Abstract
Objectives:
There are several studies from sub-Saharan Africa on postpartum urinary incontinence and anal incontinence, but very rare in pregnancy. Such data will guide obstetric caregivers in providing appropriate counseling to the women as well as in minimizing the risk factors. This study aimed to determine the comparative effects of different trimesters of pregnancy on urinary incontinence and anal incontinence, and their possible risk factors.
Methods:
The study was longitudinal in design, and the study population consisted of 223 pregnant women receiving care at the two largest tertiary health institutions in Enugu, South-East Nigeria. The recruitment was in the first trimester and the women were followed up to term. Interviews were conducted at specific times in the three trimesters and data regarding urinary incontinence and anal incontinence symptoms were obtained using validated questionnaires.
Results:
The incidence of urinary incontinence increased across the trimesters: 22%, 30.5%, and 48% in the first trimester, second, and third trimesters, respectively, with a cumulative incidence rate of 50.2%. The incidence of anal incontinence also increased across the trimesters but not as high as urinary incontinence: 1.7%, 3.6%, and 5.8%, respectively, with a cumulative incidence rate of 6.7%. The risk factors for urinary incontinence were maternal age >35 years, multiparity, previous prolonged second-stage labor, and previous history of neonatal macrosomia, while that of anal incontinence were previous instrumental vaginal delivery and previous prolonged second stage of labor.
Conclusion:
Our study demonstrated an increase in the incidence of urinary incontinence and anal incontinence as pregnancy advances. Obstetricians are therefore encouraged to discuss these pelvic floor issues during antenatal care services and make more efforts toward reducing the modifying obstetric risk factors.
Introduction
Pregnancy is a potential risk factor for pelvic floor disorders including urinary incontinence (UI) and anal incontinence (AI). 1 Pregnant women frequently develop UI and AI symptoms, demonstrating the additional and as of yet unexplained roles that predisposing pre-pregnancy maternal, genetic, or hormonal variables play. 2 It is also of great concern as to how pregnancy affects the pelvic floor and whether some obstetric procedures or risk factors could be modified to shield the pelvic floor from potential harm. 3 While studies abound on the postpartum incidence of UI and AI, they are very limited during pregnancy, especially in low- and middle-income countries (LMICs). For instance, recent studies observed that childbirth has a significant impact on UI and AI in LMICs, but did not determine the possible effects of pregnancy on these maternal morbidities. 1 Pregnancy could influence the development of UI and AI by its direct and indirect effects on supporting pelvic musculature and nerves. 1 The occurrence of UI and AI could result in a loss of self-esteem and might hinder the mother’s emotional and physical capacities to care for her newborn baby at birth. 1 Also they could predispose her to anxiety and depression. 3 Despite the above potential effects of pregnancy on the pelvic floor, this issue is rarely discussed during antenatal care services probably because of limited sound data in this regard.1,4 Interestingly, there are positive opinions that conversations on these topics during antenatal visits could decrease the emotional and psycho-social distress commonly associated with UI and AI.4–6 Furthermore, there is limited literature on UI and AI during pregnancy in LMICs despite the expected high burden of these morbidities because of the high fertility rate in these countries. A study on the effects of pregnancy on UI and AI will help provide evidence-based information on their variations across the trimesters and their possible risk factors.5,7,8 Such information will guide the clinicians in women’s counseling during antenatal care services.
Although a recent systematic review on UI in pregnancy by Moossdorff-Steinhauser et al. 9 reported a high prevalence rate (49%), and a rise in prevalence as pregnancy advances, the review was limited by high clinical heterogeneity of the included studies especially in “case definition” and research periods. 9 It was recommended that further studies should utilize a validated study instrument—International Consultation on Incontinence Questionnaire on UI Short Form (ICIQ-UI-SF), 6 and consider a longitudinal design for assessment of incidence across the three trimesters of pregnancy. Furthermore, of the 44 studies included in the review, 17 were from Asia, 15 from Europe, 8 from the United States, and only 3 and 1 from Africa and Oceania, respectively, indicating the need for more studies from the last two continents. Also, the only study from Nigeria 10 included in the review was “cross-sectional descriptive” in design and evaluated women at only “one point” in their pregnancies and thus could not determine the incidence rate of UI across the trimesters. A systematic review on the incidence of AI in pregnancy is on the other hand very scarce in literature probably because of limited studies in this regard. This study is therefore designed to determine the comparative effects of different trimesters of pregnancy on the incidence of UI and AI, and the risk factors of these conditions among pregnant women in Enugu, Nigeria.
