Abstract
Objectives:
Blood-derived immune-inflammatory markers have been identified as promising tools in many conditions. This study aims to determine the perioperative factors and hematologic indices associated with the occurrence of delayed recover after pancreatic and biliary surgeries (PBS).
Methods:
Included in this study were 106 patients with PBS who were divided into prolonged hospitalization group (≤5 days; n = 48) and short hospitalization group (>5 days; n = 58) between January 2020 and April 2023 in a tertiary hospital in Saudi Arabia. The clinico-demographic characteristics, laboratory parameters and blood-derived immune-inflammatory markers (neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio (PLR), systemic inflammatory index (SII), aggregate index of systemic inflammation, and systemic inflammatory response index) of the patients with and without prolonged hospitalization were compared by independent sample t-test, Mann–Whitney U-test, chi-square test, and Fisher’s exact test as appropriate. Receiver operating characteristic (ROC) analysis was used to establish a cut-off level of postoperative PLR and SII values to predict prolonged hospitalization. Perioperative factors associated with prolonged hospitalization were evaluated by univariable and stepwise multivariable logistic regression analyses.
Results:
Of the 106 patients undergoing PBS, 48 patients (45.28%) had prolonged hospitalization. Both postoperative PLR and SII in patients with prolonged hospitalization were significantly higher than those in patients without. The ROC curve showed that the optimal cut-off value of postoperative SII to predict prolonged hospitalization was 1073.17, with 0.792 sensitivity and 0.483 specificity. In addition, the optimal cut-off value of postoperative PLR to predict prolonged hospitalization was 227.55, with 0.604 sensitivity and 0.638 specificity. Stepwise multivariate analysis showed that postoperative SII (>1073.17) was an independent predictor for prolonged hospitalization (odds ratio = 3.527, 95% confidence interval: 1.491–8.434, p = 0.004), together with intraoperative blood transfusion requirement and preoperative prothrombin time.
Conclusions:
Our findings demonstrated an association between the higher level of postoperative SII and delayed recovery, suggesting that it may be a useful parameter for identifying patients at risk of delayed recovery after PBS.
Keywords
Introduction
Pancreatic and biliary surgeries (PBS) are specialized procedures performed to manage conditions such as gallstones, chronic inflammation, and bile duct obstructions. These interventions play a critical role in restoring biliary drainage, alleviating symptoms, and preventing complications associated with impaired pancreatic or biliary function, such as biliary obstruction. 1 Despite their significant clinical benefits, these surgeries carry considerable risks due to the anatomical complexity of the hepato-pancreato-biliary region. Common complications include hemorrhage, infection, bile leakage, and iatrogenic injury to adjacent structures.2,3
Systemic inflammation is a common complication after hepato-pancreato-biliary surgery that is frequently provoked by damaged tissues and bacterial translocation. When this response is exaggerated into a hyperinflammatory state, it can lead to excessive tissue damage and progress into systemic inflammatory response syndrome and ultimately multiple organ dysfunction.4,5 Several laboratory diagnostic tools exist to guide clinicians in the early detection of postoperative complications. C-reactive protein (CRP), an acute phase protein, has demonstrated a strong correlation with postoperative adverse events. However, concerns persist regarding it specificity, positive predictive value, and routine integration into daily care of surgical patients. 6
Simple blood-derived immune-inflammatory markers such as neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), systemic inflammatory index (SII), Aggregate Index of Systemic Inflammation (AISI), and Systemic Inflammatory Response Index (SIRI) are widely investigated for their diagnostic and prognostic potential in various conditions, including infections, autoimmune diseases, and malignancies.7,8 These markers are increasingly recognized their affordability, accessibility, and clinical utility, especially in countries with limited resources where access to advance diagnostic tools may be limited.9,10
In surgical settings, blood-derived biomarkers offer insights into the early detection of postoperative complications such as infections, impaired wound healing, and systemic inflammation.11,12 For instance, elevated preoperative SII levels in patients undergoing off-pump coronary artery bypass graft surgery were significantly associated with longer ICU stays and extended durations of mechanical ventilation. 13 In patients with intrahepatic cholangiocarcinoma undergoing surgical resection, an elevated NLR was independently associated with decreased overall survival. 14
To date, there are limited studies concerning the use of these blood-derived immune-inflammatory factors in the prediction of delayed recovery after PBS. In the present study, we aimed to investigate perioperative factors associated with delayed recovery after PBS, with particular emphasis on the predictive value of pre- and postoperative blood-derived immune-inflammatory markers including NLR, PLR, SII, AISI, and SIRI.
