Abstract
Objective:
The present study aims to determine the causal association between sodium-glucose cotransporter inhibitors and depression, as previous observational studies have concluded a potential link between sodium-glucose cotransporter 1/2 inhibition and depression.
Methods:
A total of 16 instrumental variables mimicking sodium-glucose cotransporter-1 inhibition and 6 instrumental variables mimicking sodium-glucose cotransporter-2 inhibition were selected for the study. Depression data from the Psychiatric Genomics Consortium and the UK Biobank (n = 500,199) was used as the primary outcome. We employed the random inverse variance weighted method as the primary Mendelian randomization analysis. Supplemental analyses were also conducted to ensure the robustness of the evidence.
Results:
Our results indicated that genetically predicted sodium-glucose cotransporter-1 inhibition was negatively related with depression risk (ORIVW = 0.78; 95% CI: 0.67–0.91, p = 0.002) in the European population. However, we did not find a causal association between sodium-glucose cotransporter-2 inhibition and depression (ORIVW = 0.98; 95% CI: 0.71–1.36, p = 0.919).
Conclusions:
The findings of this Mendelian randomization study indicate that sodium-glucose cotransporter-1 inhibition may decrease the risk of depression in the European population. Future studies must be done to clarify the mechanisms that underlie the causal relationship. Our study provides clear evidence of the potential benefits of sodium-glucose cotransporter-1 inhibition in depression.
Introduction
Sodium-glucose cotransporter (SGLT) inhibitors include SGLT2 inhibitors, such as empagliflozin and dapagliflozin, and SGLT1/2 dual inhibitors, such as sotagliflozin. 1 SGLT inhibitors were initially developed for the treatment of diabetes. 2 SGLT2 inhibitors have been shown to impede glucose reabsorption in the proximal convoluted tubule, while SGLT1 inhibitors simultaneously inhibit glucose reabsorption in both the proximal convoluted tubule and the intestinal lumen.3,4 A growing body of research has indicated the multifactorial functions of SGLT inhibitors, in addition to their glucose-lowering property. To date, clinical guidelines advocated the prescription of SGLT2 inhibitors as the first-line treatment for a wide range of heart failure, 5 and as an important therapy for chronic kidney disease. 6 Concurrently, it has been verified that SGLT2 inhibitors exert a protective effect on other cardiovascular diseases (CVDs), including atrial fibrillation, 7 coronary artery disease, 8 and myocardial infarction. 9 In addition to their recognized indications for cardiovascular and renal diseases, observational study has also identified a potential significant role for SGLT1/2 inhibitors in the management of depression. 10
Depression is a pervasive mental disorder that poses a significant threat to global public health. It is estimated that in 2025, the number of disabilities adjusted life years lost due to depression in China would exceed 11 million. 11 In the United States, the 1-year prevalence of depression among adults stands at 10.4% and around 20.6% individuals will develop major depression during their lifetime. 12 Among patients diagnosed with CVDs, approximately 45% suffer from depression. 13 It is also suggested that depression is more prevalent in conjunction with cardiac disease, and that it is the principal contributor to the morbidity and mortality of CVDs. 14 Therefore, the capacity for a pharmaceutical agent to simultaneously treat CVDs and depression is poised to generate substantial clinical significance, thereby reducing the disease burden, particularly that pertaining to cardiac conditions.
A cohort study was conducted that yielded findings indicating a 45% reduction in depression risk among diabetes patients who were administered SGLT2 inhibitors. 15 Sotagliflozin, a SGLT1/2 dual inhibitor, showed the potential to attenuate cardiac function and depression disorder in mice following myocardial infarction. 16 However, it is not possible to determine whether there is a causal association between SGLT1/2 inhibitors and depression, due to the nature of observational studies. The Mendelian randomization (MR) study is predicated on the premise that single-nucleotide polymorphisms (SNPs) are randomly assigned at the time of conception, thereby effectively excluding the potential for confounding factors to interfere. Moreover, the irreversibility of heredity serves to preclude the possibility of reverse causation. 17 Therefore, the present study aims to elucidate the causal association between SGLT1/2 inhibitors and depression by MR analysis.
