Abstract
Introduction:
Urinary tract infection remains one of the major public health problems among post-fistula patients worldwide, particularly in sub-Saharan Africa. Besides, antimicrobial resistance impedes the effective infection prevention and treatment of ever-increasing bacteria. There was a paucity of data on urinary tract infections and the antimicrobial susceptibility profile of bacteria among post-fistula patients.
Objective:
To determine the prevalence, antimicrobial susceptibility pattern, and associated factors of urinary tract infections among women with post-fistula attending public health facilities, Harar, eastern Ethiopia.
Methods:
An institutional-based cross-sectional study was conducted among 146 consecutively enrolled women with post-fistula from February 2017 to December 2018. Risk factors were collected using a structured questionnaire. The midstream urine was collected, cultured and bacteria species were identified by using standard culture methods. Antimicrobial susceptibility testing was done by the disk diffusion technique. The association between independent and outcome variables was computed by using logistic regression analysis. A
Results:
The prevalence of urinary tract infections was 28.8% (95% confidence interval: 23.4, 33.6).
Conclusion:
Gram-negative organisms were the commonest cause of urinary tract infections in women with post-fistula.
Introduction
A fistula is an abnormal opening that connects the vagina to another organ, such as the bladder, rectum, or colon. It is a severe medical condition in which a fistula (hole) develops between the bladder and the vagina (vesicovaginal fistula), the rectum and the vagina (rectovaginal fistula), the urethra and the vagina (urethrovaginal fistula), the colon and the vagina (colovaginal fistula), the small intestine and the vagina (enterovaginal), and the duct and the vagina (ureterovaginal). Fistulas are usually caused by poorly performed abortion, sexual abuse, surgery, circumcision, prolonged or delayed obstructed labor, an infection, and radiation treatment.1,2
Women with post-fistula can have frequent urinary tract infections (UTIs) as a result of incontinence of urine and/or feces.
3
It is a condition in which bacteria are established and multiply within the urinary tract (the urethra, bladder, ureters, kidney, and other associated appendages).
4
An infection occurs when bacteria from hematogenous or lymphatic routes or the digestive tract or vagina cling to the urinary tract and begin to multiply in the urinary tract.5,6 UTIs are pronounced as inflammation of the vagina, burning, itching, strange-smelling discharge, chills, pain in the lower abdomen, and sometimes painful urination.3,7,8 Prolonged hospitalization and more intensive care like catheterization expose fistula patients to more severe UTIs.1,2 Untreated UTI can result in serious complications such as kidney damage, renal scarring, and renal failure.2,3
UTI is treated generally by broad-spectrum antimicrobials, and treatment is started empirically without bacterial culture isolations and antimicrobial susceptibility tests. Such inappropriate prescription of antimicrobial enhances the development of antimicrobial resistance in bacterial strains.9–11 In Ethiopia, purchasing and using drugs without a prescription is a common practice, thereby contributing to the emergence and spread of antimicrobial resistance.11,12 Moreover, poor hygiene and limited infection control practices play a major role in an increased prevalence of resistant pathogenic bacteria. 13 The resistance properties easily transfer between bacteria of different genera through plasmids and other mobile genetic elements. 14
There is a paucity of data on UTIs and the antimicrobial susceptibility profile of bacteria from post-fistula patients worldwide, particularly in developing countries. Previous studies addressed these problems in pregnant women, diabetes patients, and other risk groups. 15 – 17 UTI in fistula patients is an area that is frequently hidden under the broad rubric of maternal infection and can be described as a neglected infection, 18 even in recent initiatives launched in Ethiopia to improve maternal and child health. 19 A day-to-day observation has also shown fistula patients who presented with atypical UTIs are considered as having UTIs and treated empirically. This study aimed to determine the prevalence, antimicrobial susceptibility pattern of the bacteria, and associated factors among women with a post-fistula at public health facilities, Harari Regional State, eastern Ethiopia.
Methods
Study setting and period
An institutional-based prospective cross-sectional study was conducted in public health facilities in the Harari Regional State, Ethiopia from February 2017 to December 2018. Harar, the capital city of Harari Regional State, is located on a hilltop in the eastern extension of the Ethiopian highlands about 526 km away from Addis Ababa. In the city, there are six hospitals, eight health centers, and one fistula center. Jugal Hospital, Hiwot Fana Specialized University Hospital, and the Hamlin Fistula Center serve as referral and teaching health facilities. They are well organized and provide health-care services for more than 6,305,367 (females: 3,572,998) population of the Harari Regional State, East Hararghe zone, Dire Dawa Administration, and Somali Regional State. 20
Source population
All women with post-fistula attended public health facilities in the Harari Regional State, Ethiopia.
