Abstract
En plaque meningioma is a rare type of meningioma characterized by an infiltrative nature, sheet-like growth, and at times invading the bone. We report here a case of en plaque meningioma. The patient was a 66-year-old woman presenting with headache and painful bilateral proptosis. Cerebral magnetic resonance imaging revealed a bilateral en plaque meningioma showed as a bilateral hyperostotic of greater wing sphenoid bone associated with bilateral thickening and enhancement of the dura in the anterior temporal area and the retroclival region invading bilaterally the cavernous sinus, the prechiasmatic portion of the optic nerve, and the lateral rectus muscle of the orbit through the superior orbital fissure causing bilateral exophthalmia. Due to invasion of the cavernous sinus and the orbital apex, a subtotal but extensive removal combined with bony decompression of the cranial nerves at the superior orbital fissure and optic canal followed by adjuvant radiotherapy frequently produces good functional and cosmetic results, and over a 3-year period, follow-up magnetic resonance imaging scans showed no obvious signs of recurrence.
Introduction
En plaque meningiomas (MEPs) constitute a rare variation of meningioma, accounting for a mere 2%–9% of all meningiomas.1,2 These unique tumors tend to exhibit a higher occurrence rate in females compared to males, particularly manifesting in the fifth decade of life. Our case serves as an illustrative example that underscores the significance of the clinical indicators and cerebral magnetic resonance imaging (MRI) findings. Notably, these findings are characterized by the distinctive “carpet-like” encroachment into surrounding bone, marked by extensive hyperostosis and dural thickening. The diagnostic process is further complicated by their atypical radiological presentation, while the surgical aspect is particularly intricate due to their proclivity for infiltrating osseous structures and intricately interweaving within nearby fissures and foramina. 3
Case report
We report a rare case of a 66-year-old woman, who exhibited bilateral proptosis, diminished visual acuity, retrobulbar pressure, headaches, and orbital pain associated with swelling of the temporal region, over the course of recent months. The patient’s condition prompted her visit to the emergency department due to worsening visual symptoms and excruciating headaches. A thorough physical and ophthalmological examination revealed stable hemodynamic and neurological status, alongside a noteworthy reduction in visual acuity and more pronounced right-sided exophthalmos.
Subsequent cerebral MRI showed bilateral hyperostotic changes of greater wing sphenoid bone displaying hypo intensity on both T1 and T2 sequences (Figure 1) associated with bilateral thickening and enhancement of the dura in the anterior temporal area invading the cavernous sinus, the prechiasmatic portion of the optic nerve, and the lateral rectus muscle bilaterally more pronounced on the right side through the superior orbital fissure causing bilateral exophthalmos (Figure 2).

Axial T2 magnetic resonance imaging image shows bilateral hyperostotic changes of the greater wing of the sphenoid bone (blue asterisk) associated with bilateral exophthalmos more pronounced on the right side (red arrow).

Post-contrast axial magnetic resonance imaging image shows bilateral thickening and enhancement of the dural in the anterior temporal area (blue arrow) invading the cavernous sinus (yellow asterisk), the prechiasmatic portion of the optic nerve, and the lateral rectus muscle bilaterally more pronounced on the right side (red arrow) through the superior orbital fissure causing bilateral proptosis.
A post-contrast sagittal MRI image showed thickening and enhancement of the free border tentorium cerebelli and the dura in the retroclival region (Figures 3 and 4).

Post-contrast sagittal magnetic resonance imaging shows significant thickening and enhancement of the dura in the retroclival region (red arrow).
