Abstract
Objectives:
This study aimed to determine the prevalence of detectable HIV-1 viral load and associated factors among pregnant and lactating women on Dolutegravir-based and non-Dolutegravir-based regimens.
Methods:
HIV-positive pregnant and breastfeeding women enrolled in a cross-sectional study design incorporating Dolutegravir versus non-Dolutegravir group for comparison purposes, and the study was conducted from September 2022 to March 2023. In total, 684 women receiving antiretroviral treatment (456 on Dolutegravir-based and 228 on non-Dolutegravir-based regimens) were enrolled across 16 prevention of mother-to-child transmission clinics. An undetectable viral load was defined as <20 RNA copies/mL. Univariable and multivariable regression models were used to determine the associations of the detectable viral load with factors such as sociodemographic, clinical, obstetric, and laboratory parameters.
Results:
The prevalence of detectable viral load on a non-Dolutegravir-based regimen was 32.9% and on a Dolutegravir-based regimen group was 19.7%. The overall prevalence was 24.1% (95% CI: 20.9–27.6). Compared with non-Dolutegravir-based regimens, Dolutegravir-based regimens were associated with 69% (AOR: 0.31; 95% CI: 0.16–0.61) lower risk of having a detectable viral load. The median viral load was significantly lower in women on a Dolutegravir-based regimen (1.66 log copies/mL) compared to those on a non-Dolutegravir-based regimen (2.11 log copies/mL; p < 0.001). A lower level of adherence was associated with 2.33 times (AOR: 2.33; 95% CI: 1.06–5.12) higher odds of having a detectable viral load and women who had a WHO clinical stage II and above were 2.71 times (AOR: 2.71: 95% CI: 1.30–5.64) more vulnerable to developing a detectable viremia than their counterparts.
Conclusion:
Despite the scale-up of Dolutegravir-based regimens, the overall prevalence of detectable viremia was 24.1%, with a higher prevalence of 32.9% among women on non-Dolutegravir-based regimens indicating a programmatic challenge that could affect mother-to-child transmission outcomes. Strengthening adherence monitoring and maintaining World Health Organization clinical stage I is crucial for achieving undetectable viral load levels in pregnant and breastfeeding women.
Introduction
According to the UNAIDS’ global estimate, approximately 39 million people are living with HIV and 53% of HIV positives are girls and women in 2022. Among the total global HIV-positive population, more than half (about 66%) are found in sub-Saharan Africa. Adolescent girls and young women are more than three times more vulnerable than their male peers in sub-Saharan Africa.1,2 Pregnant or lactating women who are living with HIV could face different types of complications including perinatal morbidity and mortality, especially if they are not provided comprehensive management regarding viral monitoring and antiretroviral (ARV) medications. ARV medications and regular HIV viral load monitoring can significantly minimize the vertical transmission of HIV to the fetus.3,4
According to the World Health Organization (WHO) recommendations, early identification and initiation of antiretroviral treatment (ART) without any preconditions, including viral suppression and CD4 count, are crucial.5,6 Moreover, the primary goal of early treatment during pregnancy and breastfeeding is to improve maternal health conditions and achieve undetectable HIV viral RNA in the plasma. In addition, ARV regimen selection has a greater effect on the significant control of HIV disease progression, opportunistic infection, risk of transmission (vertical and sexual), and ARV drug resistance.6,7 Since July 2019, the WHO has recommended DTG-based ART regimens as first-line HIV treatments instead of Efabrenz (EFV)-based regimens for pregnant and lactating women. 8 Ethiopia also adopted and included the integrase strand transfer inhibitor as a preferred first-line, Dolutegravir (DTG)-based treatment regimen (Tenofovir Disoproxil Fumarate (TDF) + lamivudine (3TC) + DTG)). 9 Some evidence showed that shifting the ART regimen from TDF + 3TC + EFV to TDF + 3TC + DTG can help persistently suppress the HIV viral load.10,11 A lower CD4 count indicates a weakened immune system, types of ARV treatment regimens, and adherence to ART drugs are basic factors to influence detectable maternal HIV viral load.5,10 Viral RNA measurement is an important tool for monitoring therapeutic response. Furthermore, HIV viral load testing is used to identify better therapeutic regimens for specific target groups, such as children, pregnant, and lactating women. Viral RNA detection with polymerase chain reaction (PCR)-based methods that address lower detection limits (<20 RNA copies/mL) is used for predicting long-term responses to ARV treatment, early identification of treatment failure, preventing inappropriate regimen changes, and protecting against drug resistance.12,13 Hence, this study aimed to assess the prevalence of detectable HIV-1 viral load and identify associated factors among pregnant and lactating women receiving DTG-based and non-DTG-based regimens. Ultrasensitive real-time PCR methods were employed to quantify plasma HIV-1 RNA levels, with a detection threshold of lower than 20 RNA copies/mL.
