Abstract
Objectives:
Routine dipstick urinalysis is a useful and inexpensive method for the early detection of asymptomatic diseases. Our study was carried out to detect the prevalence of urinary abnormalities among an apparently asymptomatic healthy female group and emphasize the value of routine dipstick urinalysis.
Methods:
A cross-sectional study was carried out among second to sixth (final)-year female medical students of Nnamdi Azikiwe University, Nnewi campus between 8 and 30 September 2019. A total of 100 subjects were enrolled. First-morning midstream urine samples were collected and promptly tested with dipstick strips. Subjects with abnormal findings were informed and treated appropriately.
Results:
In all, 54 (54%) of the subjects had urine abnormalities. Trace proteinuria was the commonest abnormality seen with a prevalence of 33%, followed by abnormal urine appearance (17%), leucocytes (14%), nitrites (8%) and urobilinogen (2%). Urine abnormalities were commoner in students who resided in the school hostel. Abnormal urine appearance was significantly associated with nitrites (0.003) and leucocytes (0.001).
Conclusion:
Routine dipstick urinalysis should be performed among students and apparently healthy individuals for early detection and diagnosis of renal diseases to facilitate quick interventions and prevent further progression.
Introduction
Dipstick urinalysis is a basic test used to determine pathological changes in a patient’s urine. Routine testing of urine with multi-parameter strips is the first step in the diagnosis of many diseases. It involves testing for the presence of glucose, proteins, ketones, ascorbic acid, haemoglobin, bilirubin, acetone, nitrites and leucocytes as well as assessing the hydrogen ion concentration (pH) and specific gravity.
Historically, urine examination involved the assessment of the appearance, smell and taste of urine, and physicians, known as Pisse Prophets, carved out lucrative careers based on urine examination (uroscopy) and its interpretation (uromancy). 1
A clean-catch (midstream) urine specimen is ideal and must be examined no later than 2 h after uromancy collection to obtain correct results. Certain foods (e.g. beetroot), medications (e.g. rifampicin), metabolites and infections can affect urine appearance. The presence of white blood cells, red blood cells, pus or bacteria can make urine turbid. It is not abnormal to find trace amounts of urobilinogen in urine. Dipstick testing is 91%–100% sensitive and 65%–99% specific for the detection of haemoglobin. 2 Dipstick tests for trace amounts of protein yield positive results at concentrations of 5 to 10 mg per dL – lower than the threshold for clinically significant proteinuria. 3 Normally, nitrites are absent in urine and their presence points to a urinary tract infection (UTI).A dipstick urinalysis positive for leucocyte esterase and/or nitrites in a midstream-void specimen reinforces the clinical diagnosis of UTI. 4
One of the most prevalent bacterial infections worldwide are UTIs. The mean peak age of incidence of uncomplicated UTIs in adult women is between 15 and 39 years. 5 Young women are at the highest risk of community-acquired UTIs. A major reason for this is the anatomical structure of the female genitourinary tract. The shortness of the urethra, as well as its closeness to the anus, makes it easy for bacteria to ascend the urinary tract. Also, altered vaginal flora, in cases of incomplete cure and recurrence of genitourinary tract infections, plays an important role in the development of UTIs.
Dipstick urinalysis is an important and effective screening tool for UTIs in females. Due to the lack of data on dipstick urinalysis among asymptomatic female medical students, the study was done to ascertain the prevalence of abnormal urine parameters among the study population. The impact of abnormal findings on women’s health is also discussed.
Method
The study was conducted from 8 to 30 September 2019, among 100 apparently healthy female medical (pre-clinical and clinical) students (second–sixth/final year) of the Faculty of Medicine, Nnamdi Azikiwe University, Nnewi Campus, Nnewi, Anambra State, Nigeria. Medical Training in the University is divided into the pre-medical, pre-clinical and clinical stages. In the first year, the medical students are exposed to pre-medical sciences on the main campus at Awka, and thereafter, Basic Medical Sciences in the second and third levels of study (pre-clinical stage) at Okofia in Otolo, Nnewi. From the fourth to the sixth years, they are exposed to clinical courses at Nnamdi Azikiwe University Teaching Hospital, Uruagu Nnewi.
Our study was descriptive and cross-sectional. Convenience sampling method was used. Students were considered eligible for entry into the study if they met the following criteria: female, age ⩾ 17 years, apparently healthy, medical students (second–sixth years) in the 2018/2019 Academic Session in the Faculty of Medicine, Nnamdi Azikiwe University. Exclusion criteria included first-year pre-medical students, male students, female students who did not give consent, those involved in vigorous exercises (e.g. racing), cigarette smoking, use of oral contraceptives, use of antibiotics and NSAIDS, as well as those who were menstruating or menstruated within a week before the study.
Sample size determination
The following formula 6 was used to determine the sample size
where
n = Minimal sample size
Z = Standard normal deviation at 95% confidence level = 1.96
P = Prevalence = 5.25% = 0.0525 7
1 − P = 1 − 0.0525 = 0.9475
d = Level of precision required = 0.05
Thus,
n = 76.4
n = approx. 76.
Attrition = 10% of 76 = 7.6
Hence, minimal sample size = 7.6 + 76.0 = 83.6
n = approx. 84
To improve the reliability of the study results, the sample size was increased to 100.
Bio data (age, gender, residence, level of study) of the subjects were obtained using anonymous questionnaires to ensure confidentiality. The aim of the study and the contents of the questionnaires were explained to the students. Written informed consent was obtained from each subject. Their participation in this study was entirely voluntary, and they were free to leave at any time. After filling the questionnaires, each of the participants was given a sterile urine container which was properly labelled with her serial number for urine sample collection The subjects were instructed on how to obtain an early morning urine specimen thus – to clean their perineum, separate the labia and collect midstream urine. Urine specimens were transported to the laboratory and analysed immediately after collection using the G. SURETEST 11S reagent kit for urinalysis (European Authorized Representative, EC Rep – Wellkang Ltd, UK), used for testing 11 components in urine: specific gravity, leucocytes, nitrite, pH, blood, protein, glucose, ascorