Abstract
Objectives:
Preeclampsia (PE) is a major contributor to maternal and foetal morbidity and mortality. Early identification of women at risk is crucial for timely intervention, yet a cost-effective and reliable screening tool remains unavailable. This study aimed to evaluate the role of platelet-related parameters—platelet count (PC), mean platelet volume (MPV), plateletcrit (PCT), platelet distribution width (PDW), neutrophil-to-lymphocyte ratio (NLR), and platelet-to-lymphocyte ratio (PLR)—in predicting the development and severity of PE.
Methods:
This prospective observational cohort study was conducted at AIIMS Patna between January and November 2021. Pregnant women enrolled before 14 weeks of gestation were followed throughout pregnancy. Platelet parameters were measured at three gestational intervals: 10 to 14 weeks, 24 to 28 weeks, and 32 to 36 weeks. Data were analysed using SPSS v22, employing chi-square tests, t-tests, and logistic regression.
Results:
Among 728 participants, 9.3% developed PE. Risk was significantly higher in primigravida women (p = 0.040), twin pregnancies (p = 0.027), and those with low educational status (p = 0.006). Significant differences were observed in PCT (p = 0.000) and NLR (p = 0.009) at 10 to 14 weeks, PCT (p = 0.034) at mid-trimester, and MPV (p = 0.005) at 32 to 36 weeks. Although MPV showed a consistent association with PE across trimesters, regression models did not achieve overall statistical significance.
Conclusion:
Platelet indices, particularly PCT and NLR, demonstrated a significant association with PE in early pregnancy, while MPV was significantly elevated in late pregnancy. These findings suggest potential utility in risk stratification, though further studies are required to validate their predictive value before routine use in early screening.
Introduction
Preeclampsia (PE) is a pregnancy-specific hypertensive disorder that typically arises after 20 weeks of gestation and is characterized by elevated blood pressure and proteinuria or signs of systemic organ dysfunction.1 –4 It remains one of the leading causes of maternal and perinatal morbidity and mortality globally, with an estimated prevalence ranging from 2% to 8% of all pregnancies.1 –5 In India, the reported incidence is approximately 4.8% to 7.5%. 2
The pathophysiology of PE is multifactorial and not yet fully understood. 4 It is thought to involve abnormal placental implantation, systemic endothelial dysfunction, immune dysregulation, and an exaggerated inflammatory response.1,2 A two-stage model is commonly proposed, with the first stage involving poor trophoblast invasion and spiral artery remodelling, leading to placental ischemia. The second stage is characterized by the release of antiangiogenic and inflammatory factors into the maternal circulation, leading to systemic endothelial damage and clinical manifestations of PE.2 –5
Recent research has focused on identifying reliable and cost-effective biomarkers for the early prediction of PE, allowing for timely intervention. Among these, platelet-related parameters have gained interest due to their involvement in coagulation, inflammation, and vascular function—all of which are altered in PE.2,3,6,7 Parameters such as mean platelet volume (MPV), platelet distribution width (PDW), platelet count (PC), and plateletcrit (PCT), along with inflammatory ratios like the neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), are readily obtainable from routine complete blood count (CBC) tests. 3 Studies have shown conflicting results regarding the predictive value of these markers. While some report elevated MPV and PDW in preeclamptic women, others have not found significant differences.2,3 Likewise, the utility of NLR and PLR as early indicators of systemic inflammation in PE remains a topic of debate. 1
Given the inconsistent evidence and lack of a definitive early screening tool for PE, this study aims to assess the role of platelet indices (PC, MPV, PCT, and PDW) and inflammatory ratios (NLR and PLR) in predicting the development and severity of PE in a prospective cohort of pregnant women. The objective is to evaluate whether these haematological parameters, measured in early to late pregnancy, can serve as simple, accessible, and effective predictors of PE in routine clinical practice.
Materials and methods
Study design and setting
This was a prospective observational cohort study conducted in the Department of Obstetrics and Gynaecology at All India Institute of Medical Sciences (AIIMS), Patna. The study period spanned from January 2021 to November 2021.
