Abstract
Tuberculosis remains a major cause of morbidity and mortality globally, with a global total of about 10 million people falling ill with it in 2020. Until the coronavirus (COVID-19) pandemic, tuberculosis was the leading cause of death from a single infectious agent. Nasopharyngeal tuberculosis is a rare type of extrapulmonary tuberculosis that may be either primary or secondary to pulmonary tuberculosis. Nasopharyngeal tuberculosis may be mistaken for nasopharyngeal carcinoma as both conditions may present with a nasopharyngeal mass and cervical lymph node enlargement. We present a case of nasopharyngeal tuberculosis secondary to pulmonary tuberculosis who presented without any nasal or respiratory symptoms. The patient presented with a cervical lymph node enlargement and a nasopharyngeal mass was detected on nasal endoscopy. Fine needle cytology from the cervical node and the initial biopsy report from the nasopharyngeal mass were not confirmatory, causing a diagnostic dilemma. A repeat biopsy from the nasopharyngeal mass with the demonstration of caseating granuloma and acid-fast bacilli in the specimen pointed to the diagnosis of nasopharyngeal tuberculosis. The patient responded well to the standard 6-month anti-tubercular regimen. Nasopharyngeal tuberculosis may mimic nasopharyngeal carcinoma and a repeat biopsy may be necessary to confirm the diagnosis.
Keywords
Introduction
Tuberculosis remains a major cause of morbidity and mortality globally. A global total of about 10 million people fell ill with tuberculosis in 2020. 1 Until the coronavirus (COVID-19) pandemic, tuberculosis was the leading cause of death from a single infectious agent, ranking above HIV/AIDS. 1 Tuberculosis is a multi-systemic disease affecting almost any part of the body. Nasopharyngeal tuberculosis is a rare type of extrapulmonary tuberculosis, comprising less than 1% of tuberculosis cases. 2 Nasopharyngeal tuberculosis may be either primary or secondary to pulmonary tuberculosis. Although there are few reported cases of primary nasopharyngeal tuberculosis,2–6 nasopharyngeal tuberculosis usually occurs secondary to pulmonary tuberculosis as a result of upper respiratory tract contact with the lung secretions. 6 Primary nasopharyngeal tuberculosis is described as an isolated infection of the nasopharynx in the absence of pulmonary or systemic disease. 2 Recent multiple case reports indicate either an increased awareness or incidence of this disease. 7
Initial symptoms of nasal and nasopharyngeal tuberculosis are nonspecific, which makes early diagnosis challenging. 3 Nasopharyngeal tuberculosis with cervical lymph node can be confused with a cervical malignant disease 8 and can mimic nasopharyngeal carcinoma (NPC). 9
We present a case of nasopharyngeal tuberculosis that presented with a right neck lump which was initially treated as lymphadenitis. Nasal endoscopy on a subsequent visit revealed a nasopharyngeal mass mimicking NPC with neck metastasis. A repeat biopsy of the nasopharyngeal lesion helped in the diagnosis.
Case report
A 32-year-old woman presented to the Ear, Nose and Throat clinic with a tender lump on her right side of the upper neck that had appeared for 2 weeks. She had a mild fever initially but did not have a loss of appetite, weight loss or night sweats. She also did not experience any cough, chest pain, nasal obstruction or otologic symptoms. There was no recent contact history of tuberculosis. She was treated for cervical lymphadenitis with amoxicillin by a general practitioner before coming to us. However, her neck lump persisted. On examination, there was a firm 1.5 × 1 cm right upper neck lump which was mildly tender. We performed nasal endoscopy, which showed a nasopharyngeal mass almost obliterating the fossa of Rosenmüller on both sides (Figure 1).
