Abstract
Posterior reversible encephalopathy syndrome is a clinical-neuroradiological syndrome with typical neuroimaging features of posterior cerebral white matter changes that are usually reversible. However, there are only few reports of burns with posterior reversible encephalopathy syndrome in the literature. Hence, it is a clinical entity that many burn medicine physicians may be unfamiliar with. We report a case of severe burns complicated by posterior reversible encephalopathy syndrome in a 14-month-old male patient. On the eighth day of hospitalization, the child had persistent fever, occasional convulsions, eyes staring to the right, and high-pitched cry. Magnetic resonance imaging on day 10 showed the diagnosis is posterior reversible encephalopathy syndrome. We used hormone therapy to reduce cerebral oedema, oxcarbazepine to control convulsions, and multiple other drugs and physical measures to treat fever. The symptoms, signs, and imaging abnormalities of his posterior reversible encephalopathy syndrome were rapidly reversed in a short period of time. At the 1-year follow-up, the patient had recovered completely with no residual neurological signs and symptoms. To our knowledge, the patient may be the youngest recorded patient with both burns and posterior reversible encephalopathy syndrome. Careful observation, computed tomography, and magnetic resonance imaging can achieve early detection, early diagnosis, and early treatment of posterior reversible encephalopathy syndrome, which facilitates the achievement of desired therapeutic results. Further investigation is required to determine whether burns can serve as an independent posterior reversible encephalopathy syndrome causative factor and clarify the underlying pathogenesis mechanism.
Introduction
Posterior reversible encephalopathy syndrome (PRES) is a clinical-neuroradiological syndrome with typical neuroimaging features of posterior cerebral white matter changes that are usually reversible. The main clinical manifestations of PRES are headache, seizures, visual changes, and consciousness disorder. 1 This syndrome is associated with hypertension, eclampsia, renal insufficiency, connective tissue disease, and the use of cytotoxic and immunosuppressive drugs. Thus far, most reports of PRES have been reported in adults; reports of PRES in children are limited to nephropathy, allergic diseases, autoimmune diseases, and the use of immunosuppressive and biologic drugs. 2 Reports of burn-related PRES are very rare in the literature. Therefore, it is a clinical entity that many burn medicine physicians may be unfamiliar with.
Herein, we report a case of severe burns complicated by PRES in a 14-month-old male patient and discuss the treatment and 1-year follow-up. Treatment and care were challenging given the patient’s young age. We analysed and summarized the treatment experience of this patient, combined with the review of relevant literature, and speculated on the pathogenesis, hoping to help clinicians recognize the possibility of PRES in burns and provide effective and timely treatment.
Case presentation
A 14-month-old male patient was admitted to hospital on 19 May 2021 for multiple burns all over his body. The burn area was 25% (superficial second-degree and deep second-degree) of the total body surface area (TBSA). His burns were located on the head, face, neck, chest, abdomen, back, and upper extremities. Deep second-degree burns (5% TBSA) were mainly located on the face, chest, and abdomen. Most of the burns in the remaining areas were superficial second-degree (11% TBSA). The child was healthy in the past and had no history of food and drug allergies.
