Abstract
Objective:
Coronavirus disease causes a wide spectrum of clinical picture in pregnant women. There are still a few studies concerning laboratory parameters to differentiate the severity of coronavirus disease-19 in pregnant patients. This article aims to identify the cut-off on laboratory parameters between pregnant women with asymptomatic/mild/moderate and severe/critical coronavirus disease-19 illness.
Methods:
All coronavirus disease-19 pregnant women coming to Dr. Cipto Mangunkusumo hospital between January and August 2021 were recruited into this cross-sectional study. All data extracted from medical records were classified into demographic characteristics and laboratory parameters. We defined demographic characteristics as age, parity, systolic, and diastolic blood pressure, heart rate, temperature, respiratory rate, and oxygen saturation. Laboratory parameters measured in this study consisted of complete blood count, renal function test including urea, creatinine, liver function test including aspartate transferase, alanine transferase, infection marker test including procalcitonin, C-reactive protein, interleukin-6, cycle threshold values, ferritin, coagulation test including d-dimer, fibrinogen, random blood glucose, albumin, electrolytes. Data analysis was performed using SPSS statistics.
Results:
There were 159 cases assessed in this study consisting of 130 (81.8%) patients with asymptomatic/mild/moderate illness and 29 (18.2%) patients with severe/critical illness of coronavirus disease-19. Of 28 laboratory parameters, 12 of them were significant statistically; thus, it resulted to make a cut-off using receiver operating characteristic curve and show relative risk, and 95% confidence interval. In multivariate analysis that aspartate transferase, D-dimer, and albumin were laboratory parameters impacted to the severity of coronavirus disease-19 in pregnant women. The area under receiver operating characteristic curve was 0.834 (95% CI 0.736–0.932).
Conclusion:
Of all laboratory parameters, urea, aspartate transferase, alanine transferase, procalcitonin, C-reactive protein, lactate dehydrogenase (LDH), ferritin, D-dimer, random blood glucose, cycle threshold values, albumin, and chloride differs between asymptomatic/mild/moderate, and severe/critical illness coronavirus disease-19 in pregnant women; aspartate transferase, D-dimer, and albumin were laboratory parameters impacted the most to the severity of coronavirus disease-19 in pregnant women.
Introduction
World Health Organization (WHO) stated there have been 260,867,011 confirmed cases of COVID-19 including 5,200,267 deaths until the end of November 2021.
1
Pregnant women are susceptible to the virus and they have greater risk due to special physiological conditions. The rate of COVID-19 in pregnant women admitted to hospital for any reason was around 10%.
2
In Indonesia,
About two-thirds of pregnant women with COVID-19 have no symptoms at all, and most pregnant women who have symptoms only mild cold or flu-like symptoms. Pregnant women who got COVID-19 are slightly increased risk of becoming severely compared to non-pregnant women, particularly in the third trimester. 4 Since Delta variant identified in March 2021 in the United States, Delta variant is 60% more transmissible and resulted in higher numbers of cases, hospitalizations, and deaths. 5 Study by Wang et al. 6 compared to the beginning of COVID-19, fourth wave caused more likely symptomatic pregnant women and they required oxygen support more often.
During pregnancy, there are anatomical and physiological changes in order to nurture and accommodate the developing fetus. These have begun since conception and affect every organ system in the body. 7 It also impacts to laboratory parameters. A specific cut-off value of laboratory parameters in pregnant women with COVID-19 is not yet known. There are still few studies concerning laboratory parameters to differentiate asymptomatic/mild/moderate and severe/critical illness of COVID-19. 8 Meanwhile, clinical and laboratory parameters play a vital role for control and treatment of COVID-19 in pregnancy. 9 Therefore, this article aims to identify the cutoff on laboratory parameters between pregnant women with asymptomatic/mild/moderate and severe/critical COVID-19 illness.
Methods
Study design and sample recruitment
This article aims to identify the cutoff on laboratory parameters between pregnant women with asymptomatic/mild/moderate and severe/critical COVID-19 illness. All COVID-19 pregnant women coming to Dr. Cipto Mangunkusumo Hospital between January and August 2021 were recruited into this cross-sectional study. The minimum sample size needed to have a 95% confidence interval (95% CI) with margin of error 5% and COVID-19 prevalence in pregnant patient coming to hospital 10% was 139 subjects. Based on the calculation and justification of the sample size selected for this study, the author included total sampling with a total of 159 patients in the study divided into 130 patients in asymptomatic/mild/moderate illness and 29 patients in severe/critical illness through medical record, retrospectively. We excluded the patients who come as death on arrival due to COVID-19. This study has been accepted under ethical committee Faculty of Medicine Universitas Indonesia. Informed consent was not sought for this study because this is a retrospective study using medical records without any intervention to subjects.