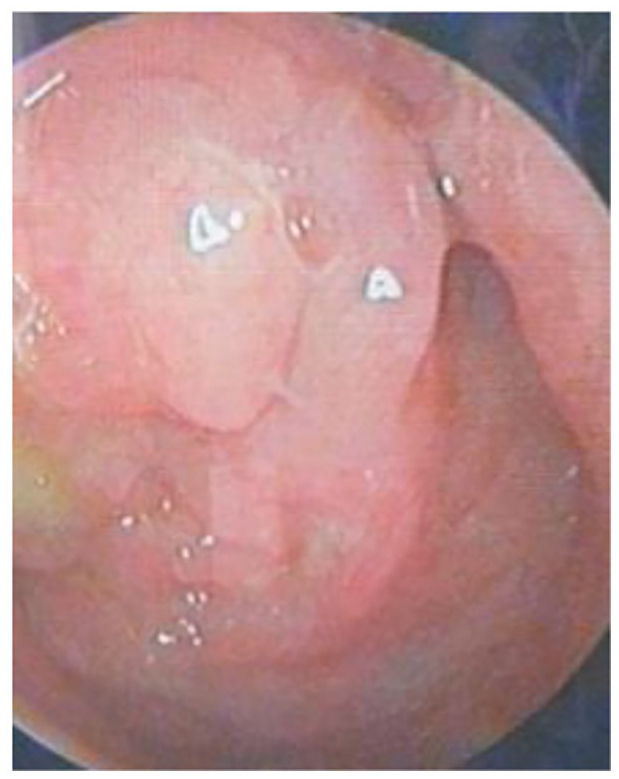
Nasopharyngeal mass seen on nasal endoscopy.
The surface of the mass looked irregular with yellowish secretions on it. The rest of the ear, nose and throat examinations were unremarkable. Our working diagnosis was NPC with neck metastasis. We ordered an ultrasound scan of the neck and fine needle aspiration cytology (FNAC) of the neck lump. A biopsy of the nasopharyngeal mass was taken. The ultrasound scan reported multiple hypoechoic enlarged cervical nodes bilaterally with distortion of the fatty hilum. A computed tomography (CT) scan of the nasopharynx and neck was ordered to further evaluate the neck lump and nasopharyngeal mass.
The FNAC was reported as showing 2 foci of atypical cells arranged in a sheet, composed of enlarged hyperchromatic nuclei. The nuclear membranes were found to be irregular with moderate cytoplasm in the background of abundant polymorphous lymphoid cells and red blood cells. The Ziehl–Neelsen (ZN) stain of the FNAC was negative for acid-fast bacilli (AFB). Metastatic carcinoma of the neck node was suspected. The CT scan showed a heterogeneously enhancing soft tissue mass measuring 2 × 1.4 × 3.9 cm in the nasopharynx, obliterating the bilateral fossa of Rosenmüller and extending inferiorly to the upper part of the oropharynx (Figure 2). Multiple enlarged, homogenously enhancing cervical nodes were seen on both sides. The visualized apical lungs showed minimal consolidation and centrilobular nodules with tree-in-bud appearances.
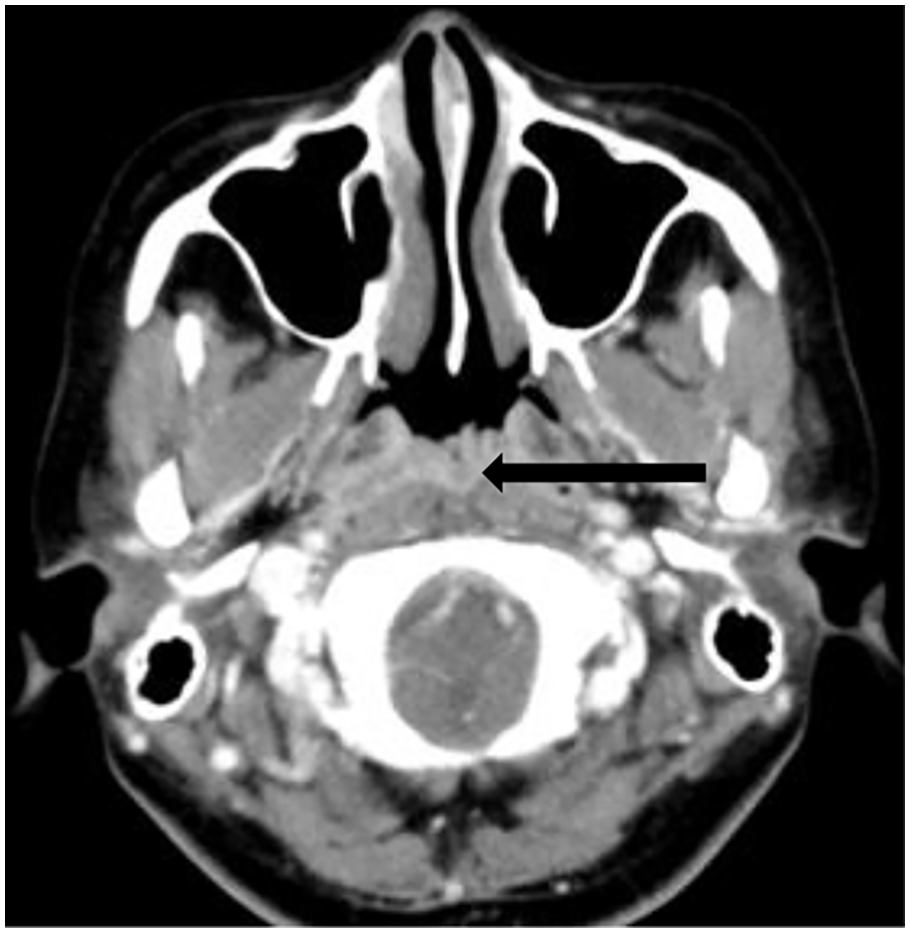
Computed tomography scan showing nasopharyngeal mass (arrow).
The radiologist’s impression from the CT scan finding was that of nasopharyngeal cancer with multiple cervical lymphadenopathy. Lesions in the lung apices were suspected of being active lung infections. Histopathological examination of the nasopharyngeal mass biopsy revealed necrotizing granulomatous inflammation with epithelioid histiocytes and multinucleated giant cells. No malignant cells were seen. Stains for AFB and Grocott methenamine silver (GMS) stain for fungal hyphae were negative.
A repeat biopsy of the nasopharyngeal mass was sent due to strong suspicion of nasopharyngeal cancer with neck metastasis. The repeat biopsy showed caseating granuloma with dense chronic inflammatory cells forming lymphoid follicles and the ZN stain demonstrated a few AFB (Figure 3(a)–(c)). Malignant cells were again not detected and GMS staining was negative for fungal hyphae.

Epithelioid granuloma with Langhans giant cells. (a) Hematoxylin & eosin stain ×100. (b) Hematoxylin & eosin stain ×400. (c) Ziehl–Neelsen stain: ×1000. Acid Fast Bacilli (arrow).
The repeat biopsy was again negative for malignant cells and fungal hyphae. We ordered sputum smears for microscopy and a chest x-ray to exclude pulmonary tuberculosis. The chest x-ray was normal, but her sputum smears for AFB tested positive. Her white blood cell (WBC) count was 6.99 × 109/L, neutrophil count 70.4%, lymphocyte count 19%, Hb level was 12.2 g/dL, erythrocyte sedimentation rate was (ESR) 60 mm/h and anti HIV and HBsAg tests were negative. A diagnosis of nasopharyngeal tuberculosis secondary to pulmonary tuberculosis was made. We referred her to a pulmonologist colleague who put her on first-line anti-tubercular drugs. The patient was admitted to the tuberculosis ward for 2 weeks to be observed for any drug reactions and acute side-effects as per the protocol. The patient responded well to standard treatment of a 2 months intensive phase (isoniazid, rifampicin, pyrazinamide, ethambutol), and a 4 months continuation phase (isoniazid and rifampicin). Her neck node was not palpable after 2 months of treatment and the nasopharyngeal mass disappeared in 3 months. She remained asymptomatic at 6 months follow-up.
