Abstract
Objective:
To identify the association between sex hormones and Sjogren’s disease.
Methods:
A case-control study investigating the relationship between sex hormones and Sjogren’s disease was conducted at Nanjing Drum Tower Hospital from January 2018 to January 2024. Two-sample Mendelian randomization was then performed to identify the causal association by using the public genome-wide association study data from the UK Biobank and the FinnGen consortium.
Results:
In the case-control study, a total of 93 cases diagnosed with Sjogren’s disease were compared to 90 Sjogren’s disease-like non-Sjogren’s disease controls from a population of naturally postmenopausal women. An association was observed between E2 (aOR = 0.984; 95% CI: 0.971–0.997; p = 0.018), hypo-estradiol (aOR = 2.195; 95% CI: 1.156–4.165; p = 0.016), and Sjogren’s disease. Mendelian randomization analysis indicated that higher odds of Sjogren’s disease were associated with decreased E2 in females, as estimated by the inverse-variance weighted (OR = 0.954; 95% CI: 0.917–0.992; p = 0.019).
Conclusion:
Our study demonstrates that Sjogren’s disease leads to reduced E2. This indicates that low E2 is a consequence of the disease rather than a causative factor.
Keywords
Introduction
Sjogren’s disease (SjD) is a systemic autoimmune disorder characterized by lymphocyte infiltration in exocrine glands.1,2 Although the exact pathogenesis of SjD remains unknown, epidemiological features of SjD, such as a higher prevalence in women, onset usually around menopause, and often triggered by significant stressful events, suggest an important role of hormones in the development of SjD.3–6
Previous studies have shown that reduced production of ovarian estrogens (E) and adrenal pro-hormone dehydroepiandrosterone (DHEA) is closely related to the onset of SjD, especially in postmenopausal women.6–10 However, the findings of different studies are controversial, and the specific association between E2 and SjD remains unclear. Based on the previous findings, we conducted a matched case-control study and hypothesized that there is an association between low E2 and an increased risk of SjD. To validate the results, the Mendelian randomization (MR) method was further used to analyze the exact causal relationship between sex hormones (testosterone, E2, and sex hormone-binding globulin (SHBG)) and SjD.
Materials and methods
A matched case-control study was performed. The Ethics Committee at the Nanjing Drum Tower Hospital approved this study, for which all patients provided written informed consent to participate. Our study was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 11 The checklist is presented in Supplemental Table S1.
Selection of cases and controls
A total of 189 cases were randomly selected from Nanjing Drum Tower Hospital between January 2018 and January 2024. Of these, 6 cases with missing data on key variables were excluded, leaving 183 valid samples for subsequent analysis. All cases consisted of postmenopausal women. The sample size was calculated using G*Power software with an effect size (Cohen’s d) of 0.8, an alpha level of 0.05, and a power (1 − β) of 0.95. With a 1:1 allocation between SjD patients and controls, the minimum required sample size was 44 participants per group. All patients were assessed by two experienced rheumatologists. The case group (n = 93) comprised newly diagnosed SjD patients. The control group comprised sex- and age-matched individuals exhibiting similar or suspected Sjögren-like symptoms, such as dry mouth and eyes, fatigue, joint pain, and weight loss, but who were excluded on screening for SjD and other autoimmune diseases. In both groups, blood samples were collected during the patients’ first visit to our department, before any treatment was administered. The timing of menopause in all cases fell within the natural age range for menopause based on a large population-based epidemiological survey. 12 The classification of SjD was based on the 2002 American-European Consensus Group criteria 13 or 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome, 14 with all cases categorized under ICD-10 M35.0. Individuals with other autoimmune disorders, premature ovarian failure, estrogen replacement therapy, polycystic ovarian syndrome, and hysterectomy without bilateral oophorectomy were all excluded from the controls/cases.