Methods
Study setting
The study was conducted in Addis Ababa, Ethiopia’s capital city. The city is administratively organized into 11 Sub-cities and based on the 2021 Ethiopian Statistical Service, formerly known as the Ethiopian Statistics Agency (ASA) estimated that 1,782,000 males and 1,992,000 females, for a total of 3,774,000 people reside in the city. According to the 2017/18 Ethiopian Health and Health-Related Indicators 13 governmental hospitals and 98 Health centers are found in Addis Ababa. Almost all hospitals and health centers were given basic maternal and child health services including prevention of mother-to-child transmission (PMTCT) services.14,15 The 2021 Ethiopian Public Health Institute (EPHI) Addis Ababa Health Atlas report also revealed that HIV/AIDS resulting in other diseases was the leading cause of disability and death in Addis Ababa. 16
Sampling and sample size determination
A facility-based cross-sectional study stratified with DTG-based and non-DTG-based regimens for comparison purposes was conducted from September 2022 to March 2023 in Addis Ababa, Ethiopia, across 16 healthcare facilities (5 hospitals and 11 health centers). Due to limited data on women receiving non-DTG-based regimens, and the larger scale-up of DTG-based regimens for pregnant and breastfeeding women as the preferred first-line treatment, option additional data from 188 women were incorporated for comparison. These data were obtained from the national viral load database of the 2020 budget year and taken from the same health facility’s data. The assumption was made that there would be no significant difference in virologic outcomes among women on non-DTG-based regimens between 2020 and 2022, allowing for the inclusion of this supplemental data. Based on our inclusion criteria, women who had been on non-DTG-based treatment for at least 6 months were eligible for the study. A total of 632 women met these criteria based on the database records. Then, 188 women were selected using systematic random sampling to ensure a representative group comparison. The study included pregnant and breastfeeding women who had been on ART for at least six consecutive months.
The sample size was determined using the double population proportion formula using Epi-info Version 7.2.0.1 (Atlanta, GA: CDC) statistical software with considering a 95% confidence level, a power of 80%, the proportion of detectable viremia among pregnant women on DTG-based regimens was 2.5%, and the proportion of detectable viremia among pregnant women on non-DTG based regimens was 9.0% based on a VESTED clinical trial. 17 We used a 1:2 ratio between the non-DTG and DTG-based regimen groups. To ensure sample representativeness, we used 1.5 for the design effect, and the final sample size was 684 (456 DTG-based regimen groups and 228 non-DTG-based treatment regimen groups).
Exclusion criteria
Pregnant or lactating women who had not been taking ART for at least 6 months and were unable to respond to the interview due to being critically ill were excluded from this study.
Variables
The main outcome variable was HIV-1 viral load suppression, defined as achieving undetectable HIV-1 viral load status. Independent variables were duration of ARV treatment, adherence to ART, and CD4 count after ART treatment, as it reflects immune recovery and WHO clinical stage as defined by the clinical guidelines of the country. 9 Sociodemographic factors include age, educational status, marital status, employment status, and family size. Clinical and drug-related data including spouse’s HIV Status, type of ARV drugs that participants were taking, and the duration of ART were also considered to be independent variables. Obstetric-related information such as gravidity types, gestational age, nutritional status, and hemoglobin were considered independent variables.
Data collection was conducted for all recruited pregnant and breastfeeding women who provided consent to participate. Data were collected using structured questionnaires administered through Open Data Kit Collect version 1.13 (Get ODK Inc. Seattle, Washington, USA) applications and paper-based forms.
Laboratory methods
Venous whole blood (10 mL) was collected through Ethylene diamine tetra acetic acid tubes, and plasma was separated immediately after collection. All the plasma samples were sent to the EPHI HIV National Reference Laboratory for HIV viral load analysis within 5 days. If samples were not tested within 5 days, they were stored at −70°C for long-term preservation. CD4 count and hemoglobin tests were either conducted at the health facility or referred to other health facilities through the existing referral system.
