Abstract
Objective:
Listeria monocytogenes is a major cause of miscarriage and postpartum infections in infant. Determining antibody levels against listeriolysin O can be valuable for diagnosing both invasive listeriosis and febrile gastroenteritis. However, serological methods that detect antibodies against incomplete forms of listeriolysin O can be more specific. The objective of this study was to identify (Listeria monocytogenes) in the serum of pregnant women using serological and culture methods.
Methods:
Clinical samples (120 cases) were collected from pregnant women with a gestational age of less than 20 weeks. Diagnosis of Listeria monocytogenes was conducted using culture methods to identify anti-Listeria antibodies. Statistical analysis of the results was conducted using IBM SPSS Statistics version 23.0 (New York, USA), Pearson’s Chi Square and fisher tests.
Results:
The number of positive samples by culture and ELISA was 24.16% (29) and 28.3% (34), respectively. Out of the 29 positive sample by the culture method, 10 individuals had no abortion history, 16 and 3 individuals had 1 and 2 abortions and no sample had 3 abortions. Maybe, the more abortions a person has had, the less likely they are to be infected. In the Enzyme Linked Immuno-Sorbent Assay (ELISA) method, 13 individuals tested positive for both IgG and IgM antibodies and 38 individuals tested negative. Additionally, among the positive individuals with 1, 2, and 3 miscarriages, 0, 17, and 3 people were positive for the IgG antibody and 0, 18, and 3 individuals were positive for the IgM antibody. The analysis results indicated that there was no significant relationship between culture and abortion history (p = 0.316), IgG ELISA and history of miscarriage (p = 0.672) and IgM ELISA and history of miscarriage (p = 0.552).
Conclusion:
There was no significant relationship between infection with Listeria monocytogenes and abortion (p ⩾ 0.05) in our samples. These results should be interpreted with caution due to the limitation of our small sample size.
Introduction
Abortion refers to the loss of a fetus. Recent research has been concerned on the causes, incidence, and harmful effects of abortion in both humans and animals.1,2 Infectious agents are the most frequently considered cause of abortions in humans and domestic animals. 3 Various factors contribute to miscarriages, including genetic disorders, chromosomal disorders, immunological problems, the mothers advance age, anatomical and hormonal problems, environmental factors, and infection. It has been observed that 5% of abortions are caused by infection factors. Conversely, infectious factors can also lead to abortion.2,3
Listeria monocytogenes (L. monocytogenes) is a bacterium that can cause diseases in both animals and humans.4,5 The Listeria genus consists of six species: L. monocytogenes, Listeria ivanoi, Listeria siligri, Listeria inocera, Listeria welshii, and Listeria spp. Among these, L. monocytogenes and L. ivanoi are known to be pathogenic. 6
