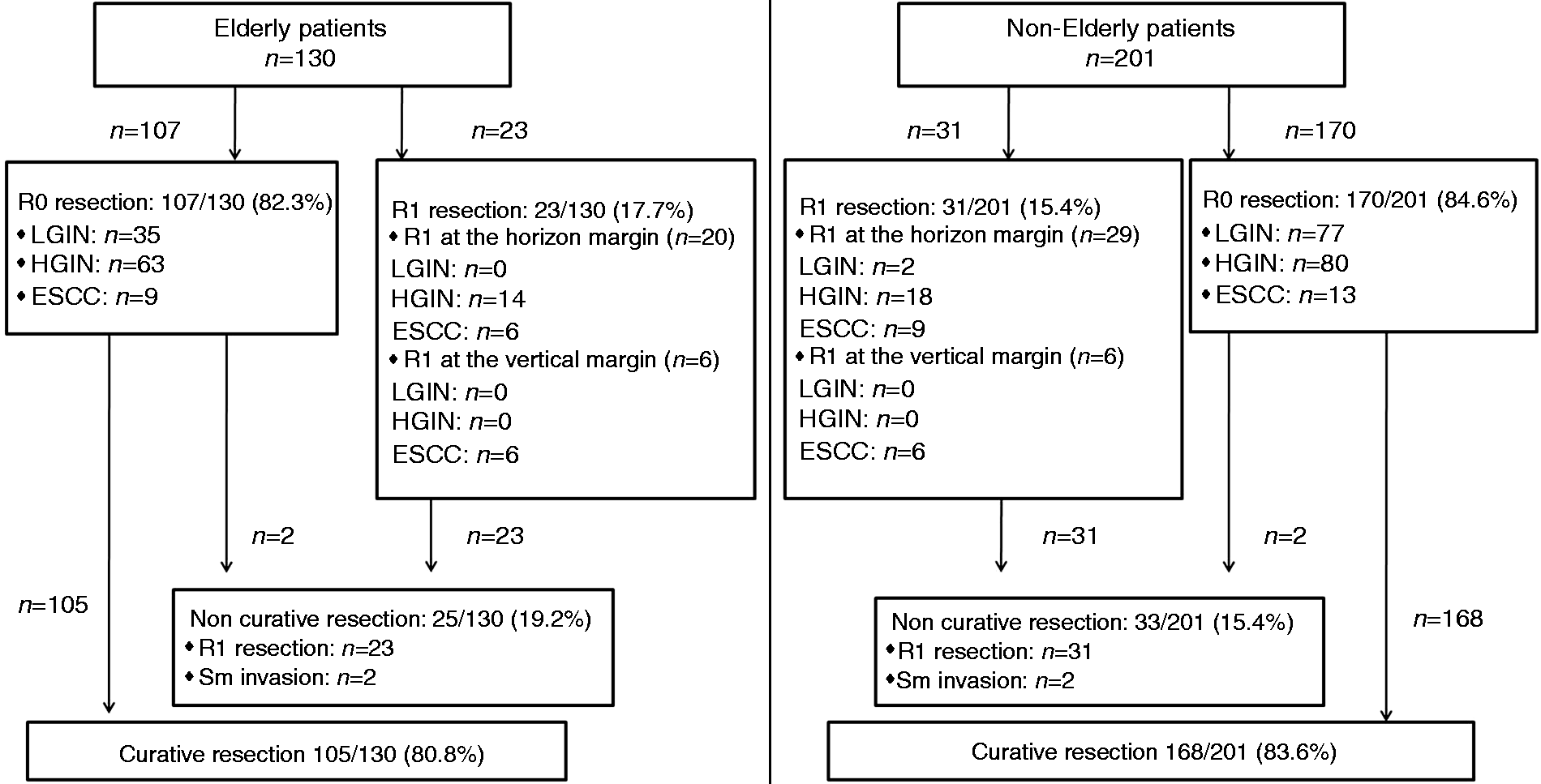
Resection rates of endoscopic submucosal dissection in all patients with superficial squamous esophageal neoplasms.
En bloc resection was achieved in all cases in the two groups. The total R0 resection rate was similar in group A (82.3%) and group B (84.6%) (p = 0.585). According to post-ESD histological type, the R0 resection rate was not significantly different between group A and group B (LGIN: 100% vs. 97.5, p = 0.478; HGIN: 81.8% vs. 81.6%, p = 0.975; ESCC: 50.0% vs. 54.2%, p = 0.789). More remarkable, the R0 resection rate in group A was higher for LGIN than for HGIN and ESCC, which was similar to group B. On the other hand, four specimens (2 ESCCs in group A and 2 ESCCs in group B) showed submucosal invasions and, therefore, were judged to be non-curative. The total curative resection rate was slightly lower in group A (80.8%) than group B (83.6%), but the difference was not statistically significant (p = 0.511). Curative resection for ESCC was achieved only in 38.9% (7/18) of group A patients and 45.8% (11/24) of group B patients (p = 0.653).