Methods
The study was conducted in Enugu state, Nigeria. The state is one of the five states in the South-East geopolitical zone of the country and its capital city is Enugu. The reported postpartum incidence rates of UI and AI in the state are 12.2% and 13.5%, respectively. 1
The study was carried out at the University of Nigeria Teaching Hospital (UNTH) Ituku-Ozalla and Enugu State University Teaching Hospital (ESUTH) Parklane between January 2021 and June 2022. The UNTH Enugu is the pioneer teaching hospital in South-East Nigeria, and it is owned by the federal government of Nigeria. It offers antenatal and postnatal care services to pregnant women in Enugu state and handles an average of 1500 deliveries annually, with a 28-bed capacity obstetrics unit. It practices the traditional mode of antenatal care whereby women are generally seen monthly until 28 weeks of gestation, fortnightly until 36 weeks, and then weekly until delivery. The ESUTH-Parklane is a tertiary health institution owned by the Enugu state government. The hospital handles an average of 1800 deliveries annually, with a 26-bed capacity obstetrics unit. It also practices the traditional mode of antenatal care. In both hospitals, after a normal delivery, women are generally observed for 48 h before discharge, and then seen after 6 weeks at the postnatal clinics.
Prior to the commencement of the study, ethical clearance was obtained from the institutional review board of the UNTH, Ituku-Ozalla, Enugu (application reference no: NHREC/05/01/2008B-FWA00002458-1RB00002323), and ESUTH-Parklane (application reference no: ESUTHP/C-MAC/RA/034/Vol.11/57.4). A written informed consent was also obtained from each participant before enrollment into the study. The study was longitudinal in design, and the study population consisted of pregnant women attending antenatal care at the two study centers. All pregnant women booking for antenatal care in the first trimester (T1) in the two study centers were eligible for the study. However, women with symptoms of urinary tract infection, multiple gestation, or serious medical disorders including severe hypertensive disease, severe renal disease, or severe cardiac disease were excluded from the study. Also, women who were incontinent of urine or feces prior to pregnancy were excluded. The eligible participants were consecutively recruited as they came for the antenatal booking until the desired sample size was achieved. Using a UI rate of 12.2% obtained from a previous related study from the study population 1 at a 95% confidence interval and 5% sampling error (precision), the calculated minimum sample size (n) was 165. However, considering a possible 20% attrition rate, 270 women were approached for the study.
The recruitment was in the T1 at 13 weeks of gestation or less and at the booking clinic. Each recruited participant was followed up to term. Thus, the participants for the T1, second trimester (T2), and third trimester (T3) were the same. At recruitment, the baseline socio-demographic data of the participants were taken. The pelvic floor symptoms including UI and AI were obtained using validated questionnaires—ICIQ-UI-SF 6 and Anal Incontinence Questionnaire (AIQ).1,7
Patients were informed that each set of questionnaires would take approximately 45 min to complete. It took this long because this work was an excerpt from a larger work for a PhD which had other objectives not included in this study. The questionnaires contained a cover page explaining the study’s purpose and some definitions to assist the participants in answering the questions. Following the booking, the participants were generally followed up in line with the two hospitals’ (traditional) protocol of antenatal care visits—monthly till 28 weeks of gestation, fortnightly till 36 weeks of gestation, and then weekly till delivery. During the T2 between 20 and 24 weeks, and in the T3 between 36 and 38 weeks, interviews were repeated to obtain data on the participants’ UI and AI symptoms using the same validated questionnaires.