L. monocytogenes is a type of bacteria that are short, Gram-positive bacilli (coccobacilli) without branches. They have rounded ends and can appear Gram-variable and cocci-shaped in older cultures. These bacteria can be found alone or in short chains, and sometimes in filaments. In certain situations, when they are in pairs, they may be mistaken for pneumococci.7,8 Similar to Corynebacterium, these bacteria are catalase positive, do not have a capsule, are nonacid fast, oxidase negative, and lack mycolic acid in their cell wall. 9 They grow best at temperatures between 30 and 37°C. One distinguishing characteristic of Listeria bacteria is their ability to move at room temperature (20–25°C), which sets them apart from Corynebacterium. 10
L. monocytogenes causes various infections in humans, including symptoms similar to a cold, primary meningitis, encephalitis, or septicemia. In pregnant women, this bacterium typically causes a flu-like disease known as bacteremia. If left untreated, it can lead to inflammation of the placenta or amniotic membrane and infection of the fetus, potentially resulting in miscarriage or premature birth. Individuals who have undergone lymphoma or AIDS treatment are particularly susceptible to infections caused by L. monocytogenes. The bacterium’s tendency to affect the central nervous system leads to an acute infection, which is often fatal.6,11 Pregnant women are at a 20 times higher risk than the general population, accounting for 27% of all listeriosis cases. 12
Currently, the standard method for detecting L. monocytogenes is through culture. However, the use of antibiotics in animal feed and the potential for intracellular growth of bacteria greatly reduces the sensitivity of culture. 13 Another drawback is that the cultivation method takes several days and hours. Other diagnostic methods include nucleic acid hybridization immunoassay, phase typing, and polymerase chain reaction. 14 Serological response to whole cell antigens cannot be used for diagnosis because there is antigenic cross-reaction between L. monocytogenes and other Gram-positive bacteria such as staphylococci, enterococci, and bacilli. Additionally, patients with culture-confirmed listeriosis have undetectable antibody levels. Determining antibody levels against listeriolysin O can be valuable for diagnosing both invasive listeriosis and febrile gastroenteritis. However, serological methods that detect antibodies against incomplete forms of listeriolysin O can be more specific. 15
In the culture method, samples are taken from sterile sites such as blood, cerebrospinal fluid, placental amniotic fluid, or fetal body tissue. These samples are then cultured on blood agar and incubated for 18–24 h at 35°C–37°C. On the other hand, samples taken from nonsterile sites such as anal swabs, feces, urine, and the female genital tract are cultured on Palcom or Oxford agar differential media or blood agar culture media. Differential diagnostic tests are required to identify bacteria. The spread and distribution of this bacterium in infections caused by it in Iran are unknown. From this perspective, epidemiological investigations of this bacterium are crucial, especially in high-risk populations, such as pregnant women. This will help emphasize the importance of this bacterium among other factors in prevention and treatment programs.14,15 Therefore, the aim of this study is to identify L. monocytogenes in the serum of pregnant women using serological methods and compare it with the culture method.
