Abstract
Hepatocellular carcinoma (HCC) is a common and aggressive malignancy. While common metastatic sites of hepatocellular carcinoma include the lung, bone, and lymph nodes, pharyngeal metastasis is extremely rare. This case report presents a unique case of a patient with hepatocellular carcinoma who developed pharyngeal metastasis. We describe the patient’s clinical course, diagnostic work-up, and treatment strategies and details. We paid special attention to diagnostic difficulties and to ways of its resolving. Additionally, a comprehensive literature review was performed to summarize the characteristics, possible mechanisms, and management of hepatocellular carcinoma pharyngeal metastasis. By presenting this case and reviewing the literature, we aim to raise awareness among clinicians about this rare metastatic manifestation of hepatocellular carcinoma and provide insights into its diagnosis and treatment.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer and third leading cause of cancer-related deaths globally. 1 It arises predominantly in the context of chronic liver disease, with key risk factors including hepatitis B virus (HBV) and C viral infections, aflatoxin exposure, alcohol-related cirrhosis, and metabolic dysfunction-associated steatotic liver disease. Despite advances in surveillance programs, the diagnosis of HCC often occurs at intermediate or advanced stages due to the limited sensitivity of current biomarkers and imaging modalities for early detection. Treatment strategies, including surgical resection, liver transplantation, locoregional therapies, and systemic agents (such as tyrosine kinase inhibitors and immunotherapies), are challenged by high recurrence rates, underlying liver dysfunction, and primary or acquired drug resistance.
Metastasis drives its poor prognosis, with lung, bone, and lymph nodes being common sites. 2 Pharyngeal metastasis is exceptionally rare; there are few reports to date. The absence of direct vascular/lymphatic connections suggests unconventional dissemination mechanisms, potentially involving Batson’s vertebral venous plexus enabling retrograde hematogenous spread. 3
Here, we present a 53-year-old male with HBV-associated HCC, pharyngeal metastasis was revealed by our team after 50 months after primary tumor removal. By integrating this case with a systematic literature review, we aim to characterize clinicopathological patterns and advance management strategies for this ultrarare metastasis entity and discuss strategies for its diagnosis and management, highlighting the importance of vigilance in the follow-up of high-risk HCC patients.
Cases report
A 53-year-old male with a 30-year history of untreated HBV infection presented with intermittent right upper abdominal distension and pain in September 2020, leading to a diagnosis of HCC following comprehensive evaluations (Figure 1(a)). The patient had no history of other diseases, and there were also no diseases of note in his family history. The patient’s alpha-fetoprotein (AFP) levels significantly elevated at initial diagnosis (2997.84ng/ml), and notes that liver function was maintained at Child-Pugh A throughout most of the clinical course. He underwent surgical treatment on September 7, 2020, with postoperative pathology revealing nodular (mass-trabecular) HCC grade III (9.5 × 9 × 6 cm) with partial clear cell differentiation, adjacent to the hepatic capsule, no vascular tumor thrombus, nerve invasion, or satellite nodules, negative resection margin, MVI = M0 (Figure 1(b)). In November 2021, he developed right hip and lower extremity pain, a routine follow-up computed tomography (CT) scan at this time also identified stable, small nodules in the right upper lung. Bone scintigraphy and CT confirm S1 vertebral bone metastasis; then he received palliative radiotherapy (48 Gy/18 fractions) combined with camrelizumab (200 mg IV), lenvatinib (8 mg/day), and zoledronic acid, achieving stable disease (SD) before discharge on December 27, 2021. During 2022 follow-up, he maintained regular camrelizumab + lenvatinib therapy, the patient’s AFP levels were slowly dropping to normal with imaging consistently showing SD, including stable right upper lung nodules, S1 vertebral metastasis in November 2022. After discharge, the patient regularly received lenvatinib (8 mg/day) for targeted therapy at a local hospital, and underwent regular CT re-examinations for evaluation. The patient’s AFP levels remained within the normal range during this time, with the efficacy assessment being SD.
