Abstract
Objectives:
Breakpoint Cluster Region (BCR)-Abelson tyrosine kinase (ABL) inhibitors are widely used in the treatment of blood cancers, particularly chronic myelogenous leukemia and are often combined with triazole antifungal agents to prevent fungal infections. However, the cardiac arrhythmia risks associated with BCR-ABL inhibitors in combination with triazole antifungal agents in real-world settings remain poorly understood. To address this gap, we conducted a pharmacovigilance study to evaluate and compare the cardiac arrhythmia profiles of BCR-ABL inhibitors when used with and without triazole antifungal agents in clinical practice.
Methods:
A disproportionality analysis was performed using the Food and Drug Administration Adverse Event Reporting System database (2004Q1-2024Q2). To identify potential signals of cardiac arrhythmias associated with BCR-ABL inhibitors, with or without triazole antifungal agents, we calculated reporting odds ratios and 95% confidence intervals. Comparisons were made between BCR-ABL inhibitor monotherapy and all other drugs in the Food and Drug Administration Adverse Event Reporting System database, as well as between BCR-ABL inhibitors combined with triazole antifungal agents and BCR-ABL inhibitor monotherapy. Additionally, the Weibull shape parameter test was also used to evaluate time-to-onset.
Results:
From 2004Q1 to 2024Q2, the Food and Drug Administration Adverse Event Reporting System database reported 21,433,114 cases, including 2666 and 68 cases of cardiac arrhythmias linked to BCR-ABL inhibitor monotherapy and its combination with triazole antifungal agents, respectively. The reporting odds ratios and their 95% confidence intervals for BCR-ABL inhibitor monotherapy, asciminib, nilotinib, and ponatinib were 1.31 (1.27–1.36), 2.11 (1.45–3.06), 2.66 (2.53–2.80), and 1.18 (1.05–1.33), respectively. Dasatinib plus triazole antifungal agents (reporting odds ratio: 2.98, 95% CI: 1.93–4.60) and ponatinib plus triazole antifungal agents (reporting odds ratio: 1.53, 95% CI: 1.08–2.16) were associated with a higher disproportionality of cardiac arrhythmias than BCR-ABL inhibitor monotherapy. The median time-to-onset was longer with monotherapy than with BCR-ABL inhibitors plus triazole antifungal agents (2.63 vs. 0.34 months, p < 0.001), both indicating an early failure type.
Conclusions:
BCR-ABL inhibitors plus triazole antifungal agents increase the risk of cardiac arrhythmia, particularly in the early stages of treatment, with the risk decreasing over time.
Keywords
Introduction
Breakpoint Cluster Region (BCR)-Abelson tyrosine kinase (ABL) inhibitors have demonstrated significant therapeutic efficacy in the treatment of chronic myeloid leukemia (CML), a myeloproliferative disorder driven by the Philadelphia chromosome, resulting from a reciprocal translocation between chromosomes 9 and 22. 1 This genetic rearrangement leads to the formation of the BCR-ABL fusion protein, characterized by constitutively active tyrosine kinase activity, which drives the uncontrolled proliferation and survival of myeloid progenitor cells. 2 BCR-ABL inhibitors specifically target the tyrosine kinase domain, inhibiting its enzymatic activity and subsequently inducing apoptosis in tumor cells. Several generations of BCR-ABL inhibitors have been developed and approved by the U.S. Food and Drug Administration (FDA) for CML therapy, including first-generation (imatinib), second-generation (dasatinib, nilotinib, bosutinib), and third-generation (ponatinib, asciminib) agents.3,4 These inhibitors have also demonstrated clinical efficacy in the management of other malignancies, including acute lymphoblastic leukemia, dermatofibrosarcoma protuberans, and gastrointestinal stromal tumors.5–7 As a result, BCR-ABL inhibitors have revolutionized cancer therapy, marking the beginning of a new era in targeted treatment strategies.
Patients with hematologic malignancies are at a significantly elevated risk of invasive fungal infections due to chemotherapy-induced neutropenia and immunosuppression. Consequently, triazole antifungal agents (TAAs), including fluconazole, itraconazole, voriconazole, posaconazole, and isavuconazole are commonly used for the treatment and prevention of Aspergillus and Candida infections. 8 It is important to highlight that BCR-ABL inhibitors undergo hepatic metabolism primarily via the cytochrome P450 enzyme system, with CYP3A4 playing a dominant role. The concurrent administration of TAAs, which act as moderate to potent CYP3A4 inhibitors, may induce significant pharmacokinetic interactions, potentially leading to severe adverse events (AEs). 9
The expanding clinical application of BCR-ABL inhibitors, both in routine practice and clinical trials, has demonstrated substantial therapeutic efficacy.10–12 However, a spectrum of treatment-related AEs has been documented, impacting critical organ systems, including the cardiovascular system, pulmonary system, and hepatic function. These AEs not only present significant health hazards but also compromise treatment adherence, thereby limiting therapeutic outcomes.13,14 The cardiovascular toxicities associated with BCR-ABL inhibitors were initially unforeseen, as these agents were designed to selectively target malignant cells without anticipated cardiotoxic effects. Although pharmacovigilance analyses of cardiac toxicity associated with BCR-ABL inhibitors have been conducted, these were done before the approval of asciminib.15,16 Furthermore, the cardiac arrhythmia profiles of BCR-ABL inhibitors in combination with TAAs in real-world settings remain poorly understood.
