Abstract
Xanthogranulomatous pyelonephritis is a rare and severe form of chronic pyelonephritis. Spontaneous pyeloduodenal fistula is an extremely rare complication of xanthogranulomatous pyelonephritis. We present a rare case of xanthogranulomatous pyelonephritis complicated by a complex pyeloduodenal and ureteromuscular fistulae with a cutaneous opening. A 57-year-old male patient presented with chronic right flank pain and cutaneous discharge at the right lower quadrant. Xanthogranulomatous pyelonephritis was suspected on preoperative computed tomographic urography owing to the presence of fat deposits and complex pyeloduodenal and ureteromuscular fistulae. Open nephrectomy and fistula closure were performed and the patient recovered uneventfully. Xanthogranulomatous pyelonephritis was confirmed pathologically.
Keywords
Introduction
Xanthogranulomatous pyelonephritis (XGP) is a rare and severe form of chronic pyelonephritis characterized by destruction of the renal parenchyma and replacement by foamy, lipid-laden macrophages.1,2 Although XGP is usually confined to the affected kidney, it occasionally extends to adjacent tissues, resulting in further complications such as perirenal abscess, adhesion to pancreas, or various forms of fistula.1,3,4 Spontaneous pyeloduodenal fistula is an extremely rare complication of XGP. We present a case of diffuse XGP complicated by a complex pyeloduodenal and ureteromuscular fistulae with cutaneous opening. To our knowledge, there are only several case reports which describe such a complex fistula in XGP in the English literature.1,2 The patient underwent total nephrectomy combined with fistula closure and recovered uneventfully.
Case presentation
A 57-year-old male patient presented with fever, general malaise, weight loss, and chronic right flank pain. He first noticed a cutaneous discharge at the right lower quadrant 3 weeks ago. His medical history included extracorporeal shock wave lithotripsy of the right kidney years ago and recurrent urinary tract infection. Laboratory studies revealed leukocytosis (14.26/mL, range 4–10/mL) and elevated C-reactive protein level (43.02 mg/L, range 0–5 mg/L). Serum creatinine level was within a normal range (95 µmol/L, range 62–106 µmol/L). Urinalysis was normal and urine culture was negative. A 1 cm cutaneous opening with yellowish discharge was seen at the right lower quadrant. Real-time polymerase chain reaction (PCR) of the urine and pus were negative for Mycobacterium tuberculosis. A chest X ray appeared normal.
Plain abdominal radiography demonstrated a small right kidney shadow with compacted stones (Figure 1). A 4-phase low-dose computed tomographic (CT) urography was indicated. Pre-contrast images revealed an atrophic and deformed right kidney with multiple nephrolithiasis, parenchymal calcification, multifocal fat deposits throughout the renal parenchyma, and perirenal fat stranding (Figure 2). Gas was not evident. On serial contrast images, the renal parenchyma was completely destroyed and the pyelocalyceal system was unrecognizable. A loss of differentiation between the anterior renal parenchyma and second portion of the duodenum was also appreciated, suggesting an adhesion. Renal function was not seen in the delayed excretory phases, implying a severely impaired kidney function. The right ureter was dilated (# 10 mm) with wall thickening and intraluminal hyper-attenuation compared to the contralateral normal counterpart (Figure 3). Hydronephrosis and perirenal abscess were not observed in all phases.
