Abstract
Objective:
Vitamin D deficiency in Chronic Kidney Disease is a known fact, but the data regarding the prevalence and extent of vitamin D deficiency in different stages of Chronic Kidney Disease is scarce. This leads to a delay in addressing this issue. We conducted this study to assess vitamin-D deficiency in different stages of Chronic Kidney Disease to improve the evidence available to Physicians and Nephrologists which can help them better address this common finding in Chronic Kidney Disease.
Methods:
This retrospective study from a tertiary care hospital in Karachi, Pakistan, included adult Chronic Kidney Disease patients who had their serum 25 Hydroxy vitamin D levels done. The patients were classified according to stages of Chronic Kidney Disease using Estimated Glomerular Filtration Rate values. Appropriate statistical tests were performed to find a correlation between the severity of vitamin D deficiency in different stages of Chronic Kidney Disease with a statistically significant p value of <0.05 and a 95% confidence interval.
Results:
In total, 148 patients were included in the study. All patients had serum 25 Hydroxy vitamin D levels less than 30 ng/ml. The severity of vitamin D deficiency was observed to increase progressively from stage 3A Chronic Kidney Disease through stage 5. Vitamin D levels in Chronic Kidney Disease stage 3 were significantly greater than in Chronic Kidney Disease stages 4 and 5 (p value < 0.001), and stage 5 patients had the lowest vitamin D levels in our study. Similar, statistically significant findings were observed for serum phosphate levels, too, with stage 5 Chronic Kidney Disease patients having a greater serum phosphate concentration than stages 3A and 3B (p value = 0.002, 0.006).
Conclusion:
All Chronic Kidney Disease patients in our study either had vitamin D insufficiency or deficiency. In addition, we observed a significant decrease in vitamin D levels from Chronic Kidney Disease stage 3 to stage 5. Greater serum phosphate levels were detected in stage 5 compared to stages 3A and 3B.
Introduction
Chronic Kidney Disease (CKD) is defined as abnormalities of kidney structure and function present for more than 3 months with implications for health and requires either an Estimated Glomerular Filtration Rate (eGFR) of less than 60 ml/min or markers of kidney damage such as albuminuria. 1 Affecting more than 35% of the population aged 70 and above in its early stages, CKD can be easily considered more prevalent than diabetes in the global population, with a prevalence of 11% to 13%, according to a meta-analysis, most of which is in stage 3 CKD. 2 Along with its deleterious effects on kidneys, CKD has been associated commonly with vitamin D deficiency, more pronounced in patients who underwent renal transplants or patients with end-stage renal disease. 3
Vitamin D is an essential fat-soluble vitamin found in the human body as Cholecalciferol (Vitamin D3) and Ergocalciferol (Vitamin D2). These two forms receive their first hydroxylation in the Liver to form 25 Hydroxy vitamin D (calcidiol), which further undergoes final hydroxylation in the kidneys to the active form of vitamin D, that is, 1,25 dihydroxy vitamin D (calcitriol). 4 The major source of vitamin D comes from the ultraviolet B induced activation of the stored forms in the adipose tissues. 5
Vitamin D plays a pivotal role in health and disease. It is linked with a reduced risk of hypertension and cardiovascular disease, diabetes, and glycemic control, improving certain cancers, improving immunity, alleviating cognitive impairment related to Parkinson’s disease, and regulating the renin-angiotensin system. 6 The prevalence of vitamin D deficiency worldwide varies among populations, such as about 20% in developed countries and up to 100% in low socio-economic countries. 4 A deficiency of this essential vitamin contributes to a higher risk of developing cardiovascular disease, fractures, autoimmune disorders, failure to fight against type two diabetes, depression, and even viral diseases such as influenza. 5 Therefore, vitamin D deficiency impacts a variety of pathophysiological mechanisms in our bodies.