Materials and methods
Study design and eligibility criteria
This study retrospectively analyzed patients who underwent PBS who were admitted to the operating room department of King Abdulaziz Medical City in Riyadh, Saudi Arabia, from January 2020 to April 2023. In this study, 1014 electronic medical records were reviewed for eligibility. The inclusion criteria include patients aged 18 years or older and those who have undergone elective PBS. Exclusion criteria include incomplete or missing data, patients under the age of 18 years, patients diagnosed with hematological disorders, and cancer patients.
Ethical approval
The current study was approved by the Institutional Review Board (IRB) at King Abdullah International Medical Research Centre (KAIMRC; IRB/0715/24). Since this is a retrospective study and all patient information was de-identified to protect patient anonymity, the KAIMRC ethics committee/IRB waived the requirement for informed consent.
Data collection
Data collection was carried out using an institutional electronic medical records system (BestCare). This study examined multiple factors, including laboratory, clinical, surgical, and demographic information. The demographic data included age, gender, and body mass index (BMI). The baseline clinical data comprised the American Society of Anesthesiologists (ASA) physical state classification, 15 presence of chronic medical conditions, including diabetes mellitus (DM), obesity, hypertension (HTN), and cardiovascular disease (CVD), intraoperative blood transfusion and type of surgery. In addition, the length of hospital stay was calculated from the date of surgery to discharge. The median length of hospital stay was calculated (5 days) and patients were categorized into Group A ≤ 5 days (n = 58) and Group B > 5 days (n = 48).
In addition, laboratory parameters were collected during the 3 days before and after surgery. This timeline was chosen in order to capture immediate pre- and post-operative immune-inflammatory and hematologic changes, reduce variability in the timing of laboratory evaluations, ensuring consistent and comparable data collection across all patients. Coagulation parameters, including prothrombin time (PT), partial thromboplastin time, and international normalized ratio (INR), as well as hemoglobin, hematocrit, platelet count, and absolute counts of neutrophils, lymphocytes, and monocytes, were retrieved from the electronic medical records system. Liver function tests were also collected including aspartate aminotransferase and alanine aminotransferase. Lastly, the following blood-derived immune-inflammatory markers were calculated using the following formulas: SII = Platelet count × Neutrophil count/lymphocytes count, NLR = Neutrophils count/lymphocytes count, PLR = Platelets counts/lymphocytes count, SIRI = Neutrophils count × monocytes count × platelet count/lymphocytes count, and AISI = Neutrophils count × monocytes count/lymphocytes count.
Statistical Analysis
Data analyses was conducted using R statistical software (V.4.1.2; R Foundation for Statistical Computing, Vienna, Austria). The distribution of continuous data was assessed using the Shapiro–Wilk test. Continuous variables were non-normally distributed, except for age and BMI, and were compared between groups using sample t-test or Mann–Whitney U-test, where appropriate. The values were presented as median and interquartile range (IQR) for non-normally distributed data or mean + standard deviation (SD) for normally distributed data. For categorical variables, values are expressed as number of cases (n) and percentage (%) and compared using the chi-square test or Fisher’s exact test, where appropriate.
Initially, descriptive analysis was generated for the entire cohort. Patients were then categorized into two groups based on the median length of hospital stay (≤5 days vs. >5 days), and baseline, clinical, and laboratory variables, including blood-derived immune-inflammatory markers were compared between groups. Immune-inflammatory markers that demonstrated significant differences (p < 0.05) in the bivariate analysis were further evaluated for their discriminatory power using receiver operating characteristic (ROC) curves analysis. For each marker, the area under the curve (AUC), optimal cut-off value, sensitivity, and specificity were calculated. Optimal thresholds were determined using Youden’s index, 16 after which patients were categorized into high-value and low-value groups accordingly.
Univariate logistic regression was then performed to examine associations between each perioperative factor and prolonged hospitalization, and crude odds ratios (ORs) with 95% confidence intervals (CIs) were reported. Variables were selected based on their clinical relevance and their significance in the bivariate comparisons between the two hospital-stay groups. Variables with p < 0.10 in the univariate analysis were considered eligible for inclusion in the multivariate model to avoid excluding potentially important predictors. A forward stepwise multivariable logistic regression approach (p for entry 0.05, p for removal 0.10) was applied to identify independent predictors of prolonged hospitalization, and only variables retained in the final model are reported. For all analyses, we considered a p-value (two-sided) of less than 0.05 to be statistically significant.
