Abstract
Background:
Pertussis remains a major public health concern in low- and middle-income countries despite the availability of effective vaccines. In Ethiopia, recurrent outbreaks continue to occur, particularly in hard-to-reach rural settings.
Objectives:
The aim of this study was to investigate a pertussis outbreak, identify associated risk factors, and evaluate the effectiveness of implemented interventions in Sayient Woreda, Northwest Ethiopia.
Methods:
An observational, community-based, unmatched case–control study with a house-to-house approach was conducted from October 25, 2018, to January 22, 2019. A total of 50 cases and 100 controls were selected using simple random sampling. Data were collected through face-to-face interviews using a pre-tested World Health Organization-adopted questionnaire. Data were cleaned, coded, and entered into Epi Info™ (version 7.2.1.0) and exported to SPSS (version 23) for analysis. Variables with a
Results:
The overall attack rate was 3.1/1000 population. The age of the affected individuals ranged from 3 months to 28 years, with a median age of 10 years (Interquartile Range (IQR): 0.7–12). Females accounted for 56.6% of the reported cases. Following the implementation of cephalexin therapy and active case search, a substantial decline in incident cases was observed. Independent risk factors for pertussis infection were inadequate awareness on transmission mode (adjusted odds ratio = 1.61, 95% confidence interval: 1.33–9.54), inadequate prevention and control measures (adjusted odds ratio = 4.64, 95% confidence interval: 2.19–9.83), household contact with a confirmed case (adjusted odds ratio = 6.99, 95% confidence interval: 1.19–41.15), being unvaccinated (adjusted odds ratio = 3.62, 95% confidence interval: 1.97–5.50), and unknown vaccination status (adjusted odds ratio = 1.77, 95% confidence interval: 1.96–16.08).
Conclusions:
A substantial pertussis outbreak occurred in Sayient Woreda, predominantly affecting children and adolescents. Inadequate community awareness, close household contact, and incomplete or unknown vaccination status significantly contributed to transmission. Strengthening routine immunization and enhancing community awareness are important for preventing future outbreaks.
Background
Pertussis is an acute, highly contagious, vaccine-preventable respiratory disease caused primarily by
Despite widespread immunization programs, pertussis remains endemic worldwide and continues to cause significant morbidity and mortality, particularly among infants and young children in low-income settings.3,7,8 Millions of cases and tens of thousands of deaths occur annually, even though pertussis surveillance is difficult.9 –11 In an unimmunized population, pertussis incidence peaks during preschool, and well over half of the children have the disease before adulthood.3,4,6,10,12 –15
Infants who are too young to be fully vaccinated and children with incomplete immunization are at the highest risk of severe disease and complications. Complications frequently occur during the paroxysmal stage, including pneumonia, neurological complications, hernia, otitis media, weight loss, dehydration, and other related complications.6,13,16,17 The likelihood-contributing factors for complications were pertussis-related malnutrition, post-tussive vomiting, distance of health facility, and poor seeking behavior of people.3,13,15,17,18
Pertussis has persisted and re-emerged as a significant public health concern in Ethiopia over the past two decades despite routine vaccination efforts. 19 A global summary by the World Health Organization (WHO) on vaccine-preventable diseases (VPDs) found that from 2000 to 2009, no reported national pertussis cases occurred, likely due to under-reporting and limited surveillance capabilities. However, a systematic review and meta-analysis of cases from 2009 to 2023 reported a total of 2824 cases and 18 deaths, with an AR of 10.8/1000 population and a case fatality rate (CFR) of 0.8%, and these outbreaks varied in magnitude and severity across different regions of Ethiopia.19,20
In South Ethiopia’s Region, 1840 suspected, probable, and confirmed pertussis cases and six deaths with an AR of 17.08/1000 population and a CFR of 0.33% were reported in Dara Malo district between September 2018 and January 2019, 19 while in Oromia region, 439 suspected pertussis cases and three deaths with an AR of 55/1000 population and a CRF of 0.7% were reported in Meda Walebu district between September 2018 and December 2019. 21
In Amhara Region, there were 215 reported cases and eight deaths with an AR of 1.3 and a CFR of 3.7% in Mekdela district from July 2015 to October 2015, 22 60 cases and two deaths with an AR of 1.3 and a CRF of 3.3% in Janamora district from April 22, 2017, to May 10, 2017, 23 122 cases with an AR of 0.86 in Dahena district from March, 27, 2019, to April 30, 2019, 18 and 43 cases in Simada district from December 3, 2020, to January 5, 2021, 24 indicating ongoing endemic circulation and gaps in routine immunization coverage, surveillance, and timely outbreak detection and response.15,18 –25
