Abstract
Atypical choroid plexus papilloma is a rare World Health Organization grade 2 intraventricular tumor arising from the epithelium of the plexus choroid with intermediate clinical-pathological features between the benign choroid plexus papilloma and the malignant choroid plexus carcinoma. The main criteria for differentiation are histopathologic, with difficulties in distinguishing it from choroid plexus papilloma based on imaging features. We report the case of a 4-year-old female presenting with headaches and altered mental status. Brain magnetic resonance imaging revealed a right lateral ventricular mass with some atypical characteristics, which were confirmed on pathological examination as an atypical choroid plexus papilloma.
Introduction
Choroid plexus tumors are rare intraventricular central nervous system (CNS) tumors of neuroectodermal origin arising from the epithelium of the choroid plexus and accounting for almost 1% of all brain tumors.1,2 They are classified as World Health Organization (WHO) grade 1 benign choroid plexus papilloma (CPP), WHO grade 2 atypical choroid plexus papilloma (aCPP), and WHO grade 3 CPC. 3 While most cases are benign CPP or intermediate aCPP, they pose problems by the resulting hydrocephalus from various mechanisms. Imaging is essential, providing a primary distinction of malignant characteristics from the benign, particularly through tumoral size, invasion of adjacent parenchyma, and suspicion of atypia based on certain features. However, anatomopathological confirmation is still required. Despite the possibility of total resection, the neurosurgical procedure is challenging, especially considering hemorrhagic risks, for which transcollation devices are now proposed. 4
Case report
A 4-year-old female presented with persistent headaches, vomiting, and altered mental status. Routine exams were performed, and the results were normal. A brain computed tomography (CT)-scan revealed a right heterogeneous lateral ventricular mass. Subsequent brain magnetic resonance imaging (MRI) showed a well-defined, solid, lobulated mass in the right ventricle. It appeared isointense on T1WI, hyperintense on Fluid-attenuated Inversion Recovery (FLAIR), containing a few cysts. The tumor exhibited mildly hyperintense areas on Diffusion Weighted sequences (DWI) with no restriction apparent diffusion coefficient (ADC) map. Upon gadolinium injection, it did not exhibit intense enhancement; however, multiple vessels surrounding the cysts were visible on the Maximak Intensity Projection (MIP) reconstructed images (Figure 1). Initially, an aCPP was suspected based on clinical details and atypical imaging features. Nevertheless, CPP and CPC were also considered. Histopathological examination, when performed, confirmed the diagnosis of an aCPP (Figure 2). The tumor consisted of a proliferation of papillae of variable size, exhibiting cytonuclear atypia with positivity for cytokeratin AE1 and AE3, associated with a Ki 67 index of 15% (Figure 3).
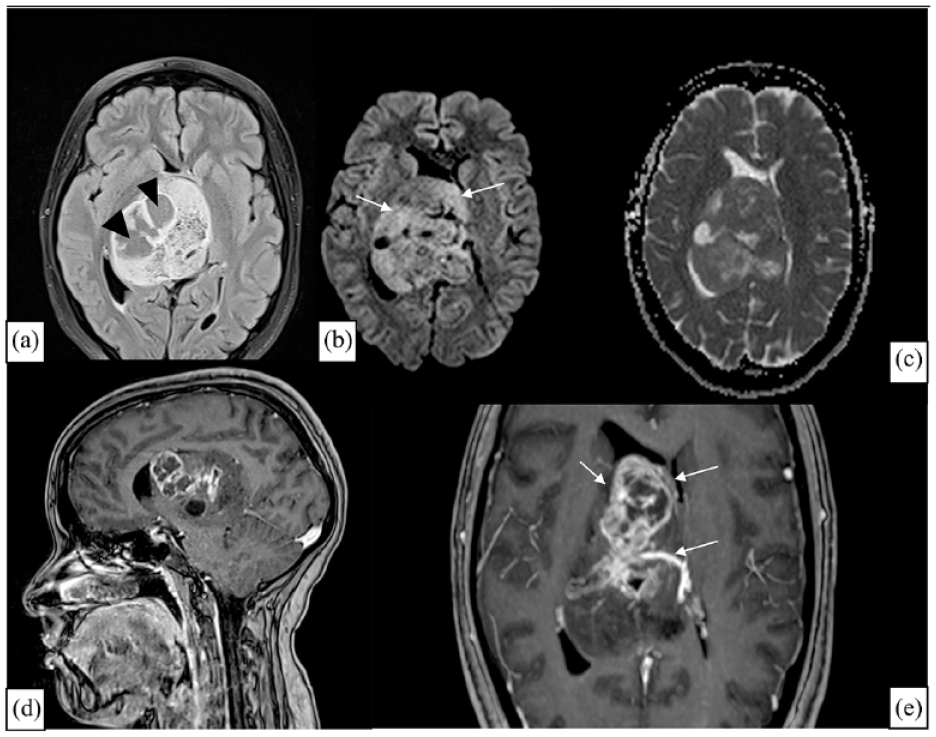
Brain magnetic resonance imaging including axial FLAIR (a), Diffusion Weighted sequences (DWI) (b) with apparent diffusion coefficient (ADC) map (c), sagittal T1-weighted-sequences with gadolinium injection (d) and axial with MIP reconstructed image (e) reveals a large well-defined, lobulated right lateral intraventricular tumor with heterogeneous structure, mainly due to the presence of cysts (arrowhead). The mass appears slightly hyperintense on DWI (arrows) with a similar appearance to brain parenchyma on ADC map. Upon gadolinium injection, a slight enhancement is observed, and curvilinear structures within the tumor revealed to be vessels at MIP reconstruction (e, arrows).

Anatomopathological examination reveals a papillomatous proliferation consisting of papillae of variable size (Image on the left). Papillae are lined with tall columnar cells showing slight cytonuclear atypia (Image on the right).

