Abstract
This report pertains to a case of nasal primary anaplastic lymphoma kinase-negative anaplastic large cell lymphoma, which was complicated by a fungal infection. The patient, a 66-year-old female, experienced pain in the left side of her face for 1 month. Pathological examination revealed the following: (1) primary systemic anaplastic lymphoma kinase-negative anaplastic large cell lymphoma. Immunohistochemical findings showed CD21(−), CD20 (clone L26) (−), CD3 (−), CD30 (+), Ki67 (60% +), CD5 (+), CD7 (−), CD56 (−), granzyme B (+), CK (−), CK5/6 (−), CD99 (+), Syn (−), S-100 (−), CD2 (+), CD4 (+), CD8 (−), CD43 (+), EMA (+), and anaplastic lymphoma kinase (5A4) (−). In situ hybridization results indicated EBER (−). (2) Microscopic findings suggested numerous fungal hyphae arranged in clusters or sheets, with PAS (+) and Grocott’s methenamine silver stain (+) staining confirming the presence of a fungal infection.
Introduction
Anaplastic large cell lymphoma (ALCL) is a distinct subtype of non-Hodgkin lymphoma. The expression of anaplastic lymphoma kinase (ALK) is a crucial prognostic factor; ALK-negative patients generally have a poorer prognosis. 1 Systemic ALK-negative ALCL is relatively rare in clinical practice and is characterized by widespread lymph node involvement, whereas extranodal involvement is less common than in ALK-positive ALCL. The most frequent extranodal sites of involvement include the skin (excluding primary cutaneous ALCL), soft tissue, lungs, and bones, while less common sites include the oropharynx, gastrointestinal tract, orbit, brain, and testes.2–6 ALCL primarily originating in the paranasal sinuses is extremely rare, with only a few case reports documented in the literature. This case report presents the diagnosis and treatment of a patient with nasal primary ALK-negative ALCL of T lymphocyte origin complicated by a fungal infection, who was admitted to our department.
The patient, a 66-year-old woman, was admitted to our department due to “left facial pain persisting for over a month.” Upon physical examination at admission, no external nose deformities were observed, nor was there any redness or tenderness in the paranasal sinus region. The nasal vestibule showed no redness, and the nasal septum was centered. However, the middle and lower bilateral turbinates were swollen, and a small amount of secretion was noted in the middle and inferior nasal meatus. The nasopharynx was poorly visualized. Notably, there was marked swelling of the hard palate and the left cheek, accompanied by significant tenderness. Imaging studies conducted after admission revealed that the tumor was situated on the anterior and inferior walls of the maxillary sinus. No obvious lesions were observed during nasal endoscopy. To confirm the diagnosis, the patient underwent a mass resection of the left nasal cavity and surgery on the left maxillary and ethmoid sinuses, both procedures performed under general anesthesia (Figure 1).

(a) A horizontal CT scan reveals increased soft tissue density in the left maxillary sinus, accompanied by calcification and surrounding bone destruction and resorption. (b) A PET/CT scan indicates abnormal uptake of FDG in the soft tissue of the left nasal cavity and the left maxillary sinus region. (c) An intraoperative endoscopic image displays fungal material and a white, flesh-like neoplasm.
With the aid of nasal endoscopy, we performed a combined opening of the left maxillary sinus through the middle nasal meatus and the lateral wall of the nasal cavity, aiming to fully expose the maxillary sinus cavity. Within the maxillary sinus cavity, we observed a mixed fungal–bacterial infection mass tissue. After cleaning the diseased tissue in the sinus cavity, we found gray–white solid tumor tissue on the surface of the sinus wall. Postoperatively, tissue from the mixed fungal–bacterial infection mass was sent for metagenomic sequencing, which identified Aspergillus flavus, Serratia marcescens, Haemophilus influenzae, and Streptococcus pneumoniae. The patient underwent antifungal therapy following surgery. Postoperative pathology revealed the following: (1) primary systemic ALCL was negative for ALK. Further immunohistochemical staining results showed CD21 (−), CD20 (clone L26) (−), CD3 (−), CD30 (+), Ki67 (60% +), CD5 (+), CD7 (−), CD56 (−), granzyme B (+), CK (−), CK5/6 (−), CD99 (+), Syn (−), S-100 (−), CD2 (+), CD4 (+), CD8 (−), CD43 (+), EMA (+), ALK (5A4) (−), and EBER (−). (2) Microscopic findings indicated numerous fungal hyphae arranged in clusters or sheets, with PAS (+) and Grocott’s methenamine silver stain (+) staining consistent with fungal infection (Figure 2). The patient received 2 weeks of antifungal treatment, followed by CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone) chemotherapy. At the 6-month follow-up, the patient’s condition remained stable (Figure 3).