Methods
Study design
We performed this MR study adhering to the Strengthening the Reporting of Observational Epidemiological Studies Using Mendelian Randomization guidelines. 18 Our study met the following three fundamental hypotheses: (1) Screened IVs significantly associated with SGLT1/2 inhibition. (2) IVs merely influenced depression through SGLT1/2 inhibition. (3) No confounder existed affecting the causal relationship between SGLT1/2 inhibition and depression (Figure 1). All the analyses were performed from March to April 2025.

Design flow chart for the MR study. MR assumptions: (1) Assumption 1 suggests that genetic variants correlated with SGLT inhibition and affect depression through SGLT1/2 inhibition. (2) Assumptions 2 and 3 indicate SNPs are not associated with any confounders and do not influence depression through other ways.
Data sources and IVs selection
The corresponding data are available from public consultation (https://gwas.mrcieu.ac.uk). The Depression data obtained from the Psychiatric Genomics Consortium (PGC) and the UK Biobank consisted of 500,199 samples, including 170,756 cases and 329,443 controls of European ancestry. T2DM GWAS data included 12,171 cases and 56,862 controls of European ancestry from the DIAGRAM consortium. Data for SGLT1/2 inhibition selection was obtained from UK biobank and consisted of 344,182 European individuals (Table S1).
We obtained the IVs for SGLT1 inhibition by following a series of steps, the details of which can also be found in a recently published study. 19 (1) An investigation was conducted into the Genotype-Tissue Expression database, with the objective of identifying loci which exhibited a correlation with the mRNA levels of SLC5A1. 20 (2) In addition, the link between specific genetic variations in SGLT1 inhibitors and glycated hemoglobin (HbA1c) was confirmed. 1 (3) We subsequently proceeded with the deletion of selected SNPs exhibiting a linkage disequilibrium coefficient r² > 0.8 within 250 kilobases and F-statistics falling below the threshold of 10. 21 As a result, 16 SNPs met the necessary criteria and were selected to mimicthe effect of SGLT1 inhibition (Table S2). As previously mentioned, the selection of IVs for SGLT2 inhibition was referenced from the aforementioned process and also validated from a previously published study. 21 Finally, we screened 6 SNPs mimicking the effect of SGLT2 inhibition (Table S3).
Statistical analysis
In the context of the MR analysis, a range of methods were employed, including the weighted median, MR Egger, simple mode, weighted mode and inverse variance weighted (IVW). However, the primary method employed for the interpretation of results was IVW. 22 It was determined that an association between variables would be considered significant if the p-value of the IVW method was less than 0.05 and the estimate direction of the four other MR methods was consistent with that of the IVW method. To evaluate the heterogeneity of IVs, a range of methodologies were employed, including the Cochran Q-test and the MR-PRESSO procedure. 23 The presence of directional pleiotropy in the genetic variants is indicated by an intercept p-value of less than 0.05. 24 In addition, a leave-one-out sensitivity analysis was undertaken to ascertain the MR result of the remaining SNPs following the removal of each SNP individually. 25 Following this extensive analysis, we visualized the results by various plots. The effectiveness of each MR approach was represented by means of scatter plots. The individual SNP estimates were displayed using forest plots, while the distribution of individual SNPs was assessed using funnel plots.
All analyses were conducted by R software (version 4.2.1), The “Two Sample MR,” “Forestploter,” and “MR PRESSO” packages were employed. This study presents estimations of effect sizes with OR accompanied by the 95% CI. A two-side approach was adopted for the statistical tests and p < 0.05 being considered to indicate statistical significance.
Results
Causal effect between SGLT1/2 inhibition and T2DM
As a positive control and for validation of the screened IVs, we analyzed the causal association between SGLT inhibition and T2DM. Seven SNPs were harmonized for SGLT1 inhibition with T2DM. The MR analysis revealed a negative correlation between SGLT1 inhibition and the risk of T2DM (ORIVW = 0.28; 95% CI: 0.11–0.70, p = 0.007) in European population (Figures 2 and S1). Furthermore, the MR-Egger intercept provided no substantial evidence of directional pleiotropy (p = 0.606), and Cochran Q-test (p = 0.905) which implied no heterogeneity. Meanwhile, three SNPs selected for SGLT2 inhibition with T2DM. The MR analysis showed that SGLT2 inhibition was also negatively associated with T2DM risk (ORIVW = 0.12; 95% CI: 0.02–0.64, p = 0.014) in European population (Figures 2 and S2). The MR-Egger intercept provided no substantial evidence of directional pleiotropy (p = 0.774), and Cochran Q-test (p = 0.934).