Measurement of exposure and outcomes
For the exposure, laboratory values including neutrophilic granulocyte, hemoglobin, platelet (PLT), complement3 (C3) and complement4 (C4), immunoglobulin G, fasting blood glucose, thyroxine levels, estradiol (E2), testosterone, SHBG, follicle-stimulating hormone, luteinizing hormone, and prolactin were all recorded. The cut-off value of E2 <89.76 pmol/L was classified as hypo-estradiol (hypoE2) by Receiver Operating Characteristic Curve (ROC) analysis. Age, menopausal age, body mass index, smoking, and tea/coffee consumption were considered possible confounding factors, along with a medical history of hypertension, diabetes, and thyroid disease. All samples for sex hormones were obtained from the patients’ early morning fasting blood. The main clinical manifestations of the patients in the case group were retrieved from the electronic medical record system. All of the laboratory results were tested by the Clinical Laboratory Center of Nanjing Drum Tower Hospital, Affiliated to Medical College of Nanjing University, according to the instructions of the corresponding kit. Anti-nuclear antibodies were determined using immunofluorescence. Antibodies against SSA (Ro/SSA) and SSB (La/SSB) were assessed by immunoblotting.
MR analyses
Supplemental Figure S1 presents a brief summary of the bidirectional MR design that investigated the relationship between sex hormones and SjD. Two MR analyses were conducted using summary statistics from a genome-wide association study (GWAS) to explore this relationship. In the forward MR analyses, sex hormones were considered as the exposure and SjD as the outcome, whereas in the reverse MR analyses, SjD was considered as the exposure and sex hormones as the outcome. For the exposures (sex hormones), sex-specific GWAS summary statistics were used, whereas only mixed-sex GWAS data were available for SjD. Therefore, the forward MR estimates represent the effects of genetically predicted male- or female-specific hormone levels on the risk of SS in the general population, whereas the reverse MR estimates reflect the potential influence of genetic susceptibility to SS on circulating sex hormone levels in males and females. MR analyses use publicly available summary statistics and do not require ethical approval and written informed consent. This part followed the Strengthening the Reporting of Observational Studies in Epidemiology–Mendelian Randomization reporting guidelines (Supplemental Table S2), available on the JAMA Network at https://jamanetwork.com/journals/jama/fullarticle/2785494). 15
Instrumental variable selection for MR analyses
For each exposure factor, SNPs were filtered based on the three primary assumptions of MR. Initially, SNPs were included if they reached genome-wide significance (p < 5 × 10−8). Due to the insufficient number of SNPs for estradiol analysis, more lenient thresholds were opted for (p < 5 × 10−7). All the F-values are greater than 10 (Supplemental Table S3). Subsequently, only variants with the lowest p-values were retained as independent instruments, considering linkage disequilibrium with an r2 threshold (r2 > 0.1 in the European 1000 Genome Reference Panel). Finally, we calculated F-statistics to assess the strength of the instrumental variables. We recommend a threshold of F-statistics >10 for MR analysis.
Sources of data and selection of instrumental variables for sex hormones
Sex hormones, including testosterone, E2, and SHBG, were obtained from the summary statistics. The summary statistics for female (N = 230,454) and male (N = 194,553) testosterone were obtained from a previous extensive GWAS that used genotype and phenotype data from the UK Biobank. 12 Estradiol summary statistics were sourced from another GWAS that used data from the UK Biobank, 16 encompassing 163,985 samples in women and 147,690 in men. Ruth et al. provided data on SHBG, which involved 214,989 samples in women and 185,221 in men. 17 Supplemental Table S4 contains comprehensive details of the summary data for all the GWAS.
Data sources and selection of instrumental variables for SjD
To minimize potential sample overlap between exposure and outcome datasets, we selected the Finnish SjD GWAS as the outcome source. Although the comprehensive GWAS by Khatri et al. 18 provides high-quality data across multiple European cohorts, it includes UK participants who also contributed to the sex hormone GWASs used in this study. Summary-level statistics for SjD were obtained from the FinnGen consortium in 2023, which included 392,423 samples (2495 cases and 389,928 controls). The complete GWAS dataset for SjD is publicly available through the FinnGen research project (https://www.finngen.fi/en), which aims to identify novel therapeutic targets by assessing genotype-phenotype correlations. The diagnosis of SjD, unspecified, was established using International Classification of Diseases codes, specifically ICD-10 M35.0.