HIV viral load
A viral load test was performed at the EPHI National HIV Reference Laboratory using the Cobas 4800 system (Roche Molecular Diagnostics, Pleasanton, CA, USA) to determine HIV-1 viral RNA from a plasma sample. The COBAS HIV-1 quantitative real-time-PCR method amplifies the HIV-1 RNA genes in clinical specimens. It employs a quantification standard to ensure quality throughout the sample extraction, amplification, and detection processes. To verify that the sample preparation, extraction, purification, and amplification processes operate as intended, this unrelated RNA sequence is amplified concurrently with the target sequence using real-time-PCR. The COBAS 4800 system detects and quantifies HIV-1 RNA levels by measuring fluorescence from labeled oligonucleotide probes that bind only to the amplified product throughout each amplification cycle. The amount of HIV-1 RNA in the sample is determined by the amplification cycle at which the fluorescence is first detected. The system uses fluorescent probes to quantify the HIV-1 target sequence in each amplification cycle, with the fluorescent signal proportional to the number of HIV-1 RNA copies present in the original sample. The amount of HIV-1 RNA copies that were present in the original sample during the amplification cycle is proportional to the fluorescent signal recorded via real-time PCR. For this analysis, a sample volume of 0.4 mL was used, and the lower detection limit is 20 RNA copies per 1 mL of plasma. 18
Operational definition
Detectable viral load: HIV-1 viral load ⩾ 20 RNA copies per 1 mL of plasma.
Adherence: Good adherence was defined as participants adhering to medication at least 95% of the number of pills, and taking their prescriptions at the times and frequencies indicated. Fair-poor adherence was also operationalized as participants adhered to their medication less than 95% of the time based on the number of pills. 9 Adherence was measured based on participants’ medication-taking behavior over the past 3 months of their treatment period.
Treatment outcome: This was defined based on viral suppression. Particularly, poor treatment outcomes were determined to be those with HIV-1 viral load ⩾ 20 RNA copies per 1 mL of plasma while the counterpart was determined to be <20 RNA copies/mL.
DTG-based regimen: Includes women on ART taking DTG-based drugs (TDF + 3TC + DTG).
Non-DTG-based regimen: Includes women on ART taking non-DTG-based regimens including, Abacavir (ABC) + (3TC) + Atazanavir/ritonavir (ATV/r), ABC + 3TC + EFV, TDF + 3TC + ATV/r, Zidovudine (AZT) + 3TC + ATV/r, ABC + 3TC + lopinavir/ritonavir (LPV/r), TDF + 3TC + EFV, AZT + 3TC + EFV, AZT + 3TC + LPV/r, TDF + 3TC + Nevirapine (NVP), and AZT + 3TC + NVP.
WHO clinical stage: This is classified as stage one (non-symptomatic or having persistent generalized lymphadenopathy) and stage two (weight loss, recurrent respiratory tract infection, fungal, and skin infection). WHO clinical stages III and IV indicate advanced and severe stages of the disease, where the immune system is significantly weakened, leading to serious infections, complications, and life-threatening conditions. 19
Immuno-suppression: level of immunosuppression defined as whose CD4 count less than or equal to 350 cells/µL showed mild to severe immunosuppression. 19
Nutritional status: Based on the Mid-Upper Arm Circumference (MUAC) in centimeters (cm), ⩽23 cm indicated that there was a severe (<19 cm) or moderate (19–23 cm) level of malnutrition, whilea MUAC > 23 cm indicated no malnutrition. 9
Anemia: Women whose hemoglobin ⩽ 11 g/dL showed mild to moderate (7–11 g/dL) or severe anemia (<7 g/dL). 9
Data quality assurance
The questionnaire and data collection tool (ODK) were pretested on 40 women (5.8% of the study population) receiving ART at nearby health centers in Addis Ababa that were not included as study sites. This pretesting was conducted during the data collection training. Data collection was done by experienced and trained data collectors (nurses/health officers and laboratory personnel), after data collection to minimize data entry errors, we followed the double entry procedures with different personnel. The laboratory quality assurance was maintained by performing daily maintenance for equipment before HIV viral load testing. Daily quality control runs were done appropriately. Additionally, the EPHI HIV national reference laboratory is participating in an external quality assurance program from One World Accuracy and Centers for Disease Control and Prevention (CDC).
Data processing and analysis
The data was exported to SPSS version 26 (IBM Corp. Armonk, New York, USA) from the ODK data set, checked and edited before performing formal analysis and clean data taken from the national HIV viral load database included in the IBM SPSS version 26. For the analysis of some figures, we used DATAtab software (DATAtab e.U. Graz, Austria). Continuous variables including median and interquartile range (IQR) were calculated, while frequencies and percentages were computed for categorical variables. Mann–Whitney U Test, Dunn–Bonferroni Tests, and crosstabulation were conducted to compare sociodemographic, clinical, and drug-related characteristics between the HIV-1 undetectable and detectable viral load groups. Binary logistic regression analysis was used to determine the associations of variables with detectable HIV-1 viral load. Variables with p-values less than 0.2 in the univariable analysis were included in the multivariable model. The multivariable analysis provided adjusted odds ratios (AORs), 95% confidence intervals, and p-values. A significance level of p < 0.05 was used for determining significant associations.