Asymptomatic is defined as individuals who test positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) using a virologic test but have no symptoms that are consistent with COVID-19. Mild illness is defined as individuals who have any of various signs and symptoms of COVID-19 without shortness of breath, dyspnea, or abnormal chest imaging. Moderate illness is defined as individuals who show evidence of lower respiratory disease during clinical assessment or imaging and who have an oxygen saturation (SpO2) ⩾94% on room air at sea level. Severe illness is defined as individuals who have SpO2 <94% on room air at sea level with a ratio of arterial partial pressure of oxygen to fraction of inspired oxygen (PaO2/FiO2) < 300 mmHg, a respiratory rate >30 breaths/min, or lung infiltrates >50%. Critical illness is defined individuals who have respiratory failure, septic shock, and/or multiple organ dysfunction. 10
Outcome measures
All data extracted from medical records were classified into demographic and clinical characteristics also laboratory parameters. We defined demographic characteristics as age, parity, systolic, and diastolic blood pressure, heart rate, temperature, respiratory rate, and oxygen saturation. Laboratory parameters measured in this study consisted of complete blood count (CBC) including hemoglobin (g/dL), hematocrit (%), leucocyte (/uL), platelet count (/uL), different count (basophil/eosinophil/neutrophil/lymphocyte/monocyte), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), renal function test including urea (mg/dL), creatinine (mg/dL), liver function test (LFT) including aspartate transferase (AST) (U/L), alanine transferase (ALT) (U/L), infection marker test including procalcitonin (PCT; ng/mL), C-reactive protein (CRP; mg/L), interleukin-6 (IL-6; pg/mL), cycle threshold values (Ct values), ferritin (ng/mL), coagulation test including d-dimer (ug/L), fibrinogen (mg/dL), random blood glucose (RBG; mg/dL), albumin (g/dL), electrolytes including natrium (mEq/L), potassium (mEq/L), and chloride (mEq/L). All COVID-19 pregnant women were classified into two groups: asymptomatic/mild/moderate illness and severe/ critical illness. The primary outcome of this study was to identify the cutoff on laboratory parameters to differentiate between severity of COVID-19 in pregnant women.
The CBC including hemoglobin, hematocrit, and platelet was performed using impedance hematology (Sysmex XN3000®); meanwhile, white blood cell was measured using flowcytometry (Sysmex XN3000). Renal function tests such urea and creatinine were measured by urease kinetic and creatinine enzymatic (Abbott Architect c8000®). LFTs such as AST and ALT were measured by NADH (Abbott Architect c8000). PCT was measured by immunofluorescent sandwich assay (Brahms Kryptor®). CRP was measured by immunoturbidimetry/nephelometry (Cobas c311/ BN ProSpec®). IL-6 was measured by ECLIA (Roche Cobas e411®). Ferritin was measured by chemiluminescence microparticle immunoassay (Abbott Architect i4000®). Coagulation tests such as d-dimer and fibrinogen were measured by immunoturbidimetry and optomechanical coagulation (Stago Starmax/Sysmex CS-5100®). Electrolyte was measured by indirect/direct ion-selective electrode (Abbott Architect c8000/Nova pHox plus L®). Lactate dehydrogenase (LDH) was measured by lactate pyruvate methods (Abbott Architect c8000®). RBG was measured by hexokinase/G6PDH (Abbott Architect c8000®). Albumin was measured by Bromocresol Green (Abbott Architect c8000®). Ct value was measured by polymerase chain reaction (PCR; Sansure, PCR mastermix®).
Statistical analysis
Data analysis was performed using SPSS statistics, version 25 (SPSS Inc, Chicago, Illnois, USA). The Kolmogorov–Smirnov test was used to check the normal distribution of continuous variables. Descriptive statistics presented were mean (standard deviation) and median (min–max) for normal and abnormal distribution. T-test test and Mann–Whitney U-test was performed to compare normal and abnormal distribution among variables; respectively.