Statistical analysis
In the case-control study, normal data were presented as the means ± standard deviations, skewed data were presented as the medians (interquartile ranges), and categorical data were presented as the absolute values. The independent-samples T test and the Mann-Whitney U test were used to compare the continuous parameters between groups, while the chi-square test was used to compare the categorical data. *p-values were adjusted for multiple comparisons using the false discovery rate method via the Benjamini–Hochberg. The logistic regression analysis was conducted to predict the relationship between sex hormones and the risk of SjD. p < 0.05 was considered significant in the two-tailed tests. Data analysis was conducted by using SPSS software (Version 26.0). In the MR study, the inverse-variance weighted (IVW) method was used to explore potential bidirectional causal links between sex hormones and SjD, with additional checks using MR-Egger, weighted median, and MR-pleiotropy residual sum, and outlier (MR-PRESSO) to address potential biases like invalid instruments and pleiotropy. A p-value exceeding 0.05 indicates the absence of horizontal pleiotropy. 19 Heterogeneity was assessed using Cochrane’s Q test. We applied the IVW random effects method 20 to estimate the main effect. MR-PRESSO identified outliers influencing heterogeneity, 21 which were subsequently excluded. A meta-analysis synthesized MR results across sexes for sex-stratified hormone and SjD using R software (Version 4.30, R Foundation for Statistical Computing, Vienna, Austria), with the two-sample MR and Meta packages utilized for the respective analyses.
Results
The relationship between sex hormones and SjD
The characteristics of the 93 patients included in the case group are shown in Table 1.
Characteristics of the patients in the case group.

Dichotomous variables date are presented as n (%), normal variables are presented as mean ± standard deviation, and non-normal variables are presented as median (interquartile). Normal range: C3 (0.80–1.60) g/L; C4 (0.20–0.40) g/L; IgG (8.00–16.00) g/L. NE: neutrophilic granulocyte; HB: hemoglobin; PLT: platelet; C3: Complement 3; C4: Complement 4; IgG: immunoglobulin G.
The distributions of all the study variables in the cases and controls are presented in Table 2. SjD cases exhibited significantly lower mean total E2 compared to controls (84.41 pmol/L versus 91.57 pmol/L; *p = 0.048). Similarly, the rate of hypoE2 was significantly higher in the SjD group than in controls (62.37% versus 46.67%; *p = 0.033). The crude odds ratio (cOR) and adjusted odds ratio (aOR) for the associations between SjD and both E2 and hypoE2 are presented in Table 3. For E2, the cOR