Abstract
Candida auris (C. auris), a globally emerging pathogen, has posed a significant threat to hospitalized individuals during the COVID-19 in Vietnam. This case series reported (1) common patterns in five patients with non-multidrug-resistant C. auris infections (multiple comorbidities, severe-to-critical illness, use of broad-spectrum antibiotics, or history of surgery/invasive procedures) and (2) high rate of C. auris-associated mortality in this medical setting (four deaths out of five cases). Further studies are needed to (1) identify risk factors for C. auris infections and mortality and (2) investigate the effects of screening and preventive measures for C. auris, especially in low-resource settings.
Introduction
Candida auris (C. auris), first reported in Japan in 2009, 1 is an emerging pathogen that poses serious threats to patient outcomes globally. 2 C. auris has been repeatedly found in many infections, including fungemia, skin and soft tissue infections, respiratory tract infections, urinary tract infections, or otomycosis.1,3 More importantly, it can cause opportunistic infections in immunocompromised individuals, 4 worsen treatment outcomes, and increase the mortality risk in critically ill patients.5,6
C. auris infections have been reported in five continents (Africa, America, Asia, Europe, and Oceania). 7 There is a growing number of countries that have isolated C. auris from patient samples.8 –10 This can increase the risk of outbreaks, as C. auris can spread rapidly and widely following the emergence of the first isolate.2,11,12 The mortality risk of this fungal infection is even more burdensome, especially in low-resource settings with poor infection control since C. auris has been shown to resist many antifungal agents.11 –15
Given the vulnerability of patients with severe medical conditions, an outbreak of C. auris infections has caused a tremendous burden on hospitalized individuals during the COVID-19 in Vietnam. As these were among the first C. auris isolates in the country, we reported a single-center case series to describe the impacts of this emerging pathogen on the course of treatment and health outcomes in a middle-income setting. We also reported the antifungal susceptibility of these isolates to clarify the resistant patterns of C. auris in this outbreak.
Case series