Ethics approval and consent to participate
Ethical approval and clearance were obtained from the Institutional Review Board (IRB) of the EPHI Ethical Review Board with the ethical approval number EPHI-IRB/436-2022. Written informed consent was obtained and documented from each study participant. In this study, all women who had no formal education were able to read and write. So, we obtained written informed consent from all participants. All participants’ personal information was kept private and confidential, accessible only to the authors.
Results
Sociodemographic, clinical, and obstetric characteristics of the study participants
A total of 684 pregnant and lactating women enrolled in the PMTCT program across 16 health facilities who had been on ART for at least 6 months were included in the final analysis. The median age of these women was 30 years (IQR: 27–35). Most participants (n = 427; 62.4%) were married, and many (n = 273; 39.9%) were housewives. Eighty-one (11.8%) participants had not received any formal education. In this study, 72.4% (247/341) of women under 30 years old were on DTG-based regimens, while 27.6% (94/341) were on non-DTG-based regimens. Women aged ⩾ 30 years old were more likely to use non-DTG-based regimens compared to their younger counterparts. Approximately, 95.9% (71/74) of women who attended higher education (university/college) and 70.5% (366/519) of women with undetectable HIV viral loads were using a DTG-based regimen. Regarding adherence, 66.5% (429/645) of women with good adherence and 69.2% (27/39) had Fair-Poor adherence were on DTG-based regimens. Similarly, participants with higher CD4 counts (⩾350 cells/µL, 71.4%) were more likely to use DTG-based regimens compared to those on non-DTG-based regimens (Table 1).
Characteristics of study participants categorized by their antiretroviral (ARV) regimen type (non-DTG vs DTG-based).
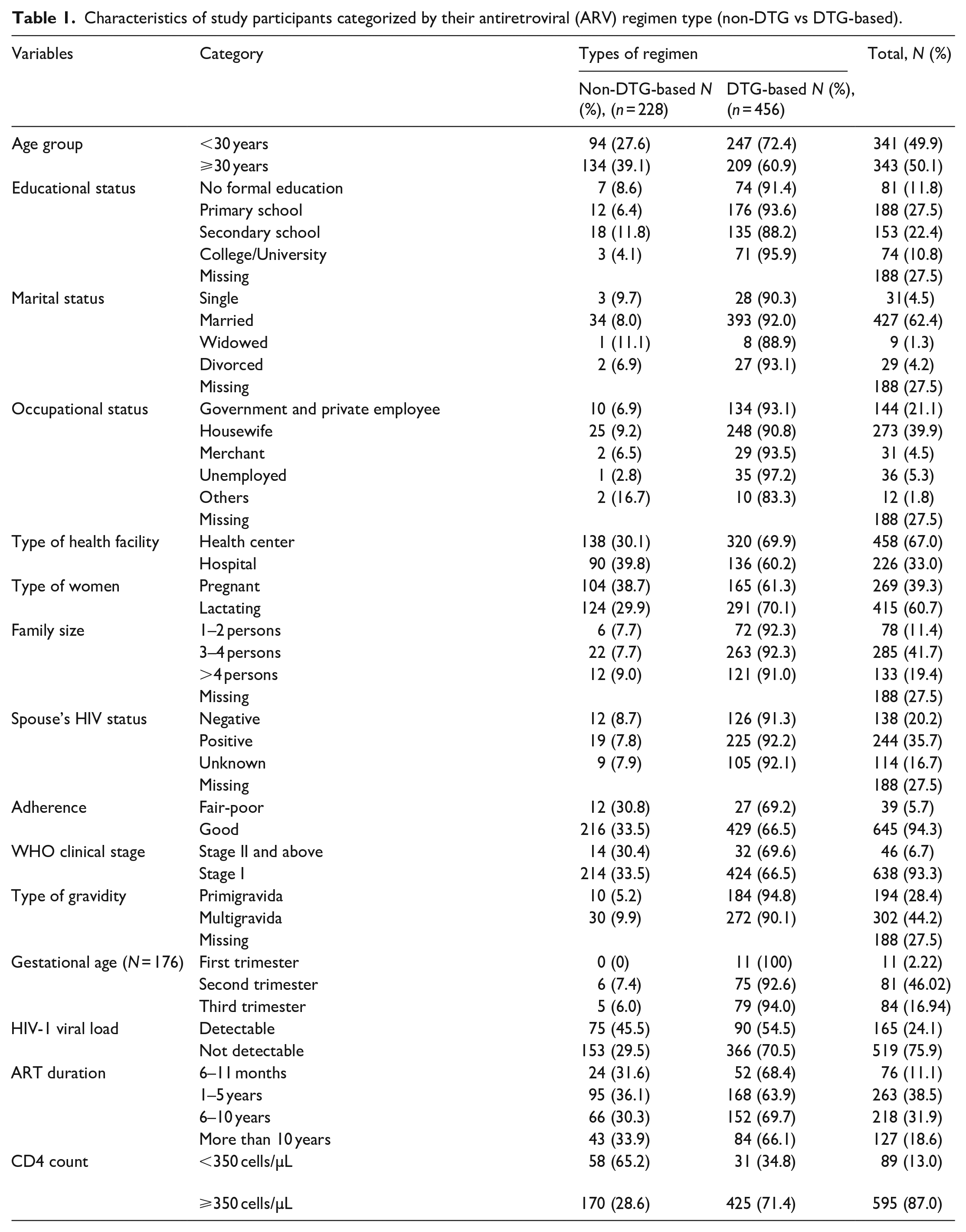
As shown in Table 1, the women’s clinical, obstetric, and drug-related characteristics are presented. Many participants were postpartum women (n = 415; 60.7%), whereas the remaining were pregnant (n = 269; 39.3%). One-third of the partners (n = 244; 35.7%) had HIV-positive results similar to those of their spouses. Most women (n = 645; 94.3%) exhibited good adherence to their ARV drug regimen. Among the women receiving treatment and care services at PMTCT clinics, 93.3% (n = 638) were in WHO clinical stage I (Table 1).