Forest plot of MR results between SGLT1/2 inhibition and depression.
Causal effect between SGLT1 inhibition and depression
After the harmonization with outcome data (depression GWAS data from PGC), 12 SNPs determined for the MR analysis of SGLT1 inhibition with depression. The MR analysis results revealed that SGLT1 inhibition was negatively correlated with depression risk (ORIVW = 0.78; 95% CI: 0.67–0.91, p = 0.002) in European population (Figure 2). Moreover, the MR-Egger intercept provided no substantial evidence of directional pleiotropy (p = 0.789), the Cochran Q-test (p = 0.952). The sensitivity analysis results also visualized in various figures (Figure 3). The MR-PRESSO further validated the causal results from IVW analysis (p = 0.0005).

Scatter plots and causal estimates from three different methods. (a) Scatter plot of SGLT1 inhibition on depression in European population; (b) Forest plot of SGLT1 inhibition on depression; (c) Funnel plot of SGLT1 inhibition on depression; and (d) Leave-one-out analysis plot of SGLT1 inhibition on depression.
Causal effect between SGLT2 inhibition and depression
Five SNPs screened for SGLT2 inhibition with depression. The MR analysis revealed that SGLT2 inhibition was not causally associated with depression risk (ORIVW = 0.98; 95% CI: 0.71, 1.36, p = 0.919) in European population (Figure 2). In addition, the MR-Egger intercept provided no obvious evidence of directional pleiotropy (p = 0.918), and Cochran Q-test (p = 0.986; Figure 4). The MR-PRESSO further corroborated the findings of the IVW analysis (p = 0.751).
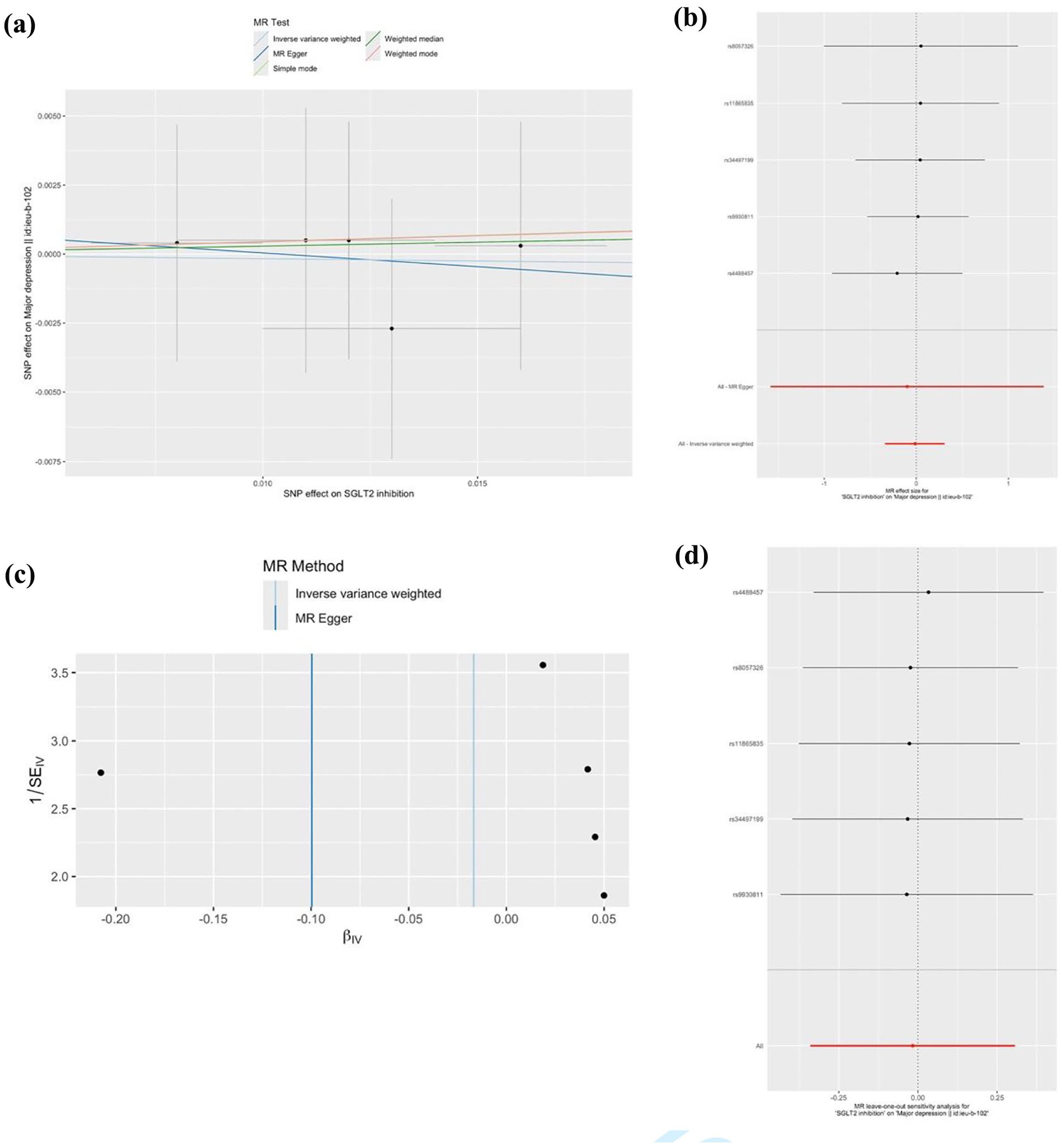
Scatter plots and causal estimates from three different methods. (a) Scatter plot of SGLT2 inhibition on depression in European population; (b) Forest plot of SGLT2 inhibition on depression; (c) Funnel plot of SGLT2 inhibition on depression; and (d) Leave-one-out analysis plot of SGLT2 inhibition on depression.
Discussion
