Abstract
Pyomyositis is a pyogenic infection of skeletal striated muscle, usually found in tropical areas, often in immunocompromised patients. We report a new observation of a nontropical Enterobacter pyomyositis occurring in an immunocompetent female in Tunisia. A 53-year-old patient presented with acute fever and intense myalgia in the right thigh. On clinical examination she had an altered general condition, a fever at 40°C and an important swelling of the lateral side of the right thigh. In biology, she had an inflammatory syndrome. Blood culture had identified Enterobacter. Muscle magnetic resonance imaging showed diffuse inflammatory involvement of the vastus lateralis muscle of the right quadriceps associated with edematous infiltration of subcutaneous fatty tissues.
Diagnosis of pyomyositis was retained. Antibiotic therapy initially probabilistic and then adapted to the antibiogram was initiated with a favorable outcome. Although rare outside the tropics, the potential severity of pyomyositis encourages its better knowledge.
Introduction
Pyomyositis was first described by Scriba in1885. 1 It is defined as a pyogenic infection of the striated skeletal muscle. It is common in Central Africa and in the South Pacific and has earned the name “tropical pyomyositis.” 2 However, in temperate countries, pyomyositis is unusual and occurs only in immunosuppressed or chronically ill adult patient. 3 On the other hand, the Staphylococcus aureus is the most common causative agent. 3 However, it is uncommon for Gram-negative organisms to cause pyomyositis. 4
Herein, we report an original case of a nontropical Enterobacter pyomyositis in an immunocompetent female from Tunisia, a temperate country in the north of Africa.
Case report
A 53-year-old female with a history of well-balanced hypothyroidism and a newly diagnosed anal fissure was admitted for acute installation symptomatology for 2 days, combining fever and pain with diffuse swelling in the right thigh. She was unable to walk. On clinical examination, the patient had a slightly altered general condition and fever at 40°C. The right thigh was notably swollen and erythematous. The underlying compartment felt firm. The cardiac auscultation was without abnormality as well as the rest of the clinical examination.