Participants
Pregnant women attending the antenatal clinic at ⩽14 weeks of gestation were screened for eligibility. Individuals who provided written informed consent and agreed to participate in regular follow-up visits were included.
Written and signed informed consent was obtained from all adult participants. For minor participants (<18 years), consent was obtained from legally authorized representatives along with participant assent where possible. For illiterate participants, the consent form was read aloud in the local language, and witnessed thumb impressions were obtained. This study was conducted in accordance with the principles of the Declaration of Helsinki (1975, revised 2024). The reporting of this study conforms to STROBE guidelines. 8
Inclusion Criteria:
Singleton or twin pregnancies
Gestational age ⩽14 weeks at enrolment
Willingness to comply with study protocol and follow-up
Exclusion Criteria: Women were excluded if they had:
Chronic hypertension, diabetes mellitus, nephropathy, or chronic kidney/liver disease
Active infections, autoimmune or haematologic disorders
Platelet count <1.5 lakh/μL at enrolment
History of smoking, alcohol use, or overt hypothyroidism
Sample size
Using the rule of 10 events per predictor variable for logistic regression and assuming a PE incidence of 9.3%, a minimum of 645 participants were required. Accounting for an anticipated 10–15% dropout rate, the target sample size was set at 750 to 800.
Sampling technique
A non-probability consecutive sampling method was used until the desired sample size was achieved.
Study procedure
The study was approved by the Institutional Ethics Committee of AIIMS Patna (Ref. No. IEC/PGTh/July19/39; dated 9 June 2020). Following ethical approval, eligible participants were enrolled after obtaining informed consent. Baseline demographic and clinical data were collected. Participants were followed throughout their pregnancy, with regular visits scheduled according to institutional protocol.
Blood sample collection and platelet parameter analysis
Venous blood (10 mL) was drawn at three gestational intervals:
First trimester: 10 to 14 weeks
Second trimester: 24 to 28 weeks
Third trimester: 32 to 36 weeks
Samples were collected in K2EDTA vacutainers and analysed within 2 h to minimize EDTA-induced changes in platelet morphology. Platelet indices—including PC, MPV, PDW, and PCT—were measured using the Sysmex XN-1000 automated haematology analyser. NLR and PLR were calculated manually from complete blood count data. Quality control for platelet indices was ensured by daily internal calibration of the Sysmex XN-1000 analyser and participation in external quality assurance programs, as per laboratory protocol, to maintain accuracy and precision of results.
Outcome measures
The primary outcome was the development of PE diagnosed according to the International Society for the Study of Hypertension in Pregnancy criteria:
Blood pressure ⩾140/90 mmHg on two occasions ⩾4 h apart after 20 weeks of gestation
Proteinuria ⩾300 mg/24 h or protein/creatinine ratio ⩾0.3 mg/dL
In the absence of proteinuria, the presence of hypertension with thrombocytopenia, renal insufficiency, hepatic dysfunction, pulmonary oedema, or neurological symptoms
Data management and statistical analysis
All patient data were anonymized and de-identified prior to analysis. Data were recorded in Google Sheets and analysed using SPSS version 22. Data were cleaned prior to analysis by excluding biologically implausible values and incomplete records. Continuous variables were expressed as mean ± standard deviation (SD) and compared using independent t-tests. Categorical variables were analysed using chi-square tests. Binary logistic regression was used to assess the predictive value of platelet parameters for the development and severity of PE. A p-value < 0.05 was considered statistically significant.
Results
Study Population: A total of 850 pregnant women were screened, and 776 met the inclusion criteria and provided consent to participate. After accounting for 48 losses to follow-up, the final study cohort included 728 women. Of these, 68 women (9.3%) developed PE, while 660 remained normotensive.
Of the 728 women followed, complete serial platelet data were available for 183 participants and included in comparative analyses presented in Tables 1–5. Participants with missing data at any time point were excluded from this subset analysis to maintain statistical robustness.