Treatment
After admission, the patient was treated with fluid rehydration, blood transfusion, antibiotics to prevent infection, tracheal intubation–assisted ventilation, and regular dressing changes on the wound areas (Table 1). From the second to seventh day of hospitalization, the patient had persistent fever; the antibiotics were changed according to drug sensitivity test results to better treat the infection. On the eighth day of hospitalization, tracheal intubation was removed, but the child still had persistent fever, occasional convulsions, eyes staring to the right, and high-pitched cry. Computed tomography (CT, day 8) showed that the intracranial brain parenchyma was scattered with patchy low-density foci, and patchy high-density shadows were seen in both lungs with blurred edges; mild cerebral oedema and pneumonia were considered in the initial diagnosis (Figure 1). On day 10, lumbar puncture and cerebrospinal fluid examination showed that the intracranial pressure was 228 mmH2O (reference range: 40–100 mm H2O), and the routine and biochemical abnormalities of cerebrospinal fluid were normal. Magnetic resonance imaging (MRI) on day 10 showed that bilateral fronto-parietal-temporal-occipital white matter and corpus callosum had symmetrical abnormal signals. The diagnosis is PRES (Figure 2). After pulse therapy with methylprednisolone (the dose was gradually reduced to oral prednisone), oxcarbazepine for anti-epileptics, and multiple other drugs and physical measures to treat fever, his limb activity, mental response, and eye gaze gradually improved. The episodes of convulsions also reduced. The burn wound healed on day 18 of admission. Intracranial pressure on lumbar puncture was 232 mmH2O on day 13 and 150 mmH2O on day 21; no other obvious abnormality was found in the cerebrospinal fluid test. Contrast-enhanced MRI performed on day 26 showed that there were multiple abnormal signals in the bilateral frontal, parietal, and occipital lobes, and the lesion range was smaller than that seen on the day 10 MRI. At this stage, PRES complicated with subacute late cerebral haemorrhage was considered. From day 27, the child had no fever, no right gaze, and no more convulsions, and his limb activities and mental reactions continued to improve further. Contrast-enhanced MRI on day 33 confirmed PRES combined with subacute late intracerebral haemorrhage; the haematoma was absorbed more than that seen in the previous MRI scan. We continued treatment with levetiracetam and piracetam for neuroprotection, oxcarbazepine for anti-epileptics, and oral prednisone to reduce cerebral oedema. On day 45 of hospitalization, the patient had no fever, convulsions, and right-eyed gaze; the fine movements of his hands improved; and he was discharged from the hospital.
Details of treatment and follow-up.

CT: computed tomography; MRI: magnetic resonance imaging; PRES: posterior reversible encephalopathy syndrome; EEG: electroencephalogram.

Computed tomography findings on day 8 from the onset: the intracranial brain parenchyma was scattered with patchy low-density foci. (As shown by the arrows).

MRI (day 10) showed bilateral fronto-parietal-temporal-occipital white matter and corpus callosum with symmetrical T1W hypointensity and T2W hyperintensity foci, enhanced sulcus lines, and patchy enhancement. (As shown by the arrows).
Follow-up
After discharge, oral prednisone (within 45 days after discharge) and oxcarbazepine (within 1 year after discharge) were continued to prevent epilepsy (Table 1). MRI performed 17 days after discharge showed that the multiple abnormal signals in the original bilateral frontal, parietal, and occipital lobes were significantly absorbed and reduced, and there were still softening foci in the bilateral frontal lobes, and most of the original bilateral frontal lobe and right occipital lobe haematoma had been absorbed. The right frontal lobe still had patchy chronic phase bleeding changes (Figure 3). CT performed 4 months after discharge showed bilateral frontal lobe softening foci (predominant on the right). The vital signs of the child were stable, and the limb movement and fine movements of the hands gradually improved. One year after discharge from the hospital, the child’s limbs and fine movements were normal; he could walk, run, and jump normally; and he could speak in short and simple sentences.

MRI (17 days after discharge) showed that the multiple abnormal signals in the original bilateral frontal, parietal, and occipital lobes were significantly absorbed and reduced. (As shown by the arrows).
Discussion
History
In 1996, Hinchey et al. 3 first proposed the concept of reversible posterior leukoencephalopathy syndrome (RPLS). In 2000, Casey et al. 4 found that this syndrome can also involve the cerebral cortex; hence, the name ‘PRES’ was proposed. Each definition has certain limitations and does not provide an accurate overview of the various symptoms of this syndrome. However, because the symptoms of most patients are reversible and the lesions mainly affect the posterior circulation area of the brain, the most commonly used name in clinical practice is still PRES. In 2020, Ganzevoort et al. 5 suggested that the earliest descriptions of PRES-related symptoms may date as far back to 1672 or even earlier. Its incidence is not known outside case reports and case series. 6 With the popularization and advancement of imaging technologies, such diseases are considerably better understood now than earlier. In addition to frequent findings in neurology, because of the particularity of the patients with this syndrome, involvement of emergency departments, cardiology, nephrology, haematology, rheumatology, oncology, transplantation, and other departments is also frequently found.
