Abstract
Sporotrichosis infections may cause cutaneous lesions mimicking other infectious or non-infectious causes such as pyoderma gangrenosum. We present a case of cutaneous sporotrichosis misdiagnosed as pyoderma gangrenosum and treated with immunosuppressants for 17 months leading to exacerbation and atypical morphology mimicking Histoplasma organisms on biopsy. Exclusion of infection prior to diagnosing pyoderma gangrenosum is important to prevent iatrogenic immunosuppression, demonstrating the challenges with application of the updated pyoderma gangrenosum diagnostic criteria.
Keywords
Introduction
Cutaneous sporotrichosis infections can be difficult to visually distinguish from other ulcerative or infiltrative lesions. The differential for ulcerative lesions includes infectious and non-infectious causes such as malignancies, vascular disorders, and inflammatory disorders. Sporotrichosis lesions have been misdiagnosed as ulcerative pyoderma gangrenosum (PG) due to similar clinical appearance or lack of positive culture or histological stains.1–8 Weenig et al. 1 found that 11/64 (17%) lesions misdiagnosed as PG were caused by primary cutaneous infections, with the pathogen being Sporothrix schenckii in 5/11 cases. As immunosuppressive therapies are standard of care for treating PG, exclusion of infectious etiology is especially important prior to starting therapy. Updated ulcerative PG diagnostic criteria require a biopsy demonstrating neutrophilic inflammation, making diagnosis of PG no longer purely a diagnosis of exclusion. 9 This case highlights the importance of exclusion of infection as a diagnostic criterion.