Socio-demographic characteristics of pregnant women among normotensive and women who developed preeclampsia (n = 183).
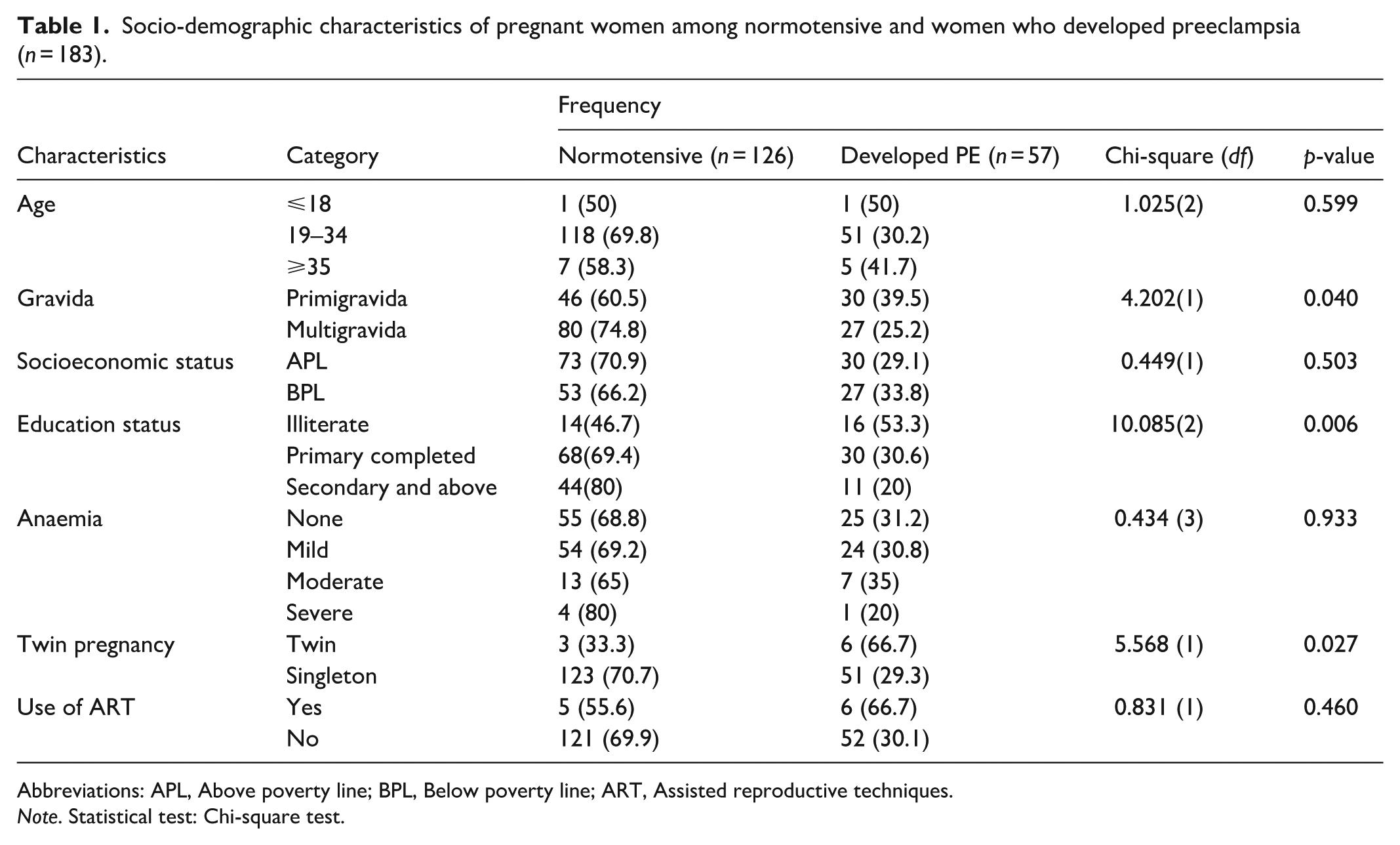
Abbreviations: APL, Above poverty line; BPL, Below poverty line; ART, Assisted reproductive techniques.