Starting from the fourth wave of COVID-19 in Vietnam (from August 2021 to January 2023), we recorded five female Vietnamese patients (Asian race; one with moderate COVID-19, one with post-COVID) whose blood samples were found positive for C. auris. C. auris was detected using real-time polymerase chain reaction and rapid clade-identification method (all being clade 1).16,17 All of these samples were drawn at the intensive care unit (ICU). The isolates of C. auris were tested for antifungal susceptibility using the broth dilution method, as described in the document of the Centers for Disease Control and Prevention. 18 The cases were reported as follows (with the characteristics and treatment timelines being summarized in Tables 1 and 2, respectively).
Characteristics of patients with Candida auris infections.
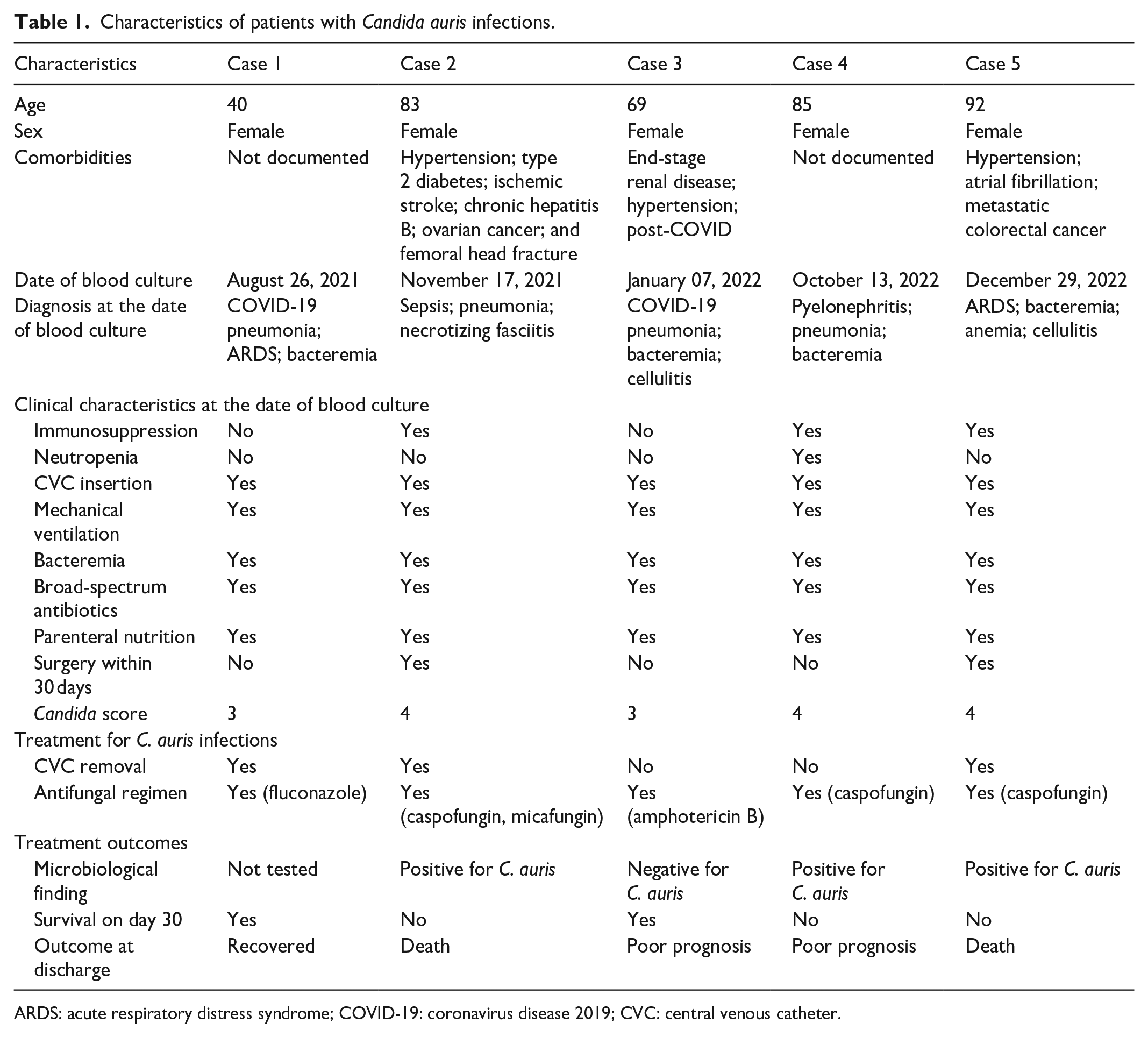
ARDS: acute respiratory distress syndrome; COVID-19: coronavirus disease 2019; CVC: central venous catheter.
Timelines of primary events of the five cases.

CVC: central venous catheter; ICU: intensive care unit.
Case 1
A 40-year-old female with no medical conditions was hospitalized for fever, cough, dyspnea, and diarrhea, and had a diagnosis of moderate COVID-19 (classified by the National Institute of Health 19 ) upon admission. As the disease progressed severely, she developed acute respiratory distress syndrome and bacteremia. The patient was inserted central venous catheter (CVC), was mechanically ventilated, and received midazolam, rocuronium, enoxaparin, broad-spectrum antibiotics (for Acinetobacter baumannii and Pseudomonas aeruginosa), dexamethasone, tocilizumab, and parenteral nutrition. On day 22, C. auris was isolated (antifungal susceptibility in Table 3). Fluconazole was initiated for C. auris on day 27 (upon receiving a positive culture result), with a loading dose of 800 mg and a maintenance dose of 400 mg, q24h. Clinical status started to improve after 3 days (ventilator weaning, clinical sobriety, and oral nutrition). By day 41, when fluconazole was discontinued, no other blood culture had been tested for C. auris. The patient recovered and was discharged on day 45. We found no re-hospitalization or any other adverse events within 90 days of discharge.
Antifungal susceptibility of Candida auris isolates.

These breakpoints were tentative at that time and did not imply any definitive resistance status of C. auris.
Only these antifungal agents were available as intravenous products at that time.
No test was conducted.
MIC: minimal inhibitory concentration; N/A: not available.
