Abstract
Alpha-fetoprotein hardly increased due to nasopharyngeal cancer. In this article, we reported a 57-year-old male nasopharyngeal carcinoma patient who had posttreatment subscapular metastasis with high serum alpha-fetoprotein but negative plasma Epstein–Barr virus DNA. Pathology results indicated that the scapular mass was undifferentiated non-keratinizing carcinoma originated in the nasopharynx. Moreover, no liver lesion was detected by imaging examination. In view of the positive alpha-fetoprotein and alpha-fetoprotein messenger RNA staining result in the right scapular mass fine needle aspiration biopsy sample, we considered the diagnosis of alpha-fetoprotein-producing nasopharyngeal carcinoma that had never been reported before.
Background
Nasopharyngeal carcinoma (NPC) is an epidemic in Southern China and the surrounding regions. 1 Plasma Epstein–Barr virus (EBV) DNA is widely used as a biomarker for NPC. 2 Alpha-fetoprotein (AFP) is a protein produced in the liver of a developing fetus and used as a tumor marker to help detect and diagnose cancers of the liver, testicles, and ovaries. 3 Previous study considered AFP nearly useless in NPC patients since NPC did not produce AFP. 4 However, we found a pathology-confirmed AFP-producing NPC patient with high serum AFP. Serum level of AFP decreased with the shrinking of tumor size and increased along with tumor progression. In this case, dynamic changes in AFP levels could predict treatment response instead of EBV infection status. We recorded this rare phenomenon and present our experience in diagnosing AFP-producing NPC.
Case presentation
In June 2019, a 57-year-old Cantonese male patient presented with a mass in the right subscapular area. His past medical history included a diagnosis of undifferentiated non-keratinizing NPC (World Health Organization type III, restaged as T3N1M0 III according to the American Joint Committee on Cancer staging manual, 8th edition) 18 months prior to the present episode of care. At that time, the pathological diagnosis was based on histopathological typing without immunohistochemical analysis. Moreover, the patient tested negative for the presence of plasma EBV DNA. After being diagnosed with NPC, he received definitive chemoradiotherapy in our hospital. Intensity-modulated radiation therapy was delivered to the primary tumor and involved lymph nodes planning target volume in 33 fractions for a total dose of 70 Gy; high- and low-risk planning target volumes received 60 and 54 Gy, respectively, in the same number of fractions. Concurrent chemotherapy regimen comprised 100 mg/m2 of cisplatin for three cycles. The patient experienced no serious adverse events during treatment and achieved complete response. After 1 year of follow-up, he remained without signs of disease progression. Three months prior to the present episode of care, the patient felt tightness in the right scapula and discovered an enlarged mass.
On the day of admission, the patient had a major complaint of paroxysmal scapula pain without other symptoms. He experienced rapid weight loss (3 kg in the month prior to admission). The patient never drank alcohol or smoked and indicated no family history of cancer, autoimmune disorders, and infectious diseases. On physical examination, his temperature was 36.6°C, heart rate was 79 beats per minute, blood pressure was 109/76 mmHg, weight was 50.3 kg, and height was 166 cm. We found no visible mass in the nasopharynx or enlarged cervical lymph nodes. The scapular mass measured 4 cm × 8 cm. His neurological examination was normal. Laboratory findings on admission are shown in Table 1. Notably, serum AFP level was 20,886 ng/mL (reference range: 0–25 ng/mL), but liver transaminases were normal. Plasma EBV DNA remained negative.
Laboratory examination findings of a 57-year-old male patient with alpha-fetoprotein–producing nasopharyngeal carcinoma.

EBV: Epstein–Barr virus; DNA: deoxyribonucleic acid; ND: not done; HBV: hepatitis B virus; HBsAg: hepatitis B surface antigen; HBsAb: hepatitis B surface antibody; HBeAg: hepatitis Be antigen; HBeAb: hepatitis Be antibody; HBcAb: hepatitis B core antibody; HCV: hepatitis C virus; HIV-Ab: human immunodeficiency virus antibody; TP: treponema pallidum; VCA-IgA: viral capsid antibody; EA-IgA: early antigen antibody; EBNA1-IgA: Epstein–Barr virus nuclear antigen 1; Zta-IgA: Zta protein antibody; Rta-IgA: Rta protein antibody.