To minimize loss to follow-up, participants’ phone numbers and home addresses were collected with their consent. Information on each participant was recorded on the follow-up summary chart indicating the days/dates for their antenatal visits. However, a day prior to the date for the visits, phone calls and text messages were used to remind the participant of her visit and to encourage her to come. Participants who failed to attend the visits despite reminders were visited at their homes to collect the relevant information. However, where home visitation was not possible, phone calls, WhatsApp charts, or emails were used to collect the necessary data where feasible. Since the study was investigating a very sensitive issue, all relevant information was collected by five research assistants (junior obstetrics and gynecology residents) who were specially trained on how to collect such sensitive data. One assistant was selected from each of the five units of the obstetrics and gynecology departments of the hospitals to cover the five (5) working days of the week for the various units so that follow-up became easier.
The first trimester (T1) was defined as the period of pregnancy from 0 to 13 weeks; T2 as the period from 14 to 27 weeks; and T3 as the period from 27 to 40 weeks. 11
The study’s validated questionnaires
(i) International Consultation on Incontinence Questionnaire on Urinary Incontinence Short Form (ICIQ-UI-SF) 6 —The ICIQ-UI-SF is a validated questionnaire for urinary incontinence (Appendix 1). It is a four (4)-item questionnaire—Questions I–IV. ICIQ-UI-SF score = sum I + II + III; ICIQ-UI-SF Score >0 = urinary incontinence; ICIQ-UI-SF Score 1–6 = mild urinary incontinence; and ICIQ-UI-SF Score >6 = moderate/severe urinary incontinence. Item IV is the unscored self-diagnostic item that helps to indicate the patient’s perception of the cause of her incontinence.
“Stress urinary incontinence (SUI) was defined as participant’s report of involuntary leakage of urine when coughing, sneezing, laughing, or exercising; urgency urinary incontinence (UUI) as participant’s report of involuntary leakage of urine before she gets to the toilet or convenience; overflow urinary incontinence (OUI) as participant’s report of involuntary leakage of urine when she is asleep, and fistulous (VVF) incontinence as participant’s report of involuntary leakage of urine all the time (continuous leakage of urine). The cumulative incidence rate of UI was defined as the proportion of participants whoever reported any type of UI in any of the three trimesters of pregnancy.” 1
(ii) AIQ—The AIQ is an 8-item validated questionnaire for anal incontinence with YES/NO answers (Appendix 2). The questionnaire has been used in two related studies in Nigeria.1,7 “A participant was said to have developed AI if she gave affirmative response (yes) to question 1 of the AI questionnaire. Flatus incontinence was defined as participant’s report of involuntary leakage of flatus only and fecal incontinence (FI) as participant’s report of involuntary leakage of feces only. Cumulative incidence rate of AI was the proportion of participants whoever reported any type of AI in any of the three trimesters of pregnancy. Double incontinence (concurrent UI and AI) rate was the proportion of participants whoever reported concurrence of AI and AI in any of the three trimesters of pregnancy.” 1
The primary outcome measures were the incidence of UI and AI in each of the trimesters. The secondary outcome measures were the risk factors for UI and AI symptoms in pregnancy.
Data analysis was done using the statistical package for social sciences version 26 (IBM Corp. Released 2019. IBM SPSS Statistics for Windows, Version 26.0. Armonk, NY: IBM Corp) for Windows. Proportions were compared using chi-square or Fisher’s exact tests as appropriate. Relationships for categorical data were expressed using relative risks (RRs) and confidence intervals (CIs), and risk factors for UI and AI were calculated by multivariate logistic regression analysis. A p-value ⩽ 0.05 was considered statistically significant.
