Abstract
Introduction:
Urinary tract infections are prevalent among pregnant women and can lead to serious maternal and neonatal complications. Hypertensive disorders of pregnancy, a leading cause of maternal morbidity, may be associated with urinary tract infections. This study investigates whether bacteriuria detected via routine urinalysis, a standard screening in Indonesia, contributes to hypertension risk during pregnancy, aiming to enhance clinical management and screening protocols.
Aim of Study:
To evaluate the association between bacteriuria detected through routine urinalysis and hypertension during pregnancy in women at Margono Hospital, Purwokerto.
Methods:
This study was conducted at Margono Hospital, Indonesia; the study included all pregnant women who delivered in 2022–2023, applying specific exclusion criteria. Urine samples were analyzed for bacteriuria, and statistical analysis determined the prevalence ratio and odds ratio for the correlation of bacteriuria and risk of hypertension.
Results:
Hypertension prevalence was 1.52 times higher in patients with bacteriuria. The odds ratio was 1.379 (confidence intervals: 0.751–2.532) with a statistically significant p-value of <0.003. These findings indicate a significant association (p < 0.05) between positive bacteriuria and increased risk of hypertension and preeclampsia.
Conclusion:
This study highlights a significant association between bacteriuria and hypertension in pregnancy, including severe complications like impending eclampsia. Pregnant women with urinary tract infections should be closely monitored for preeclampsia. Further research is needed to explore the mechanisms linking urinary tract infections and hypertension during pregnancy.
Introduction
The global prevalence of hypertensive disorders of pregnancy has shown an increase from 16.30 to 18.08 million, reflecting a 10.92% rise from 1990 to 2019. 1 Interestingly, Southeast Asia showed a decreased prevalence from 1.6 to 1.4 million during the same period. Despite this global trend, Indonesia, as a developing nation, grapples with the significant challenge of high maternal mortality rates.2,3 An average prevalence of 22.1% was found for hypertension in pregnancy among 3219 cases out of 17,771 deliveries, according to data from a multicenter trial at 11 tertiary institutions throughout Indonesia. 4 Another large-scale study using national health data and 9024 pregnant women from all provinces in Indonesia found that the prevalence of hypertension during pregnancy was 6.18%, with West Java having the highest prevalence (10.57%). 2
Pregnancy often leads to physiological and structural changes in the urinary tract. 5 Ureteral dilatation occurs in 90% of pregnant women and persists until delivery (hydronephrosis of pregnancy). Elevated urine stasis and ureterovesical reflux result from augmented bladder volume, diminished bladder tone, and reduced ureteral tone. This diminished the kidneys’ defenses against bacterial reflux. Bacterial reservoirs may be present in urinary stasis. The glomerular filtration rate and urine output increase in response to an expansion of blood volume. Urine stasis, resulting from elevated urine production and diminished ureteral tone, can lead to the dilation of the ureters, renal pelvis, and calyces. The dilatation appears to commence at 10 weeks gestation, intensify during pregnancy, and subside between 6 and 12 weeks postpartum. Progesterone induces urinary stasis by relaxing the smooth muscle of the ureters. 6 These changes can increase the risk of pathogens ascending into the urinary bladder and cause urinary tract infections (UTIs). 5 Interestingly, the incidence of UTIs is higher among pregnant women than among healthy women in the general population. UTIs, whether symptomatic or asymptomatic, are common during pregnancy and can have adverse effects on both the mother and the newborn, such as pyelonephritis, urosepsis, premature labor, and even stillbirths.7,8