Discussion
Tuberculosis can affect almost any organ, but nasopharyngeal involvement is unusual 10 and rare even in endemic areas. 4 The number of reported cases of nasopharyngeal tuberculosis has slightly increased recently which may be due to advances in diagnostic tools, although the prevalence has decreased. 6 Based on previous reports, nasopharyngeal tuberculosis is slightly more common among females. 11 Patients suffering from head and neck tuberculosis may have atypical presentation with a lack of constitutional symptoms such as weight loss, night sweats and respiratory symptoms. 12 According to previous studies, cervical tubercular lymphadenitis was responsible for the majority of head and neck tuberculosis,13–15 but other sites included the salivary glands, larynx, eyes, ears, skin, thyroid, oral cavity, nasopharynx and retropharynx. 13 The most common symptom in nasopharyngeal tuberculosis is cervical lymph node enlargement, 7 but it can present without cervical nodes.4,5 Patients with nasopharyngeal tuberculosis may also present with otologic or nasal symptoms as the primary complaint. One study has found that most patients in fact presented with otologic symptoms such as ear fullness and hearing impairment. 11 On our literature review, patients experienced nasal symptoms such as nasal obstruction,2,6,10 rhinorrhea,2,10 post-nasal drip, 4 sore throat, 16 cough, 4 and snoring.5,6,10 Otologic symptoms were ear fullness, 3 hearing impairment,10,12 ear discharge, 10 and tinnitus. 12
To avoid misdiagnosis, tissue diagnosis is important as NPC and nasopharyngeal tuberculosis can present similarly with cervical lymph node enlargement and a lesion in the nasopharynx. 7 Some of the other differential diagnoses include midline granuloma, Wegener’s granuloma, sarcoid, syphilis, lymphoma, fungal infections, leprosy and periarteritis nodosa. 16 There is no specific examination or diagnostic clues for nasopharyngeal tuberculosis except histological findings. 11 The endoscopic appearances of nasopharyngeal tuberculosis are reported as being smooth swelling,6,12 irregular mucosal thickening,4,16 polypoidal mass,9,10 granuloma-like masses, 8 and mucosal oedema and hyperaemia. 5
Tuberculosis is diagnosed by laboratory techniques that include culture, demonstration of acid-fast bacilli on microscopy, histopathology and polymerase chain reaction (PCR) analysis. 13 The demonstration of acid-fast bacilli by ZN staining is not confirmatory of tuberculosis, as bacilli may be non-tuberculous mycobacteria. 13 The absence of AFB on ZN stain, on the contrary, does not exclude tuberculosis because sensitivity is poor in patients with paucibacillary tuberculosis, including children, patients with extrapulmonary tuberculosis or those who are HIV coinfected. 17 Moreover, the presence of granuloma or its absence in histopathology does not confirm tuberculosis or exclude it, respectively. Nasopharyngeal cancer may cause granulomatous reactions in the surrounding tissues 6 and there are other conditions such as syphilis, leprosy, fungal infection, Wegener granulomatosis and sarcoidosis that cause granulomatous lesions. NPC and tuberculosis may rarely co-exist,18,19 which makes it important to exclude both conditions. In our case, it was necessary to re-biopsy the nasopharyngeal mass to exclude NPC as the ultrasound scan of the neck, the FNAC report, and the CT scan findings were suspicious of malignant disease. The second biopsy, which demonstrated caseating granuloma and AFB, and the absence of malignant cells, led us to diagnose the case as nasopharyngeal tuberculosis. Subsequently, sputum smears for AFB tested positive, which indicated the diagnosis of nasopharyngeal tuberculosis secondary to pulmonary tuberculosis. A culture for AFB and PCR analysis should have been done in our case to confirm the diagnosis of nasopharyngeal tuberculosis. However, these tests are not readily available in our resource-poor setting. Moreover, as the culture for AFB takes a long time, loss of patient follow-up is a concern in our setting where patients are mostly uneducated.
Conclusion
Nasopharyngeal tuberculosis is rare. It may be confused with NPC due to the absence of constitutional symptoms of tuberculosis, lack of characteristic endoscopic appearance and imaging findings. It may cause a diagnostic dilemma, especially in a resource-poor setting where confirmatory tests are not readily available. A repeat biopsy of the nasopharyngeal lesion may be necessary to diagnose it and exclude more common NPC. Primary infection in the lungs should be excluded by adequate assessment before labelling it as a case of primary nasopharyngeal tuberculosis.
Footnotes
Acknowledgements
We thank the patient for consenting to the publication of her anonymized story.
Author Contributions
D.P.: conception, literature review, manuscript preparation, critical revision, photography and final approval.
B.P.: manuscript preparation, design, critical revision, photography and final approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