The adverse events are shown in Table 2. The frequency of ESD-related non-cardiopulmonary adverse events was not significantly different, being 20.8% in group A and 16.9% in group B (p = 0.377). Delayed bleeding was observed exclusively in group B, without the difference being significant. All bleedings were controlled by endoscopic electrocoagulation. Perforation occurred in 5 (3.9%) patients in group A and 3 (1.5%) patients in group B (p = 0.729), all of which were successfully managed endoscopically. Eight (6.2%) patients in group A and 19 (9.5%) in group B developed symptomatic esophageal stricture (p = 0.284). To date, all these patients had complete resolution of their stricture after endoscopic balloon dilation or temporary removable esophageal stenting. Fever after ESD occurred more frequently in group A than in group B (p = 0.047).
The total incidence of cardiopulmonary adverse events did not differ between the two groups. Cardiopulmonary adverse events during ESD were seen in four (3.1%) patients in group A and three (1.5%) patients in group B (p = 0.440). After ESD, pneumonia was a major cardiopulmonary adverse event, occurring in 3% in group A and 2.6% in group B (p = 0.216). No deaths were reported because of ESD.
Additional treatment and long-term outcomes
Data on clinical follow-up and recurrences are provided in Figure 3. Additional surgical treatment was performed in 12 non-curative cases (4 in group A and 8 in group B) with ESCC after ESD. Only two cases were found to have lymph node metastases in the resected specimen after esophageal resection. Six non-curative cases (4 in group A and 2 in group B) with ESCC received radiotherapy. The remaining six non-curative cases with ESCCs (3 in group A and 3 in group B) refused additional treatment.