Note. Statistical test: Chi-square test.
Comparison of PC, MPV, PDW, PCT, NLR, and PLR across groups (n = 183).
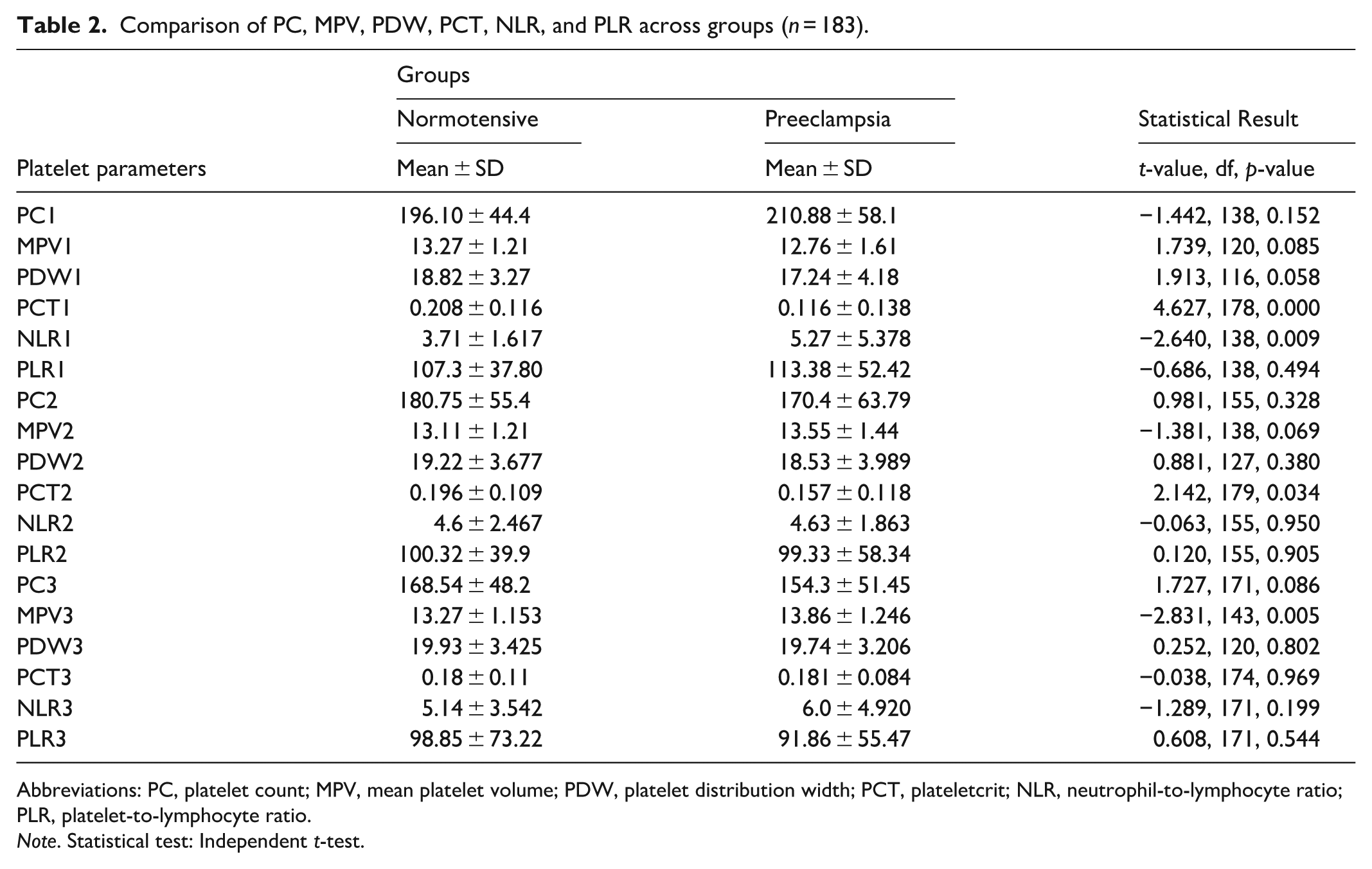
Abbreviations: PC, platelet count; MPV, mean platelet volume; PDW, platelet distribution width; PCT, plateletcrit; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Note. Statistical test: Independent t-test.