Methods
Sampling
We conducted a cross-sectional study in Iran between 2018 and 2020. One hundred twenty clinical samples were collected from referrals with a gestational age of less than 20 weeks, including those with a history of miscarriage and those without. Of the participants, 69 (57.5%) experienced cardiac abortion, while 51(42.5%) did not. Sampling was conducted based on availability and at random within the specified time frame, without any age restrictions, and with the participant’s personal consent. Pregnant women receiving antibiotic treatment were excluded from the sample. Blood serum was promptly separated from 5 ml blood samples by centrifugation at 3200 RPM for 10 min. The sera were then stored at 20°C until the day of the tests.
Isolation of L. monocytogenes positive samples by phenotypic and biochemical methods
Cultivation of samples
The next 5 ml of participants’ blood samples were used for culture experiments. The blood samples were first injected into the liquid Brain Heart Infusion Agar medium. After 48 h, they were transferred into the FRASER Listeria Selective Enrichment Broth (base), a medium prepared by Merck, Germany, specifically designed for the enrichment of L. monocytogenes. The cultivation process in this environment occurs in two-stage. Initially, the sample was cultured in FRASER broth medium with half of the culture, and then incubated at 30°C for 24 h. Next, 0.1 mL of the cultured medium from the first stage was transferred into 10 mL of FRASER broth and cultured at 35°C for 48 h. Following this, the medium from the second stage was cultured on the special Oxford agar medium. 6
Identification of L. monocytogenes colonies using phenotypic and biochemical tests
Colonies grown on Oxford medium were evaluated using Gram staining tests, growth in bile salt, catalase, oxidase, methyl red, and Vogs–Proskauer medium, movement in sulfide-indole-motility medium, and the production or absence of H2S gas. In the end, the final diagnosis was made using biochemical identification methods such as acid production tests from glucose, and rhamnose sugars, rather than the inability to consume xylose and mannitol.6,9
ELISA method
Antibodies against L. monocytogenes in serum samples were analyzed using the ELISA method (ELISA kit, DIATHEVA Co., Italy). The amount of IgG and IgM antibodies against L. monocytogenes in the serum samples of all participants were determined following the kit’s instructions. To outline the procedures, the serum samples were diluted 1:100 (volume/volume) in separate glass vials using the sample solvent. The diluted samples were thoroughly mixed (5 ml of sample + 495 ml of sample solvent). In addition, 100 μL of undiluted positive, negative, and calibrator controls were added to the wells. The diluted samples were then added to the wells in duplicate. The microtiter plates were covered and incubated at 37°C for 60 min. Subsequently, the plates were washed five