Results
Study flow and participants’ basic characteristics
In all, 270 women were approached for the study at T1 (booking clinic); however, 20 were not enrolled because of the exclusion criteria (n = 13) or because they refused to give their consent (n = 7). Among the 250 women enrolled, 223 (93.2%) completed the study, while 27 (6.8%) were lost to follow-up (11 after recruitment but before T2 questionnaire administration, and 16 before T3 questionnaire administration). All the 27 women who were lost to follow-up were excluded from the analysis. Figure 1 illustrates the specifics of the study flow. Of the 223 women who completed all three questionnaire series, 60 (26.7%) were nullipara, 143 (16.1%) were multipara, and 20 (20%) were grandmultipara (para 5 or more). The women’s ages ranged from 18 to 45 years, with an average of 30.7 ± 2.4 years, and their gestational ages ranged from 8 to 13 weeks with an average of 11.3 ± 1.8 weeks at recruitment.

Study flow chart.
Details of the baseline characteristics of the participants including age, parity, educational level, and gestational age at recruitment are shown in Table 1.
Socio-demographic characteristics of the study participants.

Urinary and AI in pregnancy
The incidence of UI increased across the three trimesters: 22% (n = 49) in T1, 30.5% (n = 68) in T2, and 48% (n = 107) in T3. The observed difference was statistically significant (χ 2 = 35.21; p < 0.001). Two-by-two comparisons showed that the incidence was significantly higher in T3 than T1 (48% vs 22%; RR = 2.18; 95% CI = 1.6–2.9; p < 0.001) and T2 (48% vs 30.5%; RR = 1.57; 95% CI = 1.2–2.0; p < 0.001). Also the incidence in T2 was significantly higher than in T1 (29.2% vs 21%; RR = 1.39; 95% CI = 1.0–1.9; p = 0.04). The cumulative incidence of UI was 50.2% (n = 112). Of the 112 women who ever developed UI in pregnancy, 67 women (30%) reported SUI, 32 (14.3%) reported UUI, and 13 (5.8%) reported OUI. None of the women experienced VVF. Regarding the severity of UI, 71 (66.4 %) reported a mild degree, while 36 (33.6%) reported a moderate–severe degree. There was a significant difference in the severity of UI across the three trimesters (4 (8.2%) vs 11 (16.2%) vs 26 (24.3%); p = 0.046). Two-by-two comparisons showed that the severity was significantly higher in T3 than T1 (24.3% vs 8.2%; RR = 2.98; 95% CI = 1.1–8.1; p = 0.03), but not significantly higher than T2 (24.3% vs 16.2%; RR = 1.50; 95% CI = 0.8–2.8; p = 0.210). There was no difference in the severity of UI between T2 and T2 (16.2% vs 8.2%; RR = 1.98; 95% CI = 0.7–5.9; p = 0.216).
The incidence of AI also increased across the three trimesters though not as high as the UI; 1.7% (n = 4) in T1, 3.6% (n = 8) in T2, and 5.8% (n = 13) in T3. Although the observed difference was not statistically significant across the three trimesters (χ 2 = 5.07; p = 0.079), two-by-two comparisons showed that the incidence was significantly higher in T3 than T1 (5.8% vs 1.7%; RR = 3.25; 95% CI = 1.1–9.8; p = 0.037). There was no significant difference in the incidence between T3 and T2 (5.8% vs 3.6%; RR = 1.6; 95% CI = 0.7–3.8; p = 0.269) and between T2 and T1 (3.6% vs 1.7%; RR = 2.0; 95% CI = 0.6–6.5; p = 0.252). The cumulative incidence of AI was 6.7 % (n = 15). The flatus incontinence, 4.9% (n = 11) was commoner than FI, 1.8% (n = 4). The incidence of double incontinence (concurrent UI and AI) was 4% (n = 9), and it also increased across the trimesters: 0.4% (n = 1) in T1, 1.3% (n = 3) in T2, and 3.0% (n = 7) in T3. Further details of the incidence of UI and AI and the severity of UI across the trimesters are shown in Table 2.
Distribution of the incidence and severity of urinary and AI across the trimesters of pregnancy*.

UI: urinary incontinence; AI: anal incontinence.