Vaccination is the most effective method for preventing pertussis in all age groups.26,27 Both diphtheria–tetanus–whole-cell pertussis (wP) and diphtheria–tetanus–acellular pertussis immunization have significantly reduced the global morbidity and mortality of pertussis. 28 High vaccine coverage leads to high protection in children under five, while minor reductions can increase cases.26 –28
Ethiopia launched the Expanded Programme on Immunization (EPI) in 1980 and provided routine immunization services free of charge at both community and health facility levels. 29 Despite the implementation of various immunization strategies, national vaccine coverage remains below the recommended 90% threshold.30 –32 Even though it remains below the national target, the full immunization coverage in Ethiopia has increased from time to time, rising from 43% in 2019 to 58.9% in 2020 at the national level and from 45.8% in 2019 to 72.5% in 2020 in the Amhara Region, yet there were great disparities across various regions in Ethiopia.33 –38 Marked geographic and socioeconomic inequities, high dropout rates, and declining trends in some poorly performing regions continue to challenge equitable and sustained immunization coverage across the country.33 –38
In Ethiopia, the national EPI program primarily targets infants, young children, and pregnant women to reduce morbidity and mortality associated with VPDs.30
–32 Under this program, infants begin vaccination at birth and receive a three dose primary series of pentavalent vaccine (Penta), which contains wP vaccine combined with diphtheria, tetanus, hepatitis B, and
The WHO recommends that all infants receive three primary doses of the pertussis-containing vaccine, followed by a booster dose between 1 and 6 years of age. 39 However, booster doses of pertussis-containing vaccine are not routinely provided beyond infancy for older children, adolescents, adults, or during pregnancy in Ethiopia. 40 The wP vaccine induces broad immune responses and has been commonly used in low- and middle-income countries, but immunity may wane over time if booster doses are not administered, leaving older children and adolescents being vulnerable to pertussis infection, particularly in settings with high population interaction and suboptimal vaccination coverage.18,22,23,35,36,40
In Sayint Woreda, routine immunization performance remained suboptimal. In the 6 months preceding the outbreak, coverage Penta-1 and Penta-3 were 77.0% and 74.0%, respectively, whereas full vaccination coverage was only 67.0%. These coverage levels fall below the thresholds required to achieve and sustain herd immunity, increasing the risk of pertussis transmission and outbreaks.18,22,23
Ethiopia’s national epidemiological surveillance system integrates case detection, reporting, and outbreak response across federal, regional, and district levels. 41 Routine laboratory confirmation for pertussis is generally not available at district level health facilities, which primarily rely on clinical diagnosis, although laboratories like the Ethiopian Public Health Institute and university-affiliated laboratories can perform confirmatory polymerase chain reaction (PCR) or culture testing. During outbreaks in rural and hard-to-reach settings, laboratory confirmation was often limited by logistical barriers, including specimen referral and transport, rather than by the absence of diagnostic capacity.15,18 –25,41 Understanding the local epidemiology, vaccination gaps, and contextual drivers of outbreaks is essential to guiding targeted interventions, particularly in resource-limited and predominantly rural settings. Therefore, this study aimed to characterize the pertussis outbreak in Sayient Woreda, identify associated risk factors, and assess the effectiveness of the control measures implemented during the outbreak response.
Methods
Study setting and period
The study was conducted in Sayient Woreda. It is an administrative district located in the South Wollo Zone of the Amhara National Region State, Northern Ethiopia. It covers an area of 1437.30 km2 (Figure 1) and has an estimated population of 144,972, with a substantial proportion aged under 15 years. The average household size is 4.14 persons, with a total of 33,604 housing units. Administratively, the woreda comprises one urban and 35 rural kebeles, of which seven rural kebeles are classified as hard-to-reach areas due to geographic and infrastructural constraints. The population is characterized by closely interactive social behavior, which may facilitate the transmission of communicable diseases. Health services infrastructure includes 32 health posts, seven health centers, and one hospital. The outbreak investigation covered the period from October 25, 2018, to January 22, 2019.
