Abstract
Objective:
Vitamin D deficiency in a patient with autoimmune rheumatic disease is the most discussed topic these days, is considered to be a pandemic, and its prevalence and associations have been studied by many countries. We aim to study the prevalence of vitamin D deficiency in autoimmune rheumatic diseases in patients visiting the rheumatology clinic at the Jigme Dorji Wangchuck National Referral Hospital in Thimphu.
Methods:
A descriptive study was conducted on 126 patients in Jigme Dorji Wangchuck National Referral Hospital, who were visiting the rheumatology clinic. Vitamin D levels were analysed among these rheumatology patients. EpiData software is used for data entry and analysis.
Results:
Out of 126 patients, 71 had rheumatoid arthritis, 34 had systemic erythema-tosus, 8 mixed connective tissue disease and 13 had other autoimmune rheumatic diseases. Among these, only 12 (9.52%) patients had normal vitamin D levels, and 114 (90.5%) patients had lower than normal serum levels of the vitamin. Among these, 75 (59.5%) were deficient and 39 (31.0%) were insufficient.
Conclusion:
The study found that the prevalence of Vitamin D deficiency is high with 59.5% among patients with autoimmune rheumatic diseases and only with 9.5% were with normal levels of vitamin D.
Introduction
Autoimmune rheumatic diseases (ARD) are a diverse category of illnesses characterised by immune system dysfunction that causes numerous organ damage. 1 Systemic lupus erythematosus (SLE), rheumatoid arthritis (RA) and systemic sclerosis (SSc) are autoimmune rheumatic disorders that primarily affect connective tissues (cartilage, joint synovium and skin). 2 It is estimated that autoimmune diseases affect more than 7% of the general population. 3 Vitamin D insufficiency increases rheumatic disease risk, potentially worsening the condition. Epidemiological research links autoimmune diseases to vitamin D deficiency, leading to increased use of vitamin D supplements as treatments and preventatives.4–6
Vitamin D is a fat-soluble hormone that aids bones in digesting calcium and phosphate. Its active form, calcitriol, denoted as 1,25-dihydroxy-vitamin D (1,25(OH)2D3), acts as an endocrine hormone, affecting the body at sub-nanomolar quantities. 7
Vitamin D deficiency is a global health concern affecting about 1 billion people, with South Asia experiencing a high rate (70%), with up to 80% of healthy South Asians having low levels, with 40% experiencing severe deficiencies, despite advancements in technology and therapies, while Southeast Asia has a range of 6%–70%.8–12 A 2019 meta-analysis of 1692 studies revealed a prevalence of poor vitamin D status ranging from 18% to 46% in 129 studies including 21,474 participants from 23 African countries. 13 India experiences vitamin D insufficiency ranging from 40% to 99%, according to a literature review. 14
Assessing vitamin D levels in rheumatologic patients involves considering factors such as sunlight avoidance, corticosteroids and hydroxychloroquine, and skin pigmentation. Higher-dose supplements should be considered for these patients. It is crucial to assess vitamin D status for all patients, regardless of their condition, and recommend adequate supplementation to avoid side effects. 15
Autoimmunity rheumatic disease patients often have vitamin deficiencies, requiring treatment for hypovitaminosis. Vitamin D supplementation, food fortification and variety are recommended for optimal health. Deficiency affects all ages and is linked to factors like time, season, ethnicity, region and latitude.16,17
A retrospective study comparing vitamin D deficiency among ARD found high prevalence in patients with SLE and RA, with similar odds in RA and SLE patients. 18 Recent research highlights vitamin D insufficiency in SLE, suggesting in vitro vitamin D supplementation corrects immunological defects. 19 Early RA patients with low serum vitamin D levels may experience negative correlations with disease severity, activity and functional impairment. 19 A study found that 84% of RA patients lack Vitamin D, potentially contributing to disease development or activity, compared to 34% of control groups.20,21 Multicentre European Survey found that vitamin D deficiency is common in RA patients, with statistically significant disparities among countries. 22
Cross-sectional research on autoimmune rheumatic disorders found that factors like young age, non-white background, body mass index, smoking, less exercise and first-time clinic visits significantly contribute to vitamin D-25 deficiency. 23 This study demonstrated that national policies, health education, and promotion programmes for the most vulnerable populations can be effective. They found that evaluating vitamin D-25 levels in individuals with rheumatic illnesses, particularly those at high risk, is beneficial.
