Abstract
Invasive fungal infections are considered a threat to hematological malignancy patients (HM). We report here a rare case of pulmonary aspergillosis in a patient diagnosed with leukemia at King Abdulaziz University Hospital. The patient received three cycles of chemotherapy and presented with febrile neutropenia and his fungal culture was repeatedly negative while signs of aspergillosis in a computed topography (CT) scan were evident. The patient was successfully recovered after 6 weeks of voriconazole treatment.
Introduction
Episodes of invasive fungal infections (IFIs) have increased substantially in patients with hematological malignancies (HM) with the incidence varying among countries.1–3 Opportunistic fungal pathogens are the main causative agents, including Candida, Aspergillus, and Cryptococcus. 4 There are multiple risk factors for IFI in patients with leukemia, including neutropenia, steroids, high-dose chemotherapy and foreign bodies medical devices, genetic susceptibility, advanced age, and the presence of comorbidities.5,6 IFI infection site may vary from superficial infections to systemic infections. However, respiratory infection was the most common. Incidence of leukemia increased worldwide. Recently, there has been an increasing number of cancer patients in the Saudi population related to various etiological factors on environmental, molecular, and genetic levels. Data from the Saudi Cancer Registry showed that leukemia is the fifth-ranking number among overall malignancies in Saudi Arabia. 7 A study from Saudi Arabia showed that leukemia and colorectal cancer were the most common types of malignancies in the male gender while among Saudi females, breast cancer, thyroid cancer, and colorectal cancer were the most prevalent. 6 There is a paucity of reported cases of pulmonary aspergillosis in leukemic patients in Saudi Arabia
Thus, over the past year 2021–2022, we searched the King Abdulaziz Hospital hematological patients’ database including clinical history, reports of radiological examination and results of the microbiological test from sputum, bronchoalveolar lavage (BAL), and blood samples, and microbiology laboratory test and found one probable case of aspergillosis in a Saudi male patient diagnosed with Acute myeloid leukemia (AML).
Case presentation
A 36-year-old Saudi male patient with AML in relapse and with a history of bacteremia related to central line infections. His chemotherapy includes an induction phase with cytarabine and daunorubicin followed by a consolidation phase with three cycles with high-dose cytarabine. Early in his induction phase, he developed oral lesions and bilateral facial palsies. A viral swab from the oral lesions was positive for herpes simplex virus-1 and he was started on acyclovir course 400mg orally twice per day and continued as prophylaxis for the rest of his leukemia treatment. He also developed catheter-related bloodstream infection with Escherichia coli and had a superficial wound on his scalp where a wound swab culture grew a multidrug-resistant Acinetobacter baumannii. The patient was on standard antifungal prophylaxis with oral fluconazole 400 mg daily for 30 days of induction. During each consolidation he got approximately 10 days of fluconazole 400 mg daily for prophylaxis during the neutropenia phase (three consolidations; Figure 1). He had no known drug allergy. The patient was presented with febrile neutropenia during the second induction course, associated with cough and shortness of breath. The patient was treated in a leukemia room in a dedicated ward for HM. The standard leukemia bed is a single-bed room with a private washroom. Positive pressure rooms and HEPA filters were available but only used for patients undergoing stem cell transplantation. In this case, the patient was not in a laminar airflow environment during chemotherapy, which could have influenced the risk of fungal infection. A septic screen with culturing the blood and urine in addition to a chest X-ray (Figure 2) was done. Frontal chest radiograph demonstrated bilateral coalescent nodular opacities in the left upper lobe. He had a high-grade fever and was started empirically on meropenem 500 mg intravenously every 6 h and intravenous vancomycin IV was added after the results of blood culture were positive for growth of methicillin-resistant Staphylococcus aureus (MRSA). He developed aspergillosis 10 days after the second consolidation with Ara-C. Fungal investigation was performed culturing the patient’s sample on Sabouraud dextrose agar supplemented with chloramphenicol yielded no fungal growth from each specimen. He improved clinically for 4 days, and the pattern of fever improved but after that started to have a dry cough and high spikes of fever. As per protocol, caspofungin (70 mg loading then 50 mg IV daily) was started to cover for possible fungal infection. Computed topography (CT) scan to look for IFIs. As the fever subsided, his respiratory symptoms continued to worsen. The requested CT of his chest showed multiple bilateral lung nodules of different sizes. The biggest nodule was located in the left upper lobe and measured 2 cm. A few of these nodules demonstrated a surrounding ground-glass appearance giving a positive hollow sign. The picture was suggestive of a lung fungal infection typical of aspergillosis (Figure 3(a)). Based on CT chest results this case was labeled a probable diagnosis of aspergillosis according to the criteria European Organization for Research and Treatment of Cancer (EORTC). 8 After completing 5 days of caspofungin he was switched to voriconazole 300 mg IV BID to complete 6 weeks on the therapeutic dose. His BAL sample culture revealed a positive growth for klebsiella pneumoniae which was sensitive to several antibiotics but resistant to ampicillin, amoxicillin, clavulanic acid, cefazolin, and cefuroxime. A month later, a BAL sample was obtained and was positive for MRSA resistance to ciprofloxacin and oxacillin but sensitive to other antibiotics. The patient was tested several times for COVID-19 and the results were negative for COVID-19 and fungal culture remained negative. Further interval improvement of the lung lesions was reported on a repeated CT scan of the chest by the end of 6 weeks (Figure 3(b)) and then he was switched to prophylactic dose voriconazole. He had an early relapse within a year from his last consolidation and was started on a second cycle of induction chemotherapy according to FLAG-Ida salvage protocol containing a combination of fludarabine, cytarabine, granulocyte-colony stimulating factor, and idarubicin. Unfortunately, despite achieving remission, he was not a candidate for an allogeneic stem cell transplant, and he relapsed after 3 months, was switched to comfort care, and succumbed to death 16 months after the initial aspergillosis diagnosis.