Geographic location of Sayint Woreda, South Wollo Zone, Amhara Region, Northern Ethiopia.
Study design
An observational, community-based, unmatched case–control study with a house-to-house approach was conducted.
Source and study population
The source population included all residents of Sayient Woreda, whereas the study population consisted of individuals residing in affected kebeles who met the WHO suspected or confirmed pertussis cases definition and individuals with no history of pertussis-like illness during the outbreak period. 39
Inclusion and exclusion criteria
Cases were all individuals in Ambasember Kebele who had a history of cough for more than 2 weeks with at least one of the following symptoms: paroxysms of coughing, inspiratory whoop, post-tussive vomiting, and apnea. Controls were individuals with no history of pertussis-like illness during the outbreak period, whereas controls that had a history of cough for at least 1 day before data collection and a history of contact with a suspected case were excluded.
Dependent and independent variables
The dependent variables are pertussis case status and intervention effectiveness, whereas the independent variables include sociodemographic characteristics (age and sex), vaccination history, knowledge about pertussis transmission and prevention, and a person’s history of contact with individuals who have pertussis within their classmates, family, and neighbors.
Sample size determination and sampling method
The total sample size was 150 (50 suspected cases and 100 controls, with one to two case–control ratios). The sample size was calculated using the Fleiss formula and the Epi Info™ (version 7.2.1.0) software, with an odds ratio (OR) of 5.859, considering one variable to establish a difference between groups, based on a two-sided 95% confidence interval (CI) and 80% power.39,42 –44 From all line-listed suspected and confirmed pertussis cases in the cluster, 50 cases were selected through active surveillance using a simple random sampling technique. For each confirmed or suspected pertussis case, two controls were selected who were present during the visited date based on the selection criteria.39,44,45
Data collection tool and procedure
Data were collected through face-to-face interviews using a pre-tested, structured questionnaire adopted from the WHO outbreak investigation tool and previously published similar studies.10,18,22,23,43,45 House-to-house visits were also performed to search active cases and controls. Cases were taken and followed daily within the study period. All the information hypothesized as risk factors for the pertussis outbreak was collected. Information collected included sociodemographic characteristics, clinical features, contact history, healthcare-seeking behavior, and knowledge of pertussis transmission and prevention (Supplemental Material). Immunization status was verified using vaccination cards when available or verbal recall obtained during household interviews using a structured survey questionnaire. Written immunization records, such as vaccination cards or health facility registers, were not consistently available or accessible at the time of the outbreak investigation. Therefore, verbal recall was used as the primary source of immunization information. This approach reflects routine practice during outbreak investigations in similar resource-limited settings. For participants younger than 16 years, legally authorized representatives were interviewed. 46
Laboratory testing procedures
During the outbreak investigation, case classification was conducted in accordance with the WHO pertussis surveillance standards, as laboratory-confirmed, epidemiologically linked, or clinically suspected.39,44,45,47 Among the total identified cases due to logistical constraints related to specimen transport and cold-chain maintenance in hard-to-reach rural settings, not all suspected cases were laboratory tested. In this investigation, 33 suspected cases of nasopharyngeal swabs were collected using a swab and placed in universal transport media for transport to the laboratory. Immediately after collection, the sample was transported to the University of Gondar Hospital Laboratory using cold packs to identify
Evaluation of intervention effectiveness
The intervention effectiveness was evaluated using both microbiological and epidemiological methods. Human clinical specimens were inoculated onto Regan–Lowe (charcoal horse blood) agar plates supplemented with selective antibiotics and incubated at 37 °C. Culture plates were examined daily for up to 7 days for the presence of characteristic
Data quality control
Appropriate quality control procedures and safety measures were carried out to ensure the reliability of the outcomes. At the end of each laboratory work, generated data on each laboratory work was recorded and checked for its completeness and consistency. The reliability of the laboratory results is also guaranteed by handling the sample carefully and performing the laboratory test from International Organization for Standardization accredited laboratory.