Chi-square test.
Risk factors for UI in pregnancy
Following a univariate analysis, maternal age ⩾35 years (p = 0.013), less than tertiary education (p = 0.044), low social class (p = 0.033), multiparity (p < 0.001), obesity (booking body mass index ⩾ 30) (p = 0.039), previous prolonged second stage of labor (⩾2 h) (p < 0.001), previous instrumental (vacuum) vaginal delivery (p = 0.021), and previous neonatal macrosomia (⩾4 kg) (p = 0.010) were significantly associated with development of UI. However, after a multivariate logistic regression, maternal age, multiparity, previous prolonged second-stage labor, and previous neonatal macrosomia, maintained a significant association with the development of UI (p < 0.05), while less than tertiary education, low social class, booking body mass index ⩾30 kg/m2, and vacuum delivery no longer had a significant association with the development of UI (p > 0.05). Details are shown in Table 3.
Association between UI and some maternal characteristics.
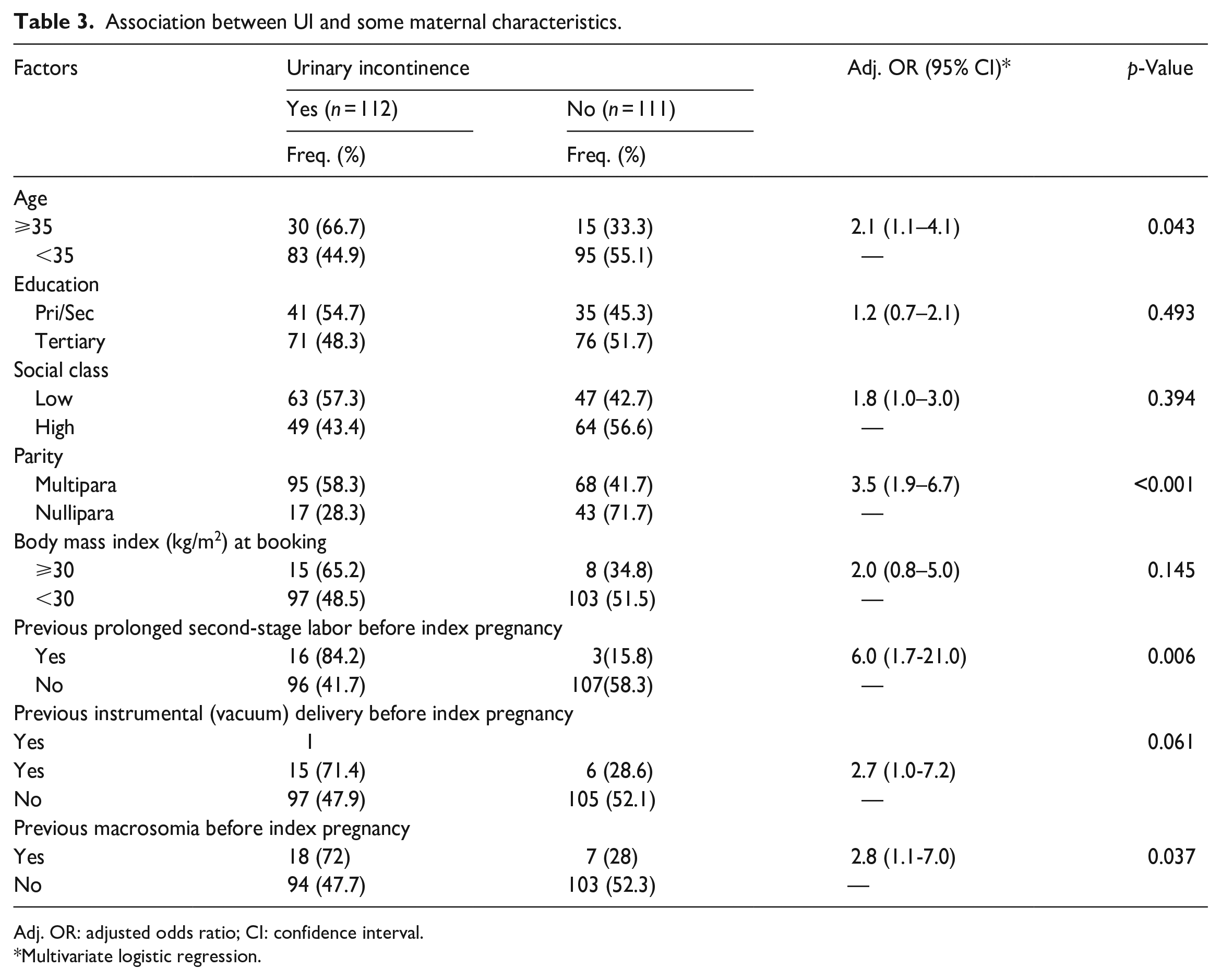
Adj. OR: adjusted odds ratio; CI: confidence interval.