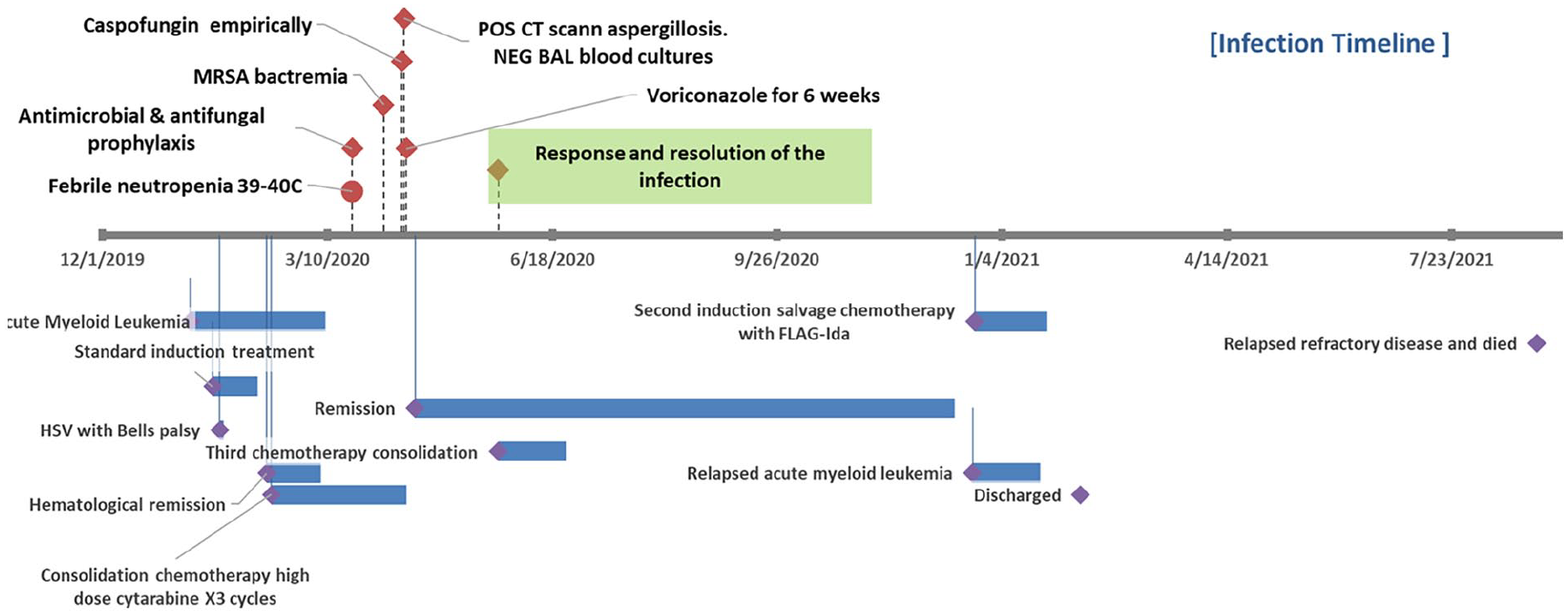
Showing the case progress of a 36-year-old relapsed AML male patient.

Frontal chest radiograph demonstrating bilateral coalescent nodular opacities; the largest is in the left upper lobe (arrow).

Axial Chest CT in lung widow showing progression/recovery before and after treatment with voriconazole. (a) On presentation demonstrating multiple bilateral variable size lung nodules. Some nodules are surrounded with ground-glass density representing the Halo Sign measuring 2 cm (arrow). (b) Two months later after treatment showing nodules resolution.