Given that a significant proportion of patients receiving BCR-ABL inhibitor therapy are immunocompromised and require concomitant antifungal treatment, a comprehensive understanding of the interactions between BCR-ABL inhibitors and TAAs is essential to ensure optimal patient safety. The concurrent administration of these agents raises significant clinical concerns due to their potential pharmacokinetic interactions. Specifically, TAAs can potently inhibit CYP3A4 activity, thereby modulating the metabolic clearance of BCR-ABL inhibitors, which may lead to clinically significant alterations in drug plasma concentrations and an increased risk of toxicity. Furthermore, both BCR-ABL inhibitors and TAAs are independently associated with an increased risk of cardiac arrhythmias. A thorough evaluation of the arrhythmogenic risk in patients receiving combination therapy is imperative, as the synergistic toxicities of these agents may substantially elevate the incidence of cardiac events beyond the baseline risk associated with either drug alone.
Therefore, we conducted a disproportionality analysis to detect cardiac arrhythmia signals associated with six BCR-ABL inhibitors, either as monotherapy or in combination with TAAs, using the latest data from FAERS database. This study aims to provide comprehensive information on the characteristics and onset timing of BCR-ABL inhibitor-related cardiac arrhythmias, with or without TAAs, and to offer valuable insights for clinical practice.
Methods
Study design and data sources
A retrospective observational pharmacovigilance study was conducted using FAERS database, 17 which includes voluntary AEs reports submitted by healthcare professionals (e.g., physicians, pharmacists, nurses), consumers (e.g., patients, family members, lawyers), and manufacturers. The large volume of data from diverse populations worldwide makes it a robust resource for pharmacovigilance studies in real-world settings. The publicly accessible FAERS database, available for download from the FDA website (https://www.fda.gov/drugs/fdas-adverse-event-reporting-system-faers/fda-adverse-event-reporting-system-faers-latest-quarterly-data-files), contains over 21 million AE reports from 1 January 2004 to 30 June 2023, across seven datasets: treatment dates (THER), patient outcomes (OUTC), adverse event coding (REAC), drug information (DRUG), patient demographics and administrative details (DEMO), reporting sources (RPSR), and drug indications (INDI), all of which were downloaded for analysis.
Data cleaning
This study used data from the FAERS database covering the period from the first quarter of 2004 to the second quarter of 2024. Following the FDA guidelines, adverse event reports with the same CASEID (used to identify a FAERS case) in the DEMO table were considered duplicates. Reports with secondary identifiers, such as FDA_DT (indicating the date the FDA received the report) or PRIMARYID (a unique identifier for FAERS reports and the primary key for linking data files), were removed. FAERS classifies drugs into four categories: PS (primary suspect), SS (secondary suspect), C (concomitant), and I (interacting). To improve analytical precision in the monotherapy analysis of BCR-ABL inhibitors, we included only AE reports where BCR-ABL inhibitors were identified as the PS drug, excluding reports involving combination therapies with TAAs. Cases were categorized into the coadministration group if either a BCR-ABL inhibitor or a TAA was designated as the primary suspect (PS), and the other drug class was also coadministered (i.e., listed in any role in the report: PS, SS, C, or I). This classification ensures that coadministration is defined solely as the simultaneous use of both drug classes. The trade and generic names of BCR-ABL inhibitors and TAAs are listed in Table S1.
FAERS follows the international safety reporting guidelines set by the International Conference on Harmonisation. AEs are coded using preferred terms (PTs) from the Medical Dictionary for Regulatory Activities (MedDRA, Version 27.1; maintained by the Maintenance and Support Services Organization [MSSO]). Relevant PTs for symptoms, signs, and investigations are grouped into narrative categories through Standardized MedDRA Queries (SMQs) to identify specific medical conditions of interest. 18 SMQs typically include two categories: “narrow” and “broad” scope. The narrow search includes terms that clearly relate to a selected event, while the broad search includes terms from the narrow scope, along with those that are potentially related but with some uncertainty. 19 Thus, the narrow SMQ for “cardiac arrhythmias” (e.g., bradyarrhythmia) is more specific, whereas the broad SMQ (e.g., central bradycardia) is more sensitive. The primary endpoint of this study was defined using the narrow SMQ category for cardiac arrhythmias, with the PTs listed in Table S2.
Statistical analysis
A descriptive analysis was conducted to examine the basic characteristics of cardiac arrhythmia reports associated with BCR-ABL inhibitor monotherapy and combination therapy with TAAs. Categorical variables, including sex, weight, age, reporter type, reporting country, and outcome of AEs, were summarized using frequencies and percentages. Fisher’s exact test was applied to compare differences in count data between groups.