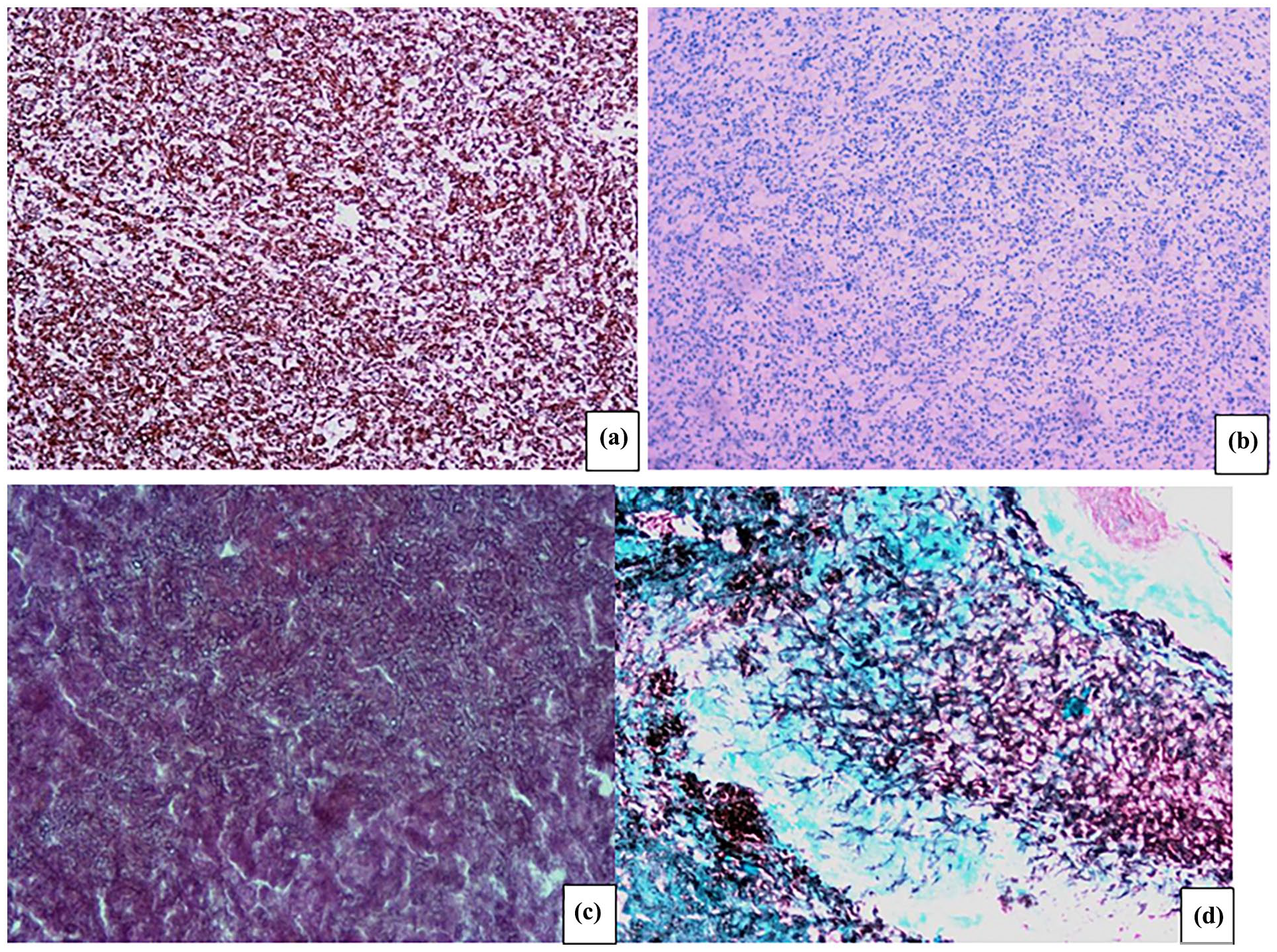
(a) IHC image demonstrating CD30 positivity. (b) IHC image indicating ALK negativity. (c) High-power microscopic view of fungi. (d) Fungal Grocott’s methenamine silver stain image (all images are magnified 400 times).

(a) A horizontal CT scan at 6 months post-follow-up reveals postoperative changes in the left maxillary sinus, including a visible soft tissue density within the sinus cavity and bone destruction of the maxillary sinus wall. (b) A PET/CT scan indicates a significantly reduced soft tissue mass in the left nasal cavity and left maxillary sinus compared to previous scans, with no abnormal FDG uptake and no radiological evidence of abnormal bone activity in the adjacent maxillary sinus wall. (c) An endoscopic image displays smooth mucosa within the maxillary sinus cavity.