Multivariate logistic regression.
Risk factors for AI in pregnancy
The following factors had a significant association with the development of AI after univariate analyses: age ⩾35 years (p = 0.023), obesity (p = 0.042), previous instrumental (vacuum-assisted) vaginal delivery (p = 0.003), previous history of neonatal macrosomia (p = 0.048), and prolonged second stage of labor (p < 0.001). After a multivariate logistic regression, only previous instrumental (vacuum-assisted) vaginal delivery (p = 0.003) and previous prolonged second stage of labor (p < 0.001) maintained a significant association with the development of AI, while age ⩾35 years, obesity, and previous history of neonatal macrosomia no longer had a significant association with the development of AI (p > 0.05). Details are shown in Table 4.
Association between AI and some maternal characteristics.

Adj. OR: adjusted odds ratio; CI: confidence interval.
Multivariate logistic regression.
Discussion
This study demonstrated that pelvic floor symptoms including UI and AI increased across the trimesters. The incidence of UI increased significantly from 22% in the first trimester, to 30.5% in the second trimester and 48% in the third trimester. A similar study from New Mexico, USA, reported a rise in the incidence as pregnancy advances and up to 69% in the third trimester. 8 These suggest that pregnancy not only affects the structures that maintain continence in women such as the pelvic floor musculatures and pubourethral ligament, but the effect increases as pregnancy advances. It is likely that the increasing uterine size that accompanies increasing gestation results in increasing intra-vesical pressure which is the utmost mechanism in the etiology of SUI. 1 The SUI accounted for approximately 60% of the UI in this study, similar to a previous study. 1 The high incidence of UI (50.2%) observed in this study suggests that this public health issue is also common in African populations. The only difference may be that African women often do not voluntarily complain about the condition, unlike their Western counterparts. 7 It is therefore imperative that clinicians enquire about this condition during pregnancy and provide appropriate counseling on its course and the measures that may ameliorate the burden. The observed high incidence is similar to reports from the Western population.5,8,9,12,13 A previous study from the study population reported a postpartum rate of 12.2% for UI 1 suggesting that the rate is less in the postpartum period.
The incidence of AI also increased across the trimesters, from 1.7% in the first trimester, to 3.6% in the second trimester, and 5.8% in the third trimester; however, the increase was not as high as observed for UI. Awareness of this negative effect of pregnancy is also necessary for women’s counseling and in allaying the related anxieties. The observed cumulative incidence rate of 6.5% recorded in this study is less than the postpartum rate of 13.5% previously reported from the study population. 1 This suggests that the pregnancy effect on pelvic floor musculature and nerves may not be as strong as that of the labor effect. The lower incidence rate of AI than UI in pregnancy, and the increase in the rates of both conditions across the trimesters are similar to reports from the Western populations.5–8
The identified risk factors for UI in this study are maternal age, multiparity, previous prolonged second-stage labor, and previous neonatal macrosomia. The weakening of the pelvic supporting structures and nerves that occur with aging may be responsible for the increased incidence of UI with advancing age.5,8,9,11,12 It has been previously reported that the damage to the pelvic structures and nerves increases with successive pregnancies1,14 and this may explain why multiparity remained a significant risk factor even after a multivariate logistic regression. Vaginal delivery has been associated with damage to pelvic floor musculature and nerves and this may likely be more profound with prolonged second stage of labor. The same mechanism may explain the observed significant effect of macrosomia in this study.14–16 These risk factors are similar to observations from other related reports.1,14,15 Clinicians’ knowledge of these predisposing factors is necessary for well-informed counseling as well as in designing policies/strategies for mitigating the burden of UI in pregnancy.