Histological examination shows positivity for cytokeratin AE1/AE3 (a). The mitotic activity, estimated with a Ki67 index, is at 15% (b).
Discussion
The choroid plexus is a richly vascularized structure in the CNS, with fenestrated and noncontinuous capillaries. Its cells form tight junctions, and the entire is lined by ependymal cells of the ventricles. 5 It can be the location of different pathological conditions. Neoplasms of the choroid plexus are classified by the WHO into three categories: CPP, atypical aCPP, and CPC, with CPP being the most prevalent. They account for 2%–6% of brain tumors in children and 0,5% in adults.1,2 The incidence of aCPP was long time underestimated, as it was not considered an independent entity until 2007. Before that, it was often misdiagnosed as a typical CPP or a CPC. 6 A current and comprehensive study on incidence of choroid plexus tumors in the United States, published in 2022, revealed that the incidence of CPP was the highest, followed by CPC and aCPP. The same incidence was higher among children less than 1 year for all the subtypes. 7
CPP may arise anywhere in the epithelium of the choroid plexus, typically in the lateral ventricles in children and the fourth ventricles in adults. 8 In the pediatric population, they represent the third most common tumor after teratoma and glioma. 1 Extra ventricular localizations are rarer and have been reported in the pineal gland, suprasellar region, cerebellum, and spine thought to be remnants of choroid plexus. 9 Case series data from the literature available for pediatric cases reveal three uncommon locations: the posterior third ventricle, the fourth ventricle, and the cerebellopontine angle, all presenting variable degrees of hydrocephalus. 10
The symptomatology in CPP typically involves progressive mental status deterioration with signs of intracranial hypertension, such as vomiting, headaches, visual defects, and lateral gaze palsy. In some cases, subarachnoid hemorrhage may serve as a revealing sign of tumor bleeding. In children, clinical presentation may include an increase in head circumference and refusal to feed. 1
The mechanism of hydrocephalus in CPP, similar to other choroid plexus tumors (CPTs), results from the combination of obstruction, hyperproduction, and impairment of reabsorption. Consequently, there is no correlation between the tumor size and the severity of hydrocephalus, unlike other intraventricular tumors. 11 Moreover, blockage of CSF absorption in the arachnoid granulations following hemorrhage or disruption of CSF dynamics by the tumor itself can lead to a reduction in “glymphatic clearance” worsening the disease. 12
It is now well recognized that similar to the peripheral lymphatic system, the CNS has its own “glymphatic system” composed of glial cells with various barrier systems responsible for maintaining homeostasis. Disorders in this system are implicated in various diseases, making it a target for various treatment pathways. This significance has led imaging techniques to explore it, yielding promising results. These include perfusion through arterial spin labeling (ASL), contrast injection through magnetic resonance lymphography, and metabolic sequences such as contrast exchange saturation transfer (CEST). Taking CPTs as an example, ASL can aid in differentiating tumoral from seeding areas from normal plexus choroid structures. CEST, as metabolic sequences, are more sensitive than spectroscopy in detecting both small exogenous and endogenous compounds. Consequently, amide proton transfer weighted imaging can better predict carcinoma with higher amounts of protein compared to plexus choroid papilloma. 13
Several genetic abnormalities are reported to be associated with the development of CPP in the pediatric population. These include Aicardi syndrome, hypomelanosis of Ito, constitutional 9p duplication, and Von-Hippel-Lindau syndrome. The germline mutation in the TP53 tumor suppressor, which is the origin of Li-Fraumeni syndrome, is more commonly associated with CPC. However, it has also been reported in some cases of CPP. 14