Discussion
ALCL is a type of peripheral T-cell lymphoma, first reported by Stein in 1985. It exhibits various pathological morphologies and an immunophenotype characterized by, strong expression of CD30 with varying degrees of loss of T-cell markers. The WHO 2022 Classification of Lymphoid Neoplasms (4th edition, revised) categorizes ALCL into four distinct entities: ALK-positive ALCL, ALK-negative ALCL, primary cutaneous ALCL, and ALCL associated with breast implants.1–3,7 In 50% of cases, there is a chromosomal translocation t(2;5) that occurs, resulting in the fusion of the ALK gene at 2p23 with the nucleophosmin gene at 5q35, leading to the expression of the ALK protein. 4 Irrespective of ALCL subtype, strong expression of CD30 can be detected on the cell membrane and Golgi region. Another peculiar feature of ALCLs is the significant repression of the T-cell expression program. Even though nearly all ALCL cases (74%–90%) show clonal T-cell receptor (TCR) gene rearrangements, both ALK positive and ALK negative ALCLs lack TCRs and related signaling molecules such as CD3, ZAP70, LAT, and SLP76. 8 ALCLs are characterized by high morphological heterogeneity, ranging from small-cell neoplasms to cases where very large and anaplastic cells predominate.
The common type of ALCL is characterized by a diffuse, solid, and cohesive proliferation of pleomorphic large cells. “Hallmark” cells (with horseshoe-shaped nuclei) and “wreath-like” multinucleated cells are frequently observed. Notably, the tumor may exhibit a sinusoidal growth pattern within the lymph node. 9
In this case, preoperative nasal endoscopy did not reveal any obvious neoplasm in the nasal cavity. Imaging suggested a combination of fungal sinusitis and a sinus tumor, prompting the need for sinus surgery to confirm the diagnosis. During the procedure, the maxillary sinus was opened, revealing a large mixed fungal–bacterial infection mass within the sinus cavity, leading to an initial diagnosis of invasive fungal sinusitis. After the complete removal of the lesion, a solid tumor was discovered in the anterior wall of the maxillary sinus and in the lacrimal recess, which was then sent for pathological examination. Before receiving the pathological results, invasive fungal sinusitis remained the primary diagnosis, and the mixed fungal–bacterial infection mass was sent for metagenomic testing, which identified A. flavus, S. marcescens, H. influenzae, and S. pneumoniae. While awaiting the pathological results, antifungal and anti-inflammatory treatments were administered. Although the pathological report confirmed ALK-negative ALCL, systemic antifungal therapy was continued for 2 weeks due to the concurrent fungal infection, followed by CHOP chemotherapy. 10 This chemotherapy regimen resulted in a favorable treatment outcome, although follow-up is ongoing to monitor for potential recurrence. Regrettably, as of the publication date of this article, we had only conducted 6 months of follow-up for the patient, which is far from sufficient for tumor surveillance. Moving forward, it is imperative to strengthen follow-up efforts to continuously monitor the patient’s treatment outcomes and quality of life in subsequent phases.
Currently, there is no standardized treatment for ALK-negative ALCL. The most commonly used regimen is CHOP chemotherapy, based on experience with aggressive B-cell lymphomas. Other chemotherapy regimens may also be effective, but none have shown ideal outcomes so far, and tumors tend to relapse. 3 Some studies suggest that in young and otherwise healthy patients with refractory or recurrent ALK-negative ALCL, high-dose chemotherapy followed by autologous stem cell transplantation after achieving complete remission may lead to better treatment outcomes. 11
The preoperative sinus CT of the patient showed typical imaging characteristics of fungal sinusitis, with irregular patchy or punctate calcifications visible within the maxillary sinuses, and areas of bone defect in the affected sites. Despite this, the possibility of a malignant tumor cannot be entirely ruled out. Nasal cavity and paranasal sinus squamous cell carcinoma, nasopharyngeal carcinoma, and sinus sarcoma cannot be excluded. According to the literature, early-stage non-Hodgkin’s lymphoma is characterized by invasive growth, roughened mucosa, contact bleeding, and some bone resorption, but without significant bone destruction or defects. This often results in mucopurulent secretions in the maxillary, ethmoid, and sphenoid sinuses. It is precisely because of the slow growth of the tumor within the sinus cavity, which affects the drainage of the sinus ostia, that secondary sinus cavity infections are triggered, thereby causing local symptoms in the patient.
In summary, primary nasal ALK-negative ALCL is a rare malignant tumor. Given the rarity of this disease, early diagnosis and specific treatment are critical. Therefore, for suspicious malignant lesions in the nasal area, ALCL could be considered as part of the differential diagnosis to enable early intervention.
Conclusion
This report concerns a case of nasal primary ALK-negative ALCL complicated by fungal infection. The patient received antifungal treatment for 2 weeks, followed by CHOP chemotherapy. A favorable outcome was achieved, which suggests that similar diseases can receive personalized treatment such as chemotherapy + antifungal treatment, which provides new treatment ideas.
Footnotes
Ethical considerations
The study was reviewed and approved by the Ethics Committee of Ganzhou People’s Hospital.
Consent for publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Author contributions
Yun Huang compiled the patient’s data, reviewed the literature, and wrote the article; Yi-bin Liu was the patient’s surgeon; Liang Luo and Long-Gui You helped perform the analysis with constructive discussions; Xin-Tao Wang was responsible for the review and revision of the article; and Huan Li helped perform the data collection. All authors have given final approval of the submitted version of the article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data presented in this study are available on request from the corresponding author.