After obtaining the significant value, we analyzed each variable using receiver operating characteristic (ROC) curve. We determined the cut-off which revealed high specificity to determine the severe/critical illness of COVID-19 among pregnant women. Comparison of proportions was performed by Pearson’s chi-square (
Results
There were 159 cases assessed in this study consisting of 130 (81.8%) patients with asymptomatic/mild/moderate illness and 29 (18.2%) patients with severe/critical illness of COVID-19. The characteristics of study population were described in Table 1. Bivariate analysis was performed to evaluate the significant difference in the severity of COVID-19 pregnant women (Table 2). Of 28 laboratory parameters, 12 of them were significant statistically; thus, it resulted to make a cut-off using ROC curve and show relative risk (RR) and 95% confidence interval (95% CI) (Tables 3 and 4). The best cut-off points used with the sensitivity ranged 50.0%–75.0% and specificity ranged 67.5%−89.6%.
Characteristics demographic of COVID-19 pregnant women.
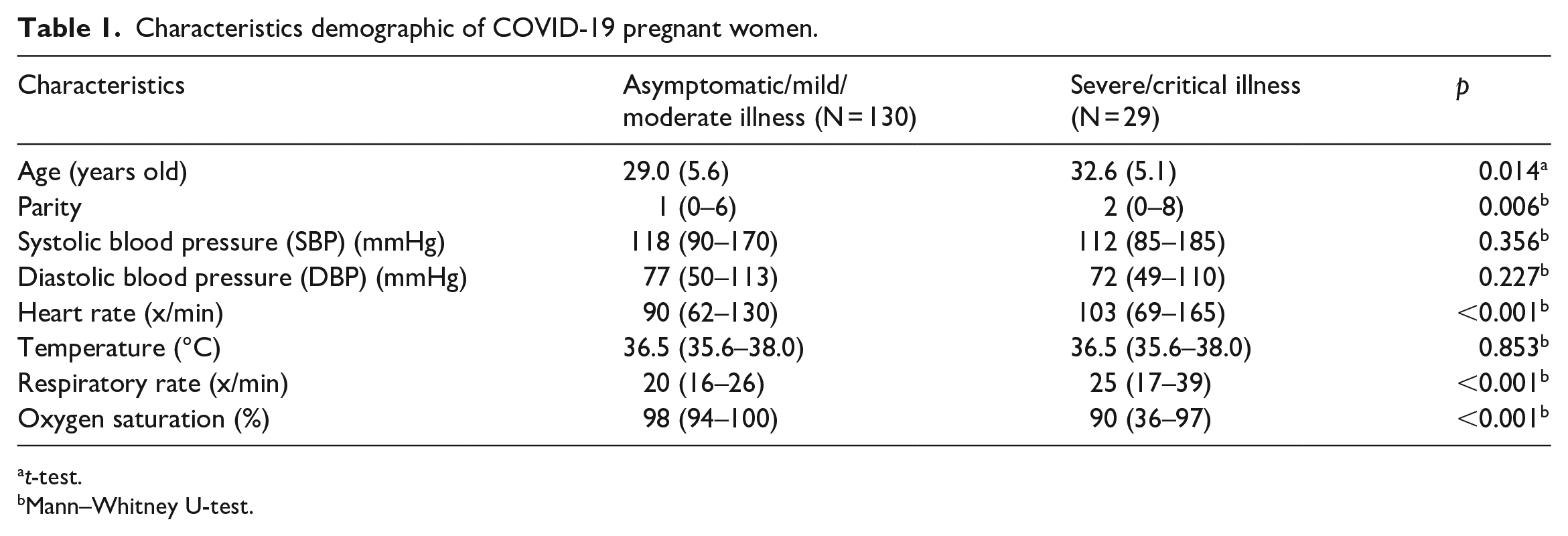
Mann–Whitney U-test.
Laboratory parameters and severity of COVID-19 pregnant women.

Mann–Whitney
Determining the cut-off of laboratory parameters to differentiate the severity of COVID-19 among pregnant women.

Abbreviation: AUC = area under receiver operating characteristic curve.
Bivariate analysis of the cut-off on clinical and laboratory parameters to differentiate the severity of COVID-19 among pregnant women.