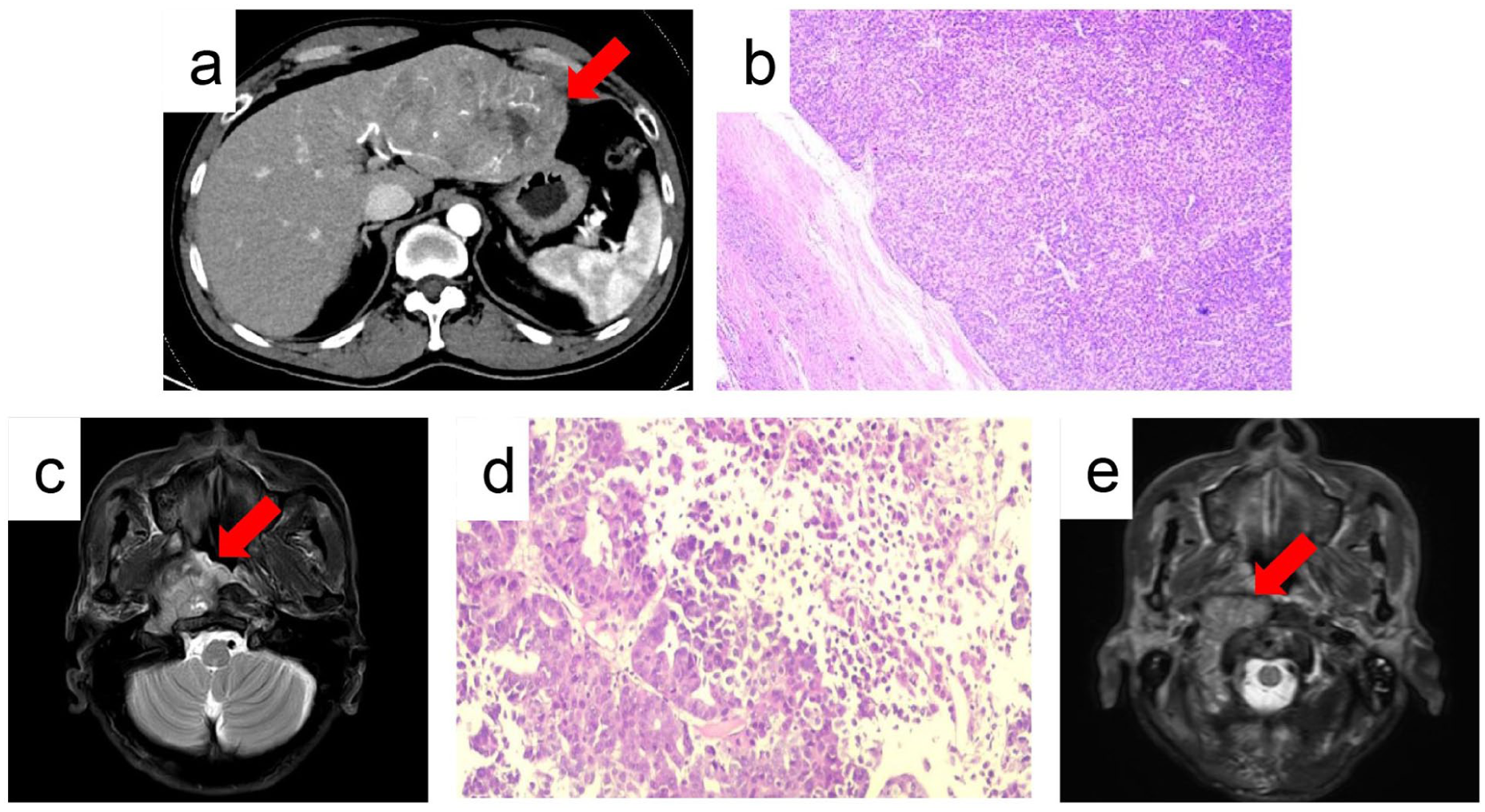
Diagnostic imaging and histopathology of HCC and its pharyngeal metastasis. (a) Arterial phase abdominal CT scan (September 5, 2020) revealing a large hepatic mass (arrow) prior to resection. (b) Photomicrograph of the resected liver specimen (H&E stain, 10× magnification) showing nodular-trabecular HCC (September 8, 2020). (c) Contrast-enhanced MRI of the head and neck (November 3, 2024) demonstrating a large, enhancing mass in the right oropharyngeal region (arrow). (d) Pathological images (November 6, 2024) obtained from ultrasound-guided needle biopsy (H&E stain, 10× magnification) confirming metastatic HCC. (e) Contrast-enhanced MRI of the head and neck (March 9, 2025) showing significant reduction in the size and enhancement of the metastatic oropharyngeal mass (arrow) following radiotherapy.