Hypertension in pregnancy is characterized by increased blood pressure, specifically a systolic blood pressure of 140 mmHg and/or diastolic blood pressure of 90 mmHg. Eclampsia is a serious development of preeclampsia, marked with widespread seizures during pregnancy or after childbirth in women exhibiting symptoms of preeclampsia. The warning signs or symptoms that imply the onset of eclampsia, such as severe headaches or neurological problems, are indicative of impending eclampsia. Gestational hypertension refers to the occurrence of chronically elevated blood pressure (⩽140/90 mmHg) after 20 weeks of pregnancy without the presence of proteinuria or any other indications of preeclampsia. Chronic hypertension refers to preexisting hypertension that manifests before pregnancy or is initially diagnosed prior to the 20th week of gestation. Superimposed preeclampsia refers to the occurrence of preeclampsia in a pregnant woman who already has chronic hypertension. 9 Preeclampsia is a pregnancy-specific condition characterized by high blood pressure (⩾140/90 mmHg) and the presence of at least one of the following: proteinuria, acute kidney injury, neurological complications, hematological complications, or uteroplacental dysfunction. Severe preeclampsia is a more severe form characterized by high blood pressure (⩾160/110 mmHg) and may include complications, such as eclampsia or HELLP syndrome (hemolysis, elevated liver enzymes, and low platelets). 10
UTIs have been found to be associated with an increased risk of developing hypertensive disorders of pregnancy, including eclampsia, impending eclampsia, gestational hypertension, chronic hypertension, superimposed preeclampsia, preeclampsia, and severe preeclampsia. UTIs contribute to the development of these conditions through various mechanisms. Infection-induced inflammation and immune responses can lead to endothelial dysfunction and impaired placental development, which is key factors in the pathogenesis of hypertensive disorders of pregnancy. 11 Additionally, UTIs can cause systemic inflammatory responses, oxidative stress, and activation of the renin-angiotensin-aldosterone system, all of which contribute to elevated blood pressure and the development of hypertension during pregnancy. 12 In this scenario, one may postulate that factors, such as infectious diseases, which elevate systemic inflammatory burden, should likewise augment the risk of preeclampsia. Given that UTI is among the most prevalent maternal infections during pregnancy, a relationship with preeclampsia is anticipated. 13
Overall, the presence of UTI during pregnancy can significantly increase the risk and severity of hypertensive disorders, highlighting the importance of early detection and appropriate management of UTIs in pregnant women to prevent adverse outcomes. 14 Further research is necessary to establish the association between bacteriuria during pregnancy and the development of preeclampsia, highlighting the need for early detection and proper management of UTIs in pregnant women. 11 This research aims to investigate the link between the presence of bacteria in routine urinalysis and the risk of hypertensive disorders like preeclampsia. It underscores the need for proper screening and timely intervention to prevent potential complications for both the mother and fetus.
Materials and Methods
Study design and settings
This cross-sectional study was conducted from October 2022 to September 2023 (1 year) at the Margono Soekarjo Hospital. The corresponding author is a Padjadjaran University Resident of Obstetrics and Gynecology currently working and studying at Margono Hospital/Jenderal Soedirman University. Padjadjaran University and Margono Hospital have established a joint partnership for research and academic studies. This study also involved a cooperating research assistant from Padjadjaran University.
Study population
Pregnant women of any gestational age were enrolled in this study. Written informed consent was collected from each participant. Participants with relevant clinical samples were admitted to the hospital on the same day. The diagnostic criteria used in this study are based on the International Society for the Study of Hypertension in Pregnancy (ISSHP) criteria for hypertension in pregnancy. 9
This study excluded pregnant women who were not aged 20–35 years, those with obesity, nulliparity, multiple gestations, a history of chronic disease (renal disease, diabetes, chronic hypertension), history of preeclampsia, and a history of other infections (premature rupture of membranes, dental infections, respiratory infections such as tuberculosis and pneumonia).