Here, we report a case of cutaneous sporotrichosis misdiagnosed and treated for 17 months as ulcerative PG, leading to exacerbation and atypical morphology mimicking Histoplasma organisms on re-biopsy.
Case report
A 78-year-old male presented with a non-healing left arm ulcer in June 2017 (Figure 1). A 5-mm punch biopsy showed extensive neutrophilic inflammation and periodic acid–Schiff (PAS) staining for fungal organisms was reported as negative, and simultaneous tissue culture was not done. The case diagnosis was compatible with PG. Various immunosuppressive treatments were tried for a year leading to worsening progression.
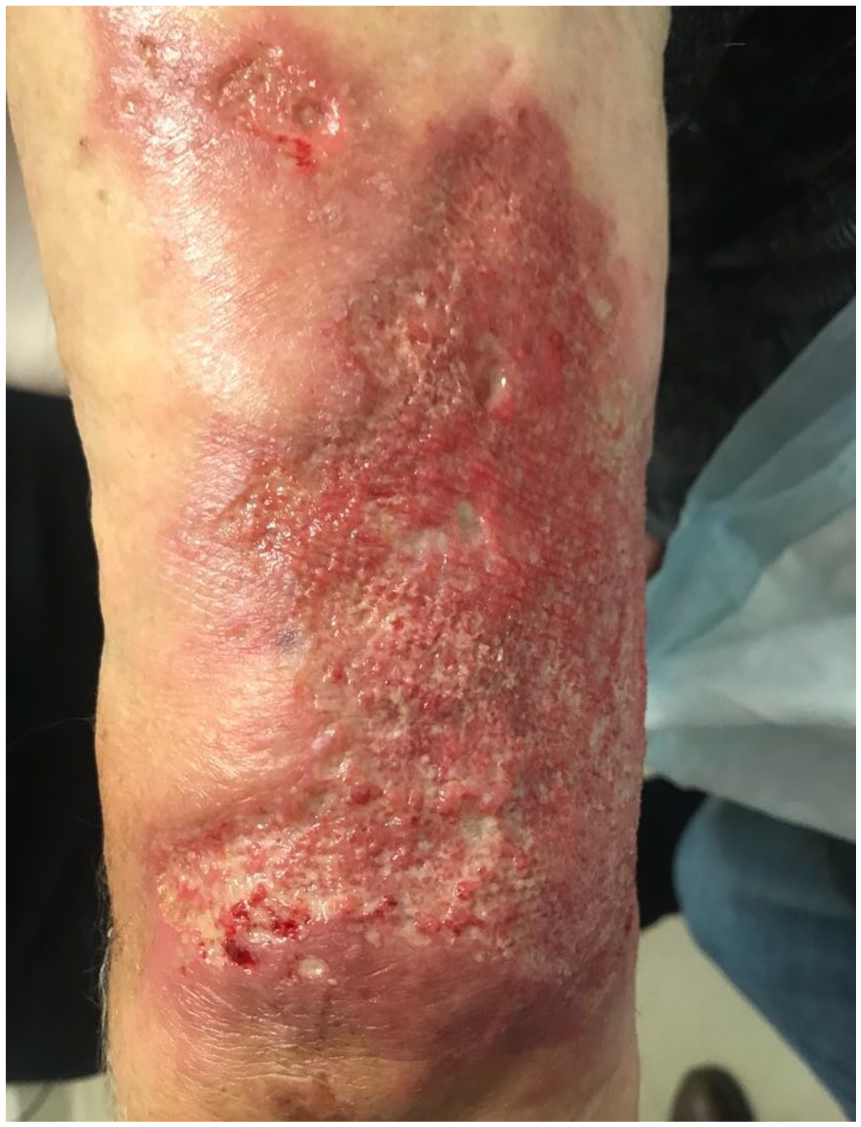
View of the patient’s left upper arm prior to antifungal treatment showing cribriform ulceration.
The patient presented to our clinic November 2018 and was being treated with IV immunoglobulin (IVIG), prednisone, and ustekinumab for 7 months with severe disease progression. Treatment with cyclosporine and mycophenolate previously failed due to hypertension and leg swelling, and gastrointestinal disturbances, respectively. No risk factors such as environmental exposures, previous occupation, and travel were reported. Lymphangitis and lymph node involvement was not present.
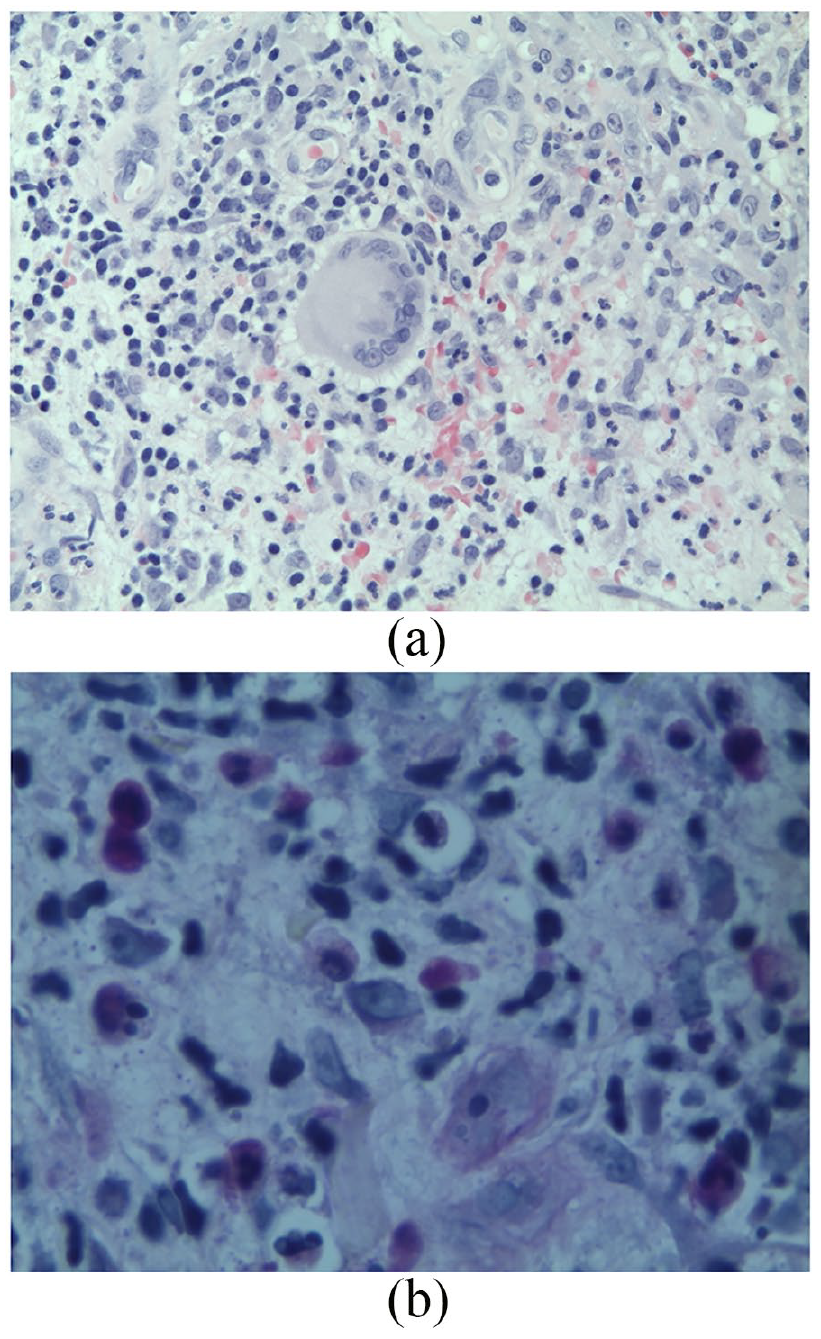
A 4-mm punch biopsy of the ulcer edge showed numerous three- to four-micron intra-cytoplasmic yeasts within histiocytes (Figure 2(a)). Hundreds of intracellular organisms were PAS stain-positive and Grocott’s methenamine silver stain-positive (Figure 2(b) and (c)), ranging from 3 to 12 microns with some budding yeasts. Despite morphological consistency with Histoplasmosis duboisii or capsulatum, tissue cultures led to isolation of S. schenckii species complex, an interesting report in histopathology.