times with X1 buffer. A measure of 100 μL of conjugated antibodies were added to each well, including the control wells. They were incubated at 37°C for 60 min and washed five times with X1 buffer 100 microliters of ABTS™ Buffer solution were added to each well, including the control wells. The plates were then incubated for 20 min at room temperature. After halting the reaction, the optical absorption of the samples was measured at a wavelength of 410 nm using an ELISA reader (BioTek, Synergy HTX, USA) within 30 min. The calculation of the average absorption for two repetitions and the determination of the INDEX value were analyzed within the following ranges: If the value was less than 0.85, it was reported as negative. If it fell between 0.86 and 1.14, it was considered suspicious, and the test was repeated. If the sample remined suspicious after the second and third repetitions, it was reported as negative. Also, if its value was greater than 1.15, it was reported as positive. The range of positive samples is between 1.15 and 5.2.
Statistical analysis
The data were analyzed using SPSS23 software. A Pearson’s, Chi Square, and Fisher tests were conducted to compare the frequency of alleles and genotypes in tumor and control samples. A significance level of p < 0.05 was utilized.
Results
In this study, 120 blood samples from pregnant mothers referred to obstetricians and gynecologists in Sanandaj were analyzed. After culturing the samples in enriching media and plating them on Oxford media, 29 positive culture samples (24.16%) were found. After isolating the colonies, the strains were identified through biochemical tests, which revealed that all 29 strains were L. monocytogenes. This bacterium is Gram-positive, oxidase-negative, catalase-positive, MRVP-positive, oxidase-negative and does not ferment mannitol or xylose sugars. However, it does ferment glucose and rhamnose sugars.
Determination of IgM and IgG antibody titers
At this stage, all samples were examined. Table 1 displays the samples that tested positive for anti-Listeria antibodies, along with the corresponding culture results listed in column 4 for comparison.
Results of ELISA and culture of samples. The second column indicates the IgG titer, the third column shows the IgM titer, and the fourth column displays the results of the same sample based on culture tests on specific media.
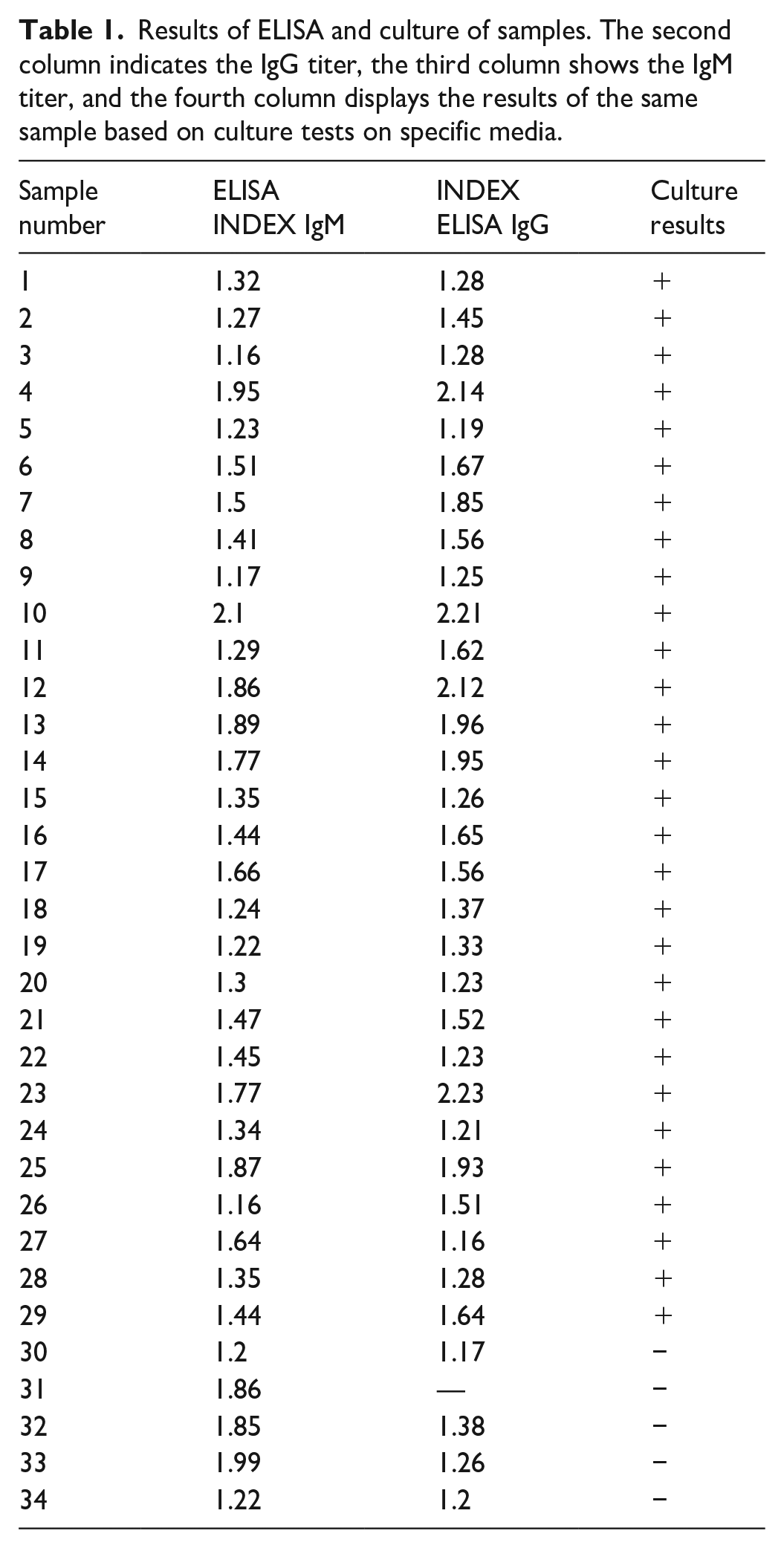
Out of the 120 serum samples, 34 (28.3%) were found to be positive for anti-Listeria antibodies according to the ELISA test. Interestingly, five of the samples that yielded negative culture results exhibited a positive anti-Listeria antibody titer. Additionally, one of the samples, had an IgG titer of zero.
Data analysis and sample specifications
Characteristics such as age, abortion history, and the number of abortions were collected for the samples using a demographic form. Out of the 120 samples, 26 were in the 15–20 age group, 79 samples were in the 20–30 age group, and 15 were in the 30–40 age group (Figure 1). The frequency of samples is displayed based on age ranges. A total of 65.83% of the samples fell within the 20–30 years range.