The study investigates vitamin D levels and their therapeutic implications in rheumatic disease severity. It aims to determine the prevalence of vitamin D deficiency and the relationship between demographic characteristics of ARD. The findings will be crucial for future research on vitamin D’s impact on disease severity, activation and progression, impacting clinical practice at Jigme Dorji Wangchuck National Referral Hospital.
Methodology
Study design
This was a cross-sectional study on the prevalence of vitamin D deficiency in autoimmune rheumatic disease visiting the rheumatology clinic at JDWNRH from October 2020 to October 2021.
Study setting
This study was carried out at the rheumatology clinic of JDWNRH.
Study population
The study population of this study is all those patients fulfilling the inclusion and exclusion criteria.
Inclusion
All new and old cases of ARD visiting the clinic.
Exclusion
Patients on Vitamin D supplementation without prior vitamin D test level.
Chronic diseases such as diabetes mellitus, chronic kidney disease, chronic liver disease.
Paediatric age group (age < 12 years)
Sample size
As per the data in the previous year, there were 266 patients with ARD visited the rheumatology clinic. Taking the population sample size as 266, assuming that there will be 50% of the patients with autoimmune rheumatic disease with vitamin D deficiency as there is a lack of any baseline data within Bhutan as well as in the region. Open Epi-software for calculating proportion was used to calculate the sample size, following equation was used to determine the sample size with a 95% confidence interval. N = zα2 × p(1−p)/d2
n = sample size
p = anticipated % frequency
DEFF = design effect
N = population size
d = desired absolute precision or absolute level of precision.
The sample size for a 95% confidence interval for the study as per the above equation calculated is 158.
Study procedure
The consecutive convenience sampling was done wherein all patients visiting the Rheumatology Clinic were screened for the inclusion and exclusion criteria. Those patients eligible for the study were provided with the information sheet along with a verbal explanation of the rationale, risk, and benefit of the study to the potential study participants and informed consent was obtained. The sample size of 126 patients was reached based on the accessible population from the census of patients visiting the clinic.
Data variables, sources and data collection method
Baseline characteristics include age, gender, education, occupation, region, diagnosis and vitamin D level. Proforma was used to collect the data from the patients at the rheumatology clinic. Serum vitamin D reports were collected from the Laboratory Department at JDWNRH. Cobas e 411 analyzers product of Roche Diagnostic for clinical laboratories test number 1500 is used for testing Vitamin D, which is intended for the qualitative determination of total vitamin D. The electrochemiluminescence binding assay is used for immunoassay analyzers. Definition of vitamin D levels: Vitamin D deficiency is defined as vitamin D level less than 20 ng/ml, vitamin D insufficiency is between 20 and 30 ng/ml and the vitamin D sufficient level is more than 30 ng/ml.
Data analysis and statistics
Data were double entered and validated using EpiData (version 3.1, EpiData Association, Odense, Denmark). Data analysis was done using EpiData analysis version 2.2.2.183 software, developed by Jens M Lauritsen, MD, PhD from Denmark. Data were described in terms of number, mean and percentages for socio-demographic profile and the diagnosis of autoimmune rheumatic disease and level of vitamin D.
Operational definition
Vitamin D levels:
Sufficient: Serum 25(OH)D level (75–250 nmol/L), (>30 ng/ml)
Insufficient: Serum 25(OH)D level (50–74 nmol/L), (20–30 ng/ml)
Deficient: Serum 25(OH) D level (<50 nmol/L), (<20 ng/ml)
Rheumatism: Refers to various painful medical conditions that affect joints, bones, cartilage, tendons, ligaments and muscles
Rheumatic diseases: Rheumatic diseases, also called musculoskeletal diseases, are characterised by pain and a consequent reduction in the range of motion and function in one or more areas of the musculoskeletal system.