The present study investigated the causal relationship between SGLT1/2 inhibition and depression by employing a MR study. The MR method is distinct from traditional observational studies in that it harnesses genetic variants as instrumental variables (IVs), thereby leveraging the inherent complexities in genetic variation to provide causal insights. Our MR analysis yielded novel findings, including the identification of a causal association between SGLT1 inhibition and depression risk, with a concomitant decrease in depression of 22% observed in the European population. Conversely, the present study did not ascertain a causal relationship between SGLT2 inhibition and depression.
SGLT2 inhibitors are highly prominent cardiovascular medication and have been extensively recommended for the treatment of heart failure, diabetes, and chronic kidney disease. Recently, the SGLT1/2 dual inhibition drug (sotagliflozin) has been developed and approved by the Food and Drug Administration for clinical usage. 26 In addition to their cardiovascular benefits, SGLT2 inhibitors have been demonstrated to play a protective role in depression. An animal study revealed that dapagliflozin has the potential to ameliorate stress-induced depression in rats by modulating neuroplasticity. 27 A population-based cohort study from Denmark revealed that SGLT2 inhibitors decrease the depression (odds ratio (OR) = 0.55, 95% CI: 0.44–0.70). 15 Another cohort study from Hong Kong demonstrated that SGLT2 inhibitors significantly reduce risk of depression incidence compared with dipeptidyl peptidase-4 (DPP4) inhibitors (OR = 0.52, 95% CI: 0.35–0.77). 10 While a Japanese retrospective study concluded that DPP4 decreases depression significantly compared with other hypoglycemic agents (OR = 0.31, 95% CI: 0.24–0.42), 28 it is challenging to ascertain the precise impact of SGLT2 inhibition on depression from the limited observational studies. Furthermore, the question of whether the glucose-lowering properties of SGLT2 inhibition contribute to an alleviation of depressive symptoms remains unresolved due to the observed concomitant anti-depressive effects of DDP4. Our MR analysis revealed that SGLT2 inhibition was not causally associated with depression risk (OR = 0.98, 95% CI: 0.71–1.36) in European population.