Study population
All women with post-repair fistula attended the selected health facilities during the study period.
Inclusion and exclusion criteria
All women with post-fistula were included in the study. Those women who were treated with antimicrobials 15 days before and during the study period and those who had revisited the respective health facility for their regular follow-up (retrieved from the medical record) during the study period were excluded from the study.
Sample size determination and sampling techniques
The sample size was determined using the single population proportion formula by taking a prevalence of UTIs (89%) from the study conducted in the Abbasi Shaheed Hospital, Karachi, Pakistan, 2 and calculated by considering the standard score of 1.96 (95% confidence level), and margin of error (5%). The total sample size with a nonresponse rate (10%) was 165. A convenient sampling technique was applied to include study participants who fulfilled the inclusion criteria until the required sample was attained.
Data collection methods
A structured questionnaire was used to collect data regarding sociodemographic characteristics, duration of fistula, clinical manifestation (frequency, urgency, nocturia, dysuria, suprapubic or loin pain with or without fever), and other related factors of UTIs. The data were collected by midwives via face-to-face interviews after obtaining informed written consent in a separate room. The physical examination was done by clinicians.
Sample collection, handling, and processing
The urine sample was collected in a well-labeled, leak-proof, reusable, and sterilizable wide-mouthed screw-capped universal container. The participants were instructed on how to collect about 5 ml of midstream urine after removing their catheters. The fresh sample was also collected aseptically from the participant’s catheter in case the participant was unable to provide midstream urine. The specimens were immediately delivered to the Microbiology Laboratory of the Department of Medical Laboratory Sciences of Haramaya University for culture identification, biochemical and antimicrobial susceptibility testing.
Culture and identification
Well-mixed urine was inoculated on Cystine-Lactose-Electrolyte-Deficient and blood agar (Oxoid, LTD, UK) using a wire loop having a capacity of containing 0.001 ml of urine. After incubation at 37°C for 24 h, the phenotypic characterization of bacteria was done using colony characteristics. The colonies were counted (a single colony represents one organism) and multiplied by 1000 to determine the number of bacteria per milliliter of the original urine sample. A count of 105 colony-forming units per milliliter of urine was considered significant bacteriuria, which indicates the presence of UTI. 4
Differentiation of the isolates to the species level was performed using the analytical profile index (bioMérieux® SA, Marcy-l’Étoile, France) biochemical reactions strips. In brief, a 0.5 McFarland bacterial suspension was prepared and dispensed to rehydrate each of the wells and the strips were incubated according to the manufacturer’s recommendation. All reactive and nonreactive reactions were compiled to obtain a profile number, which was then compared with profile numbers in the APIWEB™ database to determine the bacterial species.