and aOR were 0.988 (95% CI: 0.975–1.000; p = 0.047) and 0.984 (95% CI: 0.971–0.997; p = 0.018), respectively, indicating a significant inverse association with SjD. For hypoE2, the cOR and aOR were 1.894 (95% CI: 1.050–3.415; p = 0.034) and 2.195 (95% CI: 1.156–4.165; p = 0.016), suggesting a positive relationship with SjD. However, no significant association was found between other hormones and SjD (p > 0.05).
Descriptive statistics of the study variables in controls and cases.
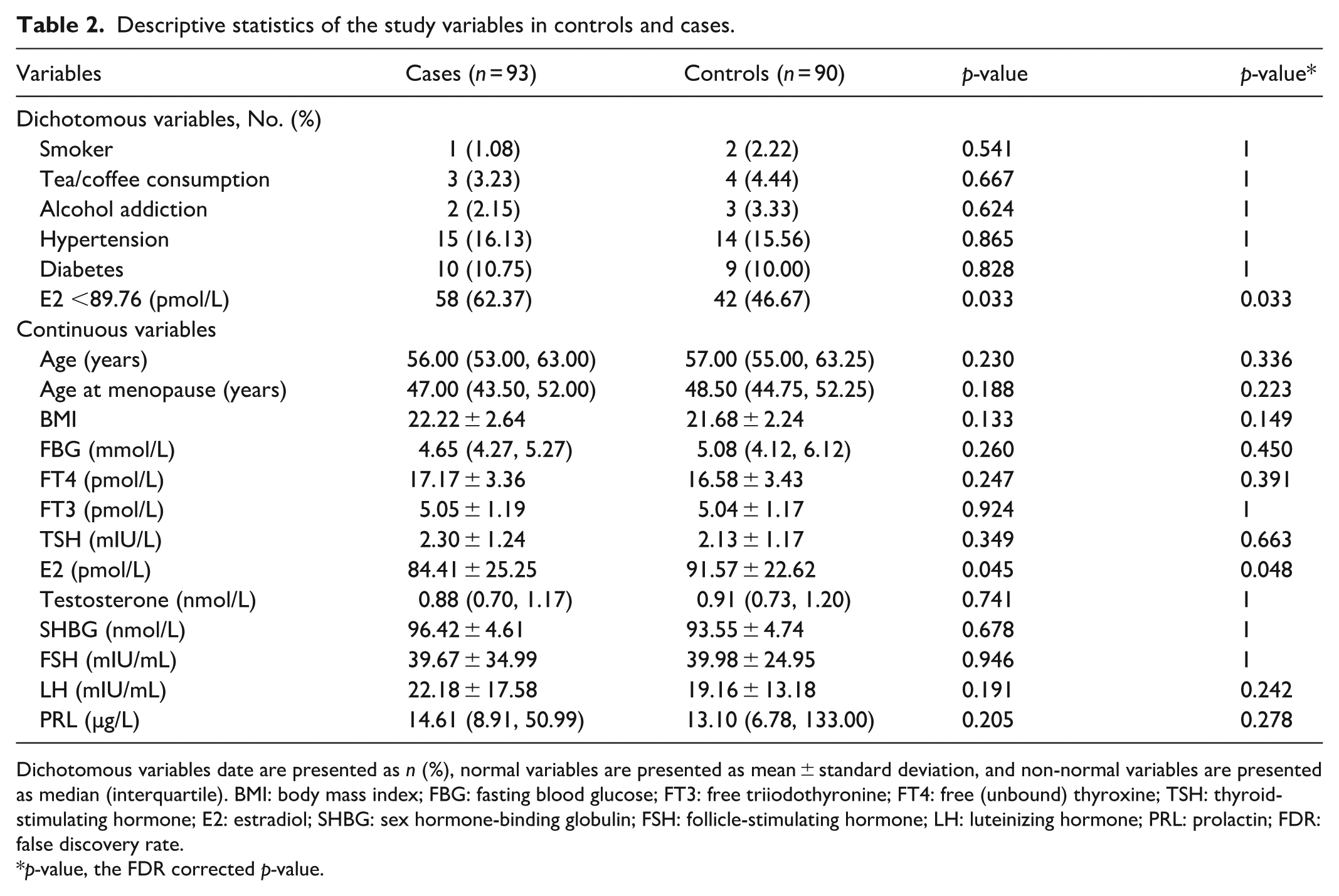
Dichotomous variables date are presented as n (%), normal variables are presented as mean ± standard deviation, and non-normal variables are presented as median (interquartile). BMI: body mass index; FBG: fasting blood glucose; FT3: free triiodothyronine; FT4: free (unbound) thyroxine; TSH: thyroid-stimulating hormone; E2: estradiol; SHBG: sex hormone-binding globulin; FSH: follicle-stimulating hormone; LH: luteinizing hormone; PRL: prolactin; FDR: false discovery rate.
p-value, the FDR corrected p-value.
Crude and adjusted OR values for the association between SjD and each hormone.

OR: odds ratio; SjD: Sjogren’s disease; aOR: adjusted odds ratio; CI: confidence interval; cOR: crude odds ratio; SHBG: sex hormone-binding globulin; FSH: follicle-stimulating hormone; LH: luteinizing hormone; E2: estradiol; PRL: prolactin; TSH: thyroid-stimulating hormone; BMI: body mass index.
The following variables were also included in each model: age, menopausal age, BMI, smoking, tea/coffee addiction, medical history of hypertension and diabetes, and thyroxine levels.
The causal effect of sex hormones on SjD
The impact of each hormone on susceptibility to SjD was examined individually. However, no significant association was observed, as shown in Figure 1. To further validate these findings, scatter plots, the leave-one-out test, funnel plot, and forest plot were employed, offering additional confirmation of the results (see Supplemental Figures S2–S7).

MR estimates for the relationship between genetically instrumented sex hormones and SjD.
The causal influence of SjD on sex hormones
Figure 2 showed that a heightened risk of SjD was associated with decreased E2 in females, as indicated by the IVW method (OR, 0.954; 95% CI: 0.917–0.992; p = 0.019). However, this effect was not observed in males. In relation to testosterone, SjD was identified as a risk factor for testosterone in both sexes (OR, 0.993; 95% CI: 0.987–1.000; p = 0.047). However, no significant difference was observed in the sex-stratified testosterone. Additional confirmation of the results was obtained through the use of scatter plots, the leave-one-out test, funnel plot, and forest plot (Supplemental Figures S8–S13).