Data processing and analysis
The collected data was cleaned, coded, and entered into Epi Info™ (version 7.2.1.0) and analyzed using SPSS (version 23). Descriptive statistics were used to summarize sociodemographic variables using tables and figures. Variables with
Operational definitions
Results
Sociodemographic characteristics and vaccination status of the participants
A total of 535 suspected pertussis cases were identified during the outbreak, with an overall AR of 3.1/1000 population. Among the 50 interviewed cases and 100 controls, 27 (54%) and 58 (58%) were females, respectively. The age range of those affected was 3 months to 28 years, with a median age of 10 years (IQR = 0.7–12), whereas the controls’ age range was between 6 months and 34 years, with a median age of 11 years (IQR = 0.9–14). The most affected age groups were 10–14 years (Table 1).
Sociodemographic characteristics and vaccination status of the participants in Sayient Woreda, Northwest Ethiopia, October 2018–January 2019.

The majority of cases, 58 (65.2%), had received three doses, and 24 (26.9%) were fully vaccinated according to the national EPI schedule. However, 35 (23.3%) of cases had unknown vaccination status, and 26 (17.3%) had no history of pertussis vaccination (Table 1).
Epidemic curve and clinical features of pertussis cases by date of onset
The first case of pertussis was reported in Mekdela Woreda, with an AR of 5.8/1000 persons. Three months later, a widespread pertussis outbreak was identified in the Sayient Woreda, which occurred from October 25, 2018, to January 22, 2019 (Figure 2). During this period, 535 suspected cases were detected through active community-based surveillance. Several cases developed clinical complications, including hernia, edema, otitis media, pneumonia, and conjunctivitis, as documented during the investigation (Figure 3).

Epidemic curve of suspected and confirmed pertussis cases by date of symptom onset, Sayient Woreda, Northwest Ethiopia, October 2018–January 2019.

Anonymized photographs illustrating clinical complications during the pertussis outbreak investigation in Sayient Woreda, Northwest Ethiopia, October 2018–January 2019.
History of pertussis and cases with contact relations
Of the 150 participants, 67 (44.7%) reported no prior history of pertussis infection, whereas 16 (10.7%) had a self-reported history of previous pertussis infection. Among the 50 confirmed cases, the predominant exposure was household contact, 29 (58%), followed by classmate exposure, 15 (30%; Figure 4). All the participants resided within the same community, and clinical assessment was conducted to document symptoms of pertussis, including paroxysmal cough and post-tissue vomiting, during active case investigation.

Epidemiological distribution of contact exposure among pertussis cases identified during the outbreak investigation in Sayient Woreda, Northwestern Ethiopia, October 2018–January 2019.
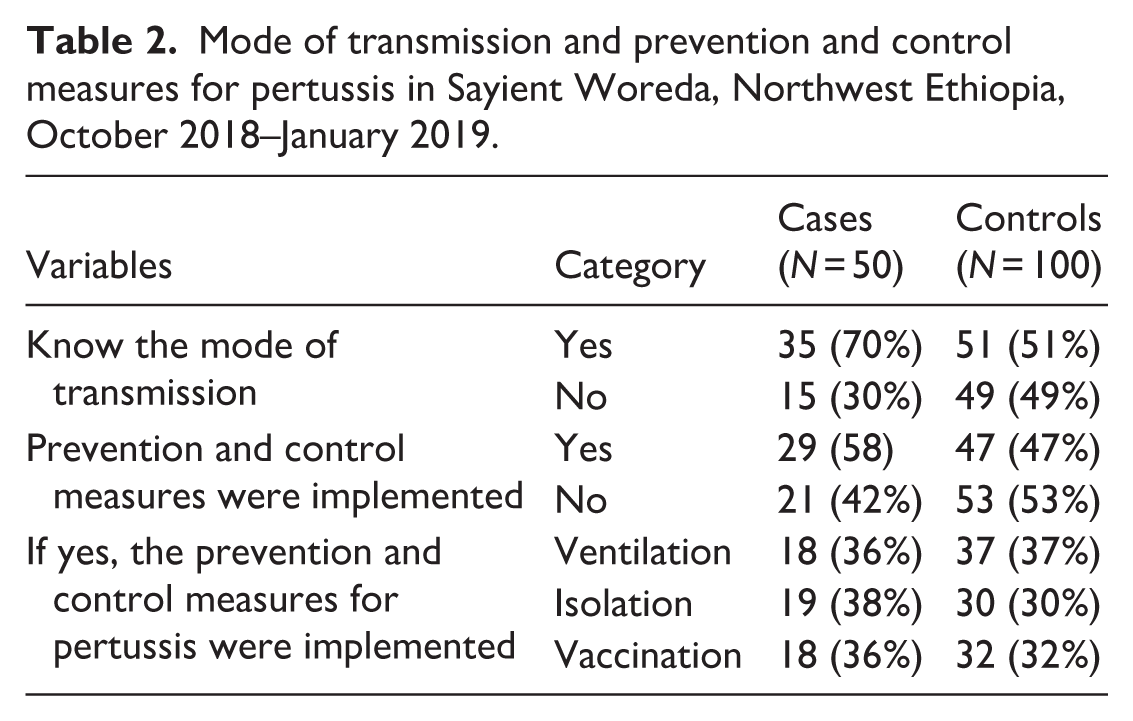
Mode of transmission, prevention, and control measures for pertussis
Of the study participants, 86 (57.3%) knew that pertussis can be transmitted from person-to-person through sneezing, coughing, or close face-to-face contact, and 74 (49.3%) had inadequate knowledge about effective ways to prevent and control the disease (Table 2).