Antimicrobial susceptibility testing
Antimicrobial susceptibility testing was done using the modified Kirby–Bauer disk diffusion method on Mueller–Hinton agar (Oxoid Ltd. Bashingstore Hampshire, UK) prepared with 4 mm thickness. First, three to five pure colonies were transferred to the tube containing sterile normal saline (0.85% NaCl) until the turbidity of growth became equivalent to 0.5 McFarland standard. A sterile cotton swab was dipped into the suspension and distributed evenly over the entire surface of the Mueller–Hinton agar. A panel of 12 antimicrobial disks (Oxoid Ltd. Bashingstore Hampshire, UK) was used for susceptibility tests. Disks for Gram-negative bacteria contain the following antimicrobials: amoxicillin (10 µg), ceftazidime (30 µg), cefoxitin (30 µg), ceftriaxone (30 µg), ciprofloxacin (5 µg), gentamicin (10 µg), and trimethoprim–sulfamethoxazole (1.25/23.75 µg). Disks for Gram-positive bacteria include penicillin G (1 unit), amoxicillin (10 µg), chloramphenicol (30 µg), ceftazidime, cefoxitin (30 µg), ceftriaxone (30 µg), ciprofloxacin (5 µg), erythromycin (15 µg), and gentamicin (10 µg). The disks were placed at least 24 mm away from each other and 15 mm from the edge to avoid the overlapping of the zone of inhibition and pressed down to ensure complete contact with the agar surface. The plates were inverted upside down and incubated aerobically at 37°C for 18–24 h. The diameter of the zone of inhibition around each disk was measured to the nearest whole number by using a digital caliper. The bacterial isolates were classified as sensitive (S), intermediate (I), or resistant (R) by comparing against the inhibition zone diameter of interpretative standards as indicated in the Clinical and Laboratory Standards Institute guideline. 21
Data quality control
A structured questionnaire first prepared in the English language was translated into Amharic and Afan Oromo by the bilingual expert, and back-translated by another bilingual expert to check the consistency. The questionnaire was pretested in 5% of the study population at Dilchora hospital and Sabian health center, Dire Dawa Administration, Ethiopia before the actual data collection. During pretesting, additional information was gathered and vague terms and phrases were modified. Data collectors were trained in data collection techniques, urine sample collection, and culture techniques. Participants were oriented on how to collect self-midstream urine samples. The specimens were transported in a cool box (2°C–8°C) and immediately processed upon reaching the laboratory. All culture media were prepared following the manufacturer’s instructions. Plates were checked for sterility and performance using
Statistical analysis
The data were coded and double entered into the Epi Info™ statistical software (version 3.5.1, Centers for Disease Control, Atlanta, GA, USA), checked for consistency and missing values, and were exported to the Statistical Package for Social Sciences version 21.0 (SPSS Inc., Chicago, IL, USA) for analysis. Descriptive statistical tools (frequency, mean, standard deviation) were used to present the data in the table and text. Data were analyzed using logistic regression models. Independent factors with a
Ethical approval
The study was ethically approved by the Institutional Health Research Ethics Review Committee (IHRERC 156/2017) of the College of Health and Medical Sciences, Haramaya University. Pertinent information regarding the purpose of the study, the procedure, and its benefits for future care was explained. The right to accept or reject their participation in the study was discussed. Privacy and confidentiality issues were strictly assured. The participation was completely voluntary and data were collected after informed written consent was made. A thumbprint was taken from individuals who were not able to read and sign. In addition, a written informed assent was also obtained from their legally authorized representatives. No reference was made in oral or written reports that could link participants in the study.
Results
Sociodemographic characteristics
A total of 146 women with post-fistula were included in this study. Nineteen participants were excluded from the study due to antimicrobial treatment before and during data collection, and/or revisited the health facilities for the same genitourinary problem; it gives rise to a response rate of 88.5%. The majority of the study participants (39.0%) were in the age group of 21–30 years with the mean age (±SD) of 29.7 ± 8.9 years. A high proportion (58.9%) of the study participants was married. Most of the study participants (35.6%) had a primary level (grade 1–8) education and were rural residents (71.2%). Almost one-third of them (23.3%) earned a low monthly income. The majority of participants had rectovaginal fistula (27.4%) (Table 1).
Sociodemographic characteristics of fistula patients attending public health facilities, Harari Regional State, eastern Ethiopia from February 2017 to December 2018 (

Daily laborer and employee.
Colovaginal and enterovaginal.