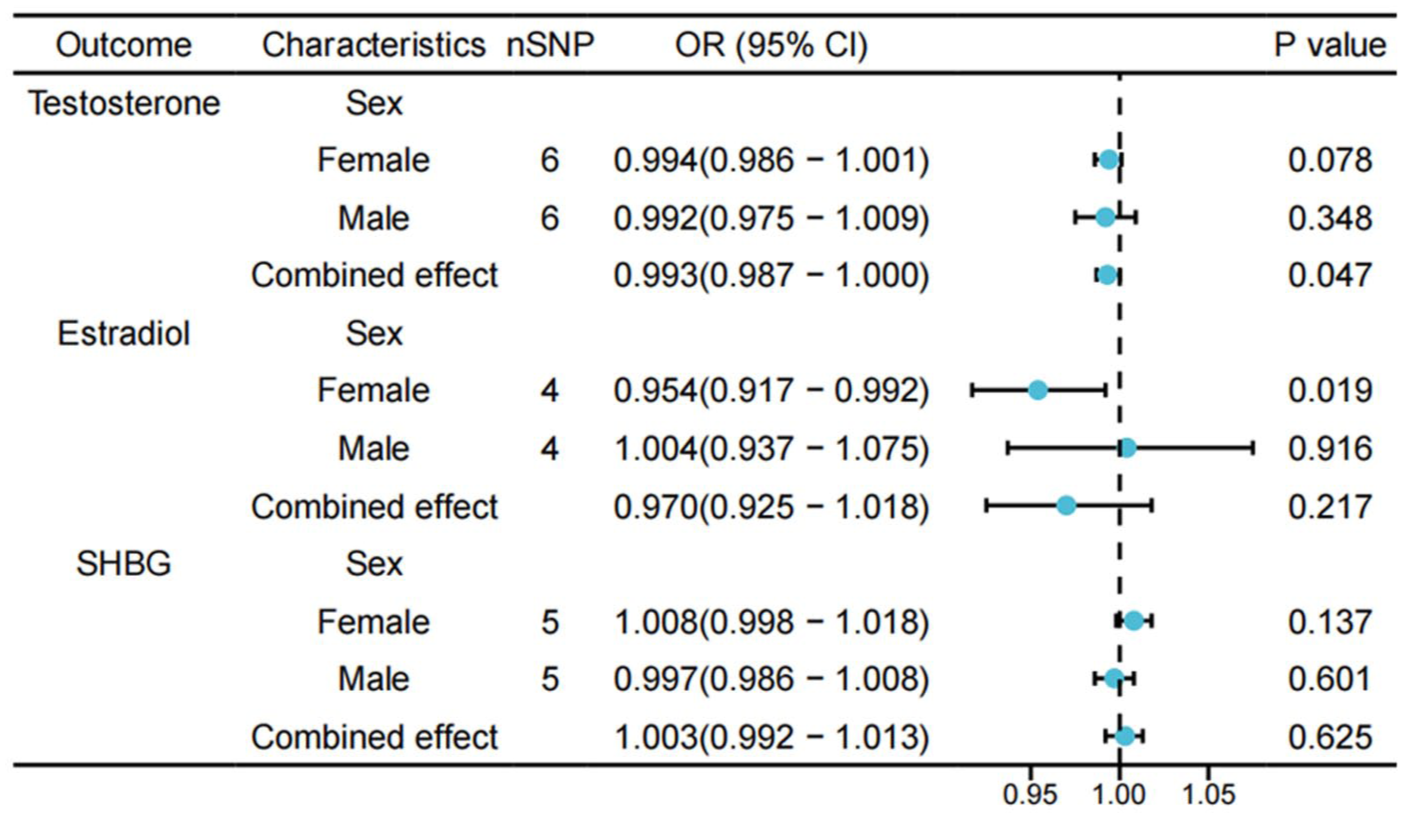
MR estimates for the relationship between genetically instrumented SjD and sex hormones.