In this study, the prevalence of detectable viral load (⩾20 HIV-1 RNA copies/mL) was 32.9% (75/228) among women on non-DTG-based regimens and 19.7% (90/456) among those on DTG-based regimens. The overall prevalence was 24.1% (n = 165/684; 95% CI: 20.9–27.6). According to Figure 1, the proportions of detectable viremia varied depending on the viral load cut-offs, with 19.3% at viral load cut-offs by 50 RNA copies/mL, 11.7% at viral load cutoffs by 200 RNA copies/mL,and 2.8% at viral load cutoffs by 1000 RNA copies/mL (Table 2 and Figure 1(a)–(d)).

Proportions of detectable viral load across different cut-off levels (a–d). (a) Viral load cut-off by 20 RNA copies/mL, (b) viral load cut-off by 50 RNA copies/mL, (c) viral load cut-off by 200 RNA copies/mL, and (d) viral load cut-off by 1000 RNA copies/mL.
Sociodemographic, clinical, obstetric, and drug-related characteristics of women classified by their HIV viral load status in the PMTCT program in Addis Ababa, Ethiopia.

This study showed that among women with detectable viral loads, the median age was slightly higher at 31 years (IQR: 28–35). The median ART duration for women with detectable viremia was 56 months (IQR: 24–120), whereas, for those with undetectable viral loads, the median ART duration was slightly longer at 72 months (IQR: 25.5–114.25), though the difference was not statistically significant (Table 2 and Figure 2(a)).

Distribution of different variables in detectable and undetectable viral load among women in PMTCT program (a–d). (a) Distribution of duration on ART in women with detectable and undetectable HIV-1 viral load, (b) distribution of CD4 count in women with detectable and undetectable HIV-1 viral load, (c) distribution of MUAC in women with detectable and undetectable HIV-1 viral load, and (d) distribution of Hgb in women with detectable and undetectable HIV-1 viral load.
The nutritional status, measured by MUAC, showed a median MUAC of 25 (IQR: 24–26) for both groups (Table 2 and Figure 2(b)). The distribution of survey time CD4 cell counts is illustrated in Figure 2(c). The median CD4 count among those with detectable viral loads was 549 cells/µL (IQR: 372–563), whereas those with undetectable viral loads had a median CD4 count of 555 cells/µL (IQR: 503–570) (Table 2). Additionally, Figure 2(d) depicts the distribution of hemoglobin levels in individuals with detectable and undetectable viremia. The results indicated that the median hemoglobin level for those with detectable viremia was 12.87 g/dL (IQR: 12–13.33), whereas for those with undetectable viremia was 12.88 (IQR:12.3–13.5) (Table 2).