To complicate the issue, the prevalence of vitamin D deficiency in patients with CKD may be prevalent up to 80% in the pre-dialysis population. 7 Studies showed that vitamin D deficiency was almost invariably seen in CKD patients who underwent peritoneal and/or hemodialysis compared to patients who did not need dialysis or received renal transplants. 8 A proposed explanation is a defective synthesis of cholecalciferol in patients undergoing hemodialysis, irrespective of the normal substrate concentration of 7-dehydrocholesterol. 9 As mentioned before, the deficiency of 25 (OH) vitamin D may lead to decreased bone mass, muscle weakness, cognitive impairment, metabolic syndrome, and an increased risk of morbidity and mortality due to cardiovascular involvement. 10 Cardiovascular disease accounts for more than half of all causes of mortality in people with CKD, which is up to 20-fold higher than those without CKD 11 Thus, targeting cardiovascular events and complications is a treatment target to reduce morbidity and mortality 12 However, simple vitamin D supplementation has shown no effect in reducing cardiovascular events. 13
Although it is known that vitamin D deficiency is common in CKD, there is little data regarding its prevalence and severity studied and compared in different stages of CKD, especially in tropical and sub-tropical, low- and middle-income countries. Knowledge of this would allow primary care physicians and nephrologists to think about vitamin D deficiency in the CKD population at an earlier stage and set therapeutic goals in such patients to curtail its long-term deleterious effects. The aim of this study is to explore the prevalence of vitamin D deficiency among different stages of CKD in a group of the South Asian population.
Methods
This retrospective observational study was conducted from October 2020 to September 2021 at Medicare Cardiac and General Hospital and Jinnah Medical and Dental College, Sohail University, Karachi, Pakistan. The Ethics Review Committee of Jinnah Medical and Dental College approved the study (protocol No: 00050/20) and waived the requirement of informed consent, as only the anonymized hospital records were included. A total of 1000 patients who presented in the nephrology department were assessed for CKD, and 390 were diagnosed with CKD. Out of those, 148 patients between the ages of 25 and 70 years fulfilled the inclusion criteria after assessing eGFR and serum 25-OH (Hydroxy)-vitamin D levels. The study’s data is present in our data extraction sheet, which is available as Supplemental File 1. All the included participants had 25-OH-vitamin D less than 30 ng/ml and an eGFR less than 60 ml/min. All those patients excluded from the study who underwent renal replacement therapy had taken vitamin D supplementation in the last 3 months at the time of enrollment in the study or were pregnant or breastfeeding. Other baseline parameters included serum calcium, phosphate, albumin, PTH, urea, creatinine, electrolytes, alkaline phosphatase, and hemoglobin levels. Demographic data, height, weight, and other clinical parameters such as blood pressure, urine protein, primary and secondary causes of renal disease, and medication list were retrieved from the patient’s record. Patients were classified into different stages of CKD using the value of eGFR into stage 3A (eGFR = 45–59 mL/min), stage 3B (eGFR = 30–44 mL/min), stage 4 (eGFR = 15–29 mL/min) and stage 5 (eGFR < 15 mL/min). A retrospective file review of all patients with CKD was done, with an eGFR of 60 mL/min/1.73 m2.
Statistical analysis
The collected data was validated and analyzed using Statistical Package for Social Sciences version 19 (IBM, USA). The categorical data were presented as frequencies (n) and percent values (%), while the continuous variables were presented as mean and standard deviation. ANOVA test with post-hoc analysis was used to compute differences between the groups where appropriate. Since the variance was unequal (suggested by the Brown-Forsythe test), a post-hoc Games-Howell test was carried out for multiple comparisons.14,15 An ordered difference between the groups was suggested; hence, the Jonckheere-Terpstra test was carried out to confirm such differences between CKD stages. 16 Since the data was skewed, the Spearman correlation was computed where appropriate. 17 Only a p-value of less than 0.05 was considered statistically significant in all analyses. A 95% confidence interval was reported where appropriate.
Results
A total of 148 patients with CKD (male-to-female ratio = 1:1) were included in the study, ranging from 25 to 70 years of age (mean ± SD; 63.55 ± 13.84), the majority (73.6%) being older than 55 years. Most of the patients had diabetes mellitus (114 participants = 77%) and/or hypertension (144 participants = 97.3%). Patients were categorized into CKD stages using the value of eGFR as stage 3A (n = 20; 13.5%), stage 3B (n = 27; 18.2%), stage 4 (n = 52; 35.1%) and stage 5 (n = 49; 33.1%). The baseline characteristics and laboratory investigations of participants are shown in Table 1.
Baseline characteristics and laboratory assessments.