Results
In total, 106 patients were analyzed. Their characteristics are shown in Table 1. The majority of all patients were females n = 66 (62.3%), and the mean age was 52.22 years (SD = 16.04). The mean BMI of the study population was 31.15 kg/m2 (SD = 6.10). Most of the patients were classified into ASA 2 (50.9%) and ASA 3 (29.2%), with more than half of the study population having chronic conditions. The majority of the patients underwent laparoscopic cholecystectomy (94.3%). Only nine patients had an intraoperative blood transfusion, and the median time of surgery was 93.30 min. Next, we stratified patients according to the median length of stay into short hospitalization group ≤5 days (Group A, n = 58) and long hospitalization group >5 days (Group B, n = 48). Patients who had longer hospitalization >5 days when compared to those in short hospitalization group were older (mean age 56.56 vs. 48.63 years, p = 0.013), had more CVD (19.0% vs. 39.6%, p = 0.030), and required more intraoperative blood transfusion (1.7% vs 16.7%, p = 0.010). Other baseline and perioperative characteristics were similar between both groups.
Baseline and perioperative characteristics.
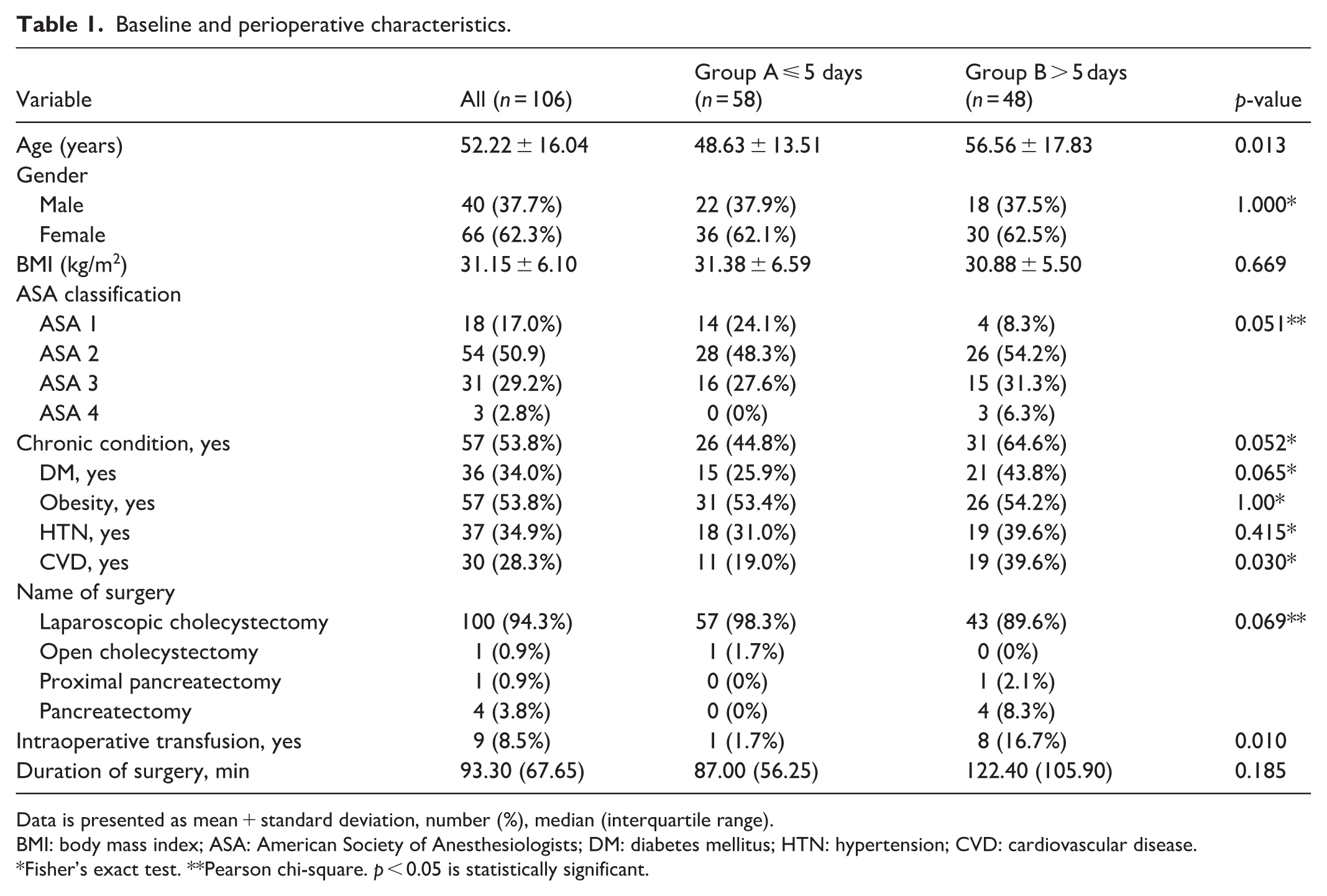
Data is presented as mean + standard deviation, number (%), median (interquartile range).
