Abstract
Background:
Urinary tract infections caused by extended-spectrum beta-lactamase organisms pose a significant concern worldwide. Given the escalating prevalence of drug resistance and the limited data on the effectiveness of oral antibiotics in treating these infections, this study aimed to assess the clinical outcomes in adult patients with extended-spectrum beta-lactamase urinary tract infections treated with oral antibiotics.
Methods:
A retrospective observational cohort study was conducted at King Abdulaziz Medical City, Saudi Arabia, from January 2018 to December 2021. It included patients ⩾18 years with complicated or uncomplicated urinary tract infections from extended-spectrum beta-lactamase Enterobacterales and treated with oral antibiotics as step-down or mainstay therapy. All-cause clinical failure within 30 days post-discharge was evaluated as the efficacy outcome. Statistical analyses were performed using SPSS software.
Results:
Out of 643 screened patients, 152 patients met the inclusion criteria. The patients were divided into oral step-down therapy (51.3%) and oral-only (48.7%) groups. The majority (69.1%) were females, with a mean age of 62 years. Complicated urinary tract infections were diagnosed in (75.5%) of cases, and the predominant pathogen was E. coli (79.6%). Clinical failure was observed in 23.1% in the oral step-down group and 13.5% in the oral-only group, with no significant difference (p = 0.128). Total antibiotics duration was significantly lower in the oral-only group (8 days vs. 12.2 days; p < 0.001). Binary logistic regression identified elder age, diabetes mellitus history, and prior extended-spectrum beta-lactamase infection as predictors of clinical failure.
Conclusion:
This study suggests that both step-down or primary oral antibiotic treatment yielded similar clinical outcomes in managing patients with extended-spectrum beta-lactamase urinary tract infections. Further prospective studies are required to validate these findings.
Graphical abstract

Introduction
Urinary tract infections (UTIs) caused by extended-spectrum beta-lactamase (ESBL)-producing Enterobacterales (ESBL-E) are a major public health concern worldwide. 1 These pathogens are responsible for numerous UTI cases in both community and hospital settings. 2 In Saudi Arabia, ESBL-producing Escherichia coli (E. coli) accounts for approximately 33% of urine isolates.3,4
ESBL genes are highly prevalent in Enterobacterales such as E. coli, Klebsiella pneumoniae, and Proteus mirabilis, with these bacteria being among the most commonly isolated ESBL producers. 5 ESBL, as a plasmid-mediated enzyme, can hydrolyze a wide range of beta-lactam antibiotics, including most penicillin, cephalosporins, and monobactams, which limit the treatment options for ESBL infections. However, non-beta-lactam antibiotics, including fluoroquinolones, trimethoprim-sulfamethoxazole (TMP/SMX), and aminoglycosides, remain unaffected by ESBL enzymes.5,6
According to the Infectious Diseases Society of America (IDSA) guidelines, the selection of empiric regimens should be guided by institutional susceptibility patterns and encourage urine cultures to be obtained for patients at risk of UTIs with multidrug-resistant (MDR) pathogens.5,6 The rising incidence of MDR infections complicates the selection of antibiotics, as ESBL-E pathogens are resistant to multiple oral antibiotics, leading to the need for parenteral options, mostly carbapenems. 5 However, parenteral antibiotics are associated with higher healthcare costs, a greater risk of complications, and reduced patient satisfaction. 7 As a result, there is a need to explore alternative treatment strategies to reduce the reliance on parenteral therapy. The current literature offers limited insight into the efficacy of oral antibiotics, particularly beyond fosfomycin and for complicated UTIs (cUTIs).8–11 Moreover, the effectiveness of oral antibiotics in our population remains understudied. Considering this knowledge gap, our study aimed to evaluate the clinical outcomes in adult ESBL UTI patients treated with oral antibiotics, either exclusively or after initial empiric intravenous (IV) therapy.