Mode of transmission and prevention and control measures for pertussis in Sayient Woreda, Northwest Ethiopia, October 2018–January 2019.
Evaluation of intervention effectiveness
Following the initiation of cephalexin treatment and intensified search for active case, a significant decrease in both culture-confirmed and clinically suspected pertussis cases and a marked reduction in the number of newly diagnosed cases were observed, indicating that the timely administration of cephalexin played a significant role in interrupting transmission and contributing to outbreak control.
Factors associated with pertussis infection among the participants
Multivariable logistic regression analysis identified several key independent predictors of pertussis infection during the outbreak after adjusting for potential confounders, including lack of awareness on transmission mode (adjusted OR (AOR) = 1.61, 95% CI: 1.33–9.54), absence of pertussis prevention and control measures (AOR = 4.64, 95% CI: 2.19–9.83), household contact with a confirmed case (AOR = 6.99, 95% CI: 1.19–41.15), being unvaccinated (AOR = 3.621, 95% CI: 1.97–5.50), and unknown vaccination status (AOR = 1.77, 95% CI: 95% CI: 1.96–16.08; Table 3).
Bivariable and multivariable analyses of factors associated with pertussis infection in Sayient Woreda, Northwest Ethiopia, October 2018–January 2019.

AOR: adjusted odds ratio; CI: confidence interval at 95% significance level; COR: crude odds ratio.
Discussion
An outbreak of pertussis was detected in Sayient Woreda, with an AR of 3.1/1000 populations, indicating lower incidence compared to Papua New Guinea (AR = 4.0), 43 Dara Malo district (AR = 17.1), 19 and Meda Walebu district (AR = 55.0) 21 and a higher rate than in Dahena district (AR = 0.86), 18 Mekdela (AR = 1.3), 22 and Janamora district (AR = 1.3). 23 The decrement of the attacking rate might be recovery in cases before case detection and under-reporting of cases or weak surveillance activities.
Females were more affected than males, which is supported by similar studies conducted in Dahena, Mekdela, and Janamora district.18 –20 This is because females are slightly more susceptible and often have a more frequent contact with children, increasing exposure to respiratory infections like pertussis.
The outbreak-affected age ranges from 3 months to 28 years, and the most affected age groups were those aged between 10 and 14 years. However, similar studies conducted in Mekdela and Janamora district indicated that children under 4 years were the most affected age groups.19,20 Most pertussis complications occurred in the four age groups, which developed to umbilicus hernia, otitis media, edema, pneumonia, and one case of kidney failure.3,13,15,17 –20 The delay in arrival at the health facility could be caused by various factors, including poor health-seeking behavior in the community, difficulties in accessing the area, the interruption of routine immunization programs, and a vulnerable population.
Treatment for pertussis is easily available and highly encouraged. The observed decline in cases following antibiotic therapy and active surveillance supports the effectiveness of timely outbreak response measures.