BMI: body mass index; ASA: American Society of Anesthesiologists; DM: diabetes mellitus; HTN: hypertension; CVD: cardiovascular disease.
Fisher’s exact test. **Pearson chi-square. p < 0.05 is statistically significant.
Tables 2 and 3 compare laboratory and immune-inflammatory markers measured during the preoperative period. In Table 2, patients in group B who had longer hospitalization of more than 5 days also had high platelet count (308.50 × 109/L vs. 253.00 × 109/L; p = 0.015), longer PT time (11.60 s vs. 11.20 s; p = 0.005), and high INR (1.02 vs. 1.06; p = 0.009) compared to those in group A. However, there were no statistically significant differences in all preoperatively measured immune-inflammatory markers between patients in both groups (Table 3). Although statistically insignificant, PLR (158.34 vs. 151.95), SII (712.94 vs. 636.01), and AISI (420.69 vs. 374.53) were slightly higher in the prolonged hospitalization group (all p > 0.05).
Preoperative complete blood count, coagulation profile, and liver function test.

PTT: partial thromboplastin time; PT: prothrombin time; INR: international normalized ratio; ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Data are presented as median (interquartile range).
p < 0.05 is statistically significant.
Preoperative measurements of blood-derived immune-inflammatory markers.
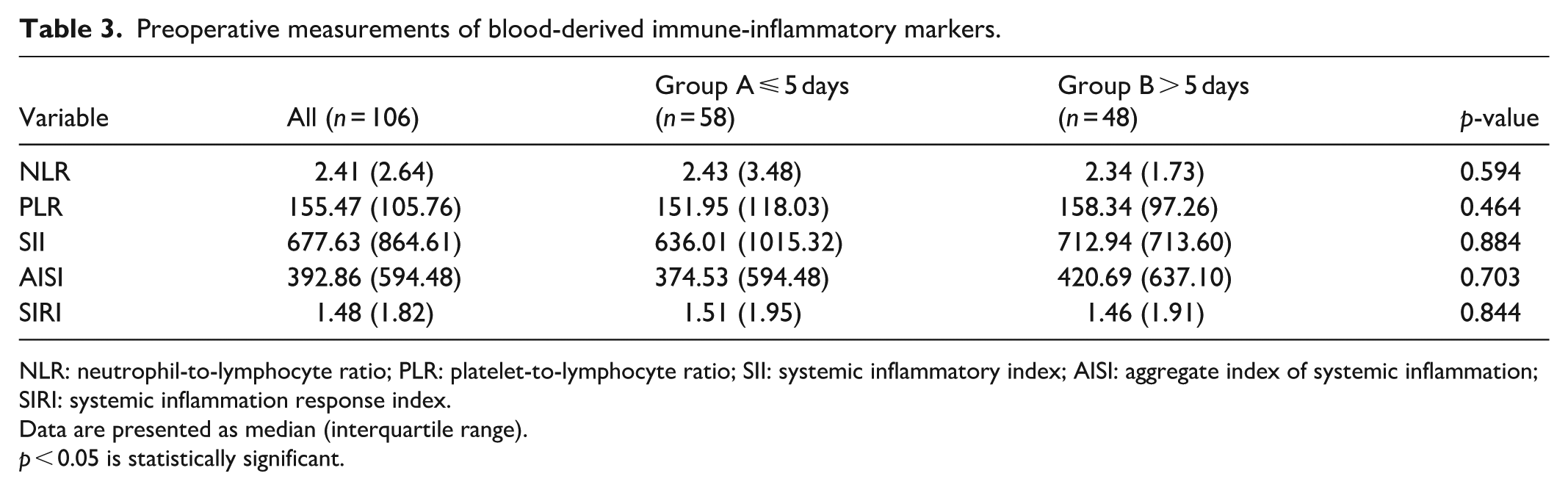
NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; SII: systemic inflammatory index; AISI: aggregate index of systemic inflammation; SIRI: systemic inflammation response index.
Data are presented as median (interquartile range).
p < 0.05 is statistically significant.