In November 2024, right head and facial pain prompted evaluation, with contrast-enhanced CT and magnetic resonance imaging (MRI) revealing a right nasopharynx/oropharynx mass with lymphadenopathy (involving C1-3 vertebrae and adjacent vessels; Figure 1(c)); The patient’s AFP levels rose again at metastasis (124575.59 ng/ml). The initial biopsy was performed using biopsy forceps under local anesthesia during nasopharyngoscopy was nondiagnostic. Subsequently, ultrasound-guided needle biopsy was performed. This re-biopsy confirmed metastatic HCC (grades II–III; Figure 1(d)). He underwent palliative pharyngeal radiotherapy (36 Gy/12 fractions) and systemic therapy adjustment to camrelizumab + regorafenib (120 mg/day), with severe mucosal reactions (grade III oropharyngeal mucositis with ulceration) managed by supportive care (recombinant human epidermal growth factor, vitamins, and nutritional supplementation) before discharge on November 30, 2024. In February 2025, right shoulder/upper extremity pain developed, the MRI examination performed in the outpatient department of our hospital suggests that possible metastases to the right humerus, olecranon, and ribs; he received ibandronate and camrelizumab at the local hospital before re-admission for further management. In March 3, 2025, a nasopharyngeal magnetic resonance examination and enhanced CT scan of the lungs was conducted, indicating that the nasopharyngeal and oropharyngeal masses had shrunk compared to before. The nasopharyngoscopy results also indicated that the mass has further shrunk, and the metastatic lymph nodes in the neck had decreased (Figure 1(e)), but the patient has metastatic tumors in both lungs. After disease progression, the patient received regorafenib maintenance treatment until May 2025. The patient was dead with disease at the last follow-up in July 2025 (Figure 2).

The course of treatment.
Discussion
HCC typically metastasizes to the lung, bone, and lymph nodes, but pharyngeal metastasis remains extremely rare, with fewer than 15 cases reported in the literature to date.4,5 Our case reported a 53-year-old male with HBV-associated HCC, pharyngeal metastasis was revealed by our team after 50 months after primary tumor removal, adds to this limited body of evidence, highlighting unique clinical and pathogenic features that align with, yet also expand, current understanding of this ultrarare entity.
Pharyngeal metastasis accounts for less than 1% of all extrahepatic HCC metastases. 6 A review of published cases (Table 1) shows a striking male predominance (100%), consistent with our patient, and a median age of 60 years (range 45–73 years), with our patient (53 years) falling within this range.7,8 Metastasis is predominantly metachronous (62.5% of cases), with an interval of 17–58 months from primary HCC diagnosis, which matches our patient’s 4-year 2-month latency. 9 Notably, our patient initially developed bone metastasis (S1 vertebra) before pharyngeal involvement, a pattern not commonly reported but consistent with HCC’s propensity for multisite dissemination. The primary tumor in our case was large (9.5 cm) and poorly differentiated (grade III) with clear cell features—factors associated with higher metastatic potential. 10 This contrasts with smaller, well-differentiated tumors in some previously reported pharyngeal metastasis cases, suggesting aggressive tumor biology may contribute to unusual metastatic tropism. 11 The pathogenesis of pharyngeal metastasis remains poorly understood, but the vertebral venous plexus (Batson’s plexus) theory is the most widely accepted. 3 This valveless venous network connects the portal system to the pharyngeal venous plexus, enabling retrograde flow of tumor cells when intra-abdominal pressure increases (e.g., during coughing or straining).
Characteristics of patients with nasopharyngeal metastasis from hepatocellular carcinoma.

TAE, transarterial embolization; RFA, radiofrequency ablation; M, male; HCC: hepatocellular carcinoma.