In biology, the initial complete blood count was remarkable for leukocytosis at 12,000/mm³ predominantly neutrophils. The C-reactive protein was elevated at 443 mg/l. The muscle enzymes were normal as well as the rest of the biological parameters. Cytobacteriological examination of urine was negative. Viral serology for HBV, HCV, and HIV were negative. Blood culture identified Enterobacter. Anti-nuclear antibodies were negative. The chest X-ray, the transthoracic ultrasound, and the abdominal ultrasound were normal.
Soft tissue ultrasound noted an heterogenous thickening of the vastus lateralis muscle with edematous sub aponevrotic infiltration consisting of an acute myositis. Muscle magnetic resonance imaging (MRI) showed diffuse inflammatory involvement of the vastus lateralis muscle of the right quadriceps associated with edematous infiltration of subcutaneous fatty tissues (Figure 1). Theses imaging findings were consistent with an acute myositis. In the muscle biopsy, there was a significant unevenness in the size of the fibers. Foci of necrosis and regeneration were noted. An important edema of the connective tissue with fragmentation of the septa and a significant increase in the intermyocytic space were associated.
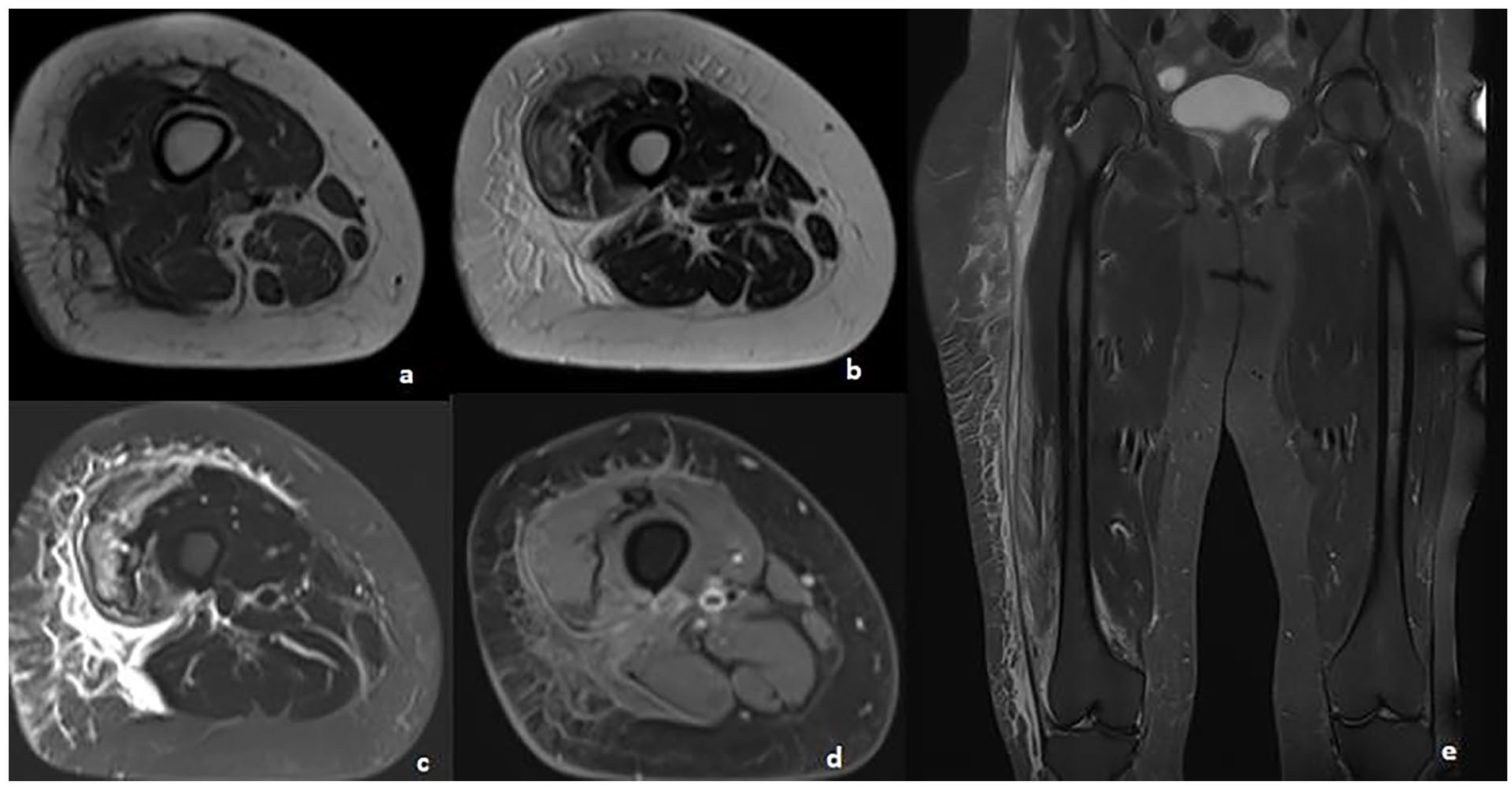
Axial-section T1 (a), T2 (b), STIR (c) and T1 FS after injection of Gadolinium (d) and coronal-section STIR (e) showing edema of the vastus intermedius, vastus lateralis, and right biceps femoris muscles, in hyposignal T1, hypersignal T2 and STIR, enhancing after injection of Gadolinium, associated with significant infiltration of the subcutaneous fat opposite.
There were multiple intra and perifascicular autophagic vacuoles testifying to myocytolysis. There was an important intraparenchymal and perivascular inflammatory infiltrate with disorganization of the parenchymal structure. The vessel wall was slightly thickened. The distribution of types 1 and 2 fibers was normal. There was no mitochondrial, glycogenic, or lipid overload (Figure 2). These histological findings were suggestive of severe inflammatory myopathy evoking pyomyositis or dermatomyositis. No muscle culture was performed at the time of the muscle biopsy. The antinuclear antibodies and the dot myositis were negative. The diagnosis of Enterobacter pyomyositis favored by anal fissure was retained.
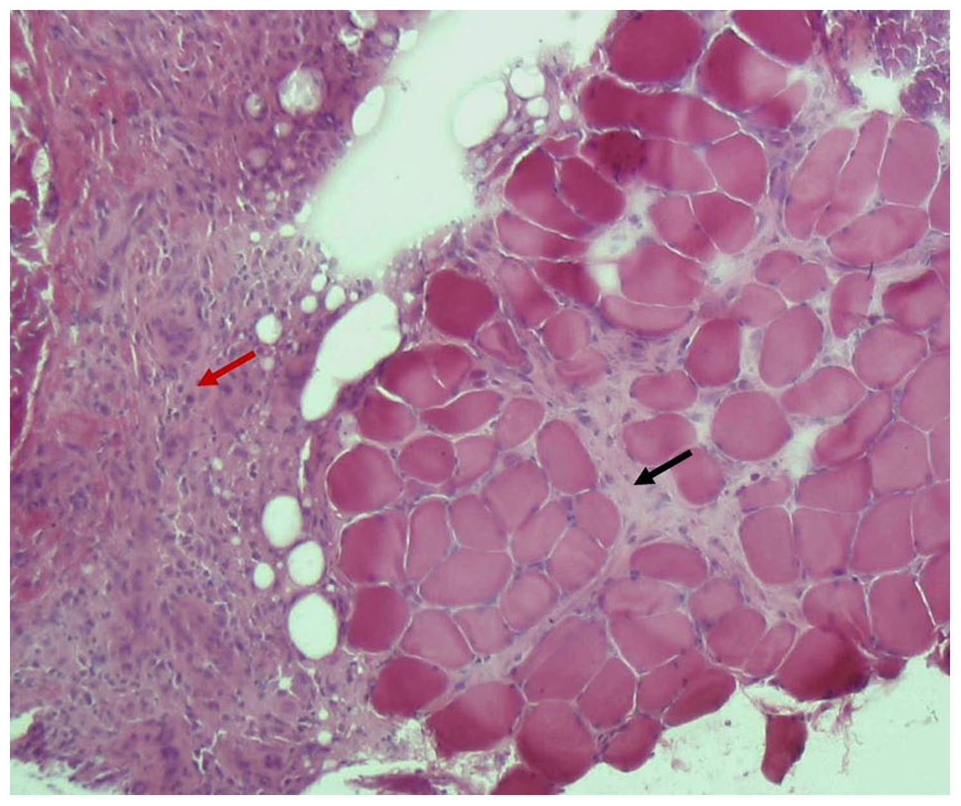
H&E-stained muscle biopsy showing endomysial inflammation and edema (black arrow) associated with severe fasciitis (red arrow).