Binary logistic regression analysis summary of various predictor variables for the development of preeclampsia at (10–14 week POG) follow-up (n = 183).

Abbreviations: PC, platelet count; MPV, mean platelet volume; PDW, platelet distribution width; PCT, plateletcrit; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Binary logistic regression analysis summary of various predictor variables for the development of preeclampsia at (24–28 week POG) follow-up (n = 183).

Abbreviations: PC, platelet count; MPV, mean platelet volume; PDW, platelet distribution width; PCT, plateletcrit; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Binary logistic regression analysis summary of various predictor variables for the development of preeclampsia at (32–36 week POG) follow-up (n = 183).
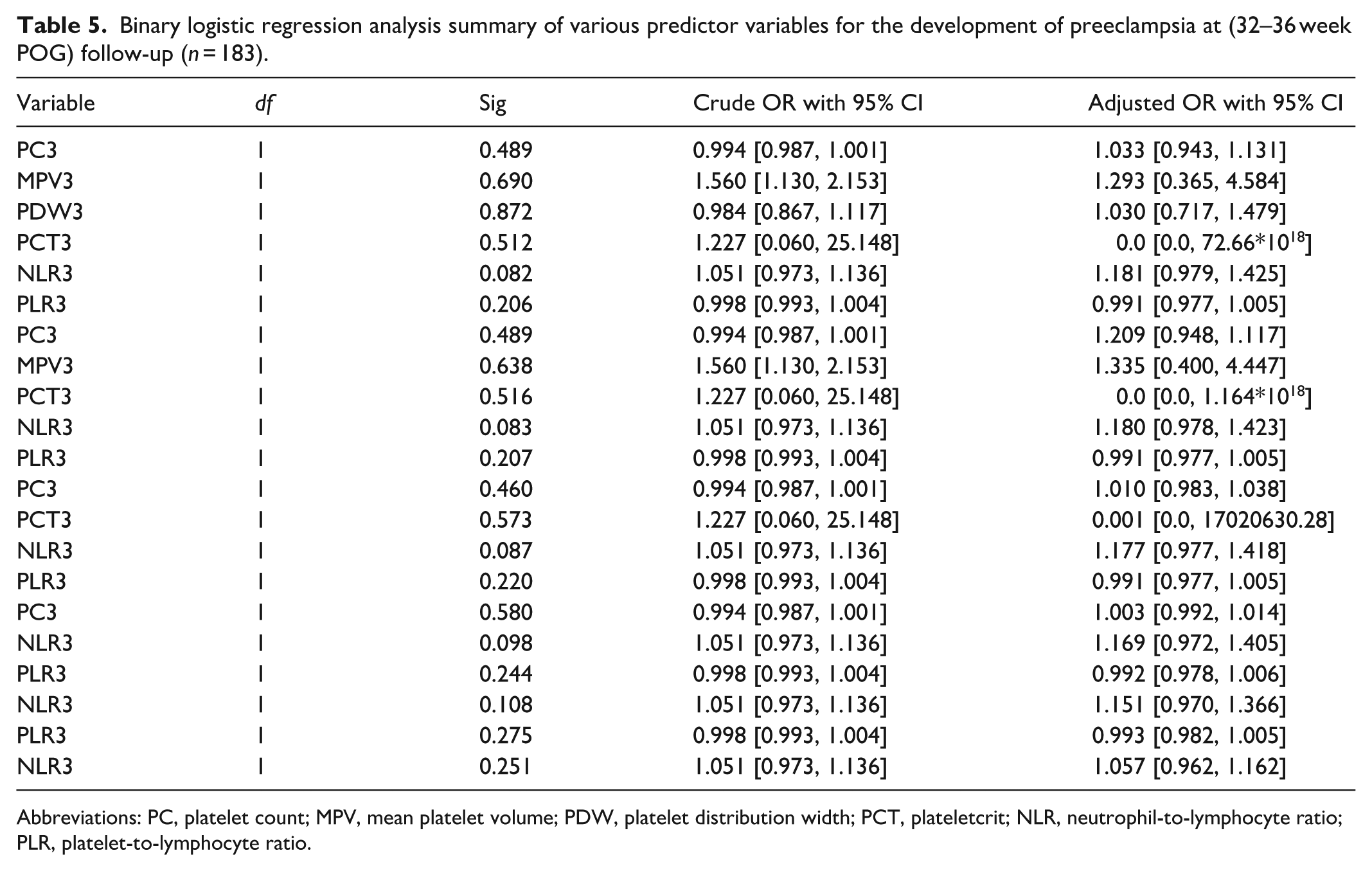
Abbreviations: PC, platelet count; MPV, mean platelet volume; PDW, platelet distribution width; PCT, plateletcrit; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Sociodemographic and Clinical Characteristics: Among the study participants, the majority (92.3%) were aged 19 to 34 years. Primigravidae constituted 41.5% of the cohort. Most women (53.3%) had completed only primary education, and 43.7% belonged to households below the poverty line (BPL), as shown in Table 1.
Chi-square analysis revealed a statistically significant association between PE and primigravida status (p = 0.040), twin pregnancies (p = 0.027), and low educational status (p = 0.006). No significant associations were found with age, anaemia severity, socioeconomic status, or use of assisted reproductive technologies (Table 1).
Platelet Parameters Across Trimesters: Independent t-tests were conducted to compare platelet-related parameters between normotensive and preeclamptic women at three gestational intervals (Table 2):