Our patient’s pharyngeal mass involved the C1-3 vertebrae and adjacent vasculature, further supporting Batson’s plexus as the route of spread, as this plexus directly communicates with the cranial venous system. 13 The absence of vascular invasion (MVI = M0) in the primary tumor suggests hematogenous spread via this alternative pathway rather than direct vascular invasion. Diagnosing pharyngeal metastasis is often delayed due to nonspecific symptoms (e.g., head/facial pain, dysphagia) and low clinical suspicion. 12 Our patient’s initial nasopharyngoscopy biopsy under local anesthesia was nondiagnostic; it may be due to limited inspection technology or superficial sampling, resulting in a negative biopsy, the report interpreted as chronic inflammation. A second ultrasound-guided needle biopsy was performed and immunohistochemical (IHC) confirmation. IHC markers such as Hepatocyte+, Arg-1+, and AFP+ were critical in confirming HCC origin, consistent with literature recommendations. 14
Treatment strategies for pharyngeal metastasis are heterogeneous. Surgical resection, as in Tohyama et al.’s long-term survivor (12 years 10 months), offers the best chance for durable control but is feasible only for localized, polypoid lesions. 4 For advanced cases like ours, palliative radiotherapy (36 Gy/12f) combined with systemic therapy (camrelizumab + regorafenib) achieved symptomatic relief and tumor shrinkage, aligning with reports that radiotherapy improves local control in 50%–70% of cases.15,16 Adverse events, such as severe mucosal reactions from regorafenib, required dose adjustments and supportive care, highlighting the need for personalized management of targeted therapy toxicities. 17 Prognosis for pharyngeal metastasis remains poor, with a median survival of 10 months (range 3 months–12 years 10 months). 18 Our patient survived 8 months after diagnosis, within this range, but the shrinkage of the pharyngeal mass and SD posttreatment suggest that aggressive multimodal therapy may extend survival in select cases. In essence, the pharyngeal metastasis did not directly kill the patient, but the severe pain and dysphagia caused by the pharyngeal mass directly led to inadequate nutritional intake. This undoubtedly accelerated the development of cachexia, a multifactorial syndrome defined by an ongoing loss of skeletal muscle mass (with or without loss of fat mass) that cannot be fully reversed by conventional nutritional support and leads to progressive functional impairment. Cachexia significantly reduces physical strength, immune function, and tolerance to anticancer therapies, thereby hastening functional decline and mortality.
This case underscores three key implications: (1) HCC surveillance should include head/neck imaging in high-risk patients (e.g., large, poorly differentiated tumors); (2) persistent head/neck symptoms in HCC patients warrant prompt nasopharyngoscopy and IHC-guided biopsy; (3) multidisciplinary collaboration (oncology, otolaryngology, radiation) is critical for optimizing treatment.19,20 In managing this exceptionally rare case, our team significantly enhanced our skills in interpreting complex head and neck imaging and mastering the multidisciplinary coordination required to manage complex toxicities from combined modality therapy.
Conclusion
HCC with pharyngeal metastasis is an extremely rare entity, with unique clinical and pathogenic features. This case highlights that pharyngeal metastasis can occur in HBV-associated HCC, even with an absence of vascular invasion in the primary tumor, supporting the role of Batson’s vertebral venous plexus in unconventional hematogenous spread. The male predominance, metachronous onset, and aggressive tumor biology (large size, poor differentiation) observed align with limited existing literature. The primary diagnostic concerns lie in the nonspecific symptoms (e.g., head and facial pain, dysphagia) that mimic benign conditions, the technical challenges of obtaining adequate tissue via endoscopic biopsy in complex anatomical sites, and the necessity of IHC confirmation (e.g., Hepatocyte+, Arg-1+, AFP+) to distinguish metastatic HCC from primary pharyngeal malignancies or other metastases. Multimodal therapy, including palliative radiotherapy combined with targeted and immunotherapy, may achieve symptomatic relief and tumor control, though prognosis remains poor. The reasons is attributable to the advanced systemic disease burden at the time of pharyngeal metastasis presentation, the aggressive biology of the primary tumor, limited curative options for unresectable lesions, and the cumulative toxicities of multimodal therapy which often compromise treatment tolerance and patient performance status. Clinicians should maintain vigilance for head/neck symptoms in high-risk HCC patients, with nasopharyngoscopy biopsy first and IHC confirmation critical for timely diagnosis. Further studies are needed to explore molecular mechanisms driving this rare metastatic tropism and optimize diagnostic and treatment strategies.
Footnotes
Acknowledgements
WCZ, LJZ, QC, TD involved in manuscript preparation. All authors have read and approved the case reports.
Ethical considerations
This research was approved by the Institutional Review Board of Hunan Provincial People’s Hospital (LL-20250725-1).
Consent for publication
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hunan Province, China (Grant No. 2025JJ60776, 2025JJ80866). Project supported by the Health Commission of Hunan Province (Grant No. W20243286), WU Jieping Medical Foundation (Grant No. 320.6750.2025-02-36).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author.