Biopsy from November 2018. (a) Hematoxylin and eosin stain (magnification 400×) showing intra-histiocytic and extracellular yeasts measuring from 3 to 5 microns, resembling Histoplasma organisms. (b) Periodic acid–Schiff stain (magnification 400×) showing yeasts measuring 3–12 microns in diameter within histiocytes. (c) Grocott’s methenamine silver stain (magnification 400×).
Prednisone was tapered and itraconazole (200 mg/day) was promptly started. Treatment continued for 4 months leading to complete wound regression and healing of the left arm (Figure 3). He received standard wound care.

View of the patient’s left upper arm 3 months post-treatment.
A second opinion of the initial biopsy in June 2017 reported the rare presence of PAS-positive yeast (Figure 4(a) and (b)) in the original biopsy.
Biopsy from June 2017. (a) Hematoxylin and eosin stain from the biopsy showing suppurative dermatitis below an ulcer, in which focally a multinucleated histiocyte is visible (magnification 400×). (b) A single PAS-positive yeast organism measuring 4–6 microns found on re-examination (magnification 640×).
Discussion
We report a case of chronic S. schenckii cutaneous infection initially misdiagnosed as PG due to a lack of tissue culture complemented with overlook in identifying PAS-positive organisms. This led to mistreatment and severe exacerbation of the patient’s lesion for 17 months. Re-biopsy showed atypical histologic morphology of S. schenckii as hundreds of intra-cytoplasmic yeasts within histiocytes, mimicking Histoplasmosis organisms. Tissue culture results confirmed S. schenckii infection. This picture is not usually seen in a biopsy of typical sporotrichosis cases and was suspected to be caused by chronic iatrogenic immunosuppression.
S. schenckii is an environmentally ubiquitous thermodimorphic fungus existing worldwide in a filamentous form at 25°C and transforming into yeast-like cells at 35°C–37°C. Common sources of infection to trauma-induced wounds include rose thorns, soil, hay, decaying vegetation, plants, animal feces, and zoonotic transmission from cats. 10
Cutaneous manifestations of sporotrichosis include lymphocutaneous, fixed cutaneous, and disseminated cutaneous. In cutaneous infections, a papulonodular lesion develops within 2–4 weeks of inoculation, followed by ulceration and purulent discharge. Lymphocutaneous infections occur with subsequent spread to lymphatic channels and accounts for up to 80% of sporotrichosis infections.11,12 Extracutaneous sporotrichosis manifestations including pulmonary, osteoarticular, mucosal, disseminated, and meningeal sporotrichosis have been reported in immunocompromised individuals.11,13
An ulcerated lesion may have an infectious or non-infectious differential (Table 1). Wound culture is the diagnostic gold standard, but may take more than 5 days to grow. A PAS-positive stain was reported in only 3.2%–19.7% of sporotrichosis infections due to low fungal burden in early infections.14,15
Differential diagnosis of cutaneous sporotrichosis.

Itraconazole is the first-line treatment for cutaneous sporotrichosis infections, with open treatment trials reporting 90%–100% success rate if continued up to 4 weeks after treatment resolution.16–18 Liposomal amphotericin B is often used for severe or extracutaneous manifestations.3,19 Local heat therapy has also been reported to be effective as temperatures >38°C minimize sporotrichosis growth.6,20
Many reported cases of sporotrichosis misdiagnosed as PG were treated with immunosuppressive treatment leading to exacerbation (Table 2). Delayed diagnosis led to reports of superinfection, disseminated sporotrichosis, and osteoarticular sporotrichosis.7,8 Because both ulcerative PG and fixed cutaneous sporotrichosis occur in absence of lymphatic involvement, the clinical picture is difficult to distinguish.3,8 In addition, 50% of sporotrichosis infections are not correlated with a history of trauma, gardening, or rose exposure. 2
Case reports of sporotrichosis infections misdiagnosed as pyoderma gangrenosum.

PG: pyoderma gangrenosum; PAS: periodic acid–Schiff; IVIG: IV immunoglobulin; BID: two times a day; TID: three times a day.
In 2018, Maverakis et al. published revised diagnostic criteria for diagnosing ulcerative PG.8 The major diagnostic criterion of histological presence of neutrophilic infiltrate at the ulcer edge must be met along with four other minor diagnostic criteria (Table 3). Under previous diagnostic criteria of exclusion, our case would not have satisfied the PG diagnostic criterion as a proper exclusion of infection was not done. However, under the new diagnostic criteria where exclusion of infectious causes is not required, potential for misdiagnosis and treatment exacerbation increases.
New diagnostic criteria for ulcerative pyoderma gangrenosum.

Source: Modified from Maverakis et al. (2018).
Ulcers occurring at trauma sites should extend past the area of trauma.
Ultimately, this case highlights the importance of tissue culture in excluding infectious causes before diagnosing PG and presents an atypical S. schenckii morphology mimicking Histoplasma organisms in chronically immunosuppressed infections.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Patient provided written and verbal informed consent for patient information and images provided (Figures 1 and ![]() ).
).