Discussion and conclusions
Generally, IFIs diagnosis is challenging, and treatment protocol must be selected carefully. Previous surveillance found mortality rates of invasive mycosis as high as 22.4%.7,9 Particularly in HM patients as they represent the top high-risk population of aspergillosis due to their low neutropenia levels. Previously, IFI has been reported in 4.5% of HM patients caused by yeast more than mold. 10 Another study reported IFI among AML to be the most common disease represented. 11 There have been significant changes in trends of hematology malignancy populations with an increase in leukemia patients in Saudi, hence, an increase in IF was predicted. More clinical studies on common fungal causative agents will help to establish an appropriate screening and diagnostic strategy and to evaluate possible environmental and genetic factors for fungal infections. 11 Many studies have reported Candidiasis in these vulnerable populations; however, mold infections remain unreported. 12 As a result, understanding invasive aspergillosis and succeeding in treatment may be hindered by available mycological diagnostic methods. In Saudi, mycological diagnosis remains a challenge, especially in case of culture failure and the absence of molecular and serological diagnostic tools. Having a trained mycologist and radiologist is crucial for applying the EORTC and Mycoses Study Group (MSG) guidelines and supporting treatment decisions. 8 Diagnosis of IFI in HM is challenging as the suspected patients may be critically ill presenting with many non-fungal specific symptoms at risk of other nosocomial coinfections. 12 There is active reporting of IFI in the United States and Europe, while limited reports are available from the Middle East and North Africa region, especially Saudi Arabia. The lack of IFI surveillance programs, especially clinical characteristics and prognostic factors, limits our understanding of the disease aspects and prevents clinicians from applying improved diagnostic tools and strategies.
In this case, we describe a successfully treated AML adult patient who developed bilateral pulmonary infiltrations which progressed to multiple cavities that regressed to scarring and fibrosis. The patient was neutropenic and received broad-spectrum antibiotics for gram-negative septicemia. The patient showed a remarkable clinical improvement accompanied by resolution of the pulmonary changes on the radiography over a period of 1 month. Our suspicion of fungal pneumonia was confirmed by a clinical and radiologic setting, especially in a highly predisposed leukemic patient with neutropenia and on broad-spectrum antibiotics.
One of the limitations in this case report is the lack of serological testing (GM antigen and B-D glucan) which is important for the diagnosis of pulmonary aspergillosis. However, limitations in the availability and accessibility of these tests in our clinical setting and many of the West region hospitals. We have reported a Saudi Arabian fungal diagnostic shortage in antifungal susceptibility testing and serological and molecular methods in previous national surveillance as they are rarely performed and mostly outsourced. Future case reports could benefit from incorporating these tests to strengthen the diagnosis.
The diagnosis was considered probable rather than proven as cultures were repeatedly negative, and no histology identification or molecular testing was performed. According to the guidelines for the diagnosis of IFIs in leukemia patients, such as those from the EORTC and the MSG, this case was probable fungal infection due to high clinical suspicion of fungal infection and supporting evidence but no direct isolation of the pathogen including. symptoms and signs consistent with fungal infection, evidence of fungal infection on chest X-ray, and CT scan.
Our case may encourage clinicians to consider advanced diagnostic tools when cultures fail to detect suspecting fungal infections in high-risk populations. While pulmonary aspergillosis is a well-recognized complication of AML, its prevalence among patients at King Abdulaziz University Hospital in Saudi Arabia appears to be limited based on our internal data and the available literature. Previous investigations conducted at our institution over the past 5 and 14 years, including a recent review of cases and diagnostic tests, have revealed a low incidence of aspergillosis with a total of three cases.
While specific data on aspergillosis in Saudi Arabia may be limited, there are ongoing efforts to establish a national surveillance system to aggregate and analyze epidemiological data on fungal infections. This system will be instrumental in providing a more comprehensive understanding of the burden of fungal diseases in the country. The inclusion of this case report contributes to the growing body of literature on pulmonary aspergillosis in the Saudi context. By highlighting the occurrence of this condition at our institution, we aim to raise awareness and encourage further research to elucidate the specific risk factors and prevalence of aspergillosis among AML patients in Saudi Arabia.
In conclusion, Early diagnosis of pathogenic mold is crucial and there is e a relatively increased incidence of fungal pulmonary complications in our patients with AML and HM in general (paper in publication process). Although this patient improved to antifungal treatment and the fungal infection completely resolved, early screening for fungal infections by using serological and molecular tools is needed for preventive measures, aiming at limiting lung damage and detecting possible resistance mutations.
Footnotes
Acknowledgements
Many thanks to parents for supporting the publication of clinical data.
Author contributions
A.M.K. wrote the manuscript; A.S.B., S.M.B., and O.O.R. reviewed the hematological aspects; G.A.A. reviewed radiological aspects; O.S. and E.I.A. reviewed the manuscript before final submission.
Data availability
All data generated or analyzed during this study are included in the submitted article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Consent for publication
All authors are in agreement with the content of the manuscript.