Disproportionality analysis, a method commonly used by the World Health Organization (WHO) and FDA, identifies positive pharmacovigilance signals in databases using the reporting odds ratio (ROR) and its 95% CI. In this study, we calculated two sets of ROR values: one for BCR-ABL inhibitors and cardiac arrhythmia-related AEs, and another for the combination of BCR-ABL inhibitors, TAAs, and the same AEs. This approach helped identify potential safety signals for BCR-ABL inhibitors both alone and in combination with interacting drugs. Tables S3 and S4 present the method for calculating the ROR for each case in a 2 × 2 contingency table. In the primary analysis, an AE was considered a positive signal if the lower limit of the 95% CI (ROR025) for the ROR exceeded 1, and the number of cases was at least 3. 20 A higher ROR value for a given drug-adverse event pair indicates a stronger disproportionality in reporting within the database. In the sensitivity analysis, we included only reports in which BCR-ABL inhibitors or TAAs were documented by healthcare professionals (HCPs) to minimize the risk of bias.
Time-to-onset analysis
Time-to-onset (TTO), defined as the interval between the START_DT (the target drugs commencement date in the THER file) and EVENT_DT (the AE onset date in the DEMO file), was assessed in our study using medians, quartiles, and the Weibull shape parameter (WSP) test.21,22 Data with inaccurate or missing input dates, as well as cases where the EVENT_DT occurred before the START_DT, were excluded. To visually present the TTO for cardiac arrhythmias associated with BCR-ABL inhibitors, both with and without TAAs, we used cumulative distribution curves for each group and compared the median TTO between them using the log-rank test.
Using the WSP test for statistical analysis, we modeled the Weibull distribution with its scale (α) and shape (β) parameters. The relatively high α value indicates that the TTO distribution of AEs spans a longer duration. Signals were classified into three types based on their predicted changes in AE risk over time: The “Early failure” type is characterized by a β parameter less than 1, with its upper 95% CI also below 1, indicating that the risk of AEs decreases over time; The “Wear-out failure” type is characterized by a β parameter greater than 1, with its lower 95% CI above 1, indicating that the risk of AEs increases over time, and the “Random failure” type is characterized by a β parameter close to 1, with its 95% CI including 1, indicating that the risk of AEs remains constant over time. 23
This study follows the Reporting of a Disproportionality Analysis for Drug Safety Signal Detection Using Individual Case Safety Reports in PharmacoVigilance (READUS-PV) guideline.24,25 In this study, p < 0.05 was considered statistically significant, and all statistical tests were two-tailed. Statistical analyses were performed using R version 4.4.1 (R Foundation for Statistical Computing, Vienna, Austria) and Microsoft Excel 2021 (Microsoft Corporation, Redmond, WA, USA). Ethical approval and informed consent were not required, as the FAERS database is publicly available and patient identity information is kept confidential.
Results
Descriptive analysis
From the first quarter of 2004 to the second quarter of 2024, a total of 21,433,114 cases were reported in the FAERS database. After excluding duplicates, 2666 cases of cardiac arrhythmias (SMQ) associated with BCR-ABL inhibitor monotherapy and 68 cases associated with BCR-ABL inhibitors combined with TAAs were included in the final analysis (Figure 1). The baseline characteristics of cases in the BCR-ABL inhibitor groups, with and without TAAs, are summarized in Table 1. Significant differences in weight, age, and outcome of AEs were observed between the two groups (p < 0.001). The proportion of males was slightly higher than that of females in the total BCR-ABL inhibitor cases, with 1128 females (41.26%) and 1327 males (48.54%). Weight data were largely missing, with only 23.92% of patients having a recorded weight. Additionally, patients aged 65 and older accounted for a larger proportion of cases compared to those aged 18–64 (34.28% vs. 26.37%), suggesting that individuals aged 65 and older may be more susceptible to BCR-ABL inhibitor-related cardiac arrhythmias (SMQ). The majority of reports were submitted by HCPs (n = 1910, 69.86%), enhancing the credibility of the data. Most cardiac arrhythmia (SMQ) cases were reported outside the United States (n = 1851, 67.70%). The most commonly reported outcomes of AEs were other serious outcomes (44.22%), followed by hospitalization (29.41%), death (11.05%), and life-threatening events (5.49%). Over a 21-year period, cardiac arrhythmias accounted for a small proportion of total AEs associated with BCR-ABL inhibitor monotherapy, ranging from 1.5% to 4.9%. In contrast, the proportion was higher in BCR-ABL inhibitors combined with TAAs, ranging from 0.0% to 29.6% (Figure 2).

Flow chart of report cases in FAERS database.
Demographic characteristics of BCR-ABL inhibitors-related cardiac arrhythmias reports with or without TAAs therapy in the FAERS database.