Reference ranges used at the Sun Yat-sen University Cancer Center and may not be appropriate for all patients.
Subsequent positron emission tomography–computed tomography (PET/CT) scanning indicated bone destruction in right scapula (Figure 1). This was considered a metastasis with the maximum standardized uptake value of 8.0. We observed soft tissue shadows in the scapular lesion, and the boundary between the inferior scapular muscle and supraspinal muscle was unclear. PET/CT and abdomen ultrasonography imaging found no lesions in the liver or signs of a tumor. Considering these results, we initiated the diagnostic assessment.

Images of the patient’s right shoulder and dynamic serum alpha-fetoprotein levels throughout the episode of care. (a) Positron emission tomography–computed tomography image on admission, (b) T1-weighted magnetic resonance imaging scan with contrast on admission, (c) disease progression after first-line chemotherapy, (d) tumor progression after second-line chemotherapy, and (e) changes in serum Epstein–Barr virus (EBV) DNA status (blue) and serum alpha-fetoprotein levels (red) during treatment.
Differential diagnosis
Viral hepatitis
In China, viral hepatitis infections incur a high burden with an annual incidence of hepatitis B of 81.57 per 100,000 persons, the second highest rate among all infectious diseases. 5 Patients with viral hepatitis or cirrhosis may have increased levels of serum AFP, although this change is brief and small in magnitude. Viral hepatitis infection is diagnosed based on a patient’s epidemiological history, clinical symptoms, and laboratory tests.
In our case, the patient indicated no history of viral hepatitis infection and had no clinical symptoms or elevated transaminase levels. Serum biochemical examination revealed no evidence of hepatic dysfunction or hepatitis B or C. Moreover, the patient tested negative for antibodies of hepatitis A, C, and E. Regarding hepatitis B, the patient also tested negative for the hepatitis B surface antigen, hepatitis Be antigen (HBeAg), and antibody to HBeAg (anti-HBe) (Table 1). Furthermore, hepatitis B virus DNA copy number was 0 IU/mL. Thus, the significant increase in AFP levels was not related to a viral hepatitis infection.
Hepatocellular carcinoma
Hepatocellular carcinoma (HCC) is mostly detected in chronic liver disease cases that have reached the stage of cirrhosis. 6 Chronic infection with hepatitis B or C viruses and excessive alcohol consumption are the most common causes of HCC worldwide.7,8 Abdomen ultrasonography, CT, and magnetic resonance imaging (MRI) are imaging surveillance tools for HCC detection. 9
AFP levels were commonly used as a serum marker for auxiliary diagnosis of HCC; 6 thus, the sharp increase in AFP levels of our patient indicated a possible HCC diagnosis. However, we found no evidence of liver lesions, viral hepatitis infection, and/or cirrhosis. The results of the abdomen ultrasonography and PET/CT imaging did not support the existence of tumors in the liver. Therefore, we should carefully consider the amplitude and dynamic changes in AFP levels and its relationship with transaminase levels, as well as combine clinical manifestations with liver imaging examination findings.
Liver metastasis from NPC
The patient had a history of NPC with complete response after chemoradiotherapy and discharge over 1 year ago. In the follow-up evaluation, PET/CT scan showed bone metastases without liver metastasis. Furthermore, the significant increase in serum AFP level and negative EBV infection status were inconsistent with the clinical manifestation of NPC liver metastases. Dynamic measurements of serum EBV DNA and AFP levels, as well as other imaging examinations, were evaluated repeatedly. Contrast-enhanced ultrasonography and MRI examinations of the liver showed no tumor site (Figure S1).
Histopathological examination
Fine needle aspiration (FNA) biopsy of the scapular mass revealed poorly differentiated cancer cells varying in size and shape. Immunohistochemistry examination indicated that the tumor was positive for pan cytokeratin (AE1/AE3) and negative for LCA, HepParl, and glypican-3 (Figure 2(m)–(t)). The tumor had a focal and patchy staining for AFP.