Clinical follow-up and recurrences data after endoscopic submucosal dissection for superficial squamous esophageal neoplasms.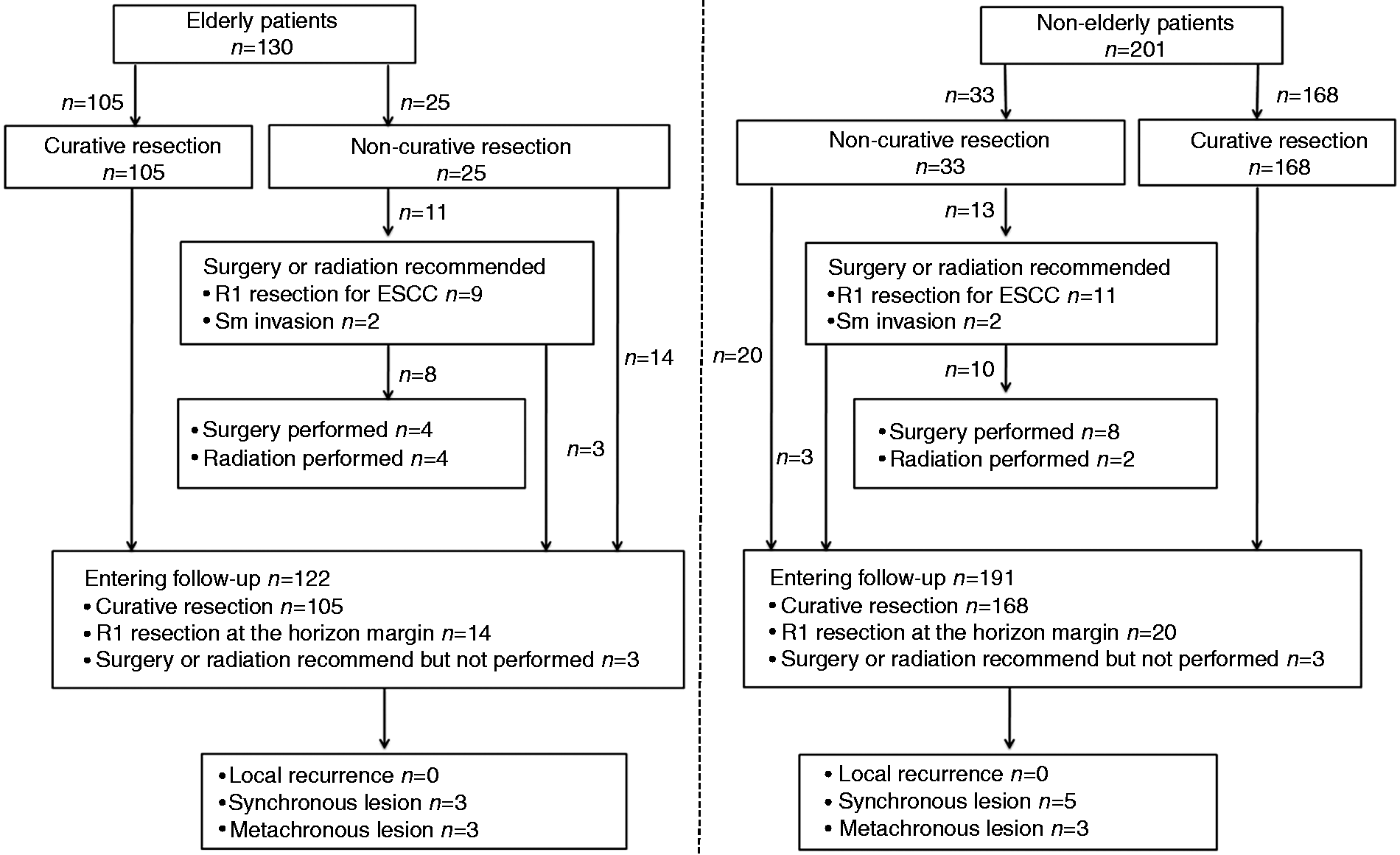
A total of 313 patients (94.6 %) were followed until death or until 1 July 2014. The median follow-up period was 15 months (IQR 12–25.7 months) in group A and 18.5 months (IQR 12.5–36.7 months) in group B (p = 0.050). No local recurrence was observed after R0 resection during follow-up. Synchronous lesions showing HGID were observed in three (2.5%) elderly and five (2.6%) non-elderly patients (p = 0.568). Of these, six patients (group A [n = 1], group B [n = 5]) were cured by ESD, and two declined repeat treatment due to advanced age (n = 1) or poor performance status (n = 1). Similarly, metachronous lesions developed in only six patients. All these lesions were HGINs; three non-elderly cases were managed with ESD, and the remaining three elderly patients refused further treatment. The cumulative incidence curve of metachronous lesions is plotted in Figure 4. The cumulative incidence of metachronous lesions at 2 years was 1.3% in both groups (p = 0.610).
Cumulative incidence of metachronous lesions after endoscopic submucosal dissection for superficial squamous esophageal neoplasms.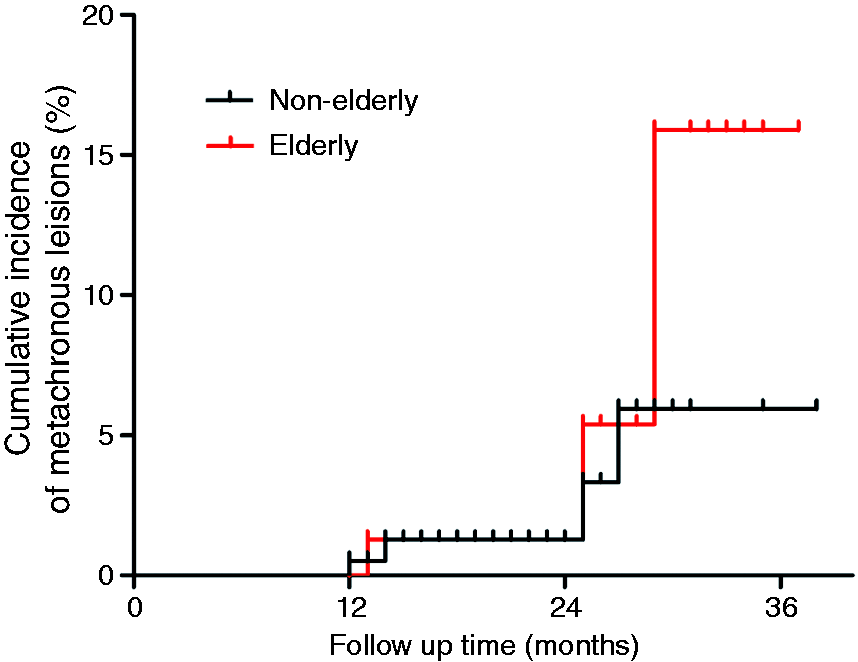
Discussion
Elderly patients with SSENs remain a medical challenge for gastroenterologists, mainly because of the frequent comorbidities. To date, few reported clinical outcome of ESD in these patients. Also, those studies included small numbers of elderly patients (65 years of age and older) and had short follow-up periods. Therefore, it is clinically important to assess the efficacy and safety of ESD for elderly patients with SSENs.