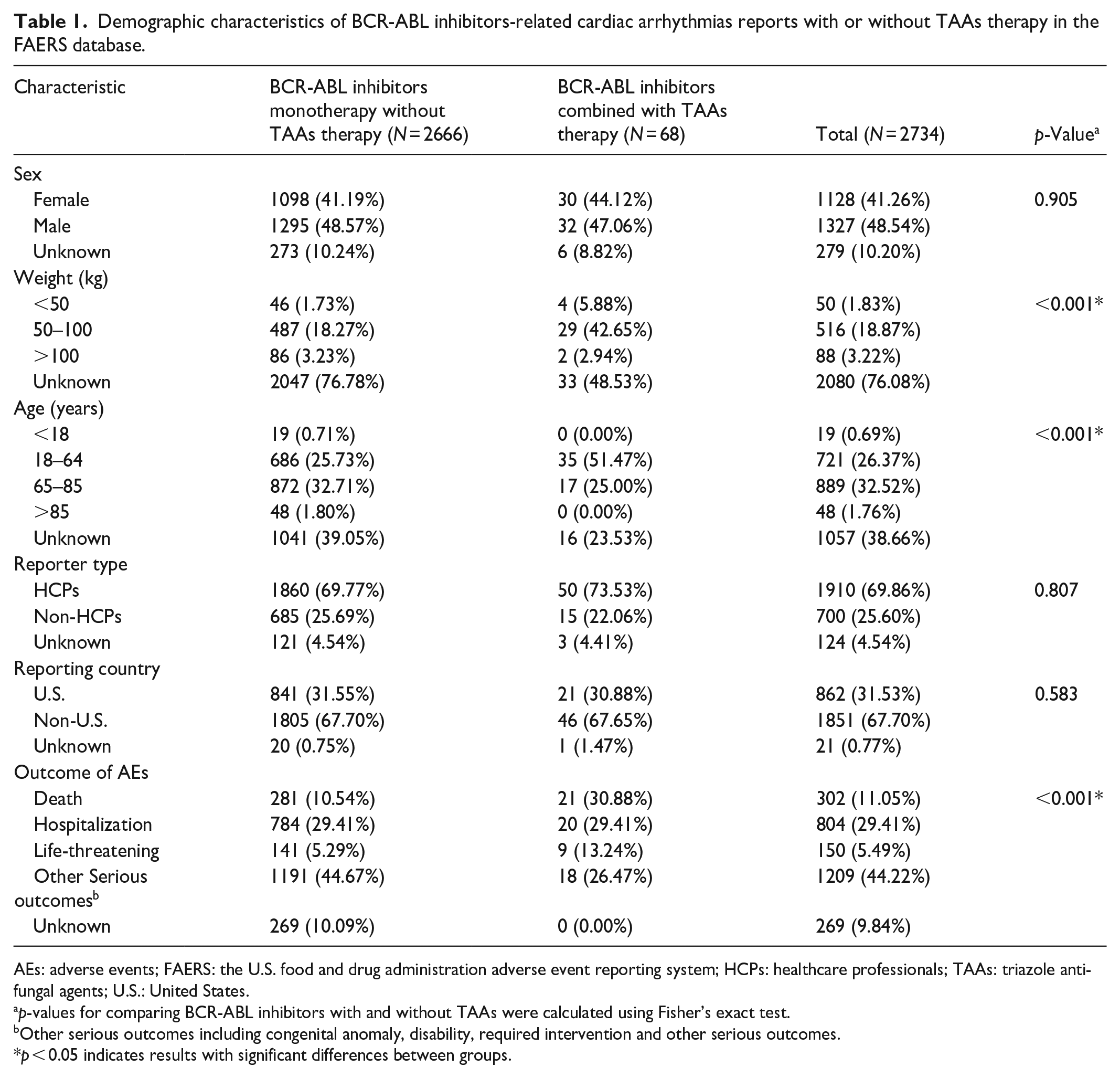
AEs: adverse events; FAERS: the U.S. food and drug administration adverse event reporting system; HCPs: healthcare professionals; TAAs: triazole antifungal agents; U.S.: United States.
p-values for comparing BCR-ABL inhibitors with and without TAAs were calculated using Fisher’s exact test.
Other serious outcomes including congenital anomaly, disability, required intervention and other serious outcomes.
p < 0.05 indicates results with significant differences between groups.

Reporting year of cardiac arrhythmias (SMQ) related to BCR-ABL inhibitors with or without TAAs therapy in the FAERS database. Statistics on BCR-ABL inhibitor-related cardiac arrhythmias (a) with monotherapy and (b) in combination with TAAs. The proportional bar chart shows the number of reports with and without cardiac arrhythmias in the FAERS database from Q1 2004 to Q2 2024, along with the overall totals. The numbers at the bottom of the chart represent the total cases of cardiac arrhythmias reported each year. The line chart displays the annual trends of reports with and without cardiac arrhythmias.
Disproportionality analysis
Signal detection of BCR-ABL inhibitors with or without TAAs related to cardiac arrhythmias (SMQ), as reported by all reporters and HCPs, is shown in Figure 3. A positive signal for cardiac arrhythmias was identified in the overall BCR-ABL inhibitor monotherapy group (ROR 1.31, 95% CI: 1.27–1.36). Among individual BCR-ABL inhibitors, the following drugs were associated with cardiac arrhythmias (SMQ): asciminib (ROR: 2.11, 95% CI: 1.45–3.06), nilotinib (ROR: 2.66, 95% CI: 2.53–2.80), and ponatinib (ROR: 1.18, 95% CI: 1.05–1.33). Nilotinib showed the strongest positive signal, while asciminib had the widest confidence interval (Figure 3(a)). In contrast, bosutinib, dasatinib, and imatinib, three of the six BCR-ABL inhibitors, showed negative signals. We summarized the PT-level cardiac arrhythmias for six BCR-ABL inhibitor monotherapies, with ROR (95% CI) results provided in Table S5. Nilotinib showed the most positive signals at the PT level, with seventeen signals detected, followed by dasatinib with five signals. Among all BCR-ABL inhibitors, electrocardiogram QT prolongation was associated with the highest disproportionality and had the highest number of reports (n = 861, ROR; 4.16, 95% CI: 3.89–4.45). This was followed by atrial fibrillation, which had the second-highest number of reports (n = 853, ROR: 1.44, 95% CI: 1.34–1.54). Further analysis revealed that compared to BCR-ABL inhibitor monotherapy, the combination of BCR-ABL inhibitors with TAAs was associated with a higher disproportionality of cardiac arrhythmias (SMQ). Specifically, dasatinib plus TAAs (ROR: 2.98, 95% CI: 1.93–4.60) and ponatinib plus TAAs (ROR: 1.53, 95% CI: 1.08–2.16) showed increased risk, while the combination of BCR-ABL inhibitors and TAAs for asciminib and bosutinib was not associated with cardiac arrhythmias (Figure 3(b)).