Assessment of heterogeneity, pleiotropy, and sensitivity analysis
In addition, we conducted sensitivity analyses to support a causal relationship between sex hormones and SjD. The Cochran’s Q test did not show any detectable heterogeneity of effects across the instrumental variables (Table 4). Furthermore, the F-statistics for all instrumental variables exceeded 10, indicating the absence of weaknesses in the selected instruments. No signs of horizontal pleiotropy were detected as the intercept of MR-Egger did not significantly deviate from zero. Additionally, the MR-PRESSO analysis did not identify any potential instrumental outliers. The leave-one-out results suggest that the causal effect was not solely influenced by a single instrumental variable.
Sensitivity analysis of the causal relationship between sex hormones and Sjogren’s disease.
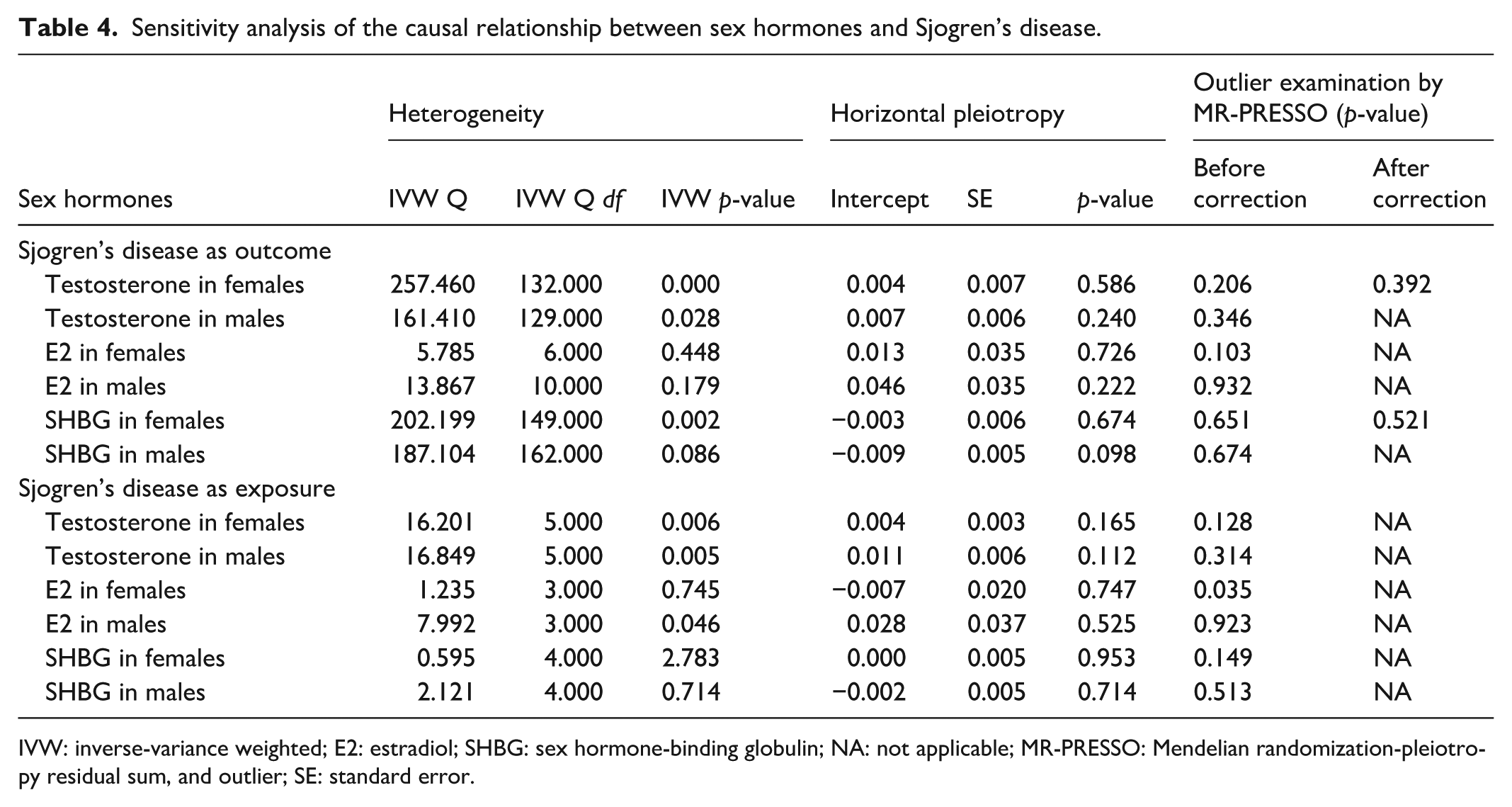
IVW: inverse-variance weighted; E2: estradiol; SHBG: sex hormone-binding globulin; NA: not applicable; MR-PRESSO: Mendelian randomization-pleiotropy residual sum, and outlier; SE: standard error.