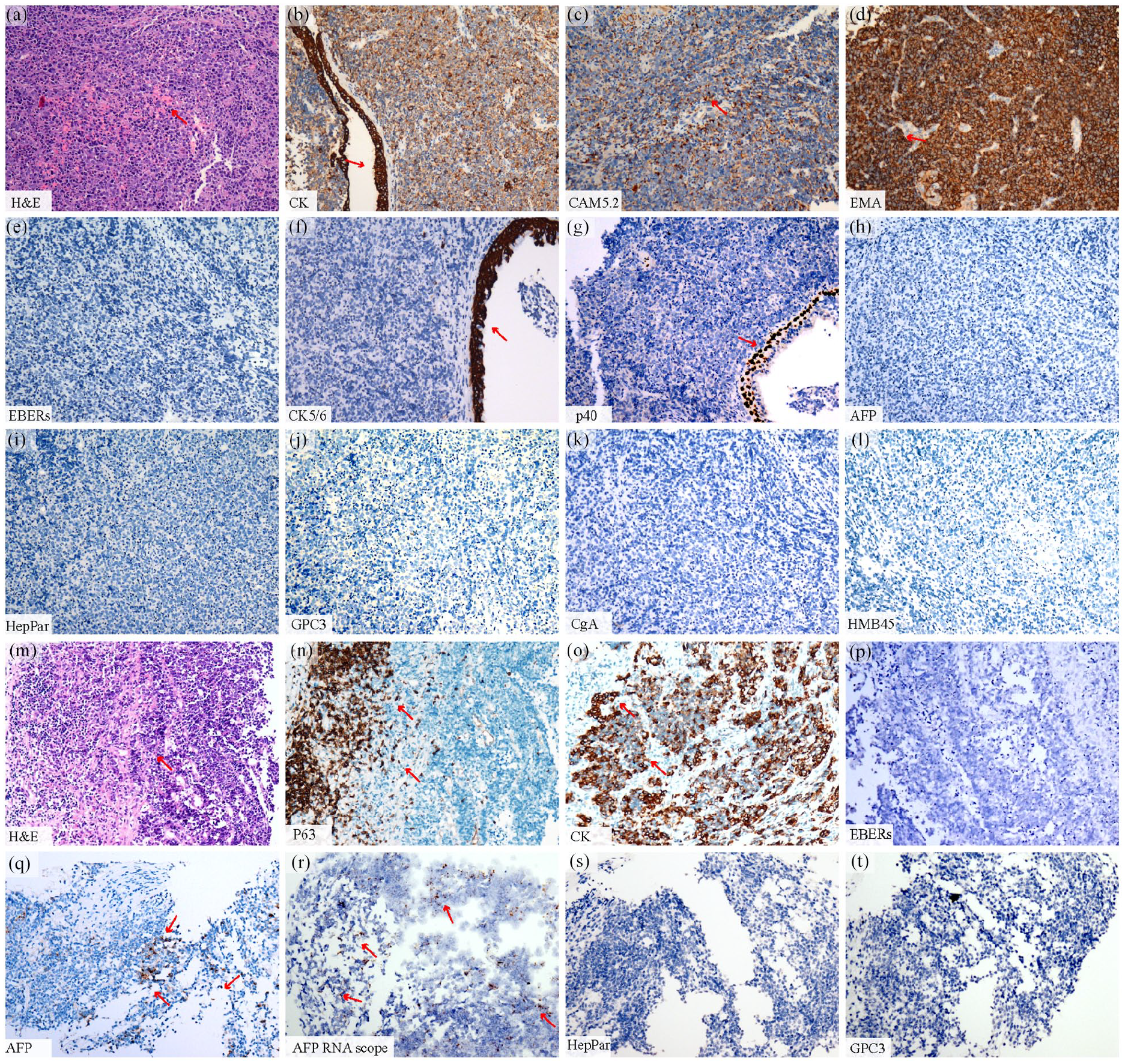
Representative images of hematoxylin and eosin staining and immunohistochemistry assay in (a–l) paraffin-embedded nasopharynx and (m–t) scapular mass tissues. (a) Hematoxylin and eosin staining of the nasopharynx sample biopsy confirmed the diagnosis of undifferentiated non-keratinizing carcinoma. Immunohistochemistry assay showed positive results for (b) CK, (c) CAM5.2, (d) EMA, (f) CK5/6, (g) and P40 in the nasopharynx sample biopsy; and negative results for (h) AFP, (i) HepPar, (j) GPC3, (k) CgA, and (l) HMB 45. (m) For the scapular mass biopsy, hematoxylin and eosin staining showed that the tumor comprised sheets of malignant cells with vesicular chromatin and prominent nucleoli; Tumor-infiltrating lymphocytes are conspicuous, maintaining identical morphology to primary lesions. Immunohistochemical staining showing positive expression of (o) CK and (n) P63. Tumor cells were negative for EBV-encoded small RNA by in situ hybridization in the (p) scapular mass and (e) nasopharynx tissue biopsies. (q) Tumors were positive for patchy alpha-fetoprotein by immunohistochemistry assay, and (r) alpha-fetoprotein mRNA was detected within the neoplasm by RNA in situ hybridization. According to immunohistochemistry assays, the tumor was negative for HepPar (s) and GPC3 (t).