Autoimmune disease: Diseases in which cells of the adaptive immune system such as autoreactive T and B cells become overreactive and produce self-reactive T-cell and antibody responses.
Autoimmunity: Refers merely to the presence of antibodies or T lymphocytes that react with self-antigens and does not necessarily imply that self-reactivity has pathogenic consequences.
Autoinflammatory diseases: Hereditary disorders characterised by recurrent episodes of severe inflammation and fever due to mutations in controls of the innate inflammatory response.
Autoimmune rheumatic disease: Any rheumatic disease where the pathogenesis is caused by autoimmune in nature.
Cytokines: Soluble proteins interact with specific cellular receptors that are involved in the regulation of the growth and activation of immune cells and mediate normal and pathologic inflammatory and immune responses.
Arthritis: Inflammation of joints of the body.
Arthralgia: Pain on the joints without signs of inflammation.
Myalgia: Aches and pain felt on the muscles of the body without signs of inflammation.
SLE: is an autoimmune disease in which organs and cells undergo damage initially mediated by tissue-binding autoantibodies and immune complexes.
SSc disease: It is an uncommon connective tissue disorder characterised by multisystem involvement, heterogeneous clinical manifestations, a chronic and often progressive course, and significant disability and mortality. Multiple genes contribute to disease susceptibility; however, environmental exposures are likely to play a major role in causing SSc.
RA: is a chronic inflammatory disease of unknown aetiology marked by symmetric, peripheral polyarthritis. It is the most common form of chronic inflammatory arthritis and often results in joint damage and physical disability.
Spondyloarthropathy: A group of overlapping disorders that share certain clinical features and genetic associations. These disorders include ankylosing spondylitis (AS), reactive arthritis, psoriatic arthritis and spondylitis, enteropathic arthritis and spondylitis, juvenile-onset spondyloarthritis (SpA) and undifferentiated SpA.
Mixed connective tissue disease (MCTD): Autoimmune disease features a combination of SLE, scleroderma (Ssc), polymyositis and RA. Positive anti-U1-RNP antibody.
Sjogren’s syndrome: It is a chronic, slowly progressive autoimmune disease characterised by lymphocytic infiltration of the exocrine glands resulting in xerostomia and dry eyes. Approximately one-third of patients present with systemic manifestations; a small but significant number of patients develop malignant lymphoma. The disease presents alone (primary Sjögren’s syndrome) or in association with other ARD (secondary Sjögren’s syndrome).
Western: Thimphu, Paro, Haa, Wangdiphodrang, Punakha, Gasa.
Central: Bumthang, Zhemgang, Dagana, Trongsa.
Eastern: Mongar, Lhuntse, Trashigang, Trashiyangtse, Pemagatshel, SamdrupJongkhar.
Southern: Sarpang Zhemgang, Sarpang, Samtse.
Ethical considerations
The ethical approval was obtained from the Interim Institutional Research Ethics Board (I-IRB) Khesar Gyalpo University of Medical Sciences of Bhutan (KGUMSB) with reference number IRB/PO20/013/471. The administrative approval to conduct the study was obtained from JDWNRH management. Informed written consent was obtained from the study participants. Data confidentiality, privacy and confidentiality of the patient were ensured by avoiding the name from the proforma. In the recruitment process, subjects were given full autonomy to enrol in the study. Even if the subjects refused to take part in the study, their treatment plans and management were not compromised. Subjects were informed that they could at any time voluntarily withdraw from the study.
Results
Demographics
A total of 126 patients were enrolled in the study, and data from these study participants were gathered to delineate the demographics and clinical characteristics. All the participants enrolled were from different regions of the country.