Methods
Study design and setting
A retrospective observational cohort study was conducted at King Abdulaziz Medical City, a tertiary academic hospital in Riyadh, Saudi Arabia. All hospital or emergency department (ED) admitted patients, who received oral antibiotics for UTIs caused by ESBL-E between January 2018 and December 2021 were identified using the hospital reporting system. The study was approved by the Institutional Review Board at King Abdullah International Medical Research Center (NRC22R/051/01). Patients were included if they were >18 years, diagnosed with uncomplicated or complicated UTIs (cystitis or pyelonephritis) due to an ESBL organism, and treated with oral antibiotics as step-down or mainstay therapy. Patients were classified to have complicated UTIs if they were male, pregnant, immunocompromised, had renal transplant, had anatomical abnormalities, or had a urinary catheter. For all included patients, a urine sample was collected, and a microbiological study was performed at the time of patient admission (or the time of symptom initiation for already-admitted patients). Patients with bacterial infections other than UTIs, polymicrobial infections, those discharged on inactive antibiotics against ESBL or prophylactic antibiotics for recurrent UTIs after completing the regimen, and those with incomplete medical records were excluded. Patient demographics, comorbidities, laboratory results, microbiology data, antibiotics regimen, duration of treatment, and clinical outcomes were collected. The primary outcome was a composite of all-cause clinical failure, defined as hospital readmission, clinic or emergency department visits, or the need for a new prescription for antibiotics due to recurrent or worsening UTI within 30 days of hospital discharge. Secondary outcomes included the individual components of the primary outcome and the duration of therapy, calculated from the first day of active therapy.
Study groups
The patients were divided into two groups. The first group was the oral step-down group, which included patients who transitioned from IV to oral antibiotics during their therapy. The second group was the oral-only group, which consisted of patients who received oral antibiotics for the entire duration of their treatment. The selection of either approach was based on the clinical judgment of the providers, taking into consideration factors such as the admission status of the patients, severity of illness, previous history of urine culture, and the ability of patients to take oral medications. The antimicrobial susceptibility test (AST) results of the infecting microorganism were not immediately available to the clinicians at the initiation of treatment. Patients were started on empirical therapy based on treatment guidelines and the best clinical judgment of the healthcare providers influenced by patient-related factors. Once susceptibility results became available, adjustments to the antibiotic regimen were made. However, for patients who were not hospitalized or were discharged before the availability of AST results, the antibiotic regimen might not have been modified.
Statistical analysis
The data were summarized using descriptive statistics. Categorical variables were compared using a Chi-square test, while continuous variables were analyzed using an independent t-test. Binary logistic regression was used to calculate the odds ratio and investigate the risk factors associated with the primary outcome based on patient characteristics. All statistical tests were two-sided, and p-values < 0.05 were considered statistically significant. All analyses were performed using SPSS software (IBM Corporation, Version 29, Armonk, NY, USA).
Results
Of the initial 643 screened patients, a total of 152 patients were included in this study (Figure 1). The oral step-down group comprised 78 patients (51.3%), and the oral-only group comprised 74 patients (48.7%). The majority of these patients were female (69.1%), with a mean age of 62 years (range: 18–96 years). Most patients had cUTIs (75.5%), with no instances of pyelonephritis diagnosed. The most prevalent comorbidities were cardiovascular diseases (67.8%), diabetes mellitus (63.8%), and the presence of a urinary catheter (39.5%). Sixty patients (39.6%) had a previous history of ESBL infection. The most commonly isolated pathogens from urinary cultures were E. coli (79.6%) and Klebsiella species (19.7%). Table 1 presents the details of the baseline characteristics.