Hypertension during pregnancy is defined as systolic blood pressure ⩾140 mmHg and/or diastolic blood pressure ⩾90 mmHg. Meanwhile, gestational hypertension is defined as new-onset or persistent hypertension after 20 weeks without proteinuria. Preeclampsia is gestational hypertension accompanied by ⩾1 of the following new-onset conditions at or after 20 weeks of gestation (proteinuria, other maternal organ dysfunction, including Acute Kidney Injury (AKI) (creatinine ⩾90 µmol/L; 1 mg/dL), liver involvement (elevated transaminases, e.g., alanine aminotransferase or aspartate aminotransferase >40 IU/L) with or without right upper quadrant or epigastric abdominal pain, neurological complications (examples include eclampsia, altered mental status, blindness, stroke, clonus, severe headaches, and persistent visual scotomata), hematological complications (thrombocytopenia–platelet count <150,000/μL, disseminated intravascular coagulation, hemolysis), and uteroplacental dysfunction (such as fetal growth restriction, abnormal umbilical artery Doppler waveform analysis, or stillbirth). 9 Severe preeclampsia includes more severe forms and may involve eclampsia or the HELLP syndrome. Eclampsia involves seizures in women with signs of preeclampsia, while impending eclampsia is indicated by symptoms such as severe headache, visual disturbances, and epigastric pain. 9
Bacteriuria denotes the existence of bacteria in the urine, which may be either symptomatic or asymptomatic. 15 In this study, bacteriuria was identified exclusively through routine urinalysis. The detection of bacteria, often quantified as the number of microorganisms per high-power field, served as the primary indicator of infection. For symptomatic individuals, any bacterial presence could suggest a UTI, while the standard threshold for significant bacteriuria is ⩾100,000 colony-forming units (CFU)/mL, represented as 5+ on urinalysis. For specific populations, such as catheterized or symptomatic patients, bacteriuria may be considered significant at a lower threshold, such as 2+ (~100 CFU/mL). 16 Both symptomatic and asymptomatic cases of bacteriuria were included in this study to provide a comprehensive overview of its prevalence and management. This distinction was not systematically analyzed but is acknowledged as a factor influencing clinical outcomes.
Ethical approval
The study was approved by the Research Ethics Committee of Margono Hospital, Purwokerto, Indonesia, Number 420/03289. All patients in this study were examined, and the ethical standards in the 1964 Declaration of Helsinki were adhered to.