The mean age at the time of the study was 41.4 years. Among the participants, 87.3% and 12.7% were women and men, among all participants 23.8% and 6.3% were housewives and dependent, 42.9% of participants did not have any education background and 36.0% were from the western part of the country.
Baseline characteristics including age, sex, occupation, education and region are shown in Table 1. Study participants included 71 RA, 34 SLE, 8 MCTD and 13 other autoimmune rheumatic diseases including AS, Sjogren’s syndrome and SSc.
Demographic characteristics of patients with different ARD (n = 126).

Clinical
Vitamin D levels
The mean (SD) vitamin D levels of these patients were 19 (11.78) ng/ml. Among these only 12 (9.52%) patients had normal vitamin D levels, 114 (90.48%) patients had lower than normal serum levels of vitamin D. Among these, 75 (59.5%) were deficient in vitamin D and 39 (30.9%) insufficient as shown in Figure 1.

Level of vitamin D in patients who attended a rheumatology clinic.
When analysing the four most frequent diagnoses (SLE, RA, MCTD, and Others), it was found that patients with SLE had a higher prevalence of vitamin D deficiency at 70.6% as shown in Figure 2.

Vitamin D deficiency and ARD.
The prevalence and severity of vitamin D deficiency in different ARDs are shown in Table 2.
Prevalence of vitamin D deficiency in different ARDs according to various categories of deficiency.

Vitamin D deficiency by age, sex, occupation and educational groups
Vitamin D levels and deficiency status according to selected patient’s demographic characteristics as shown in Table 3, in this study, 24.6% of the population aged between 12 and 29 years, whose prevalence of vitamin D deficiency was 61.3%, while the prevalence of vitamin D deficiency of the group whose age between 30 and 49 years was 58.3% and above 50 years was 60%. Patients were stratified according to age ranges and vitamin D levels were determined according to their classification as shown in Figure 3.
Vitamin D levels and deficiency status according to selected patient’s demographic characteristics.


Vitamin D status as per age groups.
When comparing vitamin D means by sex as shown in Figure 4, it was found that the average vitamin D level in women was lower than in men 17.88 (12.18) versus 19.35 (8.53). Sixty percent of the women had vitamin D deficiency compared to 56.3% of the men. However, 12.5% of men had a normal value of vitamin D compared to 9.1% of women. We also found that the deficiency was more prevalent in females (60.0%) than males (56.3%). However, the insufficiency was also more prevalent in the male population (31.3%) than in the females (30.9%).
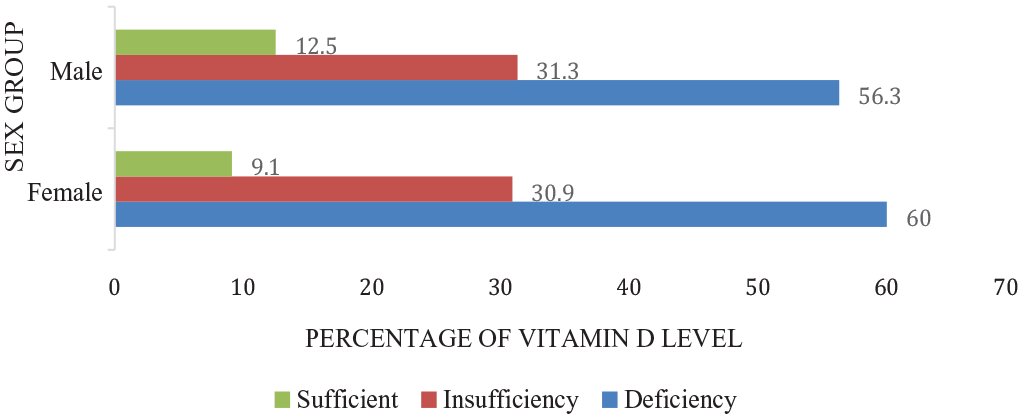
Vitamin D status as per sex groups.