10 to 14 weeks: PCT was significantly lower (p = 0.000) and NLR significantly higher (p = 0.009) in women who later developed PE.
24 to 28 weeks: PCT was significantly lower in the PE group (p = 0.034).
32 to 36 weeks: MPV was significantly higher in the PE group (p = 0.005).
No significant differences were observed for PC, PDW, or PLR at any time point.
Predictive Modelling Using Logistic Regression: Binary logistic regression models were developed at each gestational time point to evaluate the predictive potential of platelet indices (Tables 3–5):
10 to 14 weeks: Although MPV showed a significant association with PE (adjusted OR: 0.658, p = 0.022), the overall model was not statistically significant (Hosmer–Lemeshow p = 0.203) as shown in Table 3.
24 to 28 weeks: No platelet parameter significantly predicted PE. The model had poor explanatory power (Nagelkerke R² = 0.9%, p = 0.761) as shown in Table 4.
32 to 36 weeks: MPV again showed a significant association (crude OR: 1.56), but the adjusted model did not reach statistical significance (Hosmer–Lemeshow p = 0.116) as shown in Table 5.
Severity of PE: A subgroup analysis comparing women with non-severe (n = 40) and severe PE (n = 17) showed no statistically significant differences in any of the platelet parameters across all three trimesters (Table 6).
Comparison of PC1, MPV1, PDW1, PCT1, NLR1, and PLR1 across Preeclampsia sub-groups (between 10- and 14-week POG) (n = 57).

Abbreviations: PC, platelet count; MPV, mean platelet volume; PDW, platelet distribution width; PCT, plateletcrit; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Note. Statistical test: Independent t-test
Receiver operating characteristic curve analysis was not performed in the present study due to the limited number of PE cases and incomplete data for all time-points. Future studies with larger cohorts should assess sensitivity, specificity, and optimal cut-off values.