The significant risk factors for AI observed in this study were previous instrumental (vacuum-assisted) vaginal delivery and previous prolonged second stage of labor. The increased traction pull on the pelvic floor associated with vacuum delivery produces extra mechanical forces on the genital tract, which may cause damage to pelvic floor support and anal sphincter with consequent weakening of the pelvic musculature, fascia, and nerves. 1 These effects may result in AI which could manifest in the postpartum period 1 or a subsequent pregnancy as observed in this study. It therefore suggests that a predisposing factor in a previous delivery increases the risk of AI in a subsequent pregnancy. This may probably be due to further pregnancy effects on the already weakened supporting pelvic structures and nerves. This study also observed that women with previous prolonged second-stage labor had an increased risk of developing AI during pregnancy. This again may be due to the mechanical effect of labor on pelvic musculatures and nerves which is said to be more intense with a longer duration of the second stage of labor and may manifest in a subsequent pregnancy. These observations are similar to previous related studies.1,17,18
Study limitations and strength
This study does not have data on the possible effect of labor and postpartum on UI and AI symptoms. This information is necessary in determining if the observed changes would resolve or aggravate postpartum. Fortunately, the study participants are being followed up in this regard. Urodynamic studies were not performed to objectively diagnose the various types of UI that developed during the study period; however, the use of ICIQ-UI-SF as used in this study has been shown to be a reliable alternative despite some inherent measurement errors in the instrument.1,19,20 The total number of deliveries recorded in the two study institutions during the study period was not very large and this may affect the observed incidence of UI and AI reported in this study. Despite the above limitations, this study is very relevant as it is the first from sub-Saharan Africa (to the best of our knowledge) to comprehensively and prospectively evaluate the effects of pregnancy on these public health issues with a reduced likelihood of recall bias that heralded most previous designs. The study was conducted in two major tertiary health facilities in the state with high maternity flow and thus would be more representative and generalizable than a single-center study. All the instruments for data collection were validated and have been used in previously published studies.
Conclusion
The incidence of UI and AI increases across the trimesters, and UI is commoner than AI. The risk factors for these maternal morbidities are maternal age ⩾35 years, multiparity, previous prolonged second-stage labor, previous instrumental vaginal delivery, and previous neonatal macrosomia. Obstetricians are therefore encouraged to discuss these pelvic floor issues during antenatal care services and make more efforts toward reducing the modifying obstetric factors that predispose them.
Footnotes
Appendix 1
Using the “International Consultation on Incontinence Questionnaire on urinary incontinence-Short Form” (ICIQ-UI-SF)
The unscored self-diagnostic item IV helps to understand the patient’s perception of the cause of her incontinence.
Appendix 2
Anal incontinence during pregnancy using AIQ
Are you having involuntary leakage of Feces or Flatus? [ ]YES, [ ]NO
Acknowledgements
We are extremely grateful for the assistance and collaboration provided by the resident doctors, midwives, and obstetrics and gynecology consultants of the two research institutions.
Authors’ contribution
EOU and CCD were involved in the study conceptualization. EOU, CCD, and GUE were involved in the study design and data acquisition/collection. EOU, CCD, and GUE were involved in the data analysis, interpretation of findings, writing the paper for intellectual content, and approving the final paper for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Trial registration
Not applicable.
Research data availability
The research data for this manuscript is available upon request to the corresponding author.