Fischer Exact test.
Chi-square test.
Furthermore, the variables with
Discussion
This article aims to evaluate laboratory parameters in different severity of COVID-19 among pregnant women. The cutoff is used to differentiate between asymptomatic/mild/moderate and severe/critical illness COVID-19. Of 28 laboratory parameters assessed in our study, we found only12 laboratory parameters were significantly different between them. After obtaining a cutoff, several markers which elevated in severe/critical illness COVID-19 in pregnancy include urea, AST, ALT, PCT, CRP, LDH, ferritin, d-dimer, random blood glucose, and Ct values. Meanwhile, albumin and chloride decreased in severe/critical illness COVID-19.
There is renal dysfunction in COVID-19 patients; however, the mechanism is still unknown. It was speculated that SARS-CoV-2 may directly damage kidney tissue through binding to the ACE2 receptor. In our case, only urea had an association with severe/critical illness COVID-19 in pregnant women with cutoff 17.85 mg/dL. Meanwhile, there was no significant association in creatinine level. In study by Xiang et al. 11 renal dysfunction was more in severe patients than in mild patients. Renal dysfunction is positively correlated with the severity of COVID-19 patients on admission.
The liver plays an essential role in host defense against micro-organisms and involved in most systemic infections because it obtains a dual blood supply from systemic and portal circulation. Previous studies reported that 14%–76% of COVID-19 patients showed abnormal LFTs with increased levels of serum ALT and AST.12,13 The pathogenesis is still unknown; however, it suggested that liver damage in COVID-19 patients might be directly induced by liver cells’ viral infection. Apart from that the treatment of COVID-19 including antiviral drugs (lopinavir/ritonavir, hydroxychloroquine), antibiotics, antipyretics (acetaminophen), corticosteroids, and herbal medicine are potentially hepatotoxic. 14 In our cases, we determined that the cutoff of AST and ALT were 34.5 and 27.5 U/L between asymptomatic/mild/moderate and severe/critical illness COVID-19 in pregnant women. They were slightly increased with the RR 4.335 (95% CI: 2.219–8.466) and 3.317 (1.732–6.353) respectively. It was similar with study by Can et al. 15 which showed that AST and ALT elevations were generally mild (1-2x the upper limit of normal). Zhao et al. 16 concluded that abnormal LFTs in mild COVID-19 patients may not be associated with inflammatory status.
Some inflammatory biomarkers have been associated with the severity of COVID-19 including leukocytes count (lymphocyte or an elevated NLR), CRP, PCT, LDH, ferritin, and d-dimer.17,18 In our study, only CRP, PCT, LDH, ferritin, and d-dimer which has been considered differently between asymptomatic/mild/moderate and severe/critical illness COVID-19 in pregnant women. The cut-off value for CRP, PCT, LDH, ferritin, and d-dimer were 39.6 mg/L, 0.42 ng/mL, 316.0 U/L, 305.1 ng/mL, and 2730 ug/L; respectively. CRP can be used to monitor the evolution of COVID-19 disease. Study by Lombardi et al. 18 concluded that CRP was related with severity of disease or mortality. As the 116-amino acid precursor of calcitonin hormone, PCT can be synthesized in many extra thyroid tissues during bacterial infection, which is mediated by increased concentrations of tumor necrosis factor alpha (TNF-a) and interleukin (IL)-6. 19 Previous studies stated that PCT is positively associated with the severity of COVID-19.20,21 Hu et al. 22 showed that mean serum PCT levels were approximately four times higher in severe patients than in moderate patients and eight times higher in critical patients than moderate patients. It is corresponding to our result that it increased four times.
A pooled analysis by Henry et al. 23 figured out that severe infection causes cytokine-mediated tissue damage and LDH release. Elevated LDH values are associated with worse outcomes in COVID-19 patients. There was a >six-fold increase in odds of severe disease and a >16-fold increase in odds of mortality in patients with elevated LDH. Serum ferritin levels may suggest not only as an iron overload state, but also a marker of inflammatory, autoimmune, infectious, or malignant conditions. 24 The effect of COVID-19 on the immune system suggests that ferritin levels are also elevated in severe compared to moderate cases in 21 patients with COVID-19 in China. 25 D-dimer as a fibrin degradation product is used as a biomarker for thrombotic disorders. Before COVID-19, d-dimer is not considered as a useful biomarker for bacterial or viral infection. 26 However, elevated D-dimer and thrombotic complications have been reported in COVID-19 patients. Apart from that, it is associated with disease severity and hospital mortality. In non-pregnant COVID-19 patients, D-dimer level was 0.58 ug/mL in 1551 patients with mild disease and 3.55 ug/mL in 708 patients with severe disease. 27 Study by Poudel et al. 28 revealed that ROC curve for d-dimer among non-pregnant patient was 0.807 (95% CI 0.728–0.866; p < 0.001).