Prevalence and bacterial etiologies
The prevalence of UTIs was 28.8% (95% CI: 23.4, 33.6). Ten different bacteria were identified. The majority of the isolates (69.0%) were Gram-negative bacteria. Of these,
Frequency and types of bacteria species isolated (
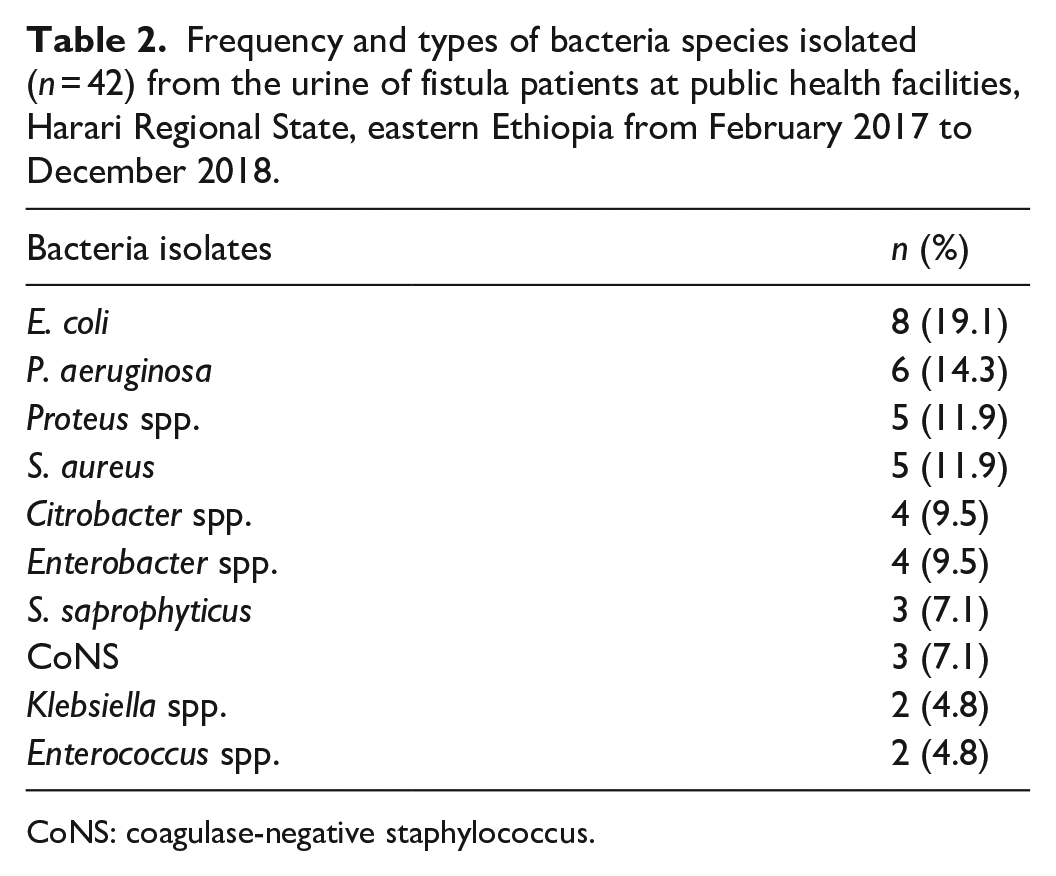
CoNS: coagulase-negative staphylococcus.
Antimicrobial susceptibility testing
Of isolates, Gram-negative bacteria showed resistance to trimethoprim–sulfamethoxazole (65.6%), amoxicillin (55.2%), and ciprofloxacin (55.2%). The majority were sensitive to ceftriaxone (86.2%), cefoxitin (79.3%), and ceftazidime (74.4%). Among the isolates,
Antimicrobial susceptibility profile of Gram-negative bacteria isolated from the urine of fistula patients at public health facilities, Harari Regional State, eastern Ethiopia from February 2017 to December 2018.

S: sensitive; I: intermediate; R: resistance; AML: amoxicillin; CAZ: ceftazidime; FOX: cefoxitin; CIP: ciprofloxacin; CN: gentamicin; CRO: ceftriaxone; STX: trimethoprim–sulfamethoxazole.
Gram-positive bacteria isolated were resistant to amoxicillin (75%), gentamicin (61.5%), and chloramphenicol (53.8%). The higher resistance was observed among
Antimicrobial susceptibility profile of Gram-positive bacteria isolated from the urine of fistula patients at public health facilities, Harari Regional State, eastern Ethiopia from February 2017 to December 2018.