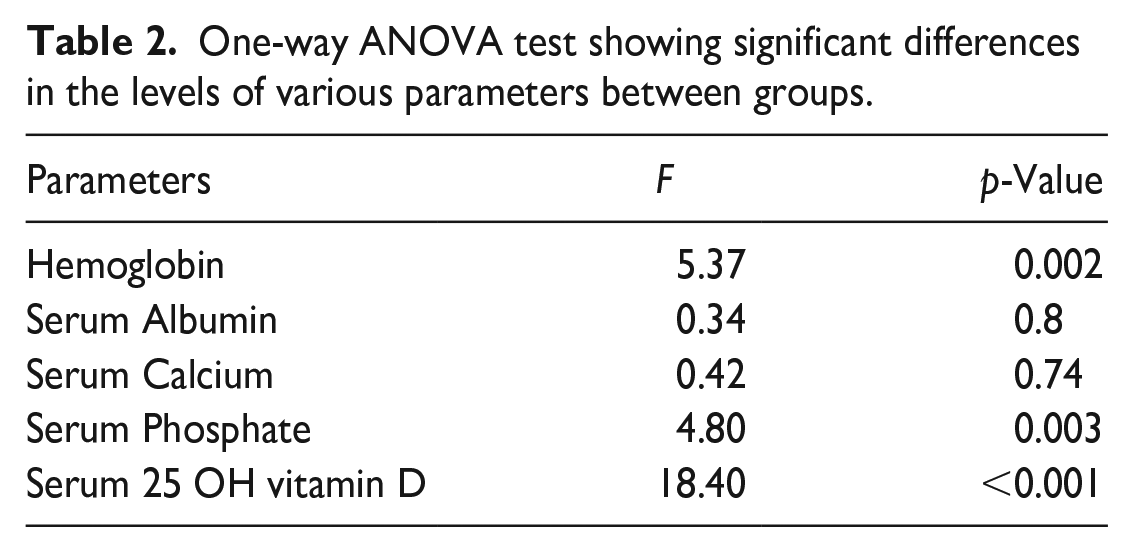
The laboratory parameters of the patients are presented in Table 2 which shows significant differences between various stages of CKD, suggesting higher levels of serum phosphate and creatinine and lower levels of serum 25-OH (Hydroxy)-vitamin D and blood hemoglobin in progressively severer form of the disease. A post-hoc analysis of the data was conducted to elaborate on the inter-group differences (Table 3) further.
One-way ANOVA test showing significant differences in the levels of various parameters between groups.
Post-hoc Games-Howell test for multiple comparisons.

The “bold” values signify the statistically significant numbers.
Figure 1 is a box and whisker plot that shows a progressive reduction in serum 25-OH-vitamin D levels in patients with CKD stage 5 as compared to stage 3A. This declining trend was confirmed with the Jonckheere-Terpstra test (J-T Statistic mean ± SD, 3917.5 ± 287.7, p < 0.001). Spearman’s Correlation was computed between various factors and is shown in Table 4, suggesting a directly proportional serum phosphate and inversely proportional 25-OH-vitamin D and calcium levels with CKD severity.

Box plot showing lower vitamin D levels (ng/ml) levels in severer forms of CKD.
Negative Spearman’s (rho) correlation test shows a decline in vitamin D levels with an increase in the severity of the disease.

Correlation is significant at the 0.05 level (2-tailed).
Correlation is significant at the 0.01 level (2-tailed).
Discussion
This study has shown that vitamin D deficiency increases with increased severity of CKD. It is also associated with lower serum calcium and increased serum phosphate levels, thus replenishing vitamin D levels could have beneficial implications in patient management, improving patient morbidity, and mortality
Decreased vitamin D would reduce calcium absorption from the intestines and kidneys, leading to increased release of parathyroid hormone and causing increased bone resorption, ultimately leading to bone weakness and pathological fractures. 5
A recent study from Karachi, Pakistan, reported that a significant number of patients (70.8%) with CKD had vitamin D deficiency. 18 This study demonstrated a significant decline in the levels of 25 OH-vitamin D as the severity of the disease increases, showing decreased levels of 25 Hydroxy vitamin D in stages 3A and 3B with much lower levels in stages 4 and 5. Thus this study not only found vitamin D deficiency in the CKD population in general but also stratified the prevalence of it in different stages of CKD.
Another study from Thailand reported significantly lower levels of 25-OH (Hydroxy)-vitamin D with increased severity of the disease. The mean vitamin D levels in stage 3A, 3B, 4, and stage 5 CKD patients were 27.84 ± 14.03 ng/mL, 25.86 ± 11.14 ng/mL, 24.09 ± 11.65, and 20.82 ± 9.86 ng/mL respectively in their study population. 19 These findings correlate with the results of our study. However, this study showed a much more severe vitamin D deficiency in all stages of CKD than in the Thai study. In this study, the mean difference in serum 25-OH-vitamin D levels between various stages of CKD was several-fold.