Out of 126 participants, in the occupation category maximum number were housewives, the average vitamin D level among these groups was 19.03 ng/ml. It was found that the deficiency level among farmers was 65.4%, and the rest of the groups were almost similar as shown in Figure 5.

Occupation groups with vitamin D levels.
Among 126 participants, 54 patients did not have any educational background and were found to have a vitamin D deficiency level of 62.9%; however, those who studied college and above had the highest vitamin D deficiency of 64.0% as shown in Figure 6.

Education groups with vitamin D levels.
Discussion
The study is the first in Bhutan to investigate vitamin D deficiency in patients with autoimmune rheumatic disorders visiting the rheumatology clinic at Jigme Dorji Wangchuck National Referral Hospital. The study found that 90.5% of patients had poor vitamin D levels, with 59.5% experiencing deficiency and 31.0% insufficiency. The highest prevalence was found in SLE (70.6%), followed by RA (57%), MCTD (50%) and other groups (46.2%).
Similarly, Toloza’s 24 research found 66.7% vitamin D insufficiency and 17.9% with deficiency. However, prevalence varied among nations, including Saudi Arabia (89.7%), Norway (82%), Poland (71%), Hongkong (27%) and the United States (20%).25–28 A study at King Abdul Aziz University Hospital 25 found 98.8% vitamin D deficiency among auto-immune rheumatic patients, while Haroon et al. 29 found 70% of new patients entering rheumatology clinics had vitamin D deficiency, nearly 10% higher than the study’s findings. Sheng et al. 30 revealed that SLE patients have a greater prevalence of vitamin D insufficiency than RA and other autoimmune rheumatic illnesses. A study in Egypt 31 found 13.2% vitamin D deficiency in SLE patients, while in China 32 27% deficiency was reported. Both studies highlight the need for improved vitamin D intake in patients with SLE. One study 33 shows vitamin D deficiency prevalence in SLE (84.3%) is higher than RA (48.8%), strongly associated with SLE. SLE patients and RA patients experience vitamin D insufficiency, with Kerr GS et al. 34 revealing a 43% prevalence in elderly RA patients. Exposure to ultraviolet light rays impacts vitamin D synthesis, as shown in studies. The higher prevalence of vitamin D deficiency among autistic rheumatic diseases is SLE. This could be because SLE patients’ photosensitivity25,35 prevents proper vitamin D synthesis, necessitating proper skin exposure for chronic treatment with corticosteroids and hydroxychloroquine.
A reduced level of vitamin D was also documented in a patient with SpA in an eight case control research, 36 which is similar to the findings of this investigation. This study discovered that younger age groups from 10 to 29 and older adults over 50 years had a higher prevalence of vitamin D deficiency with 61.3% and 60%, respectively, while age groups between 30 and 49 had 58.3%. However, Ruiz-Irastorza et al. 32 discovered a lower prevalence among older patients. The age group between 10–29 and over 50 were housewives, students, civil servants and independent; they may not have had enough morning sunshine exposure, which can decrease vitamin D synthesis. A study in Northern Taiwan 37 found that women, youths, high education, urban areas and inactivity are predictors of vitamin D deficiency. Women aged 30–39 had higher vitamin D deficiency, while those with graduate degrees had 31%. Higher education levels, college and higher, and inactivity were also found to be significant predictors. A systematic review by Sawati et al. 38 found that shift workers, healthcare workers, and indoor workers are at higher risk of vitamin D deficiency compared to outdoor workers. Nonetheless, this study reveals that outdoor workers, primarily farmers, have a high vitamin D deficiency of 65%. However, this may be due to farmers using sunscreen or being unable to work due to illness. Indoor workers, who spend most of their working hours indoors, are exposed to sunlight during the mornings and evenings.
The study found that patients from Central and Western regions had a higher prevalence of vitamin D deficiency compared to those from Eastern regions, with 65.8%, 65.2% and 47.6%, respectively. This suggests that geographic locations can impact vitamin D status, similar to a study in Korea 39 that found one to two degrees of latitude change affects vitamin D levels.