Next, we compared the postoperative laboratory data according to the median length of hospital stay (Table 4). In prolonged hospitalization group, postoperative platelets count and PT were significantly higher than those in short hospitalization group (314.50 × 109/L vs. 261.00 × 109/L and 11.75 s vs. 11.40 s, respectively; all p < 0.05).
Postoperative complete blood count, coagulation profile, and liver function test.
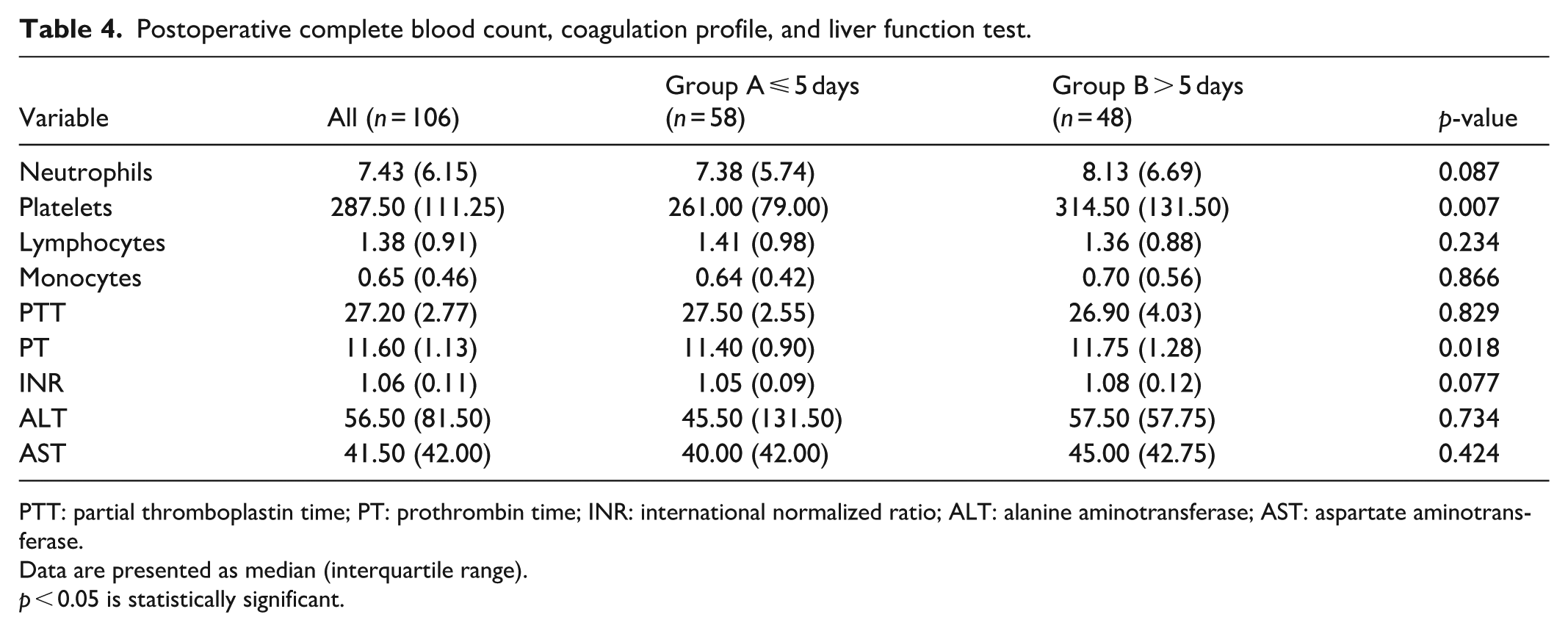
PTT: partial thromboplastin time; PT: prothrombin time; INR: international normalized ratio; ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Data are presented as median (interquartile range).
p < 0.05 is statistically significant.
Table 5 demonstrates the differences in the postoperatively measured immune-inflammatory markers. We observed that patients in group B had higher levels of PLR (median (IQR): 239.54 (172.34)) compared to Group A (202.60 (164.27)), p = 0.015. In addition, postoperative SII value in Group B was higher (1895.60 (2505.75)) compared to those in group A (1268.09 (2285.24)), p = 0.025. Although all other immune-inflammatory markers were statistically insignificant, NLR (median: 6.11 vs. 4.45), AISI (1081.10 vs. 976.05), and SIRI (3.62 vs. 3.27) were higher in the prolonged hospitalization group, all p > 0.05.
Postoperative measurements of blood-derived immune-inflammatory markers.

NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; SII: systemic inflammatory index; AISI: aggregate index of systemic inflammation; SIRI: systemic inflammation response index.
Data are presented as median (interquartile range).
p < 0.05 is statistically significant.
Following group comparison, only statistically significant immune-inflammatory variables, including postoperative PLR and SII, were further assessed for their discriminatory power (Table 6). The cut-off values obtained from ROC analysis were subsequently used to dichotomize both variables for logistic regression modeling. ROC curves of the predictive ability of postoperative PLR and SII for prolonged hospitalization. Of the two variables, the AUC value for postoperative PLR (0.638) was slightly higher with an optimal cut-off value of 227.55, and sensitivity and specificity of 60.4% and 63.8%, respectively. For postoperative SII, AUC value was 0.627 with optimal cut-off value of 1073.17, and sensitivity and specificity of 79.2% and 48.3%, respectively. Subsequently, postoperative PLR was categorized into low PLR group <227.55 and high PLR group >227.55. Similarly, postoperative SII was categorized into low SII group <1073.17 and high SII group >1073.17.
Receiver operator characteristic curve of the individual variables for predicting prolonged hospitalization.

AUC: area under the curve; Cl: confidence interval; PLR: platelet-to-lymphocyte ratio; SII: systemic inflammatory index.
p < 0.05 is statistically significant.
As shown in Table 7, univariate analysis demonstrated that age (OR = 1.033, 95% CI: 1.007–1.060, p = 0.013), presence of chronic condition (OR = 2.244, 95% CI = 1.023–4.925, p = 0.044), presence of CVD (OR = 2.799, 95% CI = 1.167–6.715, p = 0.021), intraoperative recipient of blood products (OR = 11.400, 95% CI = 1.371–94.766, p = 0.024), preoperative PT (OR = 1.970, 95% CI = 1.219–3.183, p = 0.006), preoperative INR (OR = 950.176, 95% CI = 6.966–129,608.113, p = 0.006), postoperative platelet count (OR = 1.005, 95% CI = 1.000–1.009, p = 0.035), postoperative PLR (OR = 2.689, 95% CI = 1.223–5.915, p = 0.014), and postoperative SII (OR = 3.547, 95% CI = 1.491–8.434, p = 0.004) were significantly correlated with postoperative delayed recovery.
Univariate logistic regression analysis.

OR: odds ratio; CI: Confidence interval; CVD: cardiovascular disease; PT: prothrombin time; INR: international normalized ratio; PLR: platelet-to-lymphocyte ratio; SII: systemic inflammatory index; Ref: reference category.
p < 0.05 is statistically significant.
Stepwise multiple logistic regression was then performed after including all variables from the univariate analysis. Table 8 shows the factors that remained significant in the final stepwise multiple logistic regression analysis and were independently associated with increased odds of prolonged hospitalization. These include intraoperative transfusion of blood products, preoperative PT, and postoperative SII values. Patients who had an intraoperative transfusion of blood products had 10.26 times greater odds (OR: 10.260, 95% CI: 1.059–99.380, p = 0.044) for prolonged hospitalization. In addition, preoperative PT was independently with prolonged hospitalization (OR: 1.807, 95% CI: 1.123–2.908, p = 0.004). Lastly, patients with postoperative SII value greater than 1073.17 had 3.52 times greater probability for prolonged hospitalization (OR: 3.527, 95% CI: 1.491–8.434, p = 0.004).
Stepwise multivariate logistic regression analysis.