A similar study in the United States showed that only 29% of participants with moderate CKD and 17% of participants with severe CKD had sufficient calcidiol levels. 20
The associated factors with these findings of significantly decreased levels of vitamin D among CKD patients and making them much more prone to vitamin D deficiency include decreased sun exposure, proteinuria, malnutrition due to reduced appetite, most likely due to azotemia-related effects on the gastrointestinal tract, and a diet consisting of low levels of phosphate and protein as part of a strategy commonly used to slow down the progress of CKD. 21
Although CKD would lead to vitamin D deficiency, a reverse association exists. Evidence suggests an increased risk of CKD in the general population with vitamin D deficiency. This can be explained by the effect of vitamin D on regulating blood pressure by affecting the endothelial function and renin-angiotensin-aldosterone system (RAAS). Increased activity of RAAS has been reported in vitamin D receptor knockout mice, resulting in hypertension. Interestingly, vitamin D supplementation resulted in the suppression of RAAS and blood pressure reduction. 22 However, a large meta-analysis concluded no effect of vitamin D supplementation on blood pressure regulation. This suggested that the role of vitamin D on RAAS and the regulation of blood pressure would be more complex. 23
Previous studies have shown an association between CKD and increased serum phosphate levels. 24 Hyperphosphatemia associated with CKD can be considered the root cause of cardiovascular calcifications, renal osteodystrophy, and secondary hyperparathyroidism, associated with an increased possibility of morbidity and mortality. 25 Our results correspond to previous studies mentioned above, with increasing vitamin D deficiency with increased serum phosphate levels in more severe forms of CKD.
The main strength of the current study is that it not only highlights a very prevalent issue in the CKD population, that is, vitamin D deficiency but also stratifies the data and mentioned vitamin D deficiency in different stages of CKD as well, which makes it a more specific yet comparative study useful equally for both primary care physicians and nephrologists and highlights the need of vigilant laboratorian checks on patients, especially with severe forms of CKD for such sequelae of the disease. This study is from Pakistan, a sub-tropical region where it is expected that baseline vitamin-D levels would remain normal or higher due to abundant exposure to the sun. Patients included in the study with a wider range of ages helped us assess the variation in extremes of ages.
The main limitation of our study was that it was performed only in a single tertiary care center in Karachi, Pakistan, utilizing convenience retrospective file review for sample collection. Thus, the generalizability of the results requires validation. Another limitation was the time duration of 12 months with 148 patients included in the study. We did not perform power analysis for sample size calculation; nevertheless, a sample size of 148 was comparable to contemporary studies.
Conclusion
CKD is associated with decreased vitamin D levels, more pronounced in its later stages. The data indicates a decreased 25 OH-vitamin D levels in patients with CKD along with hyperphosphatemia and anemia. All these were noted to be more pronounced in the later stages of CKD. Larger cohort studies in other populations, including larger sample size, would validate these findings.
Supplemental Material
sj-xlsx-1-smo-10.1177_20503121221148613 – Supplemental material for 25-Hydroxyvitamin-D deficiency in chronic kidney disease stages III, IV, and V in South Asian population: a retrospective cohort
Supplemental material, sj-xlsx-1-smo-10.1177_20503121221148613 for 25-Hydroxyvitamin-D deficiency in chronic kidney disease stages III, IV, and V in South Asian population: a retrospective cohort by Muhammad Raheel Abdul Razzaque, Sameer Saleem Tebha, Alaa Tukruna, Aabiya Arif, Lucas Marian Kogut, Nasir Ali Afsar, Dania Shabbir and Zain Ali Zaidi in SAGE Open Medicine
Footnotes
Author contribution
Muhammad Raheel Abdul Razzaque and Sameer Saleem Tebha: Wrote the initial draft of the manuscript and did a detailed literature review; Alaa Tukruna, Aabiya Arif, Zain Ali Zaidi, and Nasir Ali Afsar: Wrote the discussion and edited the manuscript, along with the final review and proofreading of the manuscript; Lucus Marian Kogut and Dania Shabbir: performed statistics and drafted the figures; All authors approved the final version for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from “Institutional Review Board (IRB) Jinnah Medical and Dental College, Karachi, Pakistan (APPROVAL NUMBER: 00050/20).”
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because “All the included data in our study is extracted directly from hospital records and as it is our institutional policy to take prior consent before the commencement of treatment for the use of any and all data for the purpose of academic and research activities as such the IRN board waived the requirement for individual patient consent.”
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