Figure 1 depicts a comparison of platelet indices across trimesters between normotensive and preeclamptic women.
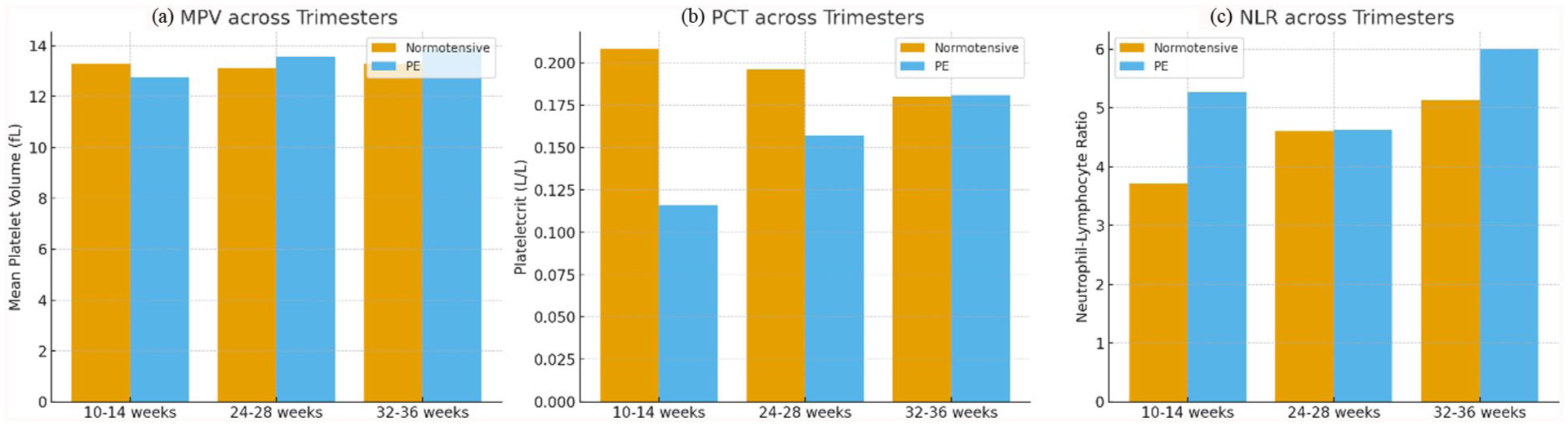
Comparison of platelet indices across trimesters between normotensive and preeclamptic women.
Discussion
This prospective cohort study aimed to assess whether platelet-related parameters could predict the development and severity of PE. Among the 728 women followed from early pregnancy to delivery, 68 (9.3%) developed PE. Our findings suggest that specific platelet indices—particularly PCT, MPV, and NLR—showed statistically significant differences between normotensive and preeclamptic women during specific trimesters. However, the predictive models based on these indices did not demonstrate consistent or statistically robust predictive capability.
Sociodemographic risk factors
We observed a significantly higher incidence of PE among primigravidae, women with twin pregnancies, and those with low educational status. These findings align with previous literature, which consistently identifies primigravidity and multiple gestation as well-established risk factors for PE. Education level, often a proxy for health literacy and access to care, also correlated with increased PE risk, as reported in other studies, including Silva et al. 9
Platelet parameters and development of PE
At 10 to 14 weeks, a significantly lower PCT and higher NLR were observed in women who eventually developed PE, consistent with findings from previous reported studies.10,11 Similarly, PCT remained lower in the mid-trimester (24–28 weeks), and MPV was higher in the third trimester (32–36 weeks) among the PE group. These trends suggest that platelet consumption and activation may begin early in the course of the disease. Several studies, including those by Thakkar et al. 2 and Walle et al., 3 have reported similar alterations in MPV and PCT values in PE, supporting their potential as early biomarkers. Although MPV physiologically tends to increase with advancing gestation, the magnitude of rise observed in our PE group was significantly higher than that in normotensive women, suggesting an association with the disease process rather than a normal gestational change.