Sample size estimation
This study uses total sampling from 2022 to 2023 at Margono Soekarjo Hospital. This study employs a total sampling of pregnant women admitted to Margono Soekarjo Hospital from January 2022 to January 2023. A total of 1840 women were initially considered; after applying exclusion criteria (maternal age <20 or >40 years, obesity, nulliparity, multiple gestations, chronic diseases, and history of other infections), only 457 participants were included. After applying the exclusion criteria, 457 out of the 1840 women qualified for the study. These participants were further divided into 2 groups: 203 with positive bacteriuria and 254 without bacteriuria. Within the positive bacteriuria group, 59 women developed hypertension, 38 preeclamptic women, while 106 remained nonhypertensive. In the no bacteriuria group, 44 women developed hypertension, 36 preeclamptic women, and 174 were nonhypertensive. Figure 1 shows the diagram of patient enrolment in this study.
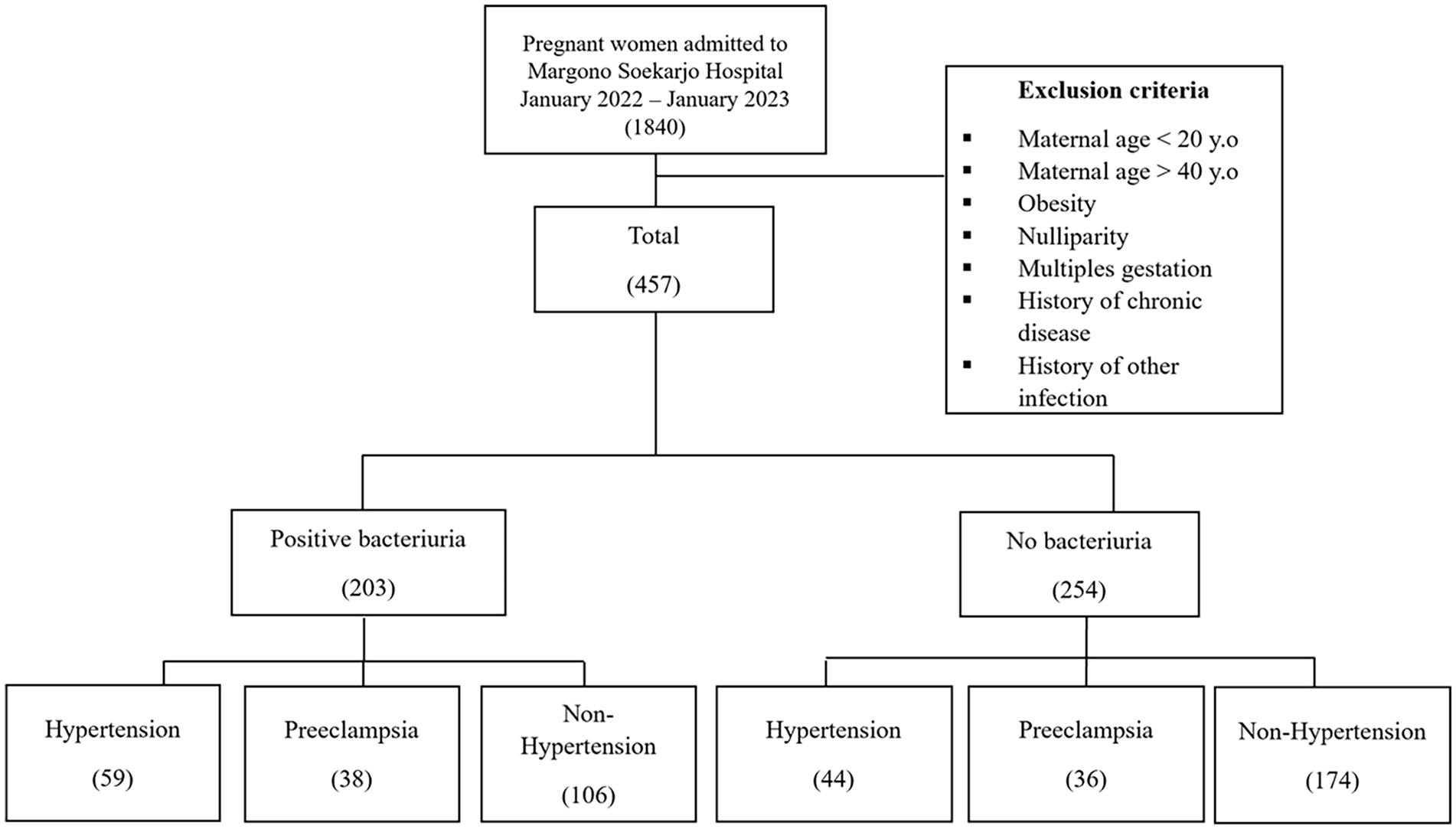
Patient enrollment process.
Data collection
Samples collected from registered pregnant women came from polyclinics or maternal emergency rooms who were hospitalized for giving birth. All patients in our study underwent blood pressure measurement and routine urinalysis simultaneously. Urinalysis is a routine examination in our setting, which is why both bacteriuria and hypertension in pregnancy were diagnosed at the same time. The observer made a conclusive diagnosis after all examinations were completed, and this was not a sequential process. The blood pressure was measured twice using a sphygmomanometer.
Pregnant women underwent laboratory examinations, including urinalysis. Initial urinalysis was conducted by explaining its purpose, obtaining informed consent, and ensuring the availability of the necessary equipment. The patients were instructed to collect a midstream urine sample in a sterile container, emphasizing cleanliness and contamination prevention. A complete urinalysis, including microscopic examination, was employed to obtain detailed and reliable results. For the microscopic examination, the urine sample was centrifuged to separate the sediment, which was then placed on a glass slide and examined under a microscope at both low-power (10×) and high-power (40×) magnification. The presence of bacteria was identified and recorded based on their shape, number, and arrangement in the field of view. This method ensured accurate detection of bacteriuria.