Factors associated with HIV-1 detectable viremia
In this study, some variables including, level of adherence, WHO clinical stage, gravidity, CD4 count, and type of ARV drug were identified with p-values < 0.2 and selected for further analysis using a multivariable model to minimize confounding effects and obtain more accurate results. The analysis revealed that pregnant and breastfeeding women taking DTG-based regimens had a 69% lower risk (AOR: 0.31; 95% CI: 0.16–0.61; p = 0.001) of experiencing a detectable HIV-1 viral load compared to those on non-DTG based drug regimens. Conversely, women with poor to fair adherence levels were 2.33 times (AOR: 2.33; 95% CI: 1.30–5.12; p = 0.036) more likely to have detectable viremia than those with good adherence. Additionally, women with WHO clinical stage II and above were 2.71 times (AOR: 2.71: 95% CI: 1.30–5.64; p = 0.008) more likely to develop detectable viremia than those with WHO stage I. On the other hand, CD4 count < 350 seemed to be associated with detectable viremia in the univariable model (p < 0.05), however, in the multivariable model the result showed non-significant associations. It could be related to other confounders. Similarly, the multigravida women’s p-value in the univariable model was <0.2, whereas in the multivariable model, the result was not retained (Table 3).
Multivariable analysis of factors associated with detectable HIV-1 viremia among women in the PMTCT program in Addis Ababa, Ethiopia.

COR: unadjustable odds ratio; AOR: adjustable odds ratio.
Significance level p < 0.05.
The HIV-1 viral load in log copies/mL was influenced by various factors, including the type of drug regimen, CD4 count, WHO clinical stage, adherence, and duration of ART. The median viral load among women on a DTG-based regimen was 1.66 log copies/mL (IQR: 1.40–2.01), whereas the median viral load among those on a non-DTG-based regimen was 2.11 log copies/mL (IQR: 1.76–2.35). The Mann–Whitney U Test indicated that this difference was statistically significant (p < 0.001) (Figure 3(a)). Women with a CD4 count below 350 cells/µL had a higher median viral load of 2.11 log copies/mL (IQR: 1.65–2.90) compared to those with a CD4 count ⩾350 cells/µL, whose median viral load was 1.89 log copies/mL (IQR: 1.45–2.18). This difference was statistically significant (p = 0.018) (Fig. 3(b)). Pregnant and lactating women with a WHO clinical stage II or higher had a higher median viral load of 2.07 log copies/mL (IQR: 1.74–3.38) compared to those at stage I, who had a median viral load of 1.89 log copies/mL (IQR: 1.46–2.18; p = 0.047) (Figure 3(c)).

Factors affecting detectable HIV-1 viral RNA log copies (a–f). (a) HIV-1 viral load between women in DTG-based and non-DTG-based treatment regimen, (b) HIV-1 viral load between Women CD4 count ⩾350 and <350 cells/µL, (c) HIV-1 viral load between women in WHO clinical stage I and those in stage II and above, (d) HIV-1 viral load between women in fair-poor adherence and good adherence, (e) HIV-1 viral load between women in primigravida and multigravida, and (f) HIV-1 viral load between women in different durations of ART treatment in months.
Another factor affecting the median viral load was adherence to ART drugs. Women with fair to poor adherence had a higher median viral load of 2.07 log copies/mL (IQR: 1.84–2.40) compared to those with good adherence. However, the Mann–Whitney U test showed that this difference did not reach the level of statistical significance (p = 0.078) (Figure 3(d)). The median viral load for multigravida women was 1.72 log copies/mL (IQR: 1.41–2.11). The median viral load between multigravida and primigravida women was almost identical (p = 0.979) (Figure 3(e)). In this study, the prevalence of detectable viremia was not significantly associated with the duration of ART. However, women who had been on ART for 6–11 months had a slightly higher median viral load of 2.09 log copies/mL (IQR: 1.89–2.90) compared to those on ART for 1–5 years or 6–10 years. Nevertheless, this difference was not statistically significant (Adjusted Dunn–Bonferroni Tests, p = 0.099) (Figure 3(f)).
Discussion
This study is institutional-based and aimed to determine the HIV-1 detectable viral load level among women on a DTG-based regimen and a non-DTG-based regimen. All women who were included in this study had received ARV drugs at least for 6 months in 16 selected PMTCT clinics in Addis Ababa, Ethiopia. Additionally, factors such as low level of adherence, WHO clinical stage II and above, and a low CD4 count, were identified as being associated with detectable viremia among pregnant and breastfeeding women.