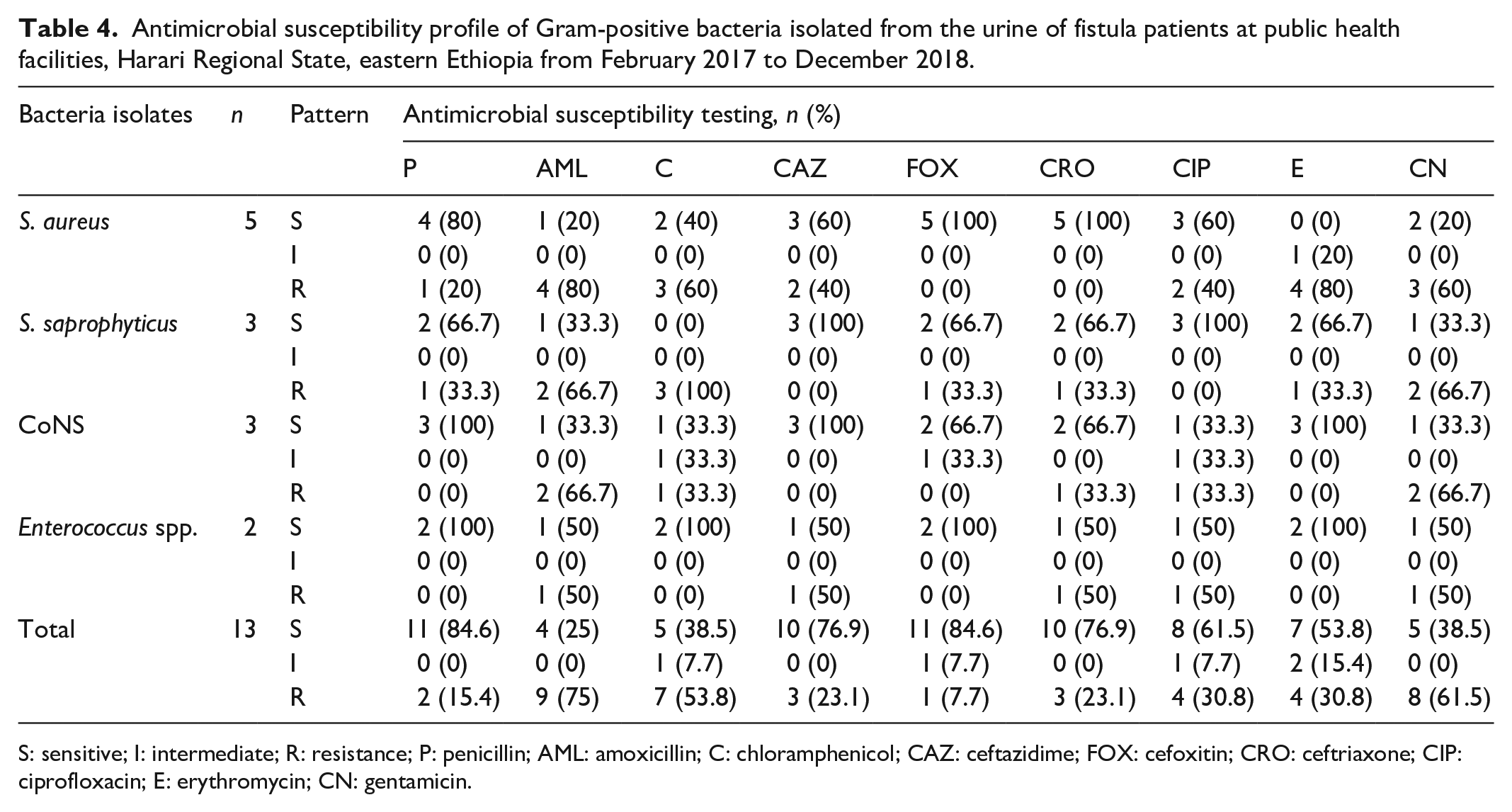
S: sensitive; I: intermediate; R: resistance; P: penicillin; AML: amoxicillin; C: chloramphenicol; CAZ: ceftazidime; FOX: cefoxitin; CRO: ceftriaxone; CIP: ciprofloxacin; E: erythromycin; CN: gentamicin.
Factors associated with UTIs
The proportion of UTI was higher among fistula patients whose age groups were greater than 30 years (30.8%), able to read and write (30.2%), have current UTI symptoms (39.3%), and rectovaginal fistula (40.0%). In bivariate logistic regression analysis, monthly income, age at first pregnancy, parity, duration of fistula, history of previous UTI, the current symptom of UTI, history of the previous catheterization, and genital wash were associated with UTIs. In multivariate analysis, giving birth to a single baby (nullipara) (AOR: 0.3, 95% CI: 0.1, 0.8), having fistula for less than 3 years (AOR: 0.2, 95% CI: 0.1, 0.6), and no previous history of catheterization (AOR: 0.3, 95% CI: 0.1, 0.9) found to decrease the odds of having UTIs. The remaining variables did not show a significant association as shown in Table 5.
Factors associated with UTI among fistula patients at public health facilities, Harari Regional State, eastern Ethiopia from February 2017 to December 2018.