Discussion
SjD is an autoimmune disease that primarily affects the exocrine glands, particularly the salivary and lacrimal glands. Follicular cells of the external glands are damaged and destroyed in SjD, resulting in reduced secretion of saliva and tear fluid. 22 In this case-control study, we observed a positive correlation between hypoE2 and SjD in postmenopausal women. We further revealed that higher odds of SjD were associated with decreased E2 in females by MR.
Estrogens exert their influence on specific intracellular estrogen receptor subtypes found within every cell of the immune system,23–25 especially in modulating the development and function of lymphocytes.26,27 They may facilitate protective effects that counteract harmful changes following inflammatory responses. For example, human lymphocyte cultures have shown a reduced CD4+/CD8+ T cell-subset ratio after estrogen treatment. 28 Estrogen can also affect the subsets of B lymphocytes. Animal studies have demonstrated that estrogens can increase the population of bone marrow progenitor B cells by protecting them from apoptosis26,29 and enhancing B cell survival. 26 Estrogens are suggested to have a protective effect on secretory glandular acinar cells by shielding them from apoptosis. Additionally, testosterone can be converted to dihydrotestosterone (DHT) in exocrine glands. DHT exhibits anti-apoptotic properties that protect acinar cells. 10 In menopausal women, the lack of local intracellular DHT may increase susceptibility to SjD when estrogen levels are low. 30 These findings have prompted a strong interest in hormone replacement therapy as a prospective treatment for SjD.31–33
In a nested case-control study (546 SjD cases and 1637 age-matched controls), the modified composite estrogen scores showed no significant association with SjD in adjusted models. 34 Both human and mouse studies indicate that the number of X chromosomes, rather than sex-steroid hormones, confers increased susceptibility to autoimmunity, particularly rheumatic diseases such as SjD. 35 Research findings suggest that the sex-based differences in the immune system can be attributed to inherent composition. Women generally exhibit a higher count of T lymphocytes CD4/CD8, B lymphocytes, and plasma cells, whereas men have a higher proportion of Natural Killer cells, CD14, and CD16 monocytes. 36 Genetic mechanisms linked to sex chromosomes have been identified as possible factors contributing to sex-biased immune responses across different age groups.37,38 Individuals with Klinefelter syndrome (47,XXY) exhibit a susceptibility to lupus similar to that of females (46, XX). 39 Conversely, females with Turner syndrome (45, X0) appear to have a protective effect. 40 In cells with multiple X chromosomes, all but one X chromosome is inactivated, though about 10% of genes escape inactivation. In the TLR7 pathway, both TLR7 and TLR adaptor, interacting with endolysosomal SLC15A4 (TASL), escaping X inactivation. Compared to male immune cells, cells with multiple X chromosomes show increased TLR7 signaling due to higher expression of these genes. X-inactive specific transcript, expressed only in cells with multiple X chromosomes, also enhances TLR7 signaling. These mechanisms may contribute to the sex bias in autoimmune diseases by mediating an X chromosome dose effect. 41
The efficacy of hormone therapy for SjD has varied significantly across clinical trials. For instance, studies in postmenopausal women have yielded conflicting results regarding the efficacy of estrogen in managing SjD symptoms. While some found that estrogen replacement therapy has no significant effect on osmolarity, tear volume, breakup time, or ocular symptoms,42,43 others reported that it could alleviate ocular symptoms.44–46 Notably, one study revealed that 3 months of estrogen replacement therapy led to the development of dry eye in postmenopausal women without SjD symptoms, while symptomatic patients showed no improvement after the same treatment period. 47 Therefore, further observations have been made regarding the interplay between estrogen treatment and the SjD signs and symptoms. In terms of androgens, individuals with SjD exhibit lower serum levels of DHEA, DHT, and DHEA sulfate compared to healthy controls. 6 For example, in a case study, the use of testosterone cream applied to the eyelids was suggested to effectively reduce SjD symptoms and improve the lipid layer breakup time and its thickness to normal levels. 48 Similarly, another case study indicated that systemic androgen and testosterone therapy reduced dry eye symptoms in postmenopausal women. 49 However, one study found no evidence for the efficacy of DHEA in SjD. Due to the lack of evidence for its efficacy, patients with SjD should avoid using unregulated DHEA supplements, as the long-term adverse effects of exposure to this hormone remain unknown. 50 Taken together, our results suggest that early sex hormone replacement therapy for normal menopausal women may not be beneficial in the prevention of SjD.