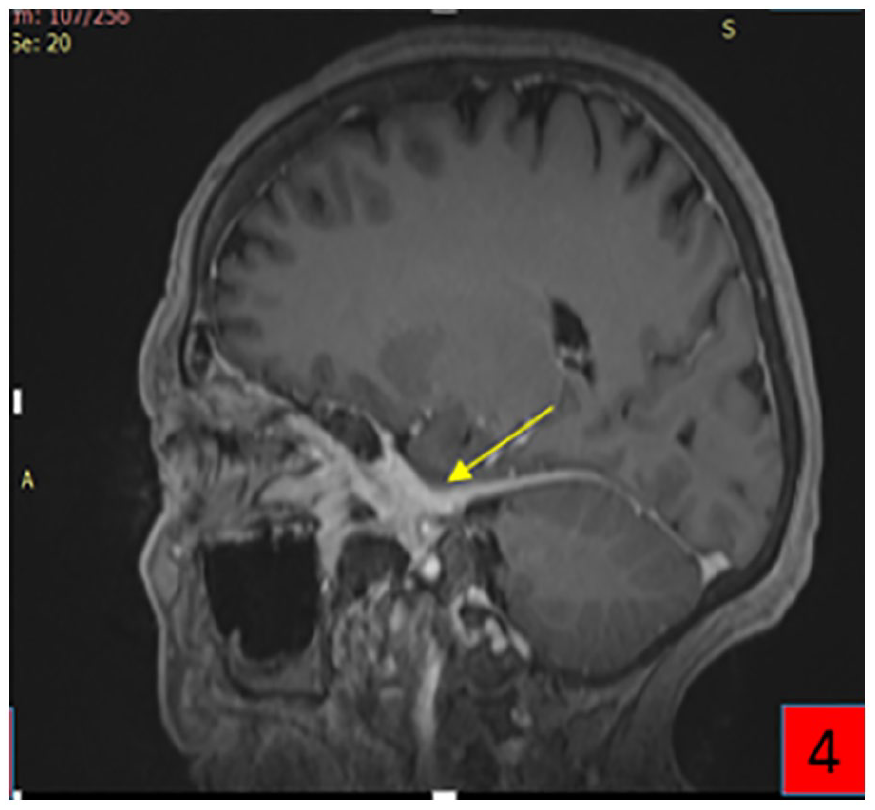
Post-contrast sagittal magnetic resonance imaging shows thickening and enhancement of the free border tentorium cerebelli (yellow arrow).
Given the bone involvement, our initial differential diagnosis encompassed Paget’s disease and fibrous dysplasia. Endoscopic transsphenoidal biopsy has been achieved to confirm the diagnosis immunohistologically. This confirmation is essential for determining the appropriate course of action depending on the nature of the lesion (benign or malignant). The pathological examination confirmed the infiltration of the bone by meningioma cells. Additional therapeutic interventions were deemed unnecessary. Following the definitive diagnosis, the patient was admitted to the neurosurgery department, where a subtotal but extensive removal combined with bony decompression of the cranial nerves at the superior orbital fissure and optic canal was achieved. Subsequently, our patient was referred to the radiotherapy department for further management, where a treatment protocol was established. The postponement of the cranioplasty to a later date was decided. Due to the increased likelihood of recurrence associated with the bilateral nature of the condition and the extent of infiltration of adjacent structures, the neurosurgery team opted for regular monitoring, with MRI scans every 3 months, over a period of approximately 2 years. If there are no signs of recurrence in the patient during this period, cranioplasty will be considered.
After surgery and having completed all his radiotherapy sessions, follow-up MRI scans showed no obvious signs of recurrence over 1.5 years.
Discussion
MEP is not greatly raised above the level of dura mater but is notoriously prone to invade the adjacent bone with accompanying hyperostosis. This group of meningioma essentially infiltrates the brain and orbit mainly by way of natural openings, such as foramina and fissures as well as the perforating blood vessels, which are also increased in number.
EPMs distinguish themselves from the more prevalent en masse meningiomas through their characteristic “carpet-like” intrusion into adjoining bone, marked by extensive hyperostosis and dural thickening. Predominantly situated in the spheno-orbital regions, MEPs are less commonly found along the cerebral convexity, temporal bone, and foramen magnum.2,3 The distinct radiological appearance of EPMs poses a diagnostic challenge, while their inclination to infiltrate osseous structures and interlace within fissures and foramina presents surgical complexities. 4
The clinical manifestation of EPMs hinges on their location and extent of spread. Symptoms arise due to direct neural compression and tumor invasion or as a consequence of bony hyperostosis, which can constrict neural passages. For instance, sphenoid wing EPMs may lead to reduced visual acuity and visual field abnormalities. Hyperostosis of orbital bones can result in proptosis, alongside complaints of retrobulbar pressure, orbital pain, and headaches.
MRI and computed tomography (CT) constitute the initial diagnostic imaging modalities. For meticulous assessment of bony involvement and hyperostosis, particularly at the skull base, certain experts advocate for thin-slice CT bone windows. MRI detects dural and intradural involvement, with post-gadolinium T1 sequences typically highlighting contrast enhancement patterns. MRI aids in comprehending tumor infiltration into the orbit, cavernous sinus, and clinoidal tissues, particularly evident through post-contrast fat suppression T1-weighted imaging that accentuates dural enhancement and soft-tissue engagement, as observed in our patient. 5
Hyperostoses observed in EPMs are characterized by surface irregularities and inward bulges of the affected structures. These subtle features aid in distinguishing hyperostosis from other conditions such as primary intraosseous meningioma, diffuse large B-cell lymphoma, plasmacytoma, meningeal fibrosarcoma, cerebral metastases, and Paget’s disease. 6
Preoperative imaging is pivotal in identifying anatomical constraints, evaluating potential bony and muscle invasion during exposure, and scrutinizing various cranial structures, including nerves, optic canal, superior orbital fissure, and orbital compression.