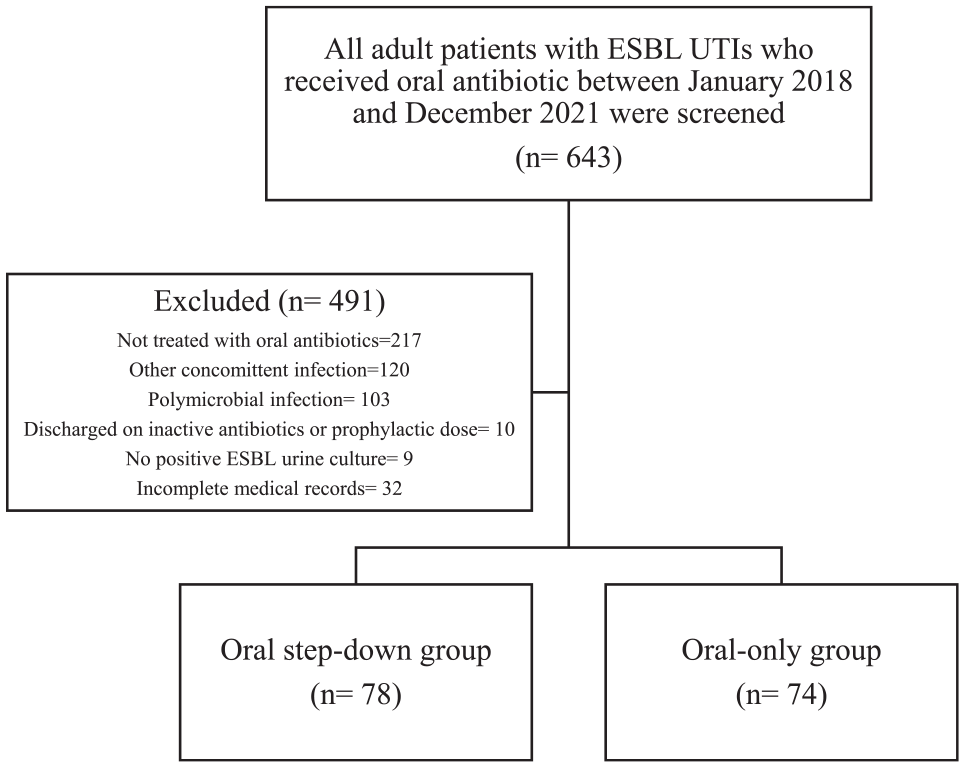
Flow diagram of study enrolment.
Baseline characteristics of the included patients.
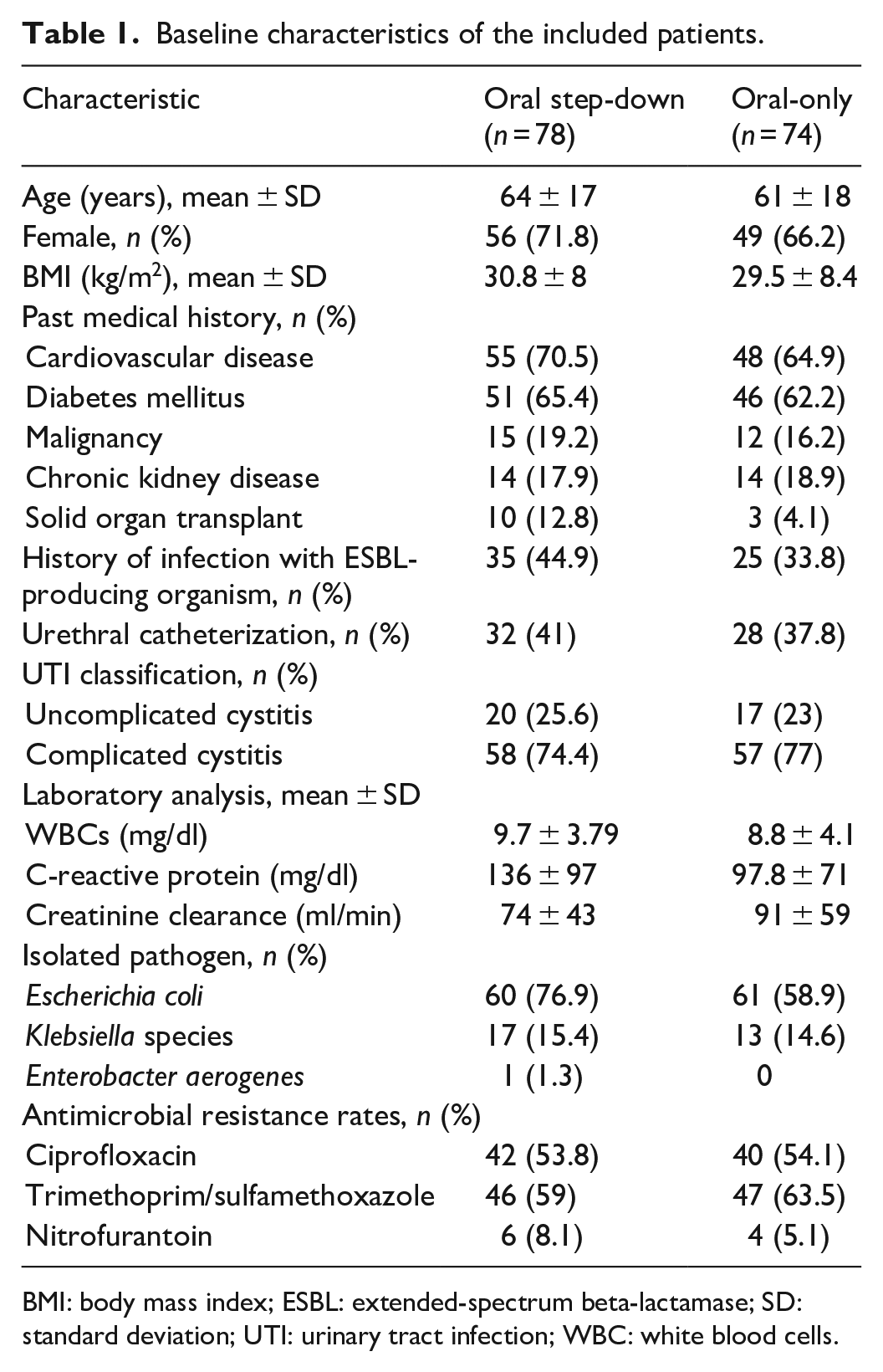
BMI: body mass index; ESBL: extended-spectrum beta-lactamase; SD: standard deviation; UTI: urinary tract infection; WBC: white blood cells.
Regarding the sensitivity profiles, results showed that nitrofurantoin had the highest sensitivity rate (94.21%) against E. coli isolates, followed by TMP/SMX (42.15%). On the other hand, ciprofloxacin displayed the lowest sensitivity rate (32.3%) against E. coli isolates. Conversely, for Klebsiella species, ciprofloxacin showed the highest sensitivity (50%), followed by nitrofurantoin (33.3%) and TMP/SMX (20%) (Supplemental Table S1).
Table 2 presents details about the management and antibiotic selection. In the step-down group, the most frequently prescribed empirical IV antibiotics were meropenem (55.1%), followed by ciprofloxacin (24.4%) and piperacillin–tazobactam (23.1%). Nitrofurantoin (43.6%) and ciprofloxacin (37%) were the most frequently prescribed oral antibiotics as step-down therapy in this group. This group’s average IV and oral antibiotics duration were 4.6 ± 2.6 and 7.6 ± 3.5 days, respectively. The average of the total duration of IV plus oral antibiotics was 12.2 ± 4 days. The most commonly prescribed oral antibiotics in the oral-only group were ciprofloxacin (50%) and nitrofurantoin (40.5%). The average duration of antibiotics in the oral group was 8.3 ± 4.1 days.
Treatment characteristics of the included patients.