Our study has several limitations. First, our collection of sex hormones was only once before SjD diagnosis and lack of follow-up, causality still needs to be clarified by designing longitudinal prospective studies or retrospective nested cohorts. Second, the GWAS datasets used in this study were not stratified by age. And the accuracy of our findings may be affected by the age-specific nature of SjD. The impact of the genetic variation may be underestimated. Third, as an exploratory analysis, our MR study did not apply corrections for multiple comparisons, such as the Bonferroni or Benjamini-Hochberg methods. Then, the limited sample size in the case-control cohort, consisting of only 93 SjD cases and 90 controls, may reduce the statistical power of our analysis and limit the generalizability of the findings. Last, the observational study used a Chinese postmenopausal female cohort, while the MR analysis relied on European GWAS summary data. Due to genetic differences across populations, this cross-ancestry approach may impact the validity of the causal inferences.
In summary, our integrated analysis combining a clinical case-control study and MR provides robust evidence supporting a unidirectional relationship between SjD and estradiol levels. Specifically, we found that SjD is associated with reduced E2 levels in postmenopausal women, rather than low E2 acting as a risk factor for the development of SjD. These findings suggest that the hormonal alterations observed in SjD patients may be a consequence of the disease process itself. Clinically, this implies that hormone replacement therapy aimed at elevating estrogen levels may not be an effective strategy for preventing SjD in menopausal women. Further research is needed to elucidate the underlying mechanisms by which SjD influences sex hormone metabolism and to explore targeted therapeutic approaches.
Conclusion
Our study demonstrates that SjD leads to reduced estradiol levels, indicating that low E2 is a consequence rather than a cause of the disease.
Supplemental Material
sj-doc-1-smo-10.1177_20503121251405020 – Supplemental material for The association between Sjogren’s disease and sex hormones: A case-control and Mendelian randomization study
Supplemental material, sj-doc-1-smo-10.1177_20503121251405020 for The association between Sjogren’s disease and sex hormones: A case-control and Mendelian randomization study by Jing Zhang, Qi Sun, Anning Wang, Mengge Pan, Wenwei Wu, Zhi Lin, Qun Chen and Shu Liu in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to extend our appreciation to the UK Biobank and the FinnGen consortium for generously sharing publicly available, summarized data derived from GWAS studies, which proved invaluable for our research.
Ethical considerations
The case-control study conformed to approved guidelines, and all experimental protocols were approved by the Ethics Committee of Nanjing Drum Tower Hospital (No. 2022-529-04). Our study complied with the Declaration of Helsinki, and all subjects provided written informed consent. In the MR study, ethical review and approval were not deemed necessary for this study involving human subjects, in accordance with local laws and institutional regulations. In addition, written informed consent for participation was not required for this study, aligning with national laws and institutional guidelines.
Author contributions
JZ and SL conceptualized and designed the study; JZ and QC conducted the statistical analysis; A-NW, M-GP, ZL, and W-WW collected the clinical specimens; JZ and QS organized the data, drafted the manuscript, and revised it; QC and SL edited the manuscript and supervised the study. All authors contributed to the article and approved its final version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Jiangsu Province (BK20250237).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
For the data of Mendelian randomization: The data that support the findings of this study are openly available in the UK Biobank and the FinnGen research project (![]() ). For the data of case-control: The data that support the findings of this study are available on request from the corresponding author, SL, upon reasonable request.
). For the data of case-control: The data that support the findings of this study are available on request from the corresponding author, SL, upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