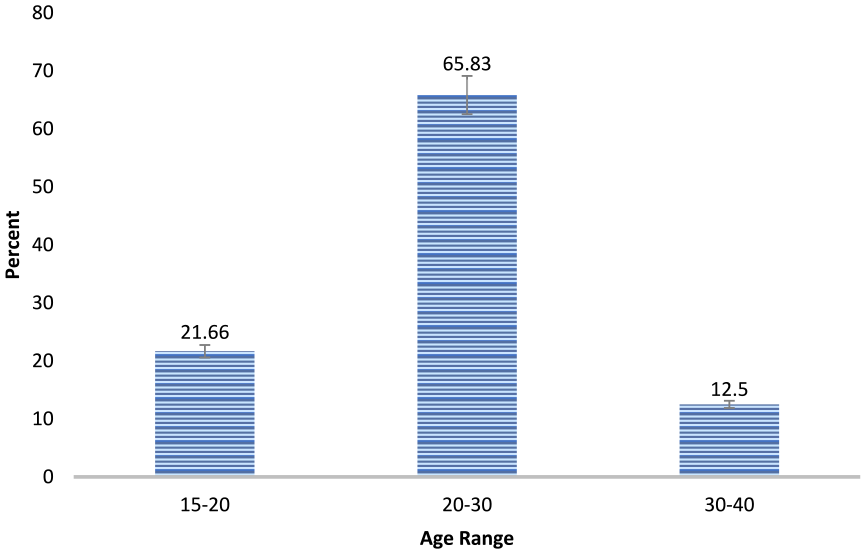
Frequency percentage of samples according to age ranges.
Also, out of the 120 samples, 69 women (57.5%) had undergone a previous abortion, while 51 women (42.5%) had no history of abortion (Figure 2). Also, 2% of the participants had previously undergone an abortion. Out of the surveyed individuals, 42.5% did not have a history of abortion, while 57.5% of the 69 individuals who did have a history of abortion had 1, 2, and 3 abortions, respectively. This breaks down to 45% (54), 10% (12), and 2.5% (3) (Figure 2).

The percentage of previous abortions in the samples; 42.5% had no history of abortion, while 45%, 10%, and 2.5% of the remaining 57.5% had 1, 2, and 3 abortions, respectively.
According to Table 2, the majority of abortions occurred among individuals aged 20–30 years, followed by those aged 30–40 years, with the lowest number of abortions observed in the15–20 age range.
Comparison of abortion and age range in the studied subjects.

Table 3 compares the number of abortions and Listeria infections based on the results of culture and ELISA tests. According to the data in the Table 3, in the culture method, out of 29 positive samples, 10 samples had no abortions, 16 samples had 1 abortion, 3 samples had 2 abortions, and no samples had 3 abortions.
Number of previous abortions based on L. monocytogenes identification tests.
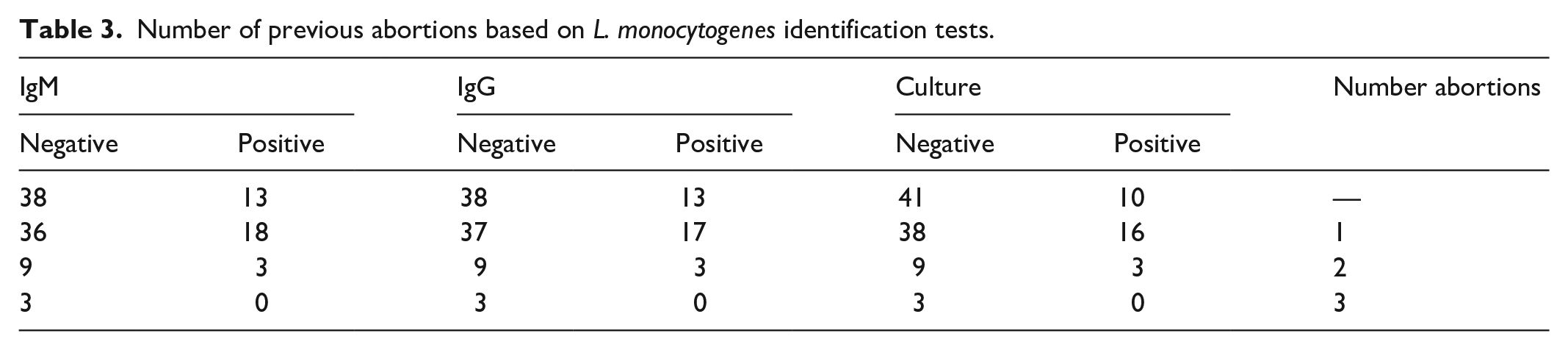
It has been observed that as the number of previous abortions increases, fewer individuals are infected with L. monocytogenes. Specifically, individuals who had undergone three abortions did not show any isolation of this bacterium. Using the ELISA method, 13 individuals with no history of abortion tested positive for both antibodies, while 38 individuals tested negative. Additionally, 17 individuals with 1 abortion, 3 individuals with 2 abortion, and 0 individuals with 3 abortions tested positive for the IgM antibody.