This study employed a qualitative methodology to categorize bacteriuria as “positive” or “negative.” Positive bacteriuria was characterized by the detection of bacteria in urinalysis, whereas negative bacteriuria indicated samples devoid of bacterial presence. Because urine culture is not a standard practice in Indonesia and is conducted solely based on clinical indications, this qualitative bacteriuria criterion was utilized as an initial screening method, especially for pregnant women upon admission. This method functions as an efficient first screening to detect probable UTIs or asymptomatic bacteriuria, enabling prompt therapeutic intervention in this demographic.
Participants with positive bacteriuria, regardless of symptom presentation, were treated with antibiotics following local clinical practices. In the Indonesian healthcare setting, routine diagnosis of UTI is typically based on microscopic analysis of urine samples to detect the presence of bacteria.
Statistical analysis
To evaluate the association between the presence of bacteriuria during routine urinalysis and the occurrence of hypertension in pregnancy, crude odds ratios (OR) were calculated along with 95% confidence intervals (CI). The OR were derived by comparing the frequency of hypertension and nonhypertension in patients with and without bacteriuria. A chi-squared test was performed to assess statistical significance, and a p-value of <0.05 was considered statistically significant.
The prevalence ratio (PR) was used to determine the likelihood of hypertension in pregnant patients with bacteriuria compared to those without bacteriuria. The statistical analysis was conducted with IBM SPSS Statistics version 29.0.1.0 (IBM Corp. Released 2023, IBM SPSS Statistics for Windows, Version 29.0; IBM Corp., Armonk, NY, USA).
Results
This study included 457 subjects: A total of 177 subjects with hypertension during pregnancy and 280 subjects without hypertension. Table 1 outlines the obstetric and clinical characteristics based on bacterial presence during routine urinalysis. Notably, bacteriuria-afflicted pregnant women were mostly aged 26–35 years, compared to the nonbacteriuria group. As seen from the BMI categories, the number of subjects was almost the same between the bacteriuria and nonbacteriuria groups. It is also the same for parity status, gestational age, and the number of subjects with each type of hypertension between the bacteriuria and nonbacteriuria groups. There were no significant differences in the characteristics between the two groups.
Obstetric and clinical information of pregnant women enrolled.


Notes:
a = individuals who are both positive for the exposure and the outcome (positive for bacteriuria and hypertensive).
b = individuals who are positive for the exposure but negative for the outcome (positive for bacteriuria but not hypertensive).
c = individuals who are negative for the exposure but positive for the outcome (hypertensive but no bacteriuria).
d = individuals who are negative for both the exposure and the outcome (no bacteriuria and not hypertensive).
The calculation and interpretation of the PR value used in the study. The formula for PR compares the proportion of individuals with hypertension among those with positive bacteriuria (exposure) to those without bacteriuria. In this case, a represents individuals who are both positive for the exposure and the outcome (positive for bacteriuria and hypertensive), while b refers to individuals who are positive for the exposure but negative for the outcome (positive for bacteriuria but not hypertensive). Similarly, c indicates individuals who are negative for the exposure but positive for the outcome (hypertensive but no bacteriuria), and d represents individuals who are negative for both the exposure and the outcome (no bacteriuria and not hypertensive). The calculation provided is PR = (a/(a + b)):(c/(c + d)), where the proportion of hypertension among women with positive bacteriuria is 97 out of 203, and the proportion of hypertension among those without bacteriuria is 80 out of 254. This yields a PR of 1.52, indicating that the likelihood of hypertension during pregnancy is 1.52 times greater in women with UTIs (positive bacteriuria) compared to those without bacteriuria. This result highlights a significant association between positive bacteriuria and an increased risk of hypertension during pregnancy.