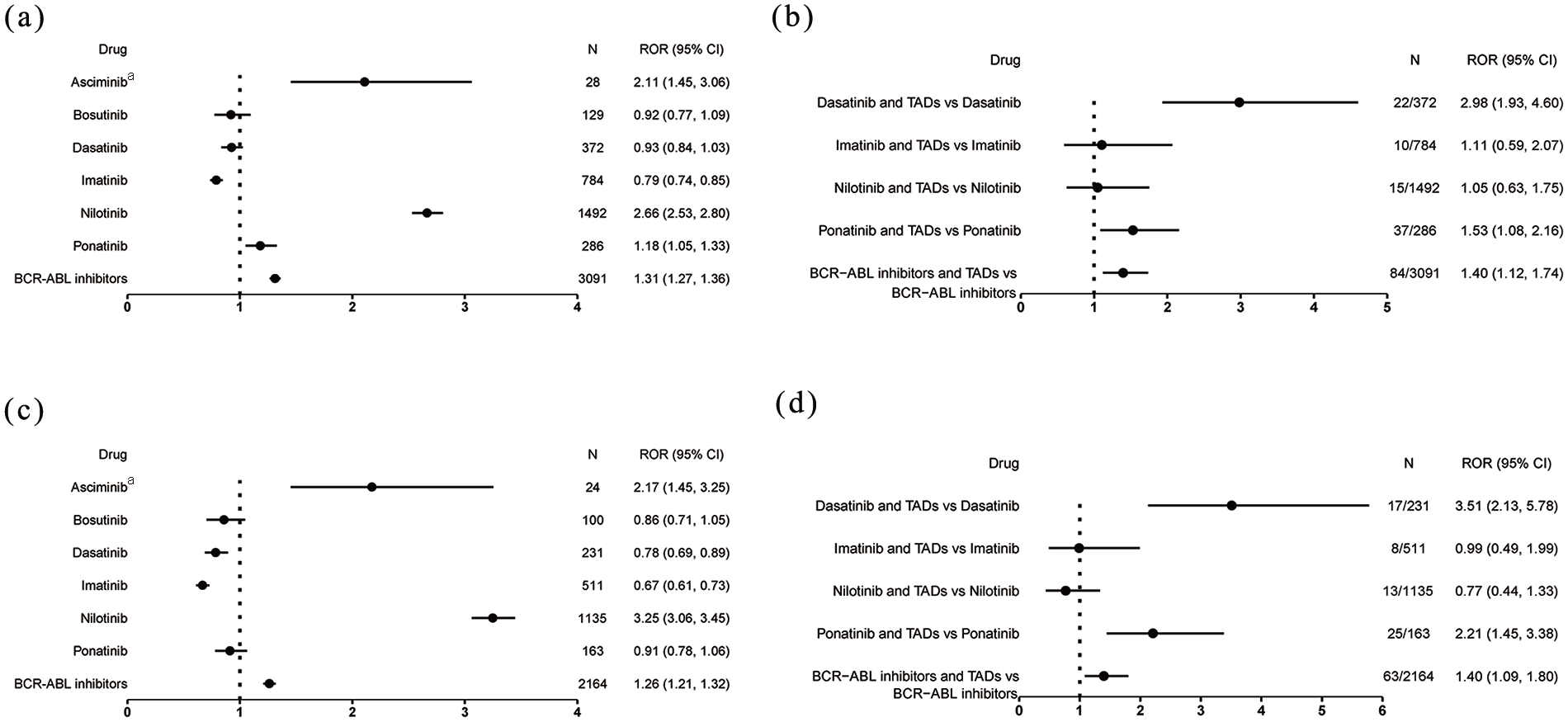
Reporting odds ratios (ROR) with 95% CI for cardiac arrhythmias (SMQ) associated with (a) BCR-ABL inhibitor monotherapy, (b) BCR-ABL inhibitors combined with TAAs, (c) BCR-ABL inhibitor monotherapy by HCPs, and (d) BCR-ABL inhibitors combined with TAAs by HCPs.
In the sensitivity analysis, cardiac arrhythmias (SMQ) remained a positive signal for overall BCR-ABL inhibitor monotherapy (ROR: 1.26, 95% CI: 1.21–1.32), asciminib (ROR: 2.17, 95% CI: 1.45–3.25), and nilotinib (ROR: 3.25, 95% CI: 3.06–3.45), but was no longer a positive signal for ponatinib (ROR: 0.91, 95% CI: 0.78–1.06) (Figure 3(c)). Compared to BCR-ABL inhibitor monotherapy, dasatinib plus TAAs continued to show the highest ROR at 3.51 (95% CI: 2.13–5.78), as reported by HCPs (Figure 3(d)).