In the current study, there were no significant differences in lesion characteristics between group A and group B. Baseline characteristics of patients differed in both groups. Nevertheless, en bloc resection rate was high and reached 100% in both groups. The total R0 resection rate was similar in both groups. Even according to different histological type, the differences in R0 resection rates in both groups showed no statistical significance. Additionally, our data for both groups showed a significant trend toward a lower R0 resection rate in patients with a higher pathological grading. All R0 resection rates, except for LGIN, were a little lower than previously published results (90%).7,15 This discrepancy was especially marked in ESCC, perhaps partly due to the fact that R0 resection in our study was strictly defined as the absence of atypical cells (LGIN, HGIN, or invasive cancer) at the horizontal and vertical resection margins. Another explanation is that the lesion size (4 cm in median diameter) was relatively large in our study. Similar results occurred in Wen’s study, 16 which showed that larger lesions and a greater depth of invasion increased the chance of residual tumor after ESD in superficial esophageal squamous cell neoplasia. Therefore, in pre-procedure evaluations of lesion size and depth invasion, it is important to choose patients who are expected to have a reasonable R0 resection rate, and to weigh cases with a lower anticipated R0 resection rate into decision-making and consent.
In the present study, ASA status was significantly higher in elderly patients versus non-elderly patients, reflecting underlying co-morbidity. However, the incidence of overall ESD-related adverse events in group A was comparable to that in group B. Only infection was more common in group A than in group B, possibly because of some relative implicating factors, e.g. an age-related natural decline in the immune status or so-called immunosenescence. In addition, in our study post-ESD stricture rate for each group was similar to reported figures of 5% to 23%,17–19 and all strictures were treated successfully with endoscopic interventions. In fact, despite different treatment and prophylactic strategies, esophageal stricture has remained a significant challenge for clinicians because it substantially decreases patients’ quality of life and requires long term endoscopic re-intervention.20–22 This may lead to greater reluctance in elderly patients and their relatives to grant consent for ESD interventions. In addition, although a significantly higher percentage of patients received anticoagulant drugs in group A than in group B, no elderly patients in either group developed delayed bleeding. This can be partly explained by the fact that anticoagulants were discontinued to eliminate the influence on bleeding during or after ESD. On the other hand, a previous study reported that poor ASA class was significantly associated with a high risk of adverse events during esophagogastroduodenoscopy and colonoscopy; 23 however, our study showed that the advanced age and poor general condition of group A patients, classified as ASA class III, did not significantly increase that group’s incidence of cardiopulmonary adverse events during or after ESD. No ESD-related mortality was observed in the two groups. Also, length of hospital stay was similar in both groups.
Although ESD can preserve the esophagus and maintain postoperative quality of life, the procedure also could increase the risk of metachronous lesions and recurrences in the remnant esophageal mucosa. The long-term oncologic outcome of ESD in elderly patients remains an unanswered question. Consistent with previous studies,6,17 all patients with pathologically complete resection in this study did not have local recurrence. The rates of synchronous lesions were similar in both groups. All these were HGIDs located in the cervical esophagus and missed by the preoperative endoscopic evaluation. Takahashi et al. reported that synchronous cancers developed in approximately 5.2% of patients with esophageal cancer. 6 Kato et al. reported that synchronous early gastric cancer had a prevalence of approximately 9%. 24 One possible reason for the risk of synchronous lesions is the endoscopist’s lack of experience. Therefore, careful preoperative examination by an experienced endoscopist is needed. Additionally, metachronous lesions of the esophagus after ESD are also a great problem. Multiple Lugol-voiding lesions might play a role in the development of metachronous lesions for patients with ESENs after endoscopic therapy. 25 Metachronous neoplasia of the esophagus has been reported to occur in as many as 10-15% of cases after endoscopic therapy and can develop any time after treatment.25–27 Kato et al. also found that the incidence of gastric metachronous cancer increased linearly after curative endoscopic resection for early gastric cancer. 24 In our study, the incidence of metachronous lesions showing HGID was only 1.3% at 2 years in each group. This difference may be explained by several factors such as number of years of data collection, types of neoplasia, and duration of follow-up periods. Therefore, appropriate follow-up strategies should occur for all patients who have undergone ESD for SSENs. In our study, most elderly patients preferred no treatment for metachronous or synchronous lesions, mainly because the elderly patients and their families considered the physical and mental burden of additional treatments.
There are limitations with our study. First, this was a single-center retrospective study, and selection bias cannot be avoided. Secondly, the generalizability of our conclusions may be limited by the fact that all procedures were undertaken by expert endoscopists who perform at least 250 ESDs per year. Therefore, the adverse event rate in this study may not be applicable to other centers. Third, the median follow-up time to evaluate long-term oncologic outcomes was comparatively short.
In conclusion, ESD is effective and safe in treating elderly patients with SSENs because it has a similar incidence of adverse event to that in the non-elderly. Therefore, the decision to undergo ESD for SSENs should be determined by careful pre-procedure evaluations, and old age per se should not be a contraindication to ESD. Appropriate endoscopic surveillance is necessary after ESD in patients with SSENs.
Footnotes
Funding
This work was supported by the Medical Science and Technology Development Projects of Nanjing City (grant number YKK 14061) and the National Natural Science Foundation of China (grant number 81201908, grant number 81201909).
Conflict of interest
None declared.