51
The microbiological findings support the epidemiological evidence, as fewer specimens yielded characteristic
Household exposure was identified as a significant determinant of pertussis infection. Individuals who had a history of contact with confirmed cases in the family were around seven times (AOR = 6.99, 95% CI: 1.19–41.15,
Participants who reported having prevention and control measures were around four times (AOR = 4.64, 95% CI: 2.19–9.83;
Participants who knew the mode of transmission were around two times (AOR = 1.61, 95% CI: 1.33–9.54;
Vaccination status was a significant determinant of pertussis infection. Booster shots may be needed throughout life to ensure that your immunity remains intact.39,49 Those who remained unvaccinated were around four times (AOR = 3.62, 95% CI: 1.97–5.50;
Incomplete vaccination and inadequate documentation can lead to partial immunity, resulting in milder or atypical pertussis symptoms in older children who may unknowingly transmit
Limitations of the study
The lack of a vaccination card hindered the ability to determine an individual vaccination history, specifically the precise date of vaccination and other relevant information, resulting in information bias. Recall bias on the date of onset by the cases, and their mothers since the investigation was conducted recently after mass cases occurred. The absence of vaccination data in affected kebeles was a problem.
Conclusion
A pertussis outbreak had occurred in Sayient Woreda, predominantly affecting children and adolescents. The overall AR was 3.1/1000 population. All complicated cases occurred under the age category of 4 years. Key factors contributing to the outbreak in the study area included exposure to infected individuals within the household, a lack of awareness about how the disease is transmitted, inadequate prevention and control measures, and incomplete or unknown vaccination status. Strengthening routine immunization services, enhancing community awareness, and ensuring timely outbreak detection and response are important for preventing future outbreaks.
Recommendation
Based on the findings to prevent subsequent pertussis outbreaks:
Health workers should strengthen health education activities for the mode of transmission and prevention measurements of pertussis.
Developing active case search surveillance for early notification of diseases/events that occur.
Routine EPI service implementation should be established and health facilities should be alerted when there is a rumor of cases/events in adjustment woredas and regions.
Supplemental Material
sj-docx-1-smo-10.1177_20503121261437154 – Supplemental material for Pertussis outbreak investigation, associated risk factors, and evaluation of intervention effectiveness in Sayient Woreda, South Wollo Zone, Northwest Ethiopia: A case–control study
Supplemental material, sj-docx-1-smo-10.1177_20503121261437154 for Pertussis outbreak investigation, associated risk factors, and evaluation of intervention effectiveness in Sayient Woreda, South Wollo Zone, Northwest Ethiopia: A case–control study by Taddie Wondmnew Kassie, Workye Tsehyu Bayu and Biset Asrade Mekonnen in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank Addis Ababa University, Amhara National Regional State Health Bureau, South Wollo Zone, and the Sayient Woreda Health Office who approved the ethical issues. Our gratitude goes to the study participants and their legally authorized representatives for their willingness to participate in this study. We wish to extend our deepest gratitude to Dr. Alemayehu Bekele and Prof. Hagos Ashenafi, for their sustained guidance and critical review throughout the work. We are ready to thank the Addis Ababa University libraries/thesis bank, which makes this thesis available online via a research collection repository to enhance the digital service that collects, preserves, and distributes important tools/theses for preserving the Addis Ababa University legacy and facilitating digital communication.
Ethical considerations
Ethical approval was obtained from the Institutional Review Board (IRB) of Addis Ababa University (reference no. APHI/PHEM/3.03/4158). A formal cooperation letter was obtained from the Amhara Public Health and submitted to the South Wollo Zone and the Sayient Woreda Health Office after a brief explanation of the study objectives. The study was conducted in accordance with the national research regulations, institutional ethical guidelines, and the principles of Declaration of Helsinki.
Consent to participate
Oral informed consent was obtained from all study participants and their legally authorized representatives prior to data collection. Data collection was started after obtaining permission and provision.
Author contributions
T.W.K.: conceptualization, data curation, formal analysis, investigation, methodology, project administration, supervision, validation, visualization, and writing—original draft preparation. W.T.B.: data curation, supervision, validation, and visualization. B.A.M.: conceptualization, data curation, formal analysis, methodology, validation, writing—original draft preparation, and writing—review and editing. All the authors agreed on the journal to which the article will be submitted and gave final approval of the version to be published.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All the data generated or analyzed during this study are included in this article and supplementary file.
Supplemental material
Supplemental material for this article is available online.