The cardiac arrhythmia (SMQ) signal spectrum for different BCR-ABL inhibitor combinations with TAAs is shown in Figure 4. Imatinib plus posaconazole (ROR = 4.07) exhibited the strongest signal, followed by dasatinib plus fluconazole (ROR = 3.91) and dasatinib plus itraconazole (ROR = 3.67). However, only the combination of BCR-ABL inhibitors with voriconazole (ROR = 2.16), rather than with other TAAs, showed a positive signal for cardiac arrhythmias (SMQ).
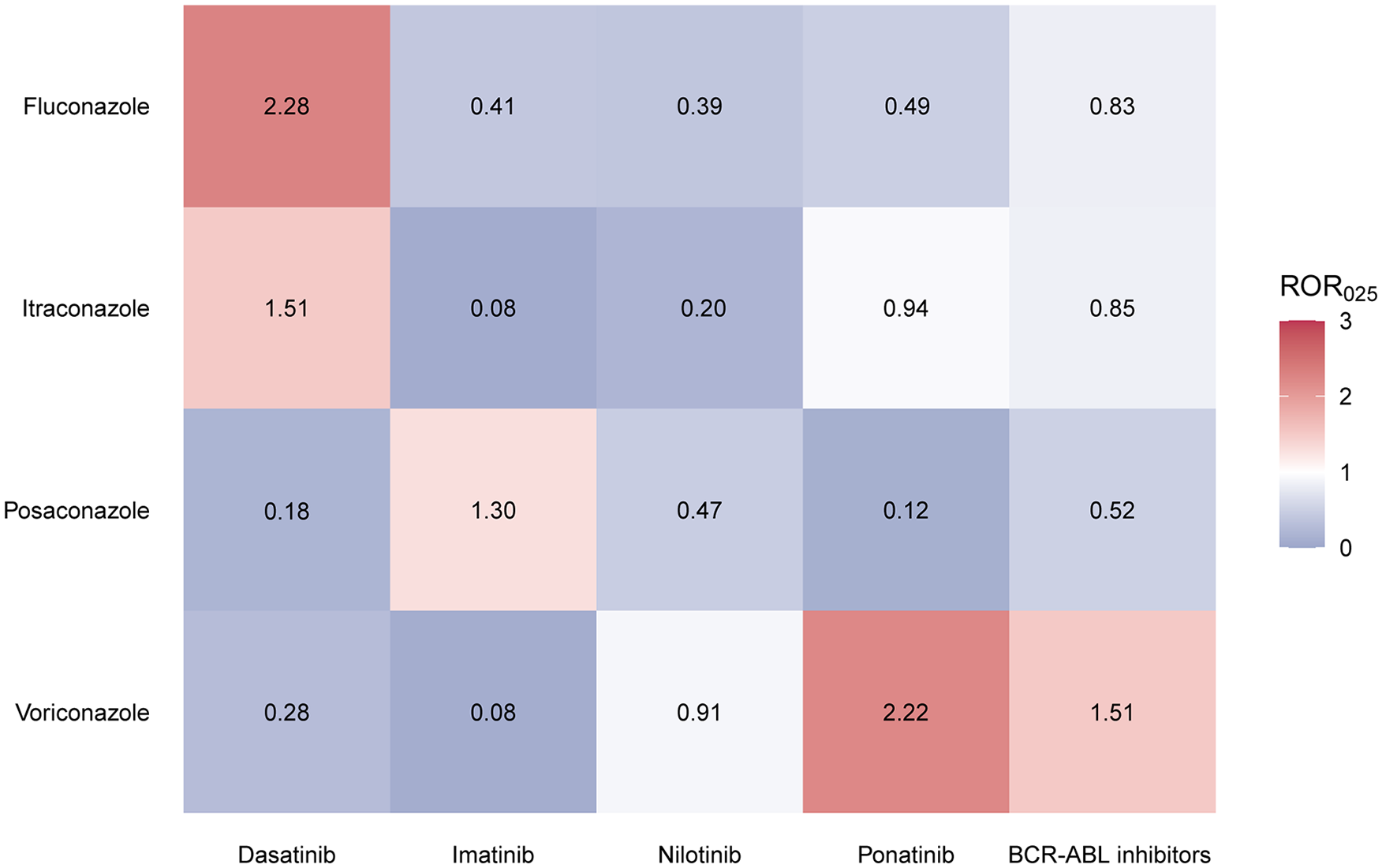
Analysis of cardiac arrhythmias (SMQ) associated with the combination of BCR-ABL inhibitors and TAAs in the FAERS database. The heatmap displays the ROR025 values for various BCR-ABL inhibitor and TAA combination treatments, compared to the corresponding BCR-ABL inhibitors. Red indicates ROR025 values greater than 1, while gray represents values less than or equal to 1, with at least three reports. The combination of asciminib and bosutinib with TAAs has not been linked to cardiac arrhythmias (SMQ), and is therefore excluded from the figure. Similarly, the combination of isavuconazole and BCR-ABL inhibitors is not associated with cardiac arrhythmias (SMQ) and is not represented.