However, while univariate comparisons showed statistically significant differences, multivariate logistic regression failed to produce models with strong predictive validity. For example, although MPV at 10 to 14 weeks was significantly associated with later PE development, the regression model as a whole was not statistically significant. Similar findings were noted for the mid- and late-trimester models.
Predictive utility and limitations of platelet parameters
Although MPV showed consistent trends across gestational windows and was also highlighted in other studies as a promising marker, its performance, either in isolation or in combination with other indices, was insufficient to justify its use as a standalone screening tool in this cohort. Our models explained very low percentages of outcome variance (Nagelkerke R² < 2%), and classification accuracy was inadequate to warrant clinical application.
Additionally, we found no statistically significant differences in platelet parameters between women with non-severe and severe PE, suggesting that while platelet changes may correlate with PE onset, they do not reliably differentiate disease severity.
The observed decrease in PCT and increase in NLR in early pregnancy are consistent with the pathophysiological model of endothelial dysfunction and subclinical inflammation preceding clinical onset of PE.10,11 Elevated MPV in late pregnancy may reflect increased platelet activation secondary to systemic inflammation and endothelial injury, as reported in Thakkar et al. 2 and Walle et al. 3 These findings collectively support the concept that platelet indices may mirror the evolving thrombo-inflammatory milieu of PE.
Our findings are consistent with the broader body of literature, indicating that platelet parameters undergo measurable changes in PE. Walle et al. 3 and Dionisio et al. 12 reported increased MPV and PDW with decreased PCs in PE cases, while others, such as Zeng et al. 13 and Cui et al., 1 have emphasized the role of NLR and PLR as inflammatory markers with diagnostic potential. However, variability in study design, population, and statistical modelling often leads to inconsistent conclusions about their predictive value.
Strengths and limitations
A significant strength of this study is its prospective design and serial assessment of platelet parameters across all three trimesters. Unlike many retrospective or cross-sectional studies, we tracked changes longitudinally, enhancing temporal association with PE onset.
However, several limitations must be acknowledged. First, the study was conducted in a tertiary care setting, which may have introduced referral bias and limited its generalizability. Second, COVID-19-related service disruptions led to missing follow-up data and potential pre-analytical errors in sample processing. Third, despite aiming for a sufficiently powered sample, the number of PE events may still have been inadequate to generate robust multivariable models.
Conclusion
This study highlights that specific platelet-related parameters—specifically PCT, MPV, and NLR—exhibit statistically significant differences between normotensive and preeclamptic pregnant women during various trimesters. MPV, in particular, demonstrated a consistent association with the development of PE. MPV was significantly elevated only in the third trimester and did not show an association in early pregnancy; hence, it may serve as an adjunct marker for ongoing disease rather than an early predictor. However, despite these associations, the predictive models constructed using these parameters lacked sufficient accuracy and statistical strength to be clinically reliable as standalone screening tools.
The incidence of PE was found to be significantly higher among primigravida, women with twin pregnancies, and those with lower educational status, underscoring the importance of demographic and social factors in risk stratification.
While platelet indices show promise as inexpensive and readily available markers, their integration into early predictive algorithms requires further validation through large-scale, multicentre studies. Future research should focus on combining these haematological markers with other clinical, biochemical, and imaging parameters to develop a more accurate and practical screening tool for early detection of PE.
Footnotes
Acknowledgements
None
Ethical considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Institutional Ethics Committee of AIIMS Patna (Ref. No. IEC/ PGTh / July19/39; dated 9 June 2020).
Consent to participate
Written informed consent was obtained from all adult participants. For minor participants (<18 years), consent was obtained from legally authorized representatives along with participant assent where possible. For illiterate participants, the consent form was read aloud in the local language and witnessed thumb impressions were obtained.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Shruti Singh, Mukta Agarwal, Shamshad Ahmad and Sudwita Sinha. The first draft of the manuscript was written by Shruti Singh and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Trial registration number
Not applicable.