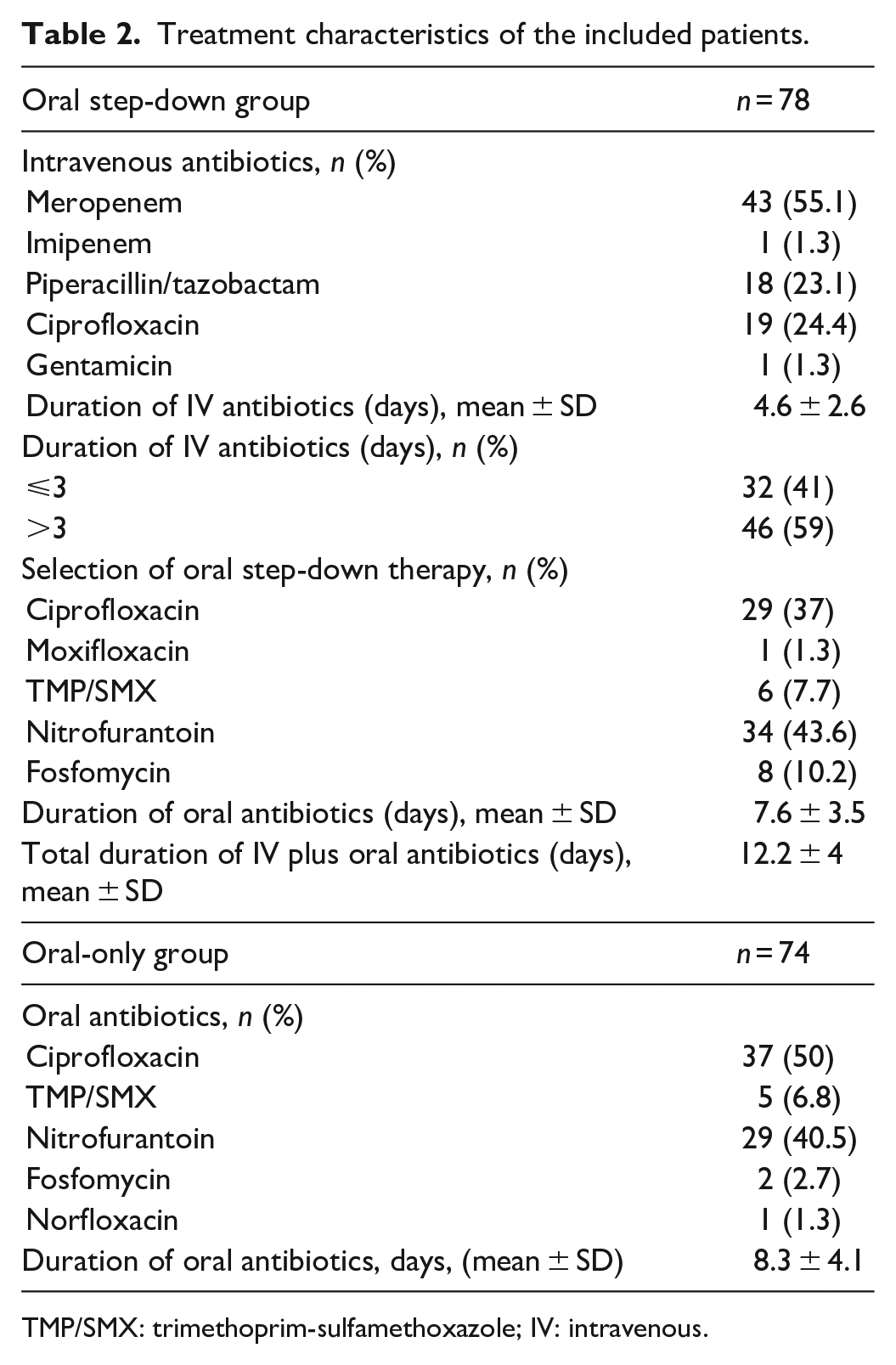
TMP/SMX: trimethoprim-sulfamethoxazole; IV: intravenous.
The composite outcomes of clinical failure occurred in 18 patients (23.1%) in the oral step-down therapy group and 10 patients (13.5%) in the oral group with non-statistically significant difference (p = 0.128) (Table 3). All secondary outcomes were non-statistically significant between the two groups except for the ED visit within 30 days due to UTI (17.9% vs. 6.8%; p = 0.037) and total antibiotics duration (12.2 days vs. 8 days; p < 0.001) were significantly higher in the oral step-down group. The binary logistic regression analysis results for clinical failure showed that advancing age, history of diabetes mellitus, and previous ESBL infection were significant predictors of clinical failure. However, receiving IV antibiotics or a longer duration of antibiotics (>7 days) did not significantly reduce the risk of clinical failure (Table 4). A subgroup analysis of patients with urinary catheters showed no significant difference in clinical outcomes or duration of therapy when compared to patients without urinary catheter (Supplemental Table S2 and S3).
Treatment outcomes.

ED: emergency department; UTI: urinary tract infection.
Logistic regression analysis for risk factors of clinical failure in patients with ESBL UTI.

BMI: body mass index; ESBL: extended-spectrum beta-lactamase; CI: confidence interval; IV: intravenous; OR: odds ratio; UTI: urinary tract infection.