Aetiology
The causes of PRES are diverse, including hypertensive encephalopathy, eclampsia, collagen disease, severe infection, anaemia, some drugs such as immunosuppressive agents, antibiotic drugs, trauma, surgery, angiography, blood stem cell transfer, rich cellular transfer, and more recently, COVID-19.7–9 PRES differs from the adult in aetiology, precipitants, and clinical manifestations. 2 The proportion of patients with combined anterior/posterior circulation was higher. 2
In 2020, Wake et al. 10 reported a case of PRES that developed during the treatment of severe burns in a 13-year-old boy. The authors believed that it was caused by an increase in blood pressure, blood vessel dilation, angioedema, and stress during skin grafting. In 2022, Ravishankar et al. 11 reported PRES which presented 2 months after burns (41% surface area) in a 20-year-old female patient. The aetiology is considered to be related to factors such as extensive spinal lesions, the use of immunoglobulin, and high blood pressure caused by burns.
To our knowledge, the patient described in this report may be the youngest recorded patient with both burns and PRES. He had hypertension at the time of onset, which may also be the cause of PRES. Sepsis (particularly gram-positive) has also been reported as an important cause of PRES. 12 However, our patient did not have ongoing sepsis. In addition, hypovolemia can also be a cause of PRES, but this patient was clinically symptomatic on day 8 of the burns when hypovolemia was no longer present. At present, there is no other evidence that the onset of PRES occurs at an earlier time. Therefore, whether burns can serve as an independent causative factor of PRES is still unclear and deserves further study.
Pathogenesis
Although extensive research has been conducted on the pathogenesis of PRES, the exact pathophysiological mechanism remains incompletely understood and is a bit controversial. Vasogenic oedema caused by perturbation of cerebral vascular autoregulatory mechanism is considered the main pathological feature of PRES. There are three main mechanisms.13–16 The first mechanism is the ‘hypoperfusion’ theory. Ischaemia caused by cerebral vasospasm, increased sensitivity to pressor substances in blood circulation, decreased vasodilatory substances, and endothelial cell dysfunction lead to changes in vascular reactivity, causing vasospasm and reduced organ perfusion that can result in extravasation of the intravascular fluid. The second mechanism is the more popular ‘hyperperfusion’ theory of cerebral perfusion pressure. 14 The acute increase in blood pressure exceeds the upper limit of cerebrovascular self-regulation and causes passive dilation of cerebral blood vessels, increased cerebral perfusion pressure, opening of tight junctions between capillary endothelial cells, increased capillary permeability, and increased blood-brain pressure. The barrier is compromised and plasma components leak into the brain parenchyma. The vascular sympathetic nerve fibres in the posterior circulation of the brain are less distributed and easy to expand, so vasogenic cerebral oedema is more likely to appear at the back of the brain. The third mechanism is the theory of vascular endothelial injury. Various causes can lead to capillary endothelial cell damage, resulting in the destruction of the blood–brain barrier and subsequent oedema.
We speculate that the pathogenesis of our patient is related to severe burns, recurrent fever (Figure 4), and hypertension (Figure 5). First, in the pathological state, the excessive fluctuation of blood pressure in a burn patient exceeds the ability of vascular self-regulation to passively dilate cerebral blood vessels, increase cerebral perfusion pressure, and open tight junctions between capillary endothelial cells. Increased capillary permeability, damaged blood–brain barrier, and leakage of plasma components into the brain parenchyma result in vasogenic cerebral oedema. Second, our patient started to experience a significant increase in blood pressure on day 8 of treatment, and systemic vasospasm may have been involved at this time. Cerebral vasospasm leads to cerebral ischaemia and cytotoxic water, which induces PRES. In addition, burns can lead to activation of the immune system, excessive release of cytokines such as tumour necrosis factor-α (TNF-α) and interleukin 1 (IL-1), the disorder of electrolyte, and the increase in C-reactive protein(CRP) (Figure 6). Both TNF-α and IL-1 activate endothelial cells, causing circulating leukocytes to produce reactive oxygen species and proteases, which, in turn, lead to endothelial damage and fluid leakage. 12 Finally, sustained high temperature also affects the thalamus, cortex, basal ganglia, and cerebellum.