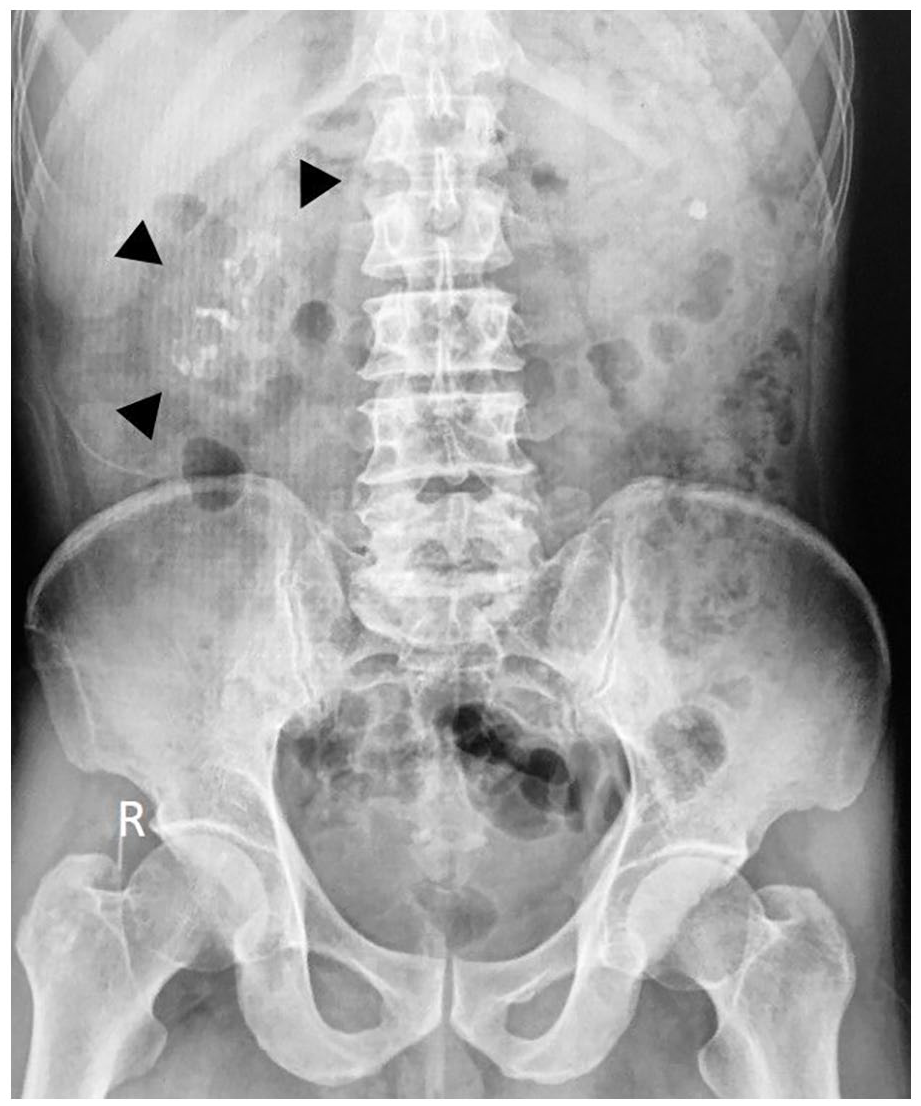
A plain abdominal radiograph shows scattered right kidney stones. Note the small right kidney shadow (arrowhead) compared to the left side and the presence of a left kidney stone.

Computed tomographic urography, non-contrast images. (a) Atrophic change and loss of reniform shape of the right kidney compared to the contralateral side. Scattered caliceal and pelvic stones and perinephric fat stranding are appreciated. (b) Parenchymal calcification (black arrow). (c) Focal fat deposit with an average density of −73.5 HU (measurement, white arrow).

Computed tomographic urography, serial contrast images. (a) and (b) Poor enhancement of the right renal parenchyma without corticomedullary differentiation on the corticomedullary phase (a) and nephrogenic phase (b) images. Note the close contact between the renal parenchyma and the D2 duodenum (long arrow) suggesting a fistula. (c) Delayed excretory phase image obtained at 60 min after contrast administration, prone position, demonstrates a dilated, pus-filled right ureter (short arrow). The ureteral wall is thickened and enhanced with surrounding fat stranding. These findings are consistent with chronic pyelonephritis and severely impaired renal function. (d) Sagittal reconstruction image reveals an intramuscular abscess formation with a cutaneous opening (black arrow).
A 7.5 cm × 6 cm × 3.5 cm fluid collection was found at the right lower quadrant, confined within the right iliac muscle. The collection had a thick enhanced wall and a 1 cm cutaneous opening. The right ureter was found to empty at the medial margin of the collection, indicating a chronic ureteral iliac muscle fistula. The “run-off” ureter was not seen on any delayed phases even with the patient was on prone position. The adjacent bony and vascular structures appeared normal. Other abnormality was a left kidney stone.
A CT fistulography was consequently performed which demonstrated a complex fistula from the intramuscular collection, connecting with the distal ureter upward to the right pyelocalyceal system and communicating with the adjacent duodenum (Figure 4), corresponding to stage III according to Malek and Elder 5 classification. There was no evidence of nephrobronchial fistula. XGP was highly suspected. Surgical exploration through a midline incision confirmed the presence of a pyeloduodenal and a ureteromuscular fistula. Right total nephrectomy, abscess evacuation, and fistula closure were performed. Escherichia coli and Streptococcus viridans were isolated on pus culture.