Case 2
An 82-year-old female with a history of hypertension, ischemic stroke, chronic hepatitis B, ovarian cancer, and femoral head fracture was hospitalized for sepsis, pneumonia, and necrotizing fasciitis in the buttocks. Treatment included debridement and drainage (for the necrotizing fasciitis), broad-spectrum antibiotics through CVC, parenteral nutrition, and mechanical ventilation. After debridement, the patient received intensive resuscitation, wound care, and low-dose norepinephrine. On day 67, C. albicans was isolated from a blood sample. Caspofungin was initiated for C. albicans on day 71 (upon receiving positive culture result), with a loading dose of 70 mg and maintenance dose of 50 mg, q24h. On day 76, C. auris was also isolated (antifungal susceptibility in Table 3), although the patient was receiving standard antifungal therapy. After a 14-day course of caspofungin, the patient was switched to micafungin 100 mg, q24h (on day 85). Despite intensive treatment, the clinical status continued to deteriorate. On day 88, the patient died of sepsis, pneumonia, multiple organ dysfunction syndrome, and fungemia.
Case 3
A 69-year-old female with post-COVID and end-stage renal disease (currently on conventional hemodialysis) was admitted to the ICU for critical COVID-19 pneumonia, bacteremia, cystitis, cellulitis in two arms, and upper gastrointestinal bleeding. The patient was mechanically ventilated and received broad-spectrum antibiotics with intermittent hemodialysis. The patient’s fevers persisted after a 7-day course of standard antibiotics (38.0°C–41.0°C). On day 16, C. auris was isolated. Amphotericin B was initiated for C. auris on day 19 (upon receiving a positive culture result), with a standard dose of 50 mg, q24h, for 15 days. Blood cultures were negative for C. auris on days 27, 33, and 45. On day 42, clinical status started to deteriorate as the patient had signs/symptoms of sepsis and a low platelet/red blood cell count. On day 47, the patient was discharged with a poor prognosis (severe sepsis and septic shock) upon the family’s request. The patient died after 1 day of discharge.
Case 4
An 85-year-old female with left ureterolithiasis and pyelonephritis was indicated by contrast-enhanced multislice computed tomography. After the procedure, the patient had symptoms of iopromide-suspected grade-2 anaphylaxis and was admitted to the ICU for further management. At the ICU, the patient’s infection progressed severely under anaphylaxis management, which required CVC insertion, antibiotics for the complicated urinary tract infection, and intensive care. The patient was then placed with a JJ stent through the left ureter. During the treatment course at the ICU, the patient was ventilated due to respiratory failure, which resulted in ventilator-associated pneumonia that required broader-spectrum antibiotics and parenteral nutrition. On day 17, caspofungin was initiated for the isolated C. auris (antifungal susceptibility available on day 28, Table 3), with a loading dose of 70 mg and maintenance dose of 50 mg, q24h. On day 24, C. auris was still isolated from the blood culture. Clinical status continued to deteriorate (septic shock on day 25). On day 27, the patient was discharged with a poor prognosis upon the family’s request. The patient died after 1 day of discharge.
Case 5
A 92-year-old female with bowel obstruction due to metastatic colorectal cancer was hospitalized for a colostomy. After the procedure, the patient was admitted to the ICU, ventilated, inserted a CVC, and received broad-spectrum antibiotics with intensive care. On day 63, caspofungin was initiated for the isolated C. auris (antifungal susceptibility in Table 3), with a loading dose of 70 mg and maintenance dose of 50 mg, q24h. Blood culture was positive for C. auris on day 66 but was negative on day 71. Caspofungin was discontinued on day 76. The patient died on day 77 with septic shock and C. auris fungemia as attributable causes.
Discussion
In this case series, we identified many patterns that have been reportedly risk factors for C. auris infections, including multiple comorbidities, severe-to-critical illness, admission to the ICU, exposure to broad-spectrum antibiotics, or history of surgery/invasive procedure.2,12,20 –23 While C. auris has been cultured from many organs or sites within the human body,24,25 we only found its isolates in the blood samples. This suggests that some invasive procedures or surgeries could have facilitated the entry of C. auris into the bloodstream or sterile sites from the internal (patient’s skin) or external sources (caregiver or hospital environment), as reported in previous studies.20,22,25,26 The risk of invasive C. auris infections is even greater if the patients have moderate-to-critical COVID-19, are on steroid therapy, or are admitted for intensive treatment at the ICU.14,22 –25,27 –30 The joint effects of these factors could be observed clearly in case 1 of this study, where the patient did not have any significant medical conditions before admission. Moreover, the use of broad-spectrum antibiotics, as for all five cases in this study, could cause dysbiosis, which is likely associated with the in vivo intestinal colonization and dissemination of C. auris. 31 Noticeably, prior use of antifungal therapy may also induce selective pressure of resistant C. auris, as reported in case 2 of this study, thus increasing the risk of invasive infections.23,24,27 These factors, combined with the inadequate infection control strategies and long survival of C. auris on many surfaces, 32 have partially explained the outbreak of this pathogen in our facility.