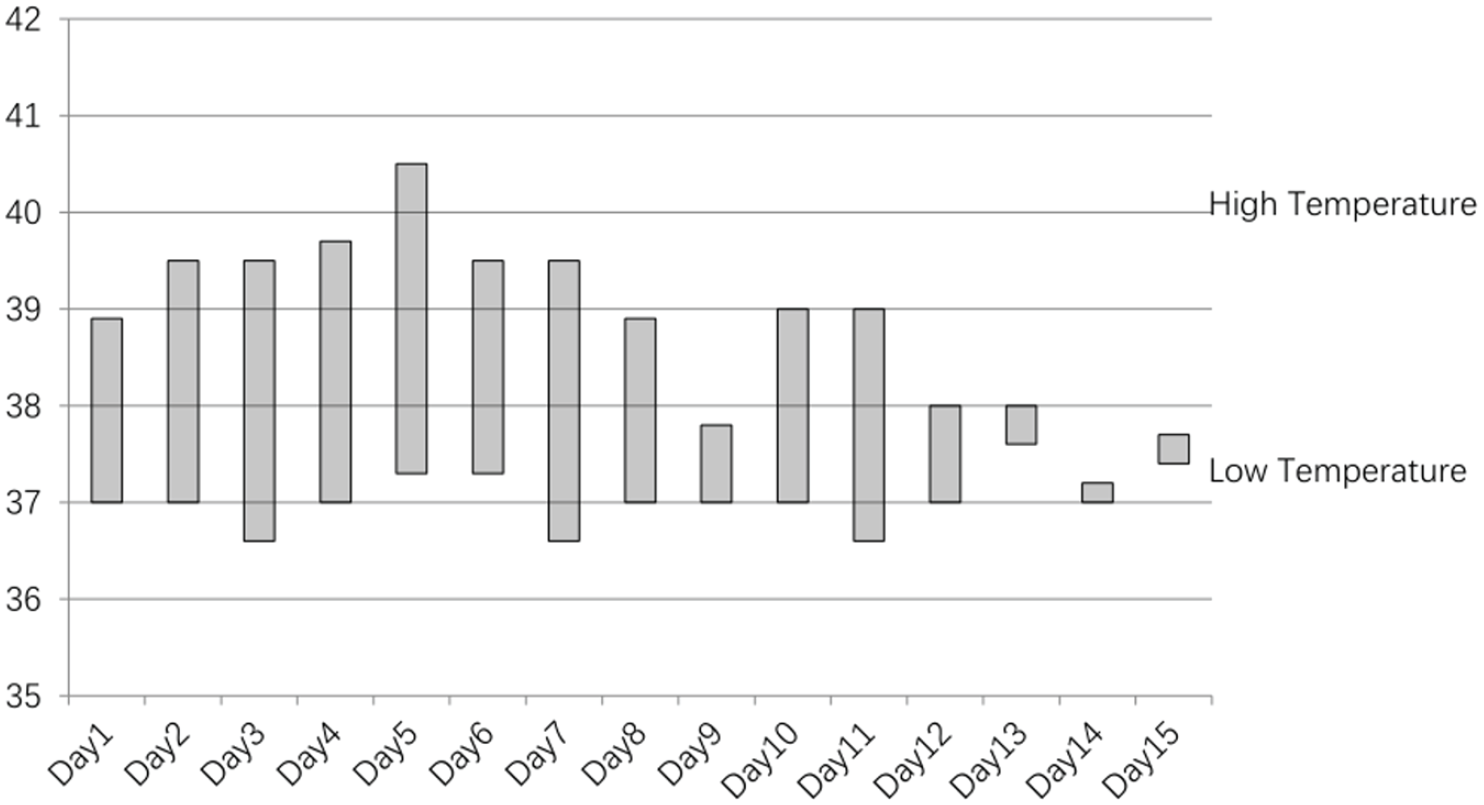
On days 1–15 of hospitalization, the child had recurrent fever.