Table 2 illustrates the crude OR for preeclampsia, hypertension, and nonhypertension based on bacteriuria status. The data are divided into two groups: positive bacteriuria (n = 203) and negative bacteriuria (n = 254). In the positive bacteriuria group, 59 patients experienced hypertension, 38 had preeclampsia, and 106 were nonhypertensive. The OR for positive bacteriuria was calculated at 1.379, with a CI of 0.751–2.532, and this result was statistically significant with a p-value of <0.003. In the negative bacteriuria group, 44 patients had hypertension, 36 experienced preeclampsia, and 174 were nonhypertensive. The OR for negative bacteriuria was 0.725, with a CI of 0.395–1.331, and this was also statistically significant with a p-value of <0.003. These findings indicate a statistically significant association (p < 0.05) between bacteriuria status and the likelihood of hypertension and preeclampsia, showing that positive bacteriuria increases the risk of these conditions compared to negative bacteriuria.
Crude odds ratio preeclampsia, hypertension, and nonhypertension from bacteriuria status.

CI: confidence interval.
p < 0.05, statistically significant result.
Discussion
Hypertension during pregnancy can lead to complications in both the mother and baby. On the other hand, UTIs are common bacterial infections in the urinary tract.9,10 While UTIs are common during pregnancy, they require attention and management. UTIs can increase the risk of complications including preterm birth and low birth weight. Pregnant women should be educated about the signs and symptoms of UTIs and encourage timely treatment. Proper diagnosis and management of UTIs in pregnant women are essential to prevent further complications. It is important to note that hypertension in pregnancy and UTIs can coexist and potentially influence each other.13,17
In our study, the positive bacteriuria cohort included 59 patients with hypertension, 38 with preeclampsia, and 106 who were nonhypertensive. The OR for positive bacteriuria was determined to be 1.379, accompanied by a CI of 0.751–2.532, and this finding was statistically significant with a p-value of <0.003. This study aligned with Yan et al. and examined the association between UTI during pregnancy and the risk of preeclampsia. An OR of 1.31 indicates a 1.31-fold increased probability of hypertension during pregnancy in women with UTIs. The chi-squared analysis produced a p-value <0.001, indicating a highly significant association between UTIs during pregnancy and elevated hypertension risk. 11
In the context of pregnancy-related hypertension, particularly preeclampsia, various hypotheses have linked infection to its pathogenesis. One theory suggests that infection contributes to preeclampsia by initiating or increasing the risk of acute uteroplacental atherosclerosis. Another hypothesis proposes that infection could directly impact trophoblast cells, leading to shallow invasion of maternal spiral arteries. Clinical and epidemiological data support these ideas, as preeclampsia is associated with the development of pathological lesions resembling atherosclerotic lesions. The role of infection in preeclampsia has been explored in retrospective, case–control, and prospective studies. Preeclampsia consistently exhibits endothelial dysfunction and excessive inflammation, and infection is believed to stimulate the immune system, contributing to the observed inflammation. Understanding the infection–preeclampsia connection offers insights into its pathophysiology and suggests potential public health measures to reduce its incidence. Preeclampsia is characterized by inflammatory responses, oxidative stress, hypertension, and proteinuria, with elevated levels of pro-inflammatory cytokines, such as TNF-α and IL-6, in affected women compared to normotensive pregnancies. Infections may trigger the release of these cytokines, intensifying inflammation in preeclampsia and causing endothelial cell dysfunction and oxidative stress.2,8
Pregnancy-related hypertension is caused by some variables, such as aberrant placentation, cardiovascular maladaptation, immune system and genetic abnormalities, increased systemic inflammatory response and angiogenic, hormonal, and dietary factors. Particularly in poor nations, these conditions, which include preeclampsia, gestational hypertension, and chronic hypertension, cause considerable morbidity and mortality in mothers and newborns. The cardiovascular, hepatic, renal, pulmonary, and central neurological systems are all affected by the associated morbidities. A history of hypertension, pregestational diabetes, multiple gestations, previous preeclampsia, nulliparity, assisted reproductive technologies, and being overweight or obese prior to pregnancy are among several risk factors. Long-term issues can develop, such as end-stage renal and cardiovascular disorders. Unfortunately, there are limited recent data on hypertensive disorder profiles in Indonesia. 18