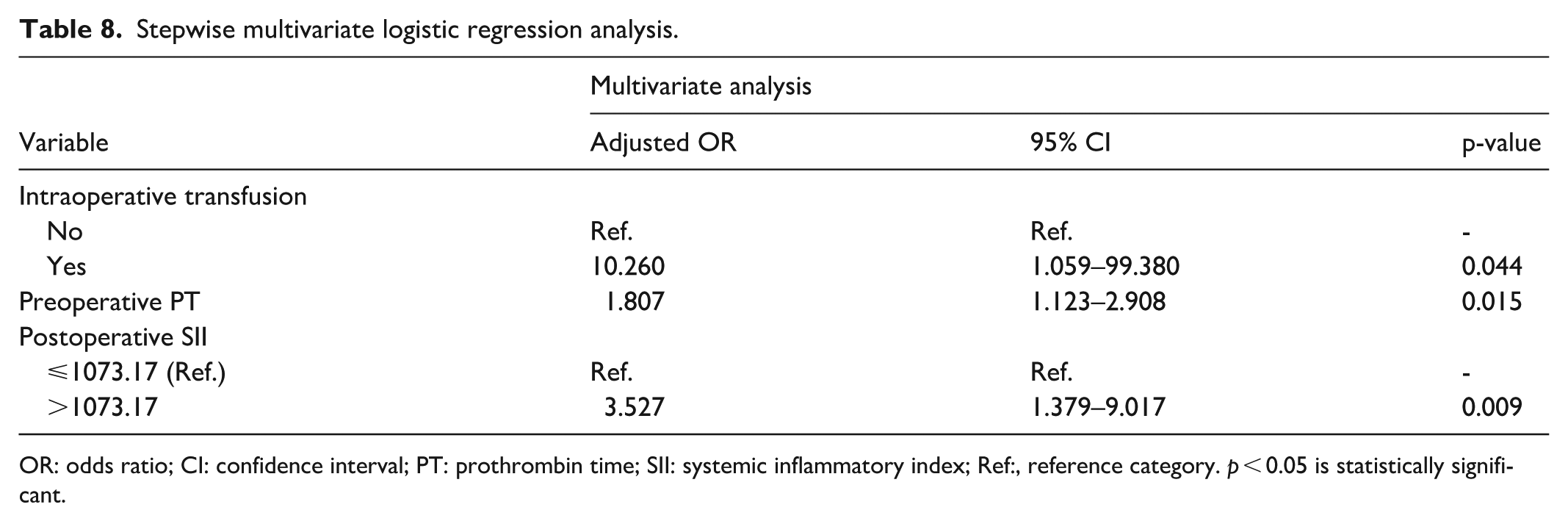
OR: odds ratio; CI: confidence interval; PT: prothrombin time; SII: systemic inflammatory index; Ref:, reference category. p < 0.05 is statistically significant.
Discussion
This study examined perioperative factors associated with prolonged hospital stay following surgery, with patients divided into two groups based on a 5-day median cut-off. Significant differences in baseline and perioperative clinical, laboratory, and immune-inflammatory markers were found between patients with prolonged hospital stay and those without. In the univariate analysis, several factors – including age, presence of chronic condition, presence of chronic CVD, intraoperative blood transfusion, preoperative PT, preoperative INR, postoperative platelet, postoperative PT, postoperative PLR, and postoperative SII – were associated with prolonged hospital stay. However, after stepwise multiple logistic regression, only the need for intraoperative blood transfusion, preoperative PT, and postoperative SII remained independent predictors of prolonged hospitalization. Furthermore, the optimal threshold for postoperative SII to predict prolonged hospital stay (>5 days) was >1073.17, with a sensitivity of 0.792, a specificity of 0.483, and an AUC of 0.627.
Our findings are consistent with previous reports showing that elevated postoperative SII independently predicts severe postoperative complications such as postoperative pulmonary complications, infection, and delirium, contributing to prolonged hospitalization and increasing economic burden. 17 In addition, Parmana et al. 18 found that increased postoperative SII was an independent predictor of prolonged mechanical ventilation and extended ICU stay in cardiac surgery. Our study extends these findings to the PBS setting, highlighting postoperative monitoring of SII as a clinically accessible, low-cost biomarker that reflects both inflammatory burden and hemostatic activation.
The prognostic role of SII also aligns with the growing recognition that multi-marker inflammatory profiling provides superior prognostic accuracy compared with single-parameter measurements such as CRP. Moreover, it has been recently reported that multiple immune-inflammatory indices could enhance the prediction of clinical outcomes in medical and surgical populations. 19 These integrative markers may therefore capture the systemic physiological stress of major abdominal surgery more accurately than single inflammatory parameters.