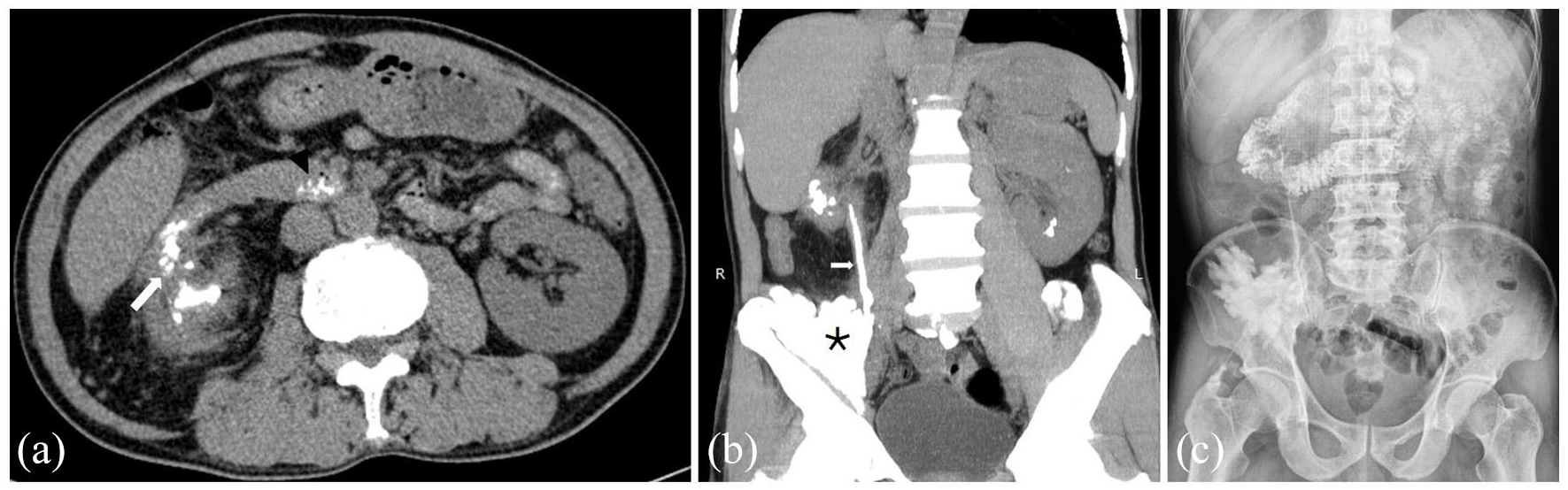
Computed tomographic fistulography. (a) Direct communication between the right renal pelvis and the D2 duodenum is clearly visualized (long arrow). Note the presence of contrast media in the distal D3 duodenum (arrowhead). (b) Coronal reconstruction with maximum intensity project image delineates a connection (short arrow) between the intramuscular abscess (asterisk), the right ureter, and the right renal pelvis. (c) A follow-up abdominal radiograph confirms a complex pyeloduodenal and ureteromuscular fistula. The distal ureter and the bladder were not visible.
Pathological examination of the resected kidney confirmed diffuse XGP. Microscopically, the lesion had three distinct zones centered by a calyx. The inner zone consisted of leukocytes, lymphocytes, plasma cells, histiocytes or macrophages, and necrosis. The middle zone consisted of granulation tissues surrounded by hemorrhage. The classic feature was the presence of lipid-laden foamy macrophages (xanthoma cells) that gave a yellow color to the tissue. The outer zone consisted of giant cells, cholesterol clefts, and fibrous tissues. In addition, various degrees of renal tubular atrophy and tubular dilatation were also observed (Figure 5). At immunohistochemistry, the lesion was positive for CD68 and vimentin, and negative for smooth muscle actin (SMA), desmin, and epithelial markers (Figure 6). Michaelis-Gutmann bodies were not evident; therefore, malakoplakia was excluded.

Pathological examination of the resected kidney. (a) The inner zone of lesion: The inflammatory infiltrates is composed of neutrophils, lymphocytes, and necrosis. (b) The outer zone of lesion: Proliferation of fibrous tissues. (c) Renal tubular atrophy, tubular dilatation at various degrees. (d) and (e) Granulation tissue with the presence of foamy macrophages and calcification (arrowhead). (f) Highest magnification shows foamy macrophages (xanthoma cells) with clear cytoplasm, eccentric nuclei at the cell periphery (arrow).