Sotagliflozin a new SGLT1/2 dual inhibitor has the capacity to target both the small intestine and the kidneys and thereby regulate blood glucose levels. 29 A basic study proved that sotagliflozin could improve depression disorder in mice after myocardial infarction by the gut-heart-brain axis. 16 Recent studies have indicated that sotagliflozin, empagliflozin, and dapagliflozin have the capacity to modulate inflammatory reactions, reduce oxidative stress, and regulate ketone metabolites.30,31 Consequently, this suggests that they may exert their neuroprotective function. Decreased tyrosine hydroxylase (TH) is associated with depression and aging, 32 supplement of TH may improve depression. 33 Herat et al. 34 discovered that sotagliflozin increased the TH level in the brain, which may have antidepressant and anti-ageing properties. In addition, SGLT2 inhibitor (dapagliflozin) was found to induce a marked upregulation of SGLT1 expression, concomitant with an increase in glucagon release. 35 A case-control study from India suggested SGLT2 inhibitors increased mild to moderate depression (OR = 1.74) and cognitive impairment (OR = 1.32) compared to controls. 36 This study’s findings contradict previous reports, but its limitations, including its small sample size and non-randomized study design, may have influenced the conclusions. While another study reported that SGLT2 inhibition could decrease depression and dementia risk, 37 the potential for SGLT2 inhibitors to induce SGLT1 levels, which may contribute to depression and cognitive dysfunction, remains to be substantiated by further research. The distribution of SGLT2 is predominantly confined to the kidneys and the retina, in contrast to SGLT1, which is distributed more extensively throughout the body. 38 SGLT1 is also highly expressed in brain and the inhibition of SGLT1 expression has been demonstrated to be efficacious in the amelioration of brain injury. 39 Interestingly, the ketogenic diet has been proposed as a metabolic therapeutic intervention targeting mood disorders. 40 Recent studies have suggested that SGLT2 inhi-bitors may enhance energy metabolism 41 and ketone production, 42 which could contribute to the pathophysiology of depression.
The present MR study demonstrated that SGLT1 inhibition led to a 22% decrease in depression risk in the European population. It is imperative that future large-scale randomized controlled trials are conducted to determine and justify the effect of SGLT1 and SGLT2 inhibitors on depression and further to verify our MR results.
Limitations
(1) The exclusive inclusion of participants of European descent give rise to questions surrounding the generalizability of the SNPs utilized in this study to other populations. (2) Despite the indications from some of the observational studies that SGLT2 inhibition may offer benefits on depression, the present MR study did not establish a causal relationship between them. (3) The number of IVs identified for SGLT1 is 16 and for SGLT2 is only 6, which is a relatively limited number. It is recommended that an updated GWAS database be utilized to validate the findings of the present study. (4) Future large-scale randomized controlled trials are needed to determine the effect of SGLT1 and SGLT2 inhibitors on depression and to further verify our MR results.
Conclusion
Our MR study indicated that SGLT1 inhibition may decrease the risk of depression in European population for the first time. As the prevalence of depression rises, particularly among individuals suffering from CVD, there is a growing need for effective treatment options. Sotagliflozin, a SGLT1/2 dual inhibitor, is a promising therapeutic agent for depression and related diseases. Future large-scale randomized controlled trials are justified to determine the effect of SGLT1 and SGLT2 inhibitors on depression and further verifying our MR results.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251352618 – Supplemental material for Sodium-glucose cotransporter-1 inhibition and depression: A Mendelian randomization study
Supplemental material, sj-docx-1-smo-10.1177_20503121251352618 for Sodium-glucose cotransporter-1 inhibition and depression: A Mendelian randomization study by Gang Fan, Hong Zuo, Xun Shi and Bo Liu in SAGE Open Medicine
Supplemental Material
sj-xlsx-1-smo-10.1177_20503121251352618 – Supplemental material for Sodium-glucose cotransporter-1 inhibition and depression: A Mendelian randomization study
Supplemental material, sj-xlsx-1-smo-10.1177_20503121251352618 for Sodium-glucose cotransporter-1 inhibition and depression: A Mendelian randomization study by Gang Fan, Hong Zuo, Xun Shi and Bo Liu in SAGE Open Medicine
Footnotes
Acknowledgements
We thank the international GWAS and IEU project for providing summary data for this analysis.
Ethical considerations
Not applicable.
Authors’ contributions
Gang Fan: Conceptualization, Writing – Original, Investigation, Software, Data Analyses, Supervision; Hong Zuo: Data Curation, Formal Analysis, Software; Xun Shi: Methodology, Validation; Bo Liu: Conceptualization, Writing – Review, Supervision.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data available on request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