Other reported C. auris outbreaks worldwide also had similar patterns.33,34 This suggests that in high-risk settings, for example, at the ICU, the emergence of one C. auris isolate may trigger a series of C. auris infections, leading to potential outbreaks in patients with multiple risk factors. While predicting such infections is challenging or even unfeasible in low-middle-income countries, it is more practical to prevent the outbreaks following these cases by implementing extensive infection control strategies. 24 However, screening for C. auris is still useful if resources are available and affordable. This is particularly important if a patient exposed to risk factors of C. auris gets transferred to another cleaned setting. The two cases reported in Australia and Taiwan are typical examples of this screening program.35,36 In the context of global threats posed by emerging pathogens, it is also essential that low-middle-income countries establish a central reporting system for effective monitoring, surveillance, and prevention of infections, ensuring timely interventions and coordinated public health responses.
As C. auris is notorious for its high resistance against many antifungal agents (especially amphotericin B, fluconazole, etc.),11 –15 treatment options are often limited, with echinocandin drug as the recommended initial therapy. 37 While all isolates in this case series were sensitive to most available antifungal agents and most patients were given optimal regimens, only one case recovered. Since this study design was not appropriate to draw any confirmative conclusion, we hypothesized that in patients with C. auris infections, effective antifungal regimens alone might not improve treatment outcomes. This is based on the fact that these infections are more likely to develop in patients with severe-to-critical illness or multiple comorbidities. These factors themselves are strongly associated with poor prognosis, which can confound the effect of antifungal regimens on survival or recovery. Therefore, management of C. auris infections should include both intensive and extensive care with multisectoral collaboration. As for our setting, we have implemented the following measures to limit and prevent other outbreaks of C. auris infections: enhancing infection control practices (hand hygiene, environmental cleaning, and isolation of infected patients), active surveillance and screening programs, and strengthening laboratory capacity for accurate and timely diagnosis.
Conclusion
In this first case series about C. auris in Vietnam, we reported five patients with severe illness who developed C. auris fungemia after more than 10 days of treatment at the ICU; four out of five patients died or were discharged with poor prognosis. We found some common patterns that could be the risk factors for C. auris infections among these cases, including multiple comorbidities, disease severity, use of broad-spectrum antibiotics, or history of surgery/invasive procedures. However, as our study was not designed or powered to detect such statistical associations, further studies are needed to confirm these hypotheses. Screening and preventive measures for C. auris should also be investigated, especially in low-resource settings.
Footnotes
Acknowledgements
None.
Author contributions
H.T.P., T.H.T., and A.H.V.: conceptualization, writing—original draft preparation, writing—review and editing, patient care. M.-T.N.S. and H.H.N.: writing—original draft preparation, writing—review and editing. M.-H.T.: conceptualization, writing—original draft preparation, writing—review and editing, supervision.
Data availability statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.T.P. reported receiving speaking fees and travel reimbursement from Servier Vietnam Ltd and Pfizer Vietnam Ltd, grants from Servier Vietnam Ltd outside the submitted work. M.-H.T. reported receiving travel reimbursement from Pfizer Vietnam Ltd, speaking fees, and grants from Servier Vietnam Ltd outside the submitted work. The other authors declare no competing interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
Ethical approval to report this case series was obtained from the Ethics Committee of Nhan Dan Gia Dinh Hospital (approval number 61-2021/NDGD-HDDD).
Informed consent
Written informed consent was obtained from the patient (for case 1) and from the legally authorized representatives (for cases 2, 3, 4, and 5 of the DECEASED subjects) for their anonymized information to be published in this article.
Consent to participate
Not applicable.