The fronto-temporo-sphenoidal craniotomy with resection of invaded tissues, including the dura mater, muscle, and intra-orbital tissues, along with dural and parietal reconstruction, is the treatment of choice.
The approaches for positioning, exposure, and craniotomy are highly individualized, tailored to the tumor’s specific location, and often involve employing techniques suited to MEP features. In some instances, extensive measures, such as employing large bi-coronal skin incisions, have been taken to procure sufficient pericranium for repairing dural defects. There have been reports of encountering bony and muscular invasion during exposure. Due to the tumor’s widespread and extensive dural infiltration, a large craniotomy is frequently necessary, particularly when infiltration occurs along the convexity dura. The squamosal and lateral sphenoid bone may display hypervascularity and hyperostosis, necessitating complete bony removal before opening the dura and resecting the tumor. If the tumor or hyperostosis extends toward the infratemporal fossa, it is the authors’ practice to inferiorly reflect the zygoma to facilitate drilling access.
All foramina involving cranial nerves should undergo proper decompression with a clear view of underlying neural structures. Consequently, osteotomies should be broad to ensure maximum symptomatic relief, as invaded and expanded osseous elements are the primary contributors to most symptoms. Consideration may be given to removing the lateral wall and roof of the orbit if there is concern for orbital invasion. Alternatively, in cases of intraorbital invasion without periorbital or orbital roof reconstruction, stripping periorbital tissue or performing aggressive coagulation of residual tumor tissue may be considered. 7
Therefore, the potential advantages of complete resection must be balanced against the heightened risk of surgical complications. Subtotal resection is commonly favored for intra-orbital tumors, those invading the cavernous sinus, extending beyond the tentorial notch, or infiltrating the superior orbital fissure. The treatment strategy for MEP associated with Cavernous sinus invasion relies on subtotal resection of all the involved bones followed by adjuvant radiotherapy. 8
Table 1 summarizes the results reported by various authors concerning the excision of spheno-orbital meningioma. There is a diversity of data, particularly with regard to cavernous sinus involvement, recurrence rates, and recurrence management options.9,10
Summary of results on spheno-orbital meningioma excision: a review of studies and reported cases.

The literature debates the necessity of reconstructive surgery after orbital resection to prevent cosmetic issues and enophthalmos/exophthalmos. Early beliefs favored reconstruction when multiple orbital walls were removed. However, recent studies without reconstruction showed no significant increase in functional or cosmetic problems, especially when preserving the periorbita. Some studies even reported the resolution of proptosis without enophthalmos, emphasizing the importance of preserving the orbital rim attached to the periorbita. Current literature suggests that reconstruction decisions should consider periorbita violation rather than bony removal. Porous polyethylene 3D-printed implants are an option in cases of periorbita violation, encouraging fibrous ingrowth for structural support without tissue ingrowth on the orbital side. Alternatives like titanium mesh and autologous bone grafts exist, with the choice influenced by surgeon preference. 10
In our case, a subtotal resection approach was coupled with postoperative adjuvant radiotherapy with improvement in clinical outcome, and follow-up MRI scans showed no evidence of recurrence of the tumor over a 1.5-year period. Bone reconstruction has not been carried out; yet, it will depend on the progression of the disease.
Conclusion
In conclusion, when there is suspicion of MEP during diagnostic evaluation, obtaining an MRI is essential to minimize the risk of misdiagnosis. Surgical resection, once the diagnosis is confirmed, is recommended to reduce the risk of cosmetic deformity and incomplete resection, thereby lowering the chances of recurrence. Intraoperatively, extensive removal of the dura is crucial to prevent recurrence, and adequate decompression of all involved cranial nerve foramina is necessary. In addition, if there is evidence of tumor invasion into the cavernous sinus or signs of recurrence on follow-up imaging, postoperative radiation therapy should be considered.
Advancements in medical imaging, surgical techniques, and therapeutic interventions have expanded the treatment options for MEPs. Further research is required to assist neurosurgeons in determining optimal treatment strategies based on individual tumor characteristics and patient presentation when dealing with an MEP.
Footnotes
Acknowledgements
We thank Professor Firdaous Touarsa’s insight vis-a-vis the case.
Author contributions
C.L., A.N., and I.H.H. contributed to the conception, acquisition, analysis, interpretation of data, and drafted the manuscript. F.T., M.F., and M.J. critically revised the manuscript and approved it.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
No ethical approval is required for de-identified single case reports based on our institutional policies.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