RNA scope method represents a significant advancement that addresses the challenges of traditional RNA in situ hybridization while providing spatial and morphological resolution at a single-cell level. 10 Thus, we used the RNA scope method, which also showed positive for AFP by in situ hybridization. Moreover, the tumor cells were negative for EBV-encoded small RNA (EBER) by in situ hybridization. Positive staining for CK, Ki67, and P63 indicated that the tumor originated in the squamous epithelium, most likely in the nasopharynx considering the patient’s medical history. 11
The negative result for EBER was unusual for NPC metastasis. Hence, we reassessed the nasopharynx biopsy performed before the chemoradiotherapy. The biopsy sample was taken from the left pharyngeal recess and diagnosed as undifferentiated non-keratinizing carcinoma by microscopic examination. We observed microscopically large tumor cells with a syncytial appearance, round to oval vesicular nuclei, and large central nucleoli. Malignant cells assumed spindle-cell features in fascicular arrangements. Immunohistochemistry and in situ hybridization assays were performed to determine whether the scapular mass originated in the pharynx. Table 2 summarized the comparison of the primary and metastatic tumor site. Immunohistochemistry assay results indicated that the tumor was positive for pan cytokeratin (AE1/AE3), CK5/6, p40, CAM5.2, EMA, and INI; and negative for Syn, CgA, CD56, MPO, CD20, CD3, CD 43, HMB 45, Melan-A, and S-100 (Figure 2(a)–(l)). Tumor cells were negative for EBER by in situ hybridization. The positive staining for CK, p40, CAM5.2, and EMA indicated a diagnosis of undifferentiated non-keratinous carcinoma. Other tumors—including lymphoma, melanoma, olfactory neuroblastoma, high-grade neuroendocrine carcinoma, and sinonasal undifferentiated carcinoma—were ruled out.
The pathology features of the primary and scapular lesion.

AFP: Alpha-fetoprotein; RNA: ribonucleic acid; EBER: EBV-encoded small RNA; EBV: Epstein–Barr virus; EMA: epithelial membrane antigen; LCA: Leukocyte common antigen; INI: integrase interactor 1.
NPCs are histologically classified into the following four subtypes based on morphologic characteristics: epithelial carcinoma (EC), sarcomatoid carcinoma (SC), mixed sarcomatoid-epithelial carcinoma (MSEC), and squamous cell carcinoma (SCC).12,13 Our case is EC subtype, most of the tumor regions are identified with round epithelial cells and vesicular nuclei phenotype appearance. Figure 2(a) and (m) represent histological view of the primary and recurrent tumors, which shows similar morphologic traits. EC subtype is more responsive to EGFR inhibitors, which is synergistically enhanced by combining with radiotherapy. Moreover, radiochemotherapy significantly improved survival compared with radiotherapy alone for patients with EC subtype.