Immunohistochemistry examination showed positive for CD68 (a and b) and vimentin (c) while negative for SMA (d and e) and AE1/3 (f).
The patient was discharged uneventfully after 10 days and was sent home on oral antibiotics. He has been doing well without recurrent fistula or abscess during 15-month follow-up.
Discussion
XGP is an atypical form of severe chronic renal parenchymal infection, accounting for approximately 1% surgically proven cases of chronic pyelonephritis. 2 The precise pathophysiology of XGP is not yet elucidated. 6 Generally, there are two forms of XGP: diffuse (90%) and focal or tumefactive form (10%). Diffuse XGP may be staged as: stage I, involvement is limited to the kidney; stage II, involvement extends to the renal pelvis or the perirenal fat within Gerota’s fascia; and stage III, involvement extends beyond Gerota’s fascia into the retroperitoneum and/or other organs. 5
CT is valuable in evaluating XGP owing to specific findings in most of the cases and preoperative planning. 7 A typical CT feature of XGP is a combination of a non-functioning enlarged kidney, a central staghorn calculus within a contracted renal pelvis, caliceal expansion, and perinephric fat stranding.3,4,7,8 The “bear’s paw sign” is a confident indicator of XGP, characterized by multiple areas of low attenuation within the renal parenchyma representing dilated, debris-filled calyces and xanthoma collections. The caliceal walls are enhanced due to inflammatory hypervascularity.7,8 CT fistulography with reconstruction images is helpful in visualizing the fistulous tracts, especially in case of complex fistulas. Before the widespread use of CT, the preoperative diagnosis of XGP is challenging due to its non-specific clinical and laboratory presentations. Malek and Elder 5 documented only 1/26 patients had a correct preoperative diagnosis of XGP. More recently, the diagnostic accuracy has been improved to 19%–22.0%.6,9 Our patient had atypical CT findings of XGP due to the absence of staghorn calculus and the bear’s paw sign. Even though the pathological hallmark of XGP is the presence of lipid-laden foamy macrophages, fat deposits representing discrete xanthomas is not frequently appreciated on CT. One study reported the prevalence of fat deposits was 27%. 8 This finding is not diagnostic of XGP; however, the presence of parenchymal fat deposits and a complex fistula in the setting of a typical chronic pyelonephritis strongly suggest XGP. 8 The differential diagnosis of XGP includes clear cell renal cell carcinoma (RCC), sarcomatoid RCC, renal abscess, tuberculosis, and angiomyolipoma. 6
Fistulas occur in up to 8% of XGP and pyeloduodenal fistula comprises about 1% of all renoalimentary fistula.2,10 The anatomic proximity of the right kidney with the second portion of the duodenum facilitates fistula formation. Contrastingly, ureteromuscular fistula is rarely documented in the English literature. The underlying pathogenesis is probably due to chronic obstruction of the ureter with concomitant infection and thus induces significant damage to the ureteral wall and resultant urinoma formation within the psoas muscle. Subsequent secondary infection of the urinoma results in psoas abscess and fistula. 11 Nephrectomy is the recommended treatment for XGP with the healing rate of 80% 6 despite encouraging outcome of conservative management has been sporadically reported. 1 In the present case, total nephrectomy was the treatment of choice owing to a non-functioning kidney along with a complex fistula.
The present case has typical histopathological findings of XGP including (1) various degrees of renal tubular atrophy and tubular dilatation; (2) inflammatory infiltration of neutrophils, lymphocytes, and necrosis; (3) proliferation of fibrous tissues; and (4) the presence of granulation tissue with foamy macrophages and calcification. As for immunohistochemistry, the lesion is positive for CD68 and vimentin while negative for SMA and AE1/3. Other malignancies, especially RCC, can be argued against by lesion morphology and immunohistochemistry (CD68, AE1/3 expression). Malakoplakia is also excluded due to the absence of Michaelis-Gutmann bodies.
Conclusion
XGP associated with complex fistula is a rare clinical entity. When CT findings are not typical, fat deposition and fistula are valuable to the preoperative diagnosis of XGP.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hue University under the Core Research Program, Grant No. NCM.DHH.2020.09.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