Tzikos et al. 20 demonstrated that PLR measured on postoperative predicted both mortality and length of stay following cardiac surgery, emphasizing its prognostic significance in assessing patient recovery. Similarly, Eren et al. 21 linked elevated PLR to longer hospitalization in patients with infected diabetic ulcers. Together, these studies suggest that increased PLR reflects persistent platelet activation and lymphocyte suppression, contributing to delayed tissue repair. This pattern aligns with our present findings and is consistent with our previous observation, where we reported a similar relationship between postoperative PLR and hospital stay in bariatric surgery patients. 22
While our study did not recognize platelets as an independent predictor of prolonged hospitalization after surgery. We noted that patients in the prolonged hospitalization group had higher platelet counts during both pre- and postoperative stages. In addition, differences in coagulation parameters may reflect variations in the postoperative inflammatory response, as both platelet activation and coagulation pathways are closely linked to surgical stress and systemic inflammation.23,24 This is consistent with prior research emphasizing the platelet prognostic value in assessing disease severity and predicting clinical outcomes. For instance, a prospective study investigating patients with community-acquired pneumonia found that patients presenting with either thrombocytopenia or thrombocytosis at admission had significantly longer hospital stays compared to those with normal platelet levels. This U-shaped relationship suggests that both abnormally low and high platelet counts are associated with increased risk of complications, thereby extending hospitalization. 25
Among the key findings of this study, it was observed that patients in the long-stay group were significantly older, had a higher prevalence of comorbidities, particularly CVD. This observation aligns closely with previous literature, emphasizing the impact of age and underlying health conditions on postoperative recovery.26,27 It was reported that prolonged stays in the cardiac intensive care unit were often attributed to cardiac decompensation, heart failure, and the complexity of surgical intervention.28,29 These findings reinforce the association between cardiovascular instability and extended hospitalization.
Moreover, the relationship between older age and prolonged hospitalization has been further supported by research highlighting the role of cognitive function, frailty, and individualized perioperative strategies in elderly surgical patients.30–33 Aging patients are more susceptible to complications such as postoperative cognitive dysfunction and delirium, both of which may not only extend hospital stays but also contribute to long-term decline in overall quality of life. 34 In this context, the current findings underscore the broader implications of age-related physiological vulnerability in determining recovery.
Nevertheless, our study has some limitations. The retrospective design limits our ability to control over residual confounding variables, the timing of blood sampling and consistency of laboratory procedures and protocols. Employing a prospective study design in future research would allow for greater control over confounding variables and the timing of sample collection, ensuring more standardized and reliable data. Additionally, most surgical procedures analyzed in the current study were laparoscopic cholecystectomy, which may limit the generalizability of our findings to all PBS. Therefore, the results should be interpreted with caution in the context of PBS. Lastly, this was a single-center study, and a sample size calculation was not performed; therefore, the results may be limited by reduced statistical power and may not be fully generalizable to other populations or settings. Conducting a multi-center study with a larger sample size would enhance the external validity of the results and provide more robust and generalizable data.
Nevertheless, one of the key strengths of this study lies in its focus on multiple blood-derived immune-inflammatory markers, which are emerging as cost effective and easily measurable tools in predicting postoperative outcomes. The analytical framework was comprehensive, incorporating a wide range of perioperative clinical, laboratory, and inflammatory parameters, enabling a multifactorial evaluation of factors associated with prolonged hospital stay. Additionally, our investigation employed stepwise multiple logistic regression, which allowed for a systematic evaluation of diverse perioperative variables and facilitating robust identification of independent predictors.
Conclusion
This study highlights the potential role of postoperative SII in predicting delayed recovery in patients after PBS. The key findings of this study highlight the potential for developing predictive tools that integrate key perioperative parameters to identify patients at risk of prolonged hospital stays after surgery. These insights may allow for the integration of a risk stratification model or scoring system into clinical practice to guide personalized care plans. For instance, high-risk patients could benefit from enhanced perioperative monitoring, targeted therapies, and early rehabilitation protocols to mitigate postoperative complications and delayed recovery.
Footnotes
Author contributions
Mohmad H. Alsabani: Conceptualization; Formal analysis; Methodology; Project administration; Supervision; Writing – original draft; Writing – review & editing. Judy S. Alkhonain: Data curation; Visualization; Writing – original draft. Mohammed S. Aldosari: Data curation; Visualization; Writing – original draft. Shaimaa F. Alqawba: Data curation; Investigation; Writing – original draft. Aljawharah K. Almughaylith: Data curation; Investigation; Writing – original draft. Ohood H. Hakami: Data curation; Investigation; Writing – original draft. Lama A. Magrashi: Data curation; Investigation; Writing – original draft. Danah A. Alyami: Data curation; Investigation; Writing – original draft. Abdulaziz S. Alorafah: Writing – original draft; Writing – review & editing. Faraj K. Alenezi: Formal analysis; Validation; Visualization; Writing – review & editing. Lafi H. Olayan: Visualization; Writing – original draft; Writing – review & editing. Mohammed K. Al Harbi: Writing – original draft; Writing – review & editing.
Ethical considerations
This study was approved by the Institutional Review Board of King Abdullah International Medical Research Center, approval number (IRB/0715/24), in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines.
Consent to Participate
Written informed consent was waived by the Institutional Review Board of King Abdullah International Medical Research Center.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