Numerous studies have shown the connection between vitamin D levels and ARD. Vitamin D supplementation 40 significantly reduced the mean disease activity in patients with SLE, and RA patients using vitamin D supplements also experienced similar reductions.41,42 This study also found vitamin D deficiency in ARD but not disease activity or correlation with supplementation. Nonetheless, similar findings and conclusions might be drawn from this study as well because all previous research and available literature have produced similar outcomes.
Limitation
The participants in this study were only representatives of autoimmune rheumatic disease visiting a rheumatology clinic at a national referral hospital, which limits to inference the results of this study to the overall population. Due to the lack of adequate testing reagents for vitamin D, it has led to limitations in recruiting adequate samples for this study. This research was conducted in a single-centre study, if there had been more centres of rheumatology clinics other than JDWNRH, more participants could have been recruited to study the status of vitamin D deficiency. Prevalence of vitamin D deficiency could be more than the current result if we have included all the patients, who were already on vitamin D supplementation prior to knowing their vitamin D level. One of the limitations is pretesting of the data collection was not done.
Conclusion
Vitamin D deficiency is prevalent among ARD. The deficiency of vitamin D is more prevalent (70.6%) among SLE. Age group 12–19 had the highest deficiency with 61.3% and a participant who studied more than college had the highest deficiency with 64% as compared to those who have no education background and who studied till higher secondary level. Geographically, Western and Eastern regions had almost 65% of deficiency as compared to the Central of the country.
This study generates local evidence of low vitamin D among autoimmune rheumatic disease patients in Bhutan like in any other country. The interventions to address the deficiency could have a positive impact on not only the ARD patients but the general population too.
Recommendation
The prevalence of vitamin D deficiency among this small participant was found to be almost 60%, if a multi-centre study in the general population and other patients could be done, to determine the prevalence, associations and disease activity that could benefit the overall population in our country. I would recommend policymakers and programme officers establish proper guidelines in screening and supplementations of vitamin D and also health promotion and education on the impact of low vitamin D on our body.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231223313 – Supplemental material for Prevalence of vitamin D deficiency in patients with autoimmune rheumatic diseases visiting the rheumatology clinic at the National Referral Hospital, Bhutan
Supplemental material, sj-docx-1-smo-10.1177_20503121231223313 for Prevalence of vitamin D deficiency in patients with autoimmune rheumatic diseases visiting the rheumatology clinic at the National Referral Hospital, Bhutan by Sangay Dorji, Sonam Yangchen and Pem chuki in SAGE Open Medicine
Footnotes
Acknowledgements
This thesis could not have been better and completed on time had it been not for the invaluable contributions made by my supervisor and co-supervisors. They have been genuinely considerate and kind enough to spare their precious time guiding me. Therefore, I would like to express my heartfelt gratitude to them.
Dr Sonam Yangchen (supervisor) and Dr Pem Chuki (co-supervisor) left no stone unturned to ensure my thesis was done on time. They sacrificed their precious limited family time and other occasions going and making necessary corrections. Without their support and unwavering assistance, this thesis would not have materialised
I would like to thank my faculty members, consultants and staff for their contributions. I am very grateful to the staff at the Rheumatology Clinic
I would like to thank Mr Tshering Choeda, Senior Programme Officer, Faculty of Postgraduate Medicine for guiding and helping me with statistical analysis and data interpretation
Finally, I would like to thank my patients and their relatives for their support and cooperation in participating in my study
Authors’ contributions
Sangay Dorji, Sonam Yangchen and Pem Chuki were involved in the conception and design of the protocol. Sangay Dorji acquired the data, performed the data analysis/interpretation and manuscript. All authors were involved in critically reviewing the paper and giving approval for the final version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethics approval
Ethical clearance from Interim Institution Research Ethics Board (I-IRB), Ref. No. INTERIM IRB/PO20/013/471.
No minors were involved as a study participant.
Ethical clearance is obtained from Khesar Gyalpo University of Medical Science of Bhutan (KGUMSB).
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