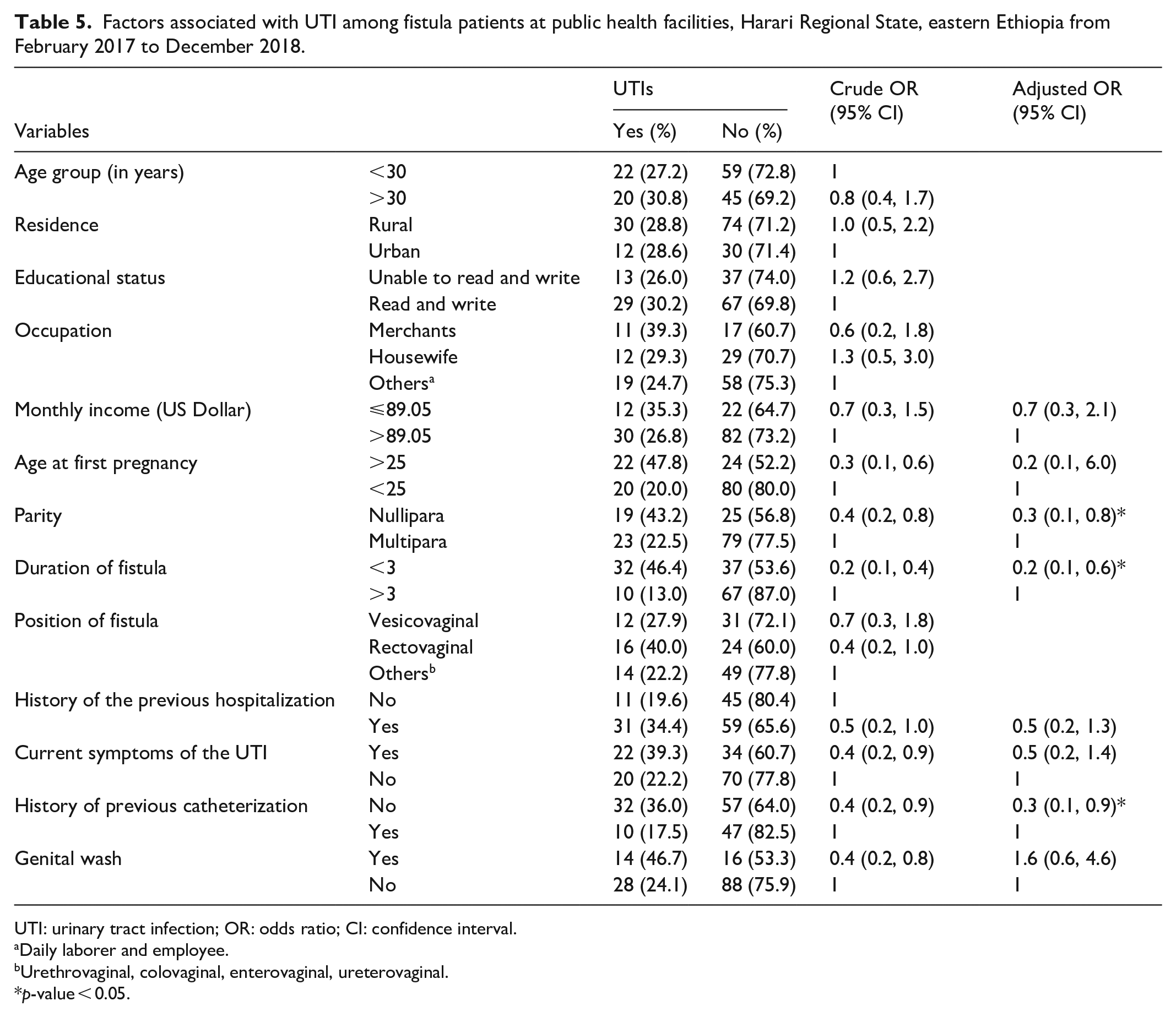
UTI: urinary tract infection; OR: odds ratio; CI: confidence interval.
Daily laborer and employee.
Urethrovaginal, colovaginal, enterovaginal, ureterovaginal.