This study showed that the prevalence of detectable viral load among women on a DTG-based regimen was 19.7%, whereas for a non-DTG-based regimen was 32.9%. DTG-based treatment regimen had a 69% lower risk (AOR = 0.31; 95% CI: 0.16–0.61) of having a detectable HIV viral load compared to those on a non-DTG-based regimen. Consistent with our findings, an Ethiopian study revealed that women on DTG-based regimens had a 31% lower risk of HIV-1 viral non-suppression compared to those on the Efavirenz-based regimen. 10 Another Ethiopian study also showed that patients on DTG-based ARV treatment had lower median viral RNA copies. 20 The results of the DolPHIN-2 trial indicated that the proportion of patients with a low level of viremia (<50 copies/mL) was higher in the DTG-based regimen group compared to the Efavirenz-based regimen group. 21
Our analysis revealed that the overall prevalence of detectable viremia (⩾20 RNA copies/mL) was 24.1% (95% CI: 20.9–27.6). This low prevalence of detectable viremia aligns with previous studies reported from Nigeria, 22 Kenya (where the prevalence of detectable viral load was 17.9%), 23 and South Africa (where the prevalence of detectable viremia was 22% among pregnant women on ART). 24 However, evidence from other regions indicates a lower prevalence of detectable viral load. For instance, in Malawi, the rate of detectable viremia was 10.8%, although this study used a higher cut-off for a detectable viral load (>400 RNA copies/mL). 25 Another study from Malawi reported a higher rate of detectable viral load (8.7%) when a cut-off of ⩾40 RNA copies/mL was used. 26 A similar result was reported in Spain, where the rate of detectable viremia among the adult population was 8.5% with a lower limit of 50 RNA copies/mL. 27 A low prevalence of detectable viremia has significant implications for reducing MTCT, minimizing the sexual transmission of HIV, and improving patient outcomes. Evidence shows that low-level viremia is related to improved CD4 counts and clinical stages.20,28 This indicates the effectiveness and success of the ART program. 25
Conversely, a higher proportion of detectable viral load was reported in Uganda (94.5%) 29 and 30.7%, 30 Rwanda (47.8%), 31 and Brazil (52%). 32 These differences might be attributed to various factors, such as lower detection limits for the assays used. Hence, most studies used 40 or 50 RNA copies/mL as the lower detection limit, whereas our study used <20 RNA copies/mL as the cutoff. Additionally, differences in the study population, study design, and types of ART medication may influence detectable viremia levels. Moreover, some studies included participants who were naïve to ARV treatment,29,31 while many studies focused on detectable viral loads among pregnant women, nearly half of MTCT occurs during breastfeeding. 33 Our study also included 415 (60.7%) breastfeeding women.
Monitoring the viral load and achieving undetectable levels in pregnant and breastfeeding women can significantly impact the prevention of MTCT of HIV and improve maternal health. Maintaining an undetectable viral load throughout pregnancy and breastfeeding can greatly reduce the risk of transmission.34,35 A study from Malawi reported that women with low levels of viremia (40–1000 RNA copies/mL) had about nine-fold greater risk of MTCT than those with undetectable viremia. 26 However, these studies highlighted that the issue of persistent undetectable viral load and viral rebound needs further investigation, as there is no definitive evidence yet. 36 Additionally, in breastfeeding women on ART, HIV proviral DNA can remain in reservoir cells within breast milk even when the maternal viral load is low. 37
The goal of ARV treatment in the “test and treat” era is for people living with HIV to improve their clinical well-being and achieve undetectable viral RNA levels in their plasma, as measured by ultrasensitive viral load assays. This signifies the concept of “Undetectable = Untransmittable (U=U),” indicating no risk of sexual transmission and minimal risk of mother-to-child transmission.38,39
This study revealed that women with fair-to-poor adherence had more than twice (AOR: 2.33; 95% CI: 1.06–5.12) at risk of having a detectable viral load, which aligns with a study conducted in Brazil, that reported higher odds of undetectable viral load with high adherence levels (>80%). 32 Similarly, a study from Malawi reported that sub-optimal adherence was associated with detectable viremia. 26 Another study from Ethiopia also revealed that low levels of adherence were significantly associated with HIV viral non-suppression. 10
In our study, WHO clinical stage II or higher had 2.71 times (AOR = 2.71; 95% CI: 1.30–5.64) greater risk of having detectable viremia compared to those with WHO clinical stage I. Similarly, HIV viral copies were significantly influenced by several factors. According to Figure 3(a)–(c), the Mann–Whitney U test result showed that the median and IQR of viral load were significantly higher (p < 0.05) among women who were on a non-DTG-based regimen, had a low CD4 count (below 350 cells/µL), and were at a higher WHO clinical stage (stage II or above). Although specific studies directly addressing these findings are limited, our results suggest that HIV viral load may be associated with immunological status, clinical progression, and the type of drug regimen used among pregnant and breastfeeding women.