Determining of correlation between L. monocytogenes culture and ELISA results and abortion history
To determine the relationship between the culture and ELISA results of L. monocytogenes and the history of abortion, Pearson’s, Chi Square, and Fisher tests were conducted. The results of this test indicate that there is no significant relationship between the isolation of L. monocytogenes bacteria and the history of abortion. This is evident from the p value, which is equal to 0.316 and greater than 0.05 (Table 4). Therefore, the relationship is not considered significant. Also, the P value for positive test IgG and IgM ELISA were 0.672 and 0.552, respectively (Tables 5), and no significant relationship was observed between the ELISA results (both IgG and IgM) and abortion history (Table 6).
Results of Pearson’s chi Square test for determining the relationship between culture and abortion history.

0 cells (0.0%) have expected count less than 5. The minimum expected count is 12.33.
Computed only for a 2 × 2 table.
The output of Pearson’s chi Square test was used to determine the relationship between IgG ELISA and history of miscarriage.

0 cells (0.0%) have expected count less than 5. The minimum expected count is 14.03.
Computed only for a 2 × 2 table.
The output of Pearson’s chi square test is used to determine the relationship between IgM ELISA and history of miscarriage.

Discussion
Based on the results of culture and confirmation of biochemical tests, 29 samples (24.16%) tested positive. When examining the samples using the ELISA method, out of 120 serum samples, 34 (28.3%) were found to be positive for anti-Listeria antibodies. Interestingly, 5 of the samples that showed negative culture results had a positive anti-Listeria antibody titer. One of these samples even had a zero IgG titer (Table 1). Demographic information revealed that out of the120 samples, 26 belonged to the 15–20 age group, 79 belonged to the 20–30 age group, and 15 belonged to the 30–40 age group (Figure 1). Furthermore, out of the 120 samples, 69 (57.7%) women had a history of previous abortions, while 51 (42.5%) had no history of abortion (Table 2). Additionally, out of the 29 positive samples, 10 had no history of abortions, 16 had one abortion, and 3 had two abortions. None of the samples had a history of three abortions (Table 3).
This result confirms that the higher the number of previous abortions, the fewer people were infected with L. monocytogenes. These bacteria were not isolated in people with three miscarriages. In the ELISA method, 13 individuals tested positive for both antibodies, while 38 people tested negative among those without a history of abortion. Also, for one, two and three abortions, 17, 0 and 3 individuals tested positive for IgG antibodies and 18, 3 and 0 people were positive for IgM antibodies. The respective proportions of positive test in the culture method, IgG, and IgM ELISA were 0.316, 0.672, and 0.552. Since all three proportions are greater than 0.05, there is no significant relationship between infection with L. monocytogenes and abortion (Tables 4–6).