Discussion
There is a lack of concrete evidence that shows the magnitude of UTIs and antimicrobial resistance in post-fistula patients worldwide. Thus, it is difficult to compare and contrast the current findings with previous reports. In the present study, the overall prevalence of UTI was 28.8% (95% CI: 23.4, 33.6). This was lower compared to a study report from Karachi, Pakistan (89%), 2 Abakaliki, Southeastern Nigeria (76.1%), 13 Addis Ababa, Ethiopia (58.1%), 11 and Gondar, Ethiopia (52.8%),3,12 but it was lower than finding reported from Kuno, Nigeria (9.2%). 22 This variation might be due to the difference in sample size, the method applied, geographical location, history of catheterization, and immune response of the study participants. The lower prevalence should not be neglected in the management of UTIs due to a higher chance to develop chronic and recurrent infections,23,24 particularly in post-fistula patients.2,12
In the present study, the rate of UTI was high among participants who had the current symptom of UTI than asymptomatic with no significant association. This finding was supported by the study done in two other places in Ethiopia where no statistically significant was reported.11,16 The finding of this study was also revealed being live with fistula for less than 3 years reduces the risk of UTI. A similar finding was reported from Addis Ababa, Ethiopia. 23 The higher rate of UTI patients who have had more than 30 years of age, rectovaginal position fistula, and regular wash of genital were recorded with no statistical significance. The possible reasons for this are not clear and may have occurred purely by chance since no statistical significance was observed in the trend. A longitudinal study design may be required to describe and provide a better understanding and interpretation of these factors.
The long-term maintenance of invasive devices, such as urethral catheters, may result in the removal of the natural protective barrier against infections, thereby, contributing to the development of UTIs (cystitis or pyelonephritis), which may progress to kidney failure.22,24–27 In this study, the rate of having a UTI was higher among fistula patients with a previous history of catheterization and/or indwelling with a significant association. The finding was almost similar to other reports that reported as catheterization is the most important risk factor for the development of UTIs.11,23,28 Therefore, the use of a urinary catheter should be considered only in exceptional cases, where other approaches have not been effective.5,27
In this study, the predominant isolates were Gram-negative bacteria (69.0%). This was consistent with other reports.11,12,17 The higher occurrence of
Up to 30% of women with UTIs develop a recurring infection in their lives.
23
These women frequently rely on long-term low-dose antibiotics.
27
While, patients with cystitis do not have recurrence or complications and hence do not require intensive treatment and management.
25
Such inconsistent misuse of a drug may lead to treatment failure.11,12 In the present study,
The strength of this study can be attributed to many factors, including the close cooperation of health institutions, health professionals in identifying women with fistula and fistula patients. The use of standardized culture isolation and antimicrobial testing techniques are also the strength of this study. However, lack of isolating of anaerobic and other uropathogenic organisms, recall bias that cannot be ruled out in a cross-sectional study and a wish to satisfy the interviewer by the participants can be taken as the limitation of the study. The generation of information from a study conducted in 2018 can also be taken as a limitation. The clinical presentation, and risk factors such as poor immune system, and concurrent fecal incontinence need further detailed study with a large sample size.
Conclusion
In conclusion, women with post-fistula are at high risk of UTIs.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221079309 – Supplemental material for Urinary tract infection, antimicrobial susceptibility pattern of isolates, and associated factors among women with a post-fistula at public health facilities, Harar, eastern Ethiopia: A cross-sectional study
Supplemental material, sj-docx-1-smo-10.1177_20503121221079309 for Urinary tract infection, antimicrobial susceptibility pattern of isolates, and associated factors among women with a post-fistula at public health facilities, Harar, eastern Ethiopia: A cross-sectional study by Dadi Marami, Degu Abate and Shiferaw Letta in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank Haramaya University for funding this study. The funder has no role in the proposal write-up, data collection, data analysis, manuscript preparation, and publication. Our sincere thanks also go to the study participants for their meticulous cooperation.
Author contributions
D.M. conceived the research title. D.M., D.A., and S.L. participated in the design, laboratory work, data analysis, interpretation, and paper write-up. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Haramaya University (HURG-2014-02-02).
Ethical approval
Ethical approval for this study was obtained from the Institutional Health Research Ethics Review Committee of the College of Health and Medical Sciences, Haramaya University (approval number: IHRERC 156/2017).
Informed consent
Informed, voluntary, written, and signed consent was obtained from all study participants before the commencement of the study. A thumbprint was taken from individuals who were not able to read and sign. In addition, written informed consent was also obtained from their legally authorized representatives. The confidentiality of the information was strictly maintained.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