The clinical manifestations of hypertension during pregnancy, oxidative stress, and proteinuria are hypothesized to result from generalized maternal endothelial dysfunction. As suggested by Kaduma et al. and Ndaboine et al., inflammation is believed to provide an oxidative insult, leading to endothelial dysfunction in preeclampsia. However, it remains unclear whether inflammation precedes the development of preeclampsia or is a consequence of the disease.19,20 Some studies have identified increased levels of inflammatory cytokines in women with preeclampsia compared to those with normotensive pregnancies. Mechanisms include association with pathologic lesions in decidual arterioles, maternal infections triggering pro-inflammatory cytokines, and potential destruction of cytotrophoblast cells or impairment of their function. 21
Kaduma et al. conducted a matched case–control study involving pregnant women with preeclampsia and without preeclampsia to investigate the association between UTIs and preeclampsia among pregnant women in Tanzania. 19 The results show that a higher proportion of pregnant women with hypertension had significant bacteriuria compared to those without hypertension during pregnancy. Further research is also recommended to investigate the maternal–fetal and neonatal outcomes of pregnant women with both preeclampsia and UTIs. 20
Karmon and Sheiner address the relationship between UTIs during pregnancy and the development of hypertension in pregnancy, which is a major cause of maternal morbidity. A study conducted at a medical facility found that pregnant women who experienced at least one UTI episode during pregnancy had a 1.3-fold higher likelihood of developing mild preeclampsia and a 1.8-fold higher likelihood of developing severe preeclampsia than women who were not exposed to UTIs. Nevertheless, the findings presented in this study were derived from univariate analysis and have not been accounted for in terms of potential confounding variables. 21
Karmon and Sheiner emphasized the significance of conducting a prospective study with sufficient power and sample size to establish the association between UTIs during pregnancy and hypertension in pregnancy. To determine whether the association between UTIs and hypertension in pregnancy is causal, spurious, or confounded, it is recommended to consider confounders, potential biases, and factors such as chronology, dose–response effects, and treatment. The authors highlight the importance of employing multivariate analysis and investigating effect modification techniques to account for confounding variables and ascertain potential factors that could influence the relationship between UTIs and hypertension during pregnancy. In summary, Karmon and Sheiner elucidated that the correlation between UTIs and the development of hypertension during pregnancy has been demonstrated in multiple studies. However, the precise nature of this link remains. 21
Asymptomatic bacteriuria is also classified as a UTI and is a common and serious condition during pregnancy that may result in increasing and severe consequences, endangering both maternal and fetal health. 22 The study by Rezavand et al. revealed a substantial disparity in asymptomatic bacteriuria with positive urine culture between the pre-eclampsia group (58 cases, 46.4%) and the control group (14 cases, 11.2%); chi-square = 37.3. The likelihood of a positive urine culture was 6.8 times greater in the pre-eclampsia group (p < 0.001). 23 A proposed notion posits that infection may elevate maternal serum cytokine levels. These cytokines impair endothelial function. The likelihood of pre-eclampsia is greatest when there is a prior history of exposure to antigens that impact the body, particularly those that influence lymphocyte function. 24
Pregnancy-related hypertension is estimated to affect 0.2%–9.2% of pregnant women, and it substantially increases maternal and perinatal morbidity and mortality, especially in developing nations. UTIs are common during pregnancy, with an estimated incidence of 20%. The management of pregnancy-related hypertension problems and tract infections can provide extra challenges in low-resource settings because of restricted access to healthcare resources and services. In these situations, offering complete prenatal care, including frequent blood pressure checks and urine tests for UTIs, is crucial.18,25
In conclusion, multiple studies have supported an association between maternal infections and hypertension during pregnancy. The presence of bacterial or viral infections has been found to increase the risk of hypertension in pregnancy. Infection may contribute to the inflammation observed in hypertension during pregnancy and potentially play a role in its pathophysiology. Further research and understanding of this association can help develop strategies to reduce the incidence of hypertension in pregnancy and its associated with maternal morbidity and perinatal mortality.