OR estimates with values of p < 0.05 were considered statistically significant.
Discussion
In this retrospective study, we assessed the clinical outcomes in patients with UTIs caused by ESBL-E, focusing on using oral antibiotics either exclusively or after initial empiric IV therapy. Our study findings suggest that using oral antibiotics as a mainstay of therapy for treating ESBL UTIs did not result in worse clinical outcomes and may be considered an effective alternative to IV therapy.
In our study, E. coli was the predominant pathogen identified in approximately 80% of the patients, consistent with the common pathogen responsible for UTIs. 6 Notably, the majority of our ESBL E. coli isolates (94%) tested susceptible to nitrofurantoin, while only approximately 33% of ESBL Klebsiella isolates demonstrated susceptibility to nitrofurantoin. Similarly, a study published in 2018 assessed the susceptibility of 464 urine cultures identifying ESBL-producing organisms and reported that 93.7% of 384 ESBL E. coli isolates were susceptible to nitrofurantoin, and 57.7% of 80 ESBL Klebsiella isolates displaying susceptibility to nitrofurantoin. 8 Several in vitro studies were done to assess the antibiotic susceptibility of ESBL-E.9–13 The results showed that Fosfomycin, nitrofurantoin, and pivmecillinam have the highest sensitivity among other oral antibiotics, such as TMP-SMX and ciprofloxacin.
The efficacy of oral antibiotics for treating UTIs caused by ESBL-E has yet to be fully established, and there are currently no randomized controlled trials comparing oral agents for the treatment of ESBL UTIs. However, the clinical outcomes of using oral antibiotics in patients with ESBL UTIs, such as fosfomycin,14–18 nitrofurantoin, 19 TMP-SMX, 20 and pivmecillinam 21 were evaluated in small observational studies. These studies found that these agents could be effective oral alternatives for the treatment of UTIs caused by ESBL-E. Of note, pivmecillinam is currently unavailable in the Saudi market.
The most recent IDSA guideline recommends several treatment oral options for patients with uncomplicated cystitis caused by ESBL-E, including nitrofurantoin and TMP-SMX as preferred treatment choices, with ciprofloxacin, levofloxacin, and fosfomycin as alternative options. 5 Furthermore, the preferred treatment options for complicated cystitis caused by ESBL-E are TMP-SMX, ciprofloxacin, or levofloxacin. Approximately three-fourths of the patients in our study had complicated cystitis, in the step-down group patients were primarily treated with meropenem before transitioning to nitrofurantoin or ciprofloxacin orally. In addition, approximately half of the patients in the oral-only group received ciprofloxacin, followed by nitrofurantoin in approximately 40% of the cases. The therapeutic approaches in our study align with current IDSA clinical recommendations for managing ESBL UTIs; however, nitrofurantoin was used for managing complicated cystitis in both groups, which is currently not recommended by the current guideline. The use of nitrofurantoin in patients with lower UTIs due to ESBL has been evaluated before in a small sample retrospective study, which included 75 patients, 14 of whom had complicated lower UTIs. 19 The results of this study found similar clinical and microbiological success rates in patients with or without complicating factors.
The all-cause clinical failure did not differ significantly in our study between the oral step-down and the oral-only groups, suggesting that the oral-only treatment approach might be a reasonable strategy. Our findings align with a recent observational study that investigated the intravenous-only treatment compared to those who were switched from intravenous to oral antibiotics, highlighting the feasibility of an oral treatment strategy. 22 They reported 28.4% clinical failure in the intravenous-only group compared to 27.6% in the intravenous to oral group (p = 0.91). In addition, the antibiotic course was shorter in our cohort of patients who received oral-only therapy as compared to the oral step-down group (8 vs. 12.2 days, p < 0.001); however, this finding might be related to the oral step-down group being sicker, which we did not evaluate comprehensively. Gamble et al. 22 also found a reduction in antibiotic duration by 2 days in patients treated with intravenous then oral therapy compared to the intravenous-only group. Moreover, we observed a reduction in the number of emergency visits due to UTI within 30 days in the oral-only group compared to the oral step-down group, which further supports better outcomes with the oral-only treatment approach. Sadyrbaeva-Dolgova et al. 23 have also demonstrated that de-escalation of intravenous antibiotics to oral options in complicated UTIs was beneficial and led to a reduction in hospital length of stay, in-hospital mortality, duration of carbapenems use, and total duration of antibiotic therapy.