The details of blood pressure.

Electrolyte and acute phase reactants.
Diagnosis
At present, there are no diagnostic guidelines for PRES. 17 The diagnosis of PRES mainly depends on initial symptoms, clinical manifestations, risk factors, and neuroimaging findings6,14 (Figure 7). The onset is often acute or subacute. Significant risk factors such as female sex, hypertension, and those currently undergoing active immunotherapy/chemotherapy are associated with PRES.6,17

Flow chart of diagnosis and treatment of PRES.
The clinical manifestations and risk factors are not sufficient to easily confirm the diagnosis. However, with the popularization of imaging examinations, the diagnosis of PRES has become easier in recent years. Careful observation, CT, and MRI can achieve early detection, early diagnosis, and early treatment of PRES, which facilitates the achievement of desired therapeutic results. CT can detect abnormal changes in the brain in a timely manner, but some diagnoses of acute intracranial pathology may be missed. MRI can be used as a gold standard for the diagnosis of PRES.17,18 Fang et al. 19 found that the serum neurofilament light (NFL) level of patients with PRES increased, which may provide an important research direction for the diagnosis and prognosis of PRES.
Treatment and prognosis
The clinical manifestations and imaging features of PRES are not necessarily reversible.14,20 If PRES is not diagnosed and treated in time, the patient’s condition may become worse or even die. 20 Therefore, PRES should be diagnosed and treated as soon as possible. The principle of treatment is the treatment of the aetiology and primary disease; symptomatic treatment includes lowering blood pressure, controlling convulsions, dehydration (lowering intracranial pressure), stopping the use of immunosuppressive drugs and chemotherapy drugs, and correcting acid–base balance and electrolyte imbalance.
In this case, we treated our patient with anti-infective agents, dressing changes, and blood transfusions and corrected the electrolyte abnormalities. At the same time, we used methylprednisone and prednisone therapy to control cerebral oedema; 21 oxcarbazepine to control convulsions; piracetam to reduce neuronal inflammation, 22 protect, and repair brain cells; continuous intravenous injection of nimodipine and sedatives to lower blood pressure steadily; and multiple drugs and physical measures to reduce the fever. The symptoms, signs, and imaging abnormalities associated with PRES were rapidly reversed in a short period of time. At the 1-year follow-up, the patient had recovered completely with no residual neurological deficit. Of course, in the future, during the treatment of burn patients, the drastic fluctuation of blood pressure should be avoided, and the haemodynamic stability of patients should be maintained to prevent the occurrence of PRES.
Conclusion
We presented a case of severe burns complicated by PRES in a 14-month-old boy, with details of 1-year follow-up. To our knowledge, the patient may be the youngest recorded patient with both burns and PRES. Although treatment and care were relatively challenging because of the patient’s young age and complicated condition, the patient recovered completely with no residual neurological deficit owing to timely and appropriate treatment. Careful observation, CT, and MRI can achieve early detection, early diagnosis, and early treatment of PRES, which facilitates the achievement of desired therapeutic results. However, further investigation is required to determine whether burns can serve as an independent PRES causative factor and clarify the underlying pathogenesis mechanism.
Footnotes
Acknowledgements
The authors are grateful to the staff of the associated centres for providing assistance.
Consent for publication
The patient’s parents signed an informed consent form to agree that the data can be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Ethical approval and consent to participate
The present study was conducted in accordance with the tenets of the 1975 Declaration of Helsinki. The patient’s parents provided written informed consent before investigations, screening, study, and treatment.
Informed consent
Written informed consent was obtained from the patient’s parents for their anonymized information to be published in this article.