Time-to-onset analysis
The results of the median TTO and WSP analysis for BCR-ABL inhibitor monotherapy and combinations with TAAs are shown in Figure 5. The median TTO for cardiac arrhythmia (SMQ) with BCR-ABL inhibitor monotherapy (2.63 months) was longer than that for the combination of BCR-ABL inhibitors with TAAs (0.34 months) (Log-rank test, p < 0.001). In the WSP test, the β value and its upper 95% confidence interval limit were both below 1, suggesting that the risk of cardiac arrhythmias is higher early in the treatment and decreases over time, which is characteristic of an “Early failure” pattern. For BCR-ABL inhibitor monotherapy, the scale parameter α was 6.73 months (95% CI: 5.85–7.60), indicating that the time-to-onset distribution of cardiac arrhythmia events is spread over a longer duration.

Time to onset and Weibull shape parameter test of cardiac arrhythmias (SMQ) associated with BCR-ABL inhibitors, with or without TAAs.
Discussion
To the best of our knowledge, this is the first pharmacovigilance study to assess drug interactions between BCR-ABL inhibitors and TAAs in relation to cardiac arrhythmias. While previous disproportionality analyses have examined cardiac arrhythmias associated with BCR-ABL inhibitors, these studies were conducted before the approval of asciminib. This study utilizes the latest real-world data from the FAERS database to provide a comprehensive evaluation of cardiac arrhythmia signals associated with BCR-ABL inhibitors, both as monotherapy and in combination with TAAs. This approach facilitates the derivation of systematic and precise conclusions. This study yielded three principal findings: (1) Monotherapy with BCR-ABL inhibitors demonstrated a positive pharmacovigilance signal for cardiac arrhythmias. (2) The combination of BCR-ABL inhibitors and TAAs was associated with an elevated risk of cardiac arrhythmia AEs compared to monotherapy with BCR-ABL inhibitors alone. (3) The median TTO of cardiac arrhythmias was shorter with the combination of BCR-ABL inhibitors and TAAs than with BCR-ABL inhibitor monotherapy, indicating an earlier onset of AEs in the combination therapy.
BCR-ABL inhibitors monotherapy: Increased cardiac arrhythmia risk
Our analysis revealed that, from 2014 to 2024, cardiac arrhythmias (SMQ) remained a relatively stable but uncommon AE associated with BCR-ABL inhibitor monotherapy. Additionally, a significant association was observed between BCR-ABL inhibitor use and cardiac arrhythmias. Similar results were observed for specific drugs, including asciminib, nilotinib, and ponatinib, with nilotinib demonstrating the strongest signal. These findings align with those of Ye et al., 26 who reported that ponatinib and nilotinib significantly increased the risk of cardiac arrhythmias. In contrast, Liu et al., 27 using FAERS data to analyze AE signals for asciminib, did not identify cardiac arrhythmias (PT) as a significant signal. This discrepancy can be attributed to the study’s reliance on data collected from October 2021 to December 2023 and the absence of analysis at the SMQ level. Additionally, the focus on reports submitted by HCPs led to the identification of a negative signal for ponatinib in this study. Another study indicated that only ponatinib was associated with hypertension (SMQ), while no signal for cardiac arrhythmias (SMQ) was identified. Despite the inclusion of other anticancer drugs in the control group, a significant signal for cardiac arrhythmias was still detected for nilotinib. 28 Although the precise mechanism of BCR-ABL inhibitor-induced cardiac arrhythmias remains elusive, a study suggests that it may be related to varying extents of hERG K+ channel inhibition by different BCR-ABL inhibitors. The study demonstrated that dasatinib, imatinib, and nilotinib inhibit hERG currents in vitro with IC50 values of 14.3, 15.6, and 0.66 μM, respectively. These in vitro findings were observed at concentrations approximately 150, 3, and 0.1 times the expected human Cmax for the three BCR-ABL inhibitors, respectively. 29
Bosutinib and dasatinib did not demonstrate significant signals for cardiac arrhythmias (SMQ); however, electrocardiogram QT prolongation was observed at the PT level for asciminib, dasatinib, imatinib, and nilotinib. Notably, only nilotinib includes electrocardiogram QT prolongation as a black-box warning in its prescribing information. Similar findings were observed in clinical trials, where any-grade electrocardiogram QT prolongation was more common with nilotinib (300 mg twice daily, 6.8%; 400 mg twice daily, 7.9%) compared with imatinib (3.9%).30,31 Furthermore, significant positive signals for atrial fibrillation were detected with bosutinib, dasatinib, nilotinib, and ponatinib. Both ponatinib and nilotinib demonstrated an increased ROR for atrial fibrillation, with a 2- to 3-fold increase. 32
BCR-ABL inhibitors combined with TAAs: Increased cardiac arrhythmia risk