Since 2018, the WHO has recommended a new DTG-based regimen in combination with a nucleoside reverse-transcriptase inhibitor (NRTI) as the preferred first-line regimen for adults and adolescents including pregnant and breastfeeding women. 8 Furthermore, the DTG-based regimen is associated with minimal adverse drug reactions and improved virological outcomes compared with regimens based on Raltegravir or Efavirenz. 40 Some studies have demonstrated that the DTG-based regimen is well-tolerated, safe, and effective in achieving an undetectable viral load.41,42
Limitations of the study
This study was conducted in an urban setting, where there is better access to health facilities to help with viral load monitoring and a good awareness of medication adherence, which affects the uptake of DTG-based regimens and potential early diagnosis linked to the WHO clinical staging. Hence, the limitations of this study, which was conducted in the capital city, and the case may be worse in rural and semi-urban areas. The other limitation of this study is that the health facilities were selected based on their high load of women in PMTCT clinics, which may introduce selection bias and might be a risk of high levels of detectable viremia in health facilities that have had low numbers of women in PMTCT clinics since efforts, experts, and infrastructures are towards high load sites. In this study, we did not match women on second-line or third-line treatment regimens in DTG-based and non-DTG-based regimens. Although the number of women was limited in second or third-line regimens, it could have some effect on the study outputs, and we considered it as a limitation.
Conclusion
In this study, the overall prevalence of detectable HIV viral load was relatively low. However, the prevalence of detectable viral load among pregnant and breastfeeding women on non-DTG-based regimens was significantly higher compared to those on DTG-based regimens. DTG-based ARV treatment regimens were 69% less likely to develop detectable viral loads. In this study, fair to poor adherence and WHO clinical stage II or above were significantly associated with detectable HIV viral loads, increasing the risk of MTCT. Therefore, regular HIV viral load monitoring, intensive adherence counseling, and special attention are crucial for these populations to minimize detectable viremia and the risk of MTCT. Despite the higher prevalence of detectable viral load among participants on non-DTG-based regimens, this highlights the need for further scale-up of DTG-based regimens for better treatment outcomes. The low level of detectable viremia among the overall pregnant and breastfeeding women implies the success of the ART program among these groups of sub-populations. Moreover, improving enhanced adherence counseling and targeting women with WHO clinical stage II or above could further improve the virologic outcome of pregnant and breastfeeding women in Ethiopia.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251320460 – Supplemental material for Role of Dolutegravir-based antiretroviral treatment in achieving an undetectable HIV-1 viral load among women attending prevention of mother-to-child transmission clinics in Addis Ababa, Ethiopia
Supplemental material, sj-docx-1-smo-10.1177_20503121251320460 for Role of Dolutegravir-based antiretroviral treatment in achieving an undetectable HIV-1 viral load among women attending prevention of mother-to-child transmission clinics in Addis Ababa, Ethiopia by Belete Woldesemayat, Yimam Getaneh, Sisay Adane, Ajanaw Yizengaw, Amelework Yilma, Shambel Araya Haile and Kidist Zealiyas in SAGE Open Medicine
Footnotes
Acknowledgements
The authors extend their sincere gratitude to all the participants in the study and the staff of the PMTCT clinics at the health centers and hospitals where data collection was conducted. We also thank the staff and management of the EPHI National HIV Reference Laboratory for their invaluable assistance and resources that made this research possible.
Authors’ contributions
BW: Conceptualization, writing the original draft, and designing this study. SA and AY contributed to collecting data and laboratory testing. BW, SAH, YG, and AY analyzed the results and wrote the manuscript. SAH, YG, and KZ revised the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Ethical approval and clearance were obtained from the Institutional Review Board (IRB) of the EPHI Ethical Review Board with the ethical approval number EPHI-IRB/436-2022. Written informed consent was obtained and documented from each study participant. All participants’ personal information was kept private and confidential, accessible only to the authors.
Informed consent
Written informed consent was obtained and documented from each study participant. In this study, all women who had no formal education were able to read and write. So, we obtained written informed consent from all participants. All participants’ personal information was kept private and confidential, accessible only to the authors.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