L. monocytogenes is a bacterium that causes miscarriage, with pregnant women accounting for approximately 30% of patients. Pregnant women are particularly vulnerable to Listeria infections, as these organisms attack the placenta and can lead to premature births or even fetal death.10–14
In a study conducted by Benshushan, 16 involving 65,022 pregnant women, it was found that 1100 of them and their babies had Listeria infection. Another study by Mylonakis et al., 17 revealed that Listeria infection can impact the fetus and result in miscarriage or premature birth. In this particular study, an indirect immunofluorescence method was utilized. Out of the 307 pregnant women who displayed no symptoms of infection, 207 experienced a miscarriage. The presence of high antibody titers in these women indicated that they had Listeria infection, which was the cause of their miscarriages. Administering treatment to the pregnant women with positive antibodies resulted in the successful birth of 152 healthy babies. 17
In Iran, a study conducted by Tahery et al., 18 evaluated the effect of L. monocytogenes on pregnant women and assessed the relationship between listeriosis and abortion in 204 women in the city of Jahrom for 1 year. The age groups most affected by contamination were 16–20 years and 25–30 years. 18 Contamination was determined based on the presence of IgG. In our study, IgG was found most frequently in the age group of 20–30 years, followed by the age group of 15–20 years. Only one positive sample was found in women over 30 years old. These findings were consistent with Tahery’s study. 18
Another study conducted by Jamshidi et al., 19 examined 250 women who had experienced spontaneous abortion and a group of 200 women who had a normal delivery. Of the women who had a miscarriage 35.6% tested positive for antibodies against L. monocytogenes, compared to 17.5% of the women who did not have a miscarriage. 19
In our study, we found a significant correlation between the presence of anti-Listeria antibodies and the absence of a history of abortion. This finding contrasts with the results of the Jamshidi 19 study. The difference may be attributed to variations in geographical location, appropriate treatment for individuals with a history of abortion or the smaller sample size in our study compared to the Jamshidi study. The primary methods or identifying L. monocytogenes are based on phenotypic characteristics. These methods involve examining biochemical, antigenic, and bacteriophage characteristics.20,21 However, these characteristics can be influenced by external conditions, developmental stage, and random genetic mutations. Consequently, the use of phenotypic tests may occasionally produce inaccurate results. 22
We did not use a specific formula to determine the sample size: sampling was based on convenience, which was one limitation of this study. Definite conclusions can be drawn from multiple large-scale studies. Collaborative studies may help overcome the limitations of individual studies.
Conclusion
Detecting L. monocytogenes in samples using standard culture methods can be challenging. The presence of other microflora in clinical samples reduces the ability to isolate L. monocytogenes, so enrichment media is used to increase isolation, especially in clinical samples. Cultivating these bacteria in enriched environments increases the speed of isolating and detecting bacteria, while also reducing the possibility of Listeria growth interfering with other bacteria in clinical samples. In this study, the clinical samples were cultured through direct enrichment in a specific environment. However, these methods are generally time-consuming and challenging, requiring at least 5 days to detect L. monocytogenes (with confirmatory tests). In some cases of contamination, immediate action is necessary; however, these methods may produce negative results if the sample is contaminated with a large amount of other bacteria, as they can interfere with the growth of L. monocytogenes. The primary methods of identifying L. monocytogenes are based on its phenotypic characteristics, such as its biochemical, antigenic, and bacteriophage characteristics. However, these characteristics can be altered by external conditions, developmental stages, and random genetic mutations leading to false results when using phenotypic tests. Therefore, it is recommended that pregnant women without a history of abortion undergo routine ELISA tests to detect anti-listeria antibodies and identify L. monocytogenes infection.
Footnotes
Acknowledgements
We would like to thank the participants for their participation in all the stages of the study.
Author contributions
FK developed the concept and design of the study, SV and MM collected and analyzed the data. All authors collaborated on preparing the first draft of the manuscript. FK and MM edited and reviewed the manuscript. All authors reviewed and approved the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This research was ethically approved by the Ethics Committee of the Sanandaj Branch, Islamic Azad University, Sanandaj, Iran (IR.2018.P.7366/100255). Also, all protocols involving human subjects comply with the requirements of the Declaration of Helsinki 23 of the Iranian Ministry of Health and Medical Education.
Informed consent
Verbal informed consent was obtained from all subjects before the study and was approved by the Institutional Review Board. Additionally, written informed consent was obtained from the Legally Authorized Representative of minor subjects (age <18 years) (two individuals).
Trial registration
Not applicable.