The combination of BCR-ABL inhibitors and TAAs is used to treat and prevent fungal infections caused by Aspergillus and Candida species in patients with hematologic malignancies.33–35 This study found that combining BCR-ABL inhibitors with TAAs increases the risk of cardiac arrhythmias compared to BCR-ABL inhibitor monotherapy. Specifically, the combination of dasatinib with either fluconazole or itraconazole was associated with a higher disproportionality of cardiac arrhythmias than dasatinib alone. Similarly, the combination of posaconazole with imatinib and voriconazole with ponatinib both elevated the risk of cardiac arrhythmias compared to their respective monotherapies. Additionally, cardiac arrhythmias occurred earlier when BCR-ABL inhibitors were combined with TAAs, as evidenced by a shorter median TTO. This phenomenon may be partially attributed to additive pharmacodynamic toxicity. Key proarrhythmic mechanisms for TAAs include spatial dispersion of repolarization, prolonged action potential configuration, and direct effects on the cardiovascular system. BCR-ABL inhibitors, on the other hand, function as blockers of the hERG potassium channel. 36 The likelihood of cardiac arrhythmias increases due to the overlapping toxicity profiles when BCR-ABL inhibitors are combined with TAAs. Additionally, BCR-ABL inhibitors are metabolized by hepatic cytochrome P450 (CYP450) enzymes. 37 Therefore, coadministration with azole antifungals, which are moderate to strong inhibitors of CYP3A4, may further contribute to the development of cardiac arrhythmias. Although fluconazole is not a potent CYP3A4 inhibitor, its moderate inhibition at higher doses may still contribute to increased systemic exposure of BCR-ABL inhibitors, thereby enhancing their hERG potassium channel-blocking effects. 38 Additionally, fluconazole is a known CYP2C9 and weak CYP2D6 inhibitor,39,40 which may further influence the metabolism of certain BCR-ABL inhibitors. Beyond metabolic interactions, fluconazole may also exert direct electrophysiological effects on cardiac ion channels, further predisposing patients to arrhythmias when combined with BCR-ABL inhibitors. Patients administered BCR-ABL inhibitors or combination therapy with TAAs necessitate vigilant surveillance for cardiac arrhythmias as a potential AE. The integration of continuous electrocardiographic (ECG) monitoring into standard care protocols is recommended, alongside proactive measures to mitigate arrhythmogenic risks. With ongoing research breakthroughs, the advent of mobile ECG monitoring technology holds promise for enhancing early detection and patient safety during BCR-ABL inhibitor or TAAs combination therapies in the oncology realm.
Limitations
In this study, we acknowledge several limitations that warrant consideration. First, the FAERS database is a spontaneous reporting system with reports originating from various countries and different types of reporters. This variability may result in incomplete data, potentially introducing reporting bias or confounding effects. Second, not all AEs are captured in the reports, limiting our ability to establish causal relationships between the combination of BCR-ABL inhibitors and TAAs and cardiac arrhythmias AEs, as well as to calculate their incidence. Third, accurately determining the time to onset of adverse events remains challenging due to incomplete temporal data, potential recall biases, and inconsistent documentation practices inherent to spontaneous reporting systems. These limitations may compromise the reliability of time-to-event analyses and affect the interpretation of temporal relationships between drug exposure and arrhythmia development. Furthermore, although a signal suggesting a potential association was observed through the ROR, this finding should be interpreted with caution. Due to the limitations of spontaneous reporting data, further investigation is essential to validate the potential link between BCR-ABL inhibitors, TAAs, and cardiac arrhythmias. Finally, the cardiac arrhythmias associated with the combination of BCR-ABL inhibitors and TAAs have not been clinically validated, and research in this area remains limited. Therefore, the findings related to these cardiac arrhythmias AEs need confirmation through large-scale prospective studies. We plan to conduct future clinical trials with standardized time-to-onset assessments to further validate our results. Despite these inherent limitations, this study provides valuable insights into the characteristics of cardiac arrhythmia AEs associated with combination therapy, which warrants further exploration in prospective studies.
Conclusions
Overall, BCR-ABL inhibitor monotherapy is associated with a potential risk of cardiac arrhythmias. The combination of BCR-ABL inhibitors with TAAs significantly increases this risk compared to monotherapy. These findings strongly support the recommendation for rigorous electrocardiographic monitoring in cancer patients receiving BCR-ABL inhibitors, whether as monotherapy or in combination with TAAs. These findings could provide valuable insights for the individualized selection of combination therapies in clinical practice. In conclusion, further research is needed to determine the optimal dosing and treatment regimens for combination therapy to maximize synergistic effects while minimizing the risk of cardiac arrhythmias.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251328762 – Supplemental material for Cardiac arrhythmias of BCR-ABL inhibitors with or without triazole antifungal agents: A real-world pharmacovigilance study based on the food and drug administration adverse event reporting system database
Supplemental material, sj-docx-1-smo-10.1177_20503121251328762 for Cardiac arrhythmias of BCR-ABL inhibitors with or without triazole antifungal agents: A real-world pharmacovigilance study based on the food and drug administration adverse event reporting system database by Peitao Xie, Lishan Lu, Yixuan Tian, Rongrong Jia, Xuemei Tian and Pu Bai in SAGE Open Medicine
Footnotes
Acknowledgements
We acknowledge and appreciate the efforts made by the staff and participants of the FDA adverse event reporting system. We are deeply grateful to Wenyue Su for her meticulous language editing, which significantly improved the article’s linguistic quality and scientific clarity.
Statements and Declarations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