Compared with the scapular mass, all the immunohistochemistry results were similar in the nasopharynx biopsy, except that AFP was not detected in the primary nasopharynx biopsy (Table 2). The above results indicated that the scapular mass originated in the nasopharynx. Additional immunohistochemistry analysis for HepParl, glypican-3, and AFP was carried out, obtaining negative results. Nonetheless, high serum AFP levels still represented an unusual presentation in NPC. In view of the positive AFP staining result in the right scapular mass FNA biopsy sample, we considered the diagnosis of AFP-producing NPC.
Gene sequencing
The FNA biopsy sample of the right scapular mass was used for next-generation sequencing testing. Microsatellite-stability status was determined, and the tumor mutational burden was low (1 Muts/Mb) with no detectable genomic alterations. Variants of unknown significance (VUS) of
The human
Discussion
Adults with elevated serum AFP levels require differential diagnosis for malignant diseases, such as liver cancer, germ cell tumors (e.g. yolk sac tumor), and metastatic lung cancer—and benign diseases, such as acute or chronic hepatitis, liver cirrhosis, and congenital biliary atresia.
3
Considering the results of pathological examinations, our patient received a final diagnosis of AFP-producing NPC with multiple metastases. AFP is mainly synthesized by fetal liver cells and yolk sac, the prognostic role of which in NPC remains unclear. AFP-producing malignancies of the ovaries or gastrointestinal tract were with a poor prognosis.22,23 In this case,
A multicenter, phase III, randomized clinical trial provided evidence that gemcitabine plus cisplatin is superior to 5-fluorouracil plus cisplatin in metastatic NPC treatment. 26 Because the patient’s plasma creatinine level was less than 60 mL/(min×1.73 m 2 ), we replaced cisplatin for capecitabine in his treatment regimen. He received two cycles of chemotherapy with gemcitabine 1000 mg/m2 on days 1 and 8 every 3 weeks, and capecitabine 1500 mg/m2 on days 1–14 every 3 weeks. During chemotherapy, he had clinical symptoms and reported dental ulcer, odynophagia, and weight loss. After chemotherapy regimen completion, the scapular mass volume slightly decreased but the patient’s scapular pain persisted and he developed a new axillary lymph node metastasis (Figure 1(c)). Therefore, we confirmed disease progression after first-line chemotherapy. Moreover, serum AFP levels remained elevated and serum EBV DNA was still negative.
Few studies have evaluated the efficacy of chemotherapy as a second-line treatment for metachronous metastasis of NPC patients. We used albumin-bound paclitaxel 100 mg/m2 every week and apatinib 500 mg once a day for two cycles (1 cycle = 3 weeks). The patient reported relief of symptoms after second-line treatment and the tumor mass and volume reduced significantly. Post-treatment MRI scan did not detect liver lesions and serum AFP level decreased from 20,886 ng/mL to approximately 5000 ng/mL. High serum AFP levels may be explained by the presence of AFP-producing NPC, as indicated by pathological examination. The patient underwent second-line treatment regimen maintenance and remained symptom-free with a stable disease and improved quality of life for 6 months. In March 2020, the right scapular mass increased again, accompanied by an increase in plasma AFP levels (Figure 1(e)). The patient refused further chemotherapy and died surrounded by his family 4 months later.
Final diagnosis
AFP-producing EBV-negative metastatic NPC, which had never been reported before. In addition, this type of NPC is with poor prognosis.
Conclusion
AFP-producing EBV-negative metastatic NPC had poor treatment response to the regular chemotherapy of NPC. In this case,
Supplemental Material
sj-pdf-1-sco-10.1177_2050313X211057704 – Supplemental material for Alpha-fetoprotein–producing recurrent nasopharyngeal carcinoma: A case report
Supplemental material, sj-pdf-1-sco-10.1177_2050313X211057704 for Alpha-fetoprotein–producing recurrent nasopharyngeal carcinoma: A case report by Zhen-Chong Yang, Mei-Juan Luo, Li-Li Liu, Mu-Yan Cai, Yu-Jing Liang, Qiu-Yan Chen, Lin-Quan Tang and Hai-Qiang Mai in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
We would like to thank the NPC patient and his family for allowing us to publish this unique case.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study had institution ethical approval for reporting this individual case.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