In a trial to determine the genetic alterations behind CPTs, a study found that pediatric CPTs lack recurrent driver alterations compared to adult CPTs, except for TP53. On the other hand, adult exhibit Telomerase Reverse Transcriptase (TERT) promoter mutations or a novel CCDC47-PRKCA gene fusion, both associated with an unfavorable prognosis. 15
Although surgery is the therapeutic strategy for all CPTs, a preoperative diagnosis of CPP is particularly useful. Preoperative embolization can be performed in cases of CPC due to their more invasive and hemorrhagic nature. 15
CPP typically presents as well-defined intraventricular masses that appear iso- or mildly hyperdense on unenhanced CT and homogeneously enhance after contrast injection, with possible calcifications observed in 4%–25% of cases.5,16,17 On MRI, typical CPPs usually appear hypointense on T1-weighted-sequences (T1WI) and hyperintense on T2-weighted sequences (T2WI). However, atypical CPPs may present differently, appearing isointense on T1W, hyperintense on T2WI, and isointense on DWI. This is in contrast to CPCs, which often exhibit various signal combinations. “Foci of high signal intensity due to intratumoral hemorrhage” and “flow-voids due to high flow feeding vessels” are possible. 18 CPTs are generally hypervascular, resulting in marked enhancement across all three categories: benign, atypical, and malignant. However, literature reports vary, noting mild, moderate, and even no enhancement in some extraventricular cases, suggesting a potential poor blood supply.19–21 The presented case demonstrates mild to no enhancement despite being intraventricular, which may be a unique observation for atypical intraventricular CPP. The presence of a cystic component, along with the aforementioned features, supports the primary diagnosis of CPP. Cystic components have been reported in 20% of cases, 22 often attributed to an overproduction of CSF or spontaneous bleeding. 23 Enhancement of the cyst wall has also been described, representing an inflammatory reaction after hemorrhage. 24 In our patient’s MRI, curvilinear vessels surrounding the cyst wall are visible on reconstructed MIP images (Figure 1). Despite being a rare pathological entity, awareness is crucial for recognizing cystic CPP as it can lead to severe obstructive hydrocephalus and even result in sudden death.25,26
While some reports have denied the existence of a correlation between signal and enhancement characteristics on MRI, 27 it has been observed that larger tumoral size, increased peritumoral edema, irregular internal morphology, and necrosis elevate the probability of malignancy. 28 These features somehow correlate with histological characteristics: higher cellular density appears to differentiate a CPP from a normal non-neoplastic choroid plexus. aCPP exhibits increased mitotic activity (as was the case in our study), while the reduction of papillary patterns, higher cellular density, necrosis, and invasion of adjacent parenchyma are features of carcinoma.29,30 A recent study 28 has further subdivided CPT based on these histological features and the appearance of the solid part on MRI into papillary, lobulated, and irregular configurations. The papillary configuration is more characteristic of CPP, featuring a single columnar epithelium surrounding a fibrovascular stalk. The increase in the solid component associated with malignancy is accompanied by the loss of this papillary configuration and increased irregularity. CPC has a different organization with higher number of vessels. ASL can help differentiate them from CPP by using the relative Cerebral Blood Flow, as previously stated. 31 Moreover, a medullary MRI is indicated if a CPC is suspected to search for metastatic lesions.