Blood glucose has an association with severity of COVID-19 patients. The underlying mechanism is still unclear. SARS-COV-2 relies on the binding to membrane angiotensin-converting enzyme 2 (ACE-2) receptor to enter pulmonary cells in the lungs. Patients with diabetes or uncontrolled glucose level might have impaired innate immunity due to dysfunction of macrophage and lymphocytes, which may lead to an increased risk of septic shock and multiple organ failures. 29 In our case, RBG 98.5 mg/dL was used as a cutoff to differentiate asymptomatic/mild/moderate and severe/critical illness COVID-19 in pregnant women with RR 2.399 (95% CI 1.232–4.668). Study by Wang et al. 30 showed that patients with an elevated level of blood glucose >6.1 mmol/L or 110 mg/dL had a 58% higher risk of disease progression and 3.22-fold higher risk of fatality. This is consistent with previous reports that a high glucose level contributes to the development of acute respiratory distress syndrome in COVID-19 patients.31,32
Hypoalbuminemia is correlated with the severity of COVID-19. Although there is no hepatocellular injury in COVID-19, hypoalbuminemia happens due to intense systemic inflammation which increased capillary permeability. 33 In our case, hypoalbuminemia with level less than 3.18 g/dL increased risk for severe/critical illness of COVID-19 among pregnant women. Study by Huang et al. 34 revealed that serum albumin level < 35 g/L at presentation independently increased risk of death in COVID-19 by at least six-fold.
Currently, RT-PCR methods for diagnosis of COVID-19 are able to give an estimate of the viral load. Ct values of PCR reaction correlate inversely with viral loads and higher viral loads have been seen to associate with disease severity and infectivity. 35 In our case, the Ct value of 31.0 was used as a cutoff for severe/critical illness of COVID-19 in pregnant women. Study in New York showed that in-hospital mortality was 35% with a high viral load (Ct < 25; n = 220), 18% with a medium viral load (Ct 25–30; n = 216), and 6% with a low viral load (Ct > 30; n = 242; p < 0.001). However, many studies did not find any difference between viral loads as determined by Ct values between symptomatic and asymptomatic patients.36,37
In study by Sahin et al., 8 CRP, NLR, ferritin, and AST are related with the severity of COVID-19 in pregnant women. They stated that combination of CRP and ferritin had higher sensitivity and negative predictive value compared with using other tests alone. Apart from that coagulation markers are not useful in predicting disease severity in pregnancy. It differs with our study that AST, D-dimer, and albumin were laboratory parameters impacted the most to the severity of COVID-19 in pregnant women.
The limitation is only few numbers of subjects included in this study because the trend of COVID-19 is varied from time to time in Indonesia. Apart from that due to cross-sectional study, we cannot predict the outcome from the severity of COVID-19. Meanwhile, the study concerning laboratory parameters in COVID-19 pregnant patients in the world is still limited; therefore, this study is still new for the obstetrician’s knowledge in facing COVID-19 pregnant patients.
Conclusion
Of all laboratory parameters, urea, AST, ALT, PCT, CRP, LDH, ferritin, D-dimer, random blood glucose, Ct values, albumin, and chloride differs between asymptomatic/mild/moderate and severe/critical illness COVID-19 in pregnant women; AST, D-dimer, and albumin were laboratory parameters impacted the most to the severity of COVID-19 in pregnant women.
Footnotes
Acknowledgements
The authors thank the Department of Obstetrics and Gynecology, Dr. Cipto Mangunkusumo Hospital, Jakarta who has supported in this manuscript preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval has been accepted by the Ethics Committee of the Faculty of Medicine, University of Indonesia under KET-528/UN2.F1/ETIK/PPM.00.02/2021
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for this study because this is a retrospective study using medical records without any intervention to subjects.