The patient was initially treated with probabilistic antibiotic therapy consisting of oxacillin (2g*4/day) intravenously associated with oral ciprofloxacine (500 mg*2/day). Amikacin 1 g/day was also associated with the five first days. Based on the antibiotic sensitivity, the current antibiotics were maintained. The whole duration of antibiotherapy was 4 weeks.
Clinical evolution was remarkable for a stable apyrexia since 72 h of antibiotherapy; then, a progressive regression of local inflammatory signs was noted. Anal fissure has also improved. A prompt recovery was obtained. Biology showed a decrease, and then a normalization of C-reactive protein and leukocyte levels. Colonoscopy performed 2 months later showed no abnormality. A control MRI, performed after 2 months of evolution, showed a discrete hypersignal T2 of the lower half of the vastus lateralis and a contrast taking of the fascia in view (Figure 3). In the subsequent 4-year-follow-up of our patient, no relapse was noted.

Control magnetic resonance imaging in axial T1-weighted (a), T2-weighted (b), T1 FS after gadolinium injection, (c) and coronal STIR (d), showing disappearance of muscle edema with normal appearance of the thigh.
Discussion
Pyomyositis is a bacterial infection of the striated skeletal muscle of acute or subacute onset. Its pathogenesis remains unclear. The occurrence of pyomyositis in nontropical areas should motivate the search for predisposing factors such as a muscle injury, surgery, or ischemia or use of intravenous drugs or a little-known immunosuppression state. 5 Pyomyositis predominantly affects the muscles of the lower limbs and specially the quadriceps and iliopsoas followed by the gluteal muscles. The muscles of the upper limb, trunk, and spine can also be involved. 2
Pyomyositis presents initially as muscular inflammation, generally of subacute onset and it later progresses to form abscesses. 6 In severe cases, pyomyositis can lead to shock and death. Differential diagnosis may include deep venous thrombosis, osteomyelitis, malignant tumors, hematoma, and septic arthritis. 7 All the authors agree that the most sensitive and contributing imaging examination to the early diagnosis of pyomyositis is the muscle MRI. When this later is not available in emergency, computed tomography (CT) scan or ultrasound can help the early recognition of this challenging and potentially life-threatening diagnosis. In fact, Nelson reported a case of pyomyositis established on the basis of the features showed by a high-frequency linear ultrasound consisting of an intramuscular abscess of the vastus lateralis. 8 However, muscular biopsy is not necessary for diagnosis in typical cases.
In cases of pyomyositis, aspirated fluid cultures are usually positive, whereas blood cultures are usually negative. 7 The S. aureus is the most common causative agent; it is responsible for up to 50%–95% of pyomyositis cases in all age groups. 2 Less frequently, streptococcus is involved. However, it is uncommon for Gram-negative organisms to cause pyomyositis.4,5 There are only a few reports of pyomyositis caused by Escherichia coli. Moreover, to our knowledge, this is the fourth case of pyomyositis due to Enterobacter in the literature. The first and the second case of such condition were reported, respectively, by Falasca 9 in 1994 and Gousseff 10 in 2013. The third case was reported by Meehan et al. 4 in 2023 in a patient with a bone marrow failure of unknown etiology.
Probabilistic broad-spectrum antibiotic therapy should be started immediately on the diagnostic suspicion of the risk of occurrence of the state of septic shock. The principal treatment for suppurative pyomyositis is surgical incision and drainage combined with intravenous antibiotic administration for 2–3 weeks. Such drainage can be performed via open surgery, aspiration under ultrasound or CT guidance, or percutaneously. 5
Conclusion
Although rare in immunocompetent adult outside the tropics, the potential severity of pyomyositis encourages its better knowledge. This condition should be considered in front of any localized myalgia associated with a fever. Soft tissue ultrasound and CT scan can contribute to the diagnosis of pyomyositis. But the MRI remains the gold standard. The identification of the causative agent is essential to adapt the probabilistic antibiotic therapy initially introduced.
Footnotes
Acknowledgements
The authors would like to thank Ms Slim Jerbi and Ms Mohamed Gannar for their kind assistance in the elaboration of this article.
Author contributions
All the authors participated in the design, performance, analysis, and drafting of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