Confounding factor
The study considered potential confounding factors in the association between bacterial presence during routine urinalysis and hypertension in pregnancy. Maternal age <20 and >35 years, obesity, nulliparity, history of other infections, and history of chronic disease were confounding factors due to their association with both bacteriuria and hypertension, necessitating statistical adjustment. These confounders were excluded from the analysis. None of the participants had a history of smoking during pregnancy.
In the previous study, confounding factors were not accounted for in the analysis. 21 In contrast, our study has addressed this limitation by excluding potential confounders, strengthening our results’ validity. By controlling these variables, we believe our findings represent an improvement over prior studies, offering a clearer understanding of the association under investigation.
Limitation of study
This study, conducted at Margono Hospital Purwokerto in Indonesia, has several limitations. The cross-sectional design prevents the establishment of causal relationships between bacteriuria in pregnancy and the development of hypertension, as it provides only a snapshot at a specific point in time. Future prospective studies with larger sample sizes and careful consideration of confounders are needed to better understand this association. Additionally, the study did not evaluate other urinalysis parameters, such as nitrite, leukocyte esterase, and proteinuria, which are commonly used to indicate UTIs. While urine culture is the gold standard for diagnosing UTIs, it is not routinely performed at the study site, limiting the diagnostic precision.
The study’s sample size was determined based on available resources and convenience rather than a formal sample size calculation, which may affect the power to detect statistically significant associations. This limitation highlights the need for future studies to perform rigorous sample size calculations to ensure robust and generalizable findings.
The use of broad exclusion criteria, including maternal age, obesity, nulliparity, multiple gestations, chronic diseases, and history of other infections, helped reduce the influence of confounders but may have limited the generalizability of the findings to the broader population of pregnant women. Incorporating these factors into statistical models in future research could enhance applicability. Furthermore, the reliance on microscopic detection of bacteria for UTI diagnosis, without quantifying bacterial load, and the lack of systematic analysis of patient symptoms, represent methodological limitations. Urinalysis alone may not adequately differentiate between contamination, asymptomatic bacteriuria, and clinically significant UTI, potentially affecting the interpretation of results. Lastly, while this study highlights the importance of early identification and management of UTIs in pregnancy to prevent hypertension, longitudinal observational studies are needed to explore the causal relationship between UTIs and hypertensive disorders during pregnancy.
Conclusion
The study concludes that there is a statistically significant association between UTIs observed in bacteriuria during pregnancy and an increased risk of hypertension in pregnancy. Pregnant women with bacteriuria have a higher odd of developing hypertension during pregnancy than those without bacteriuria. Routine urine culture and antimicrobial susceptibility testing are recommended for pregnant women with hypertension, and further research is needed to explore the outcomes for both the mother and the newborn. Overall, although the exact mechanisms linking UTIs and hypertension in pregnancy, especially preeclampsia, are not fully understood, evidence suggests a potential relationship that may contribute to the development of hypertension during pregnancy.
Footnotes
Acknowledgements
None.
Author contributions
MD was involved in the conception, design, analysis, interpretation of the data, and manuscript drafting. UA and SH collected and analyzed the data and assisted in drafting the manuscript. MD and AU collected and analyzed the data. AANP, SH, HB, and AAM developed the conception design, analyzed and interpreted the data, and revised the manuscript critically for intellectual content. The different affiliations of each author were due to the exchange of cooperation from each center.
Consent to participate
Written informed consent was obtained from the patient for participation in the study. This consent covered the patient’s agreement to provide medical information and undergo any necessary procedures, assessments, or data collection related to the study or report. The patient was informed of their right to withdraw consent at any time without affecting their care or treatment.
Consent for publication
Written informed consent was obtained from the patient for publication and any accompanying images. All patients have signed informed consent to publish their medical information and/or images in the manuscript.
Data availability statement
The data supporting this study’s findings are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy or ethical restrictions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The study obtained an approval from the Research Ethics Committee of Margono Hospital, Purwokerto, Indonesia, number 420/03289. All patients in this study were examined, and the ethical standards set out in the 1964 Declaration of Helsinki were adhered to.
Trial registration
Not applicable.