On histology, papillomas display papillary fronds lined by bland columnar epithelium and are challenging to distinguish from a normal choroid plexus. The absence of mitotic activity, nuclear pleomorphism, and necrosis are the distinguishing features of aCPP and CPC. 32 These features contribute to a five times higher risk of recurrence with aCPP compared to typical ones. A mitotic activity ⩾2 mitoses per 10 high-power fields defines an aCPP, whereas CPC has⩾5 mitoses per 10 high-power fields. 33 CPP express cytokeratin, vimentin, and S-100 with variability compared to that of CPC. The negativity of Epithelial Membrane Antigen and Glial Fibrillary Acidic Protein (GFAP) favors the diagnosis of CPP, even though GFAP may be seen focally in some cases. Moreover, immunochemistry shows positivity for vimentin, synuclein, and S-100 in aCPP. 6
It is worth knowing that there is an infrequently described benign type of CPTs in the literature known as choroid plexus adenoma. They are not recognized by the WHO classification as a separate entity but appear well-differentiated tubular glands with irregularly shaped acini, usually responsible for hydrocephalus. On CT scan, they appear as a calcified mass with solid and /or cystic components with possible absence of ventricular enlargement but with increased cerebral perfusion on MRI. They have a good prognosis after various therapeutic approaches, ranging from subtotal to complete resection. 6
The treatment of CPC involves a multimodal approach aimed at improving survival. Significant mortality is attributed to blood loss. While preoperative angiography is deemed unnecessary in general, 34 it is performed in some cases of extraventricular localizations with the aim of revealing blood supply, “blushes” of enlarged arterial feeders, or early filling of the tumor-draining veins. Preoperative embolization can minimize these risks and improve tumor respectability. The use of transcollation devices is also a recent technique performed for this purpose. 4 To prevent recurrences and prolong survival, neoadjuvant chemotherapy is used in a limited manner, while it is administered with or without radiation in cases of aCPPs with metastases and incomplete resection.1,6
Gross total surgical resection remains the gold standard treatment for CPP, and all efforts should be directed toward achieving this goal. Following the removal of the choroid plexus or other intraventricular neoplasms, it is essential to assess intracranial pressure. Resolution of hydrocephalus must be achieved, and further treatment is necessary if it is not resolved. 29 However, there have been reported cases of persistent hydrocephalus in CPP after treatment, of which management remains not well established due to lack of long-term follow-up and clinical data. 6
Conclusion
aCPP is a grade 2 WHO intraventricular tumor with intermediate malignancy between CPP and CPC, which are the other two categories of CPTs. MRI plays a crucial role in establishing the diagnosis, revealing a papillary or lobulated intraventricular tumor with homogeneous enhancement. However, the absence of enhancement and the presence of cysts may represent morphological features of atypia, allowing for the improvement of pretreatment diagnosis.
Footnotes
Authors contribution
All authors participated actively in elaboration of this scientific document: H.A., who’s the correspondent one, wrote the text; S.B.E.H. contributed to its elaboration; C.M. was the neurosurgeon who operated the patient; M.A. is the anatomopathologist who set the diagnosis; A.F. is the chief of department, who validated the case.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