In our evaluation, cases of clinical failure were linked to many contributing risk factors in a logistic regression analysis showing that elderly patients (i.e., >65 years old), patients with diabetes mellitus, and patients with a previous history of ESBL had more chances for clinical failures. These risk factors are known to be linked to the development of ESBL infections.24,25 Hence, for patients exhibiting these risk factors, it may be advisable to thoroughly assess the suitability of an oral antibiotic regimen instead of opting for IV treatment.
To the best of our knowledge, this is the first study that assesses the feasibility of managing ESBL UTIs using an exclusively oral regimen in comparison to an oral step-down regimen while also incorporating a substantial number of complicated UTI cases. This study fills a significant gap in the existing literature and introduces novel avenues for tackling this frequently encountered situation in clinical practice and advancing efforts in antimicrobial stewardship.
This study comes with several limitations. Due to the study’s retrospective design, there are risks for confounding and bias, which we attempted to control using logistic regression. The severity of the UTIs and the need for ICU stay were not evaluated, which may have limited the distinction between severe and mild UTI cases. In addition, in this study, we did not calculate the sample size; however, we retrieved all available data for the target population. This approach could potentially impact the generalizability and statistical power of the findings. Nevertheless, it is worth noting that the number of patients included in our study is comparable to the number reported in the currently available literature addressing this issue. Lastly, the fosfomycin susceptibility was not reported in this study due to the unavailability of this data in the electronic medical records.
Conclusion
Our study findings emphasize the practicality of adopting oral antibiotics as a primary approach to managing UTIs caused by ESBL-E. This adds to the growing evidence that supports the use of oral antibiotics to treat UTIs caused by ESBL-producing organisms. Nevertheless, additional research is warranted to compare the effectiveness and safety of various antibiotic options and determine the optimal treatment strategy tailored to specific patient comorbidities. Moreover, it remains imperative to investigate preventive measures to mitigate the emergence and persistence of ESBL-producing pathogens, to reduce the prevalence of UTIs caused by these organisms.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241259993 – Supplemental material for Effectiveness of oral antibiotics in managing extended-spectrum B-lactamase urinary tract infections: A retrospective analysis
Supplemental material, sj-docx-1-smo-10.1177_20503121241259993 for Effectiveness of oral antibiotics in managing extended-spectrum B-lactamase urinary tract infections: A retrospective analysis by Sumaya N Almohareb, Atheer Aldairem, Nada Alsuhebany, Omar A Alshaya, Dimah Aljatli, Hesah Alnemer, Khlood Almutairi, Norah Aljammaz and Shuroug A Alowais in SAGE Open Medicine
Footnotes
Authors’ contributions
Conceptualization: Sumaya N Almohareb, Shuroug A Alowais; Methodology: Sumaya N Almohareb, Atheer Aldairem, Shuroug A Alowais, Omar A Alshaya; Acquisition and analysis of the data: Atheer Aldairem, Dimah Aljatli, Hesah Alnemer, Khlood Almutairi, Norah Aljammaz; Writing – original draft preparation: Sumaya N Almohareb, Atheer Aldairem, Nada Alsuhebany, Omar A Alshaya, Shuroug A Alowais; Writing – review and editing: Sumaya N Almohareb, Atheer Aldairem, Nada Alsuhebany, Omar A Alshaya, Shuroug A Alowais, Dimah Aljatli, Hesah Alnemer, Khlood Almutairi, Norah Aljammaz; Supervision: Sumaya N Almohareb; All authors have read and agreed to the published version of the manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Ethical approval was received from the King Abdullah International Medical Research Center Institutional Review Board, Riyadh, Saudi Arabia (NRC22R/051/01) with the need for written consent waived by the ethical committee due to the retrospective nature of the study.
Informed consent
The need for written consent waived by the ethical committee due to the retrospective nature of the study.
Consent for publication
Not applicable.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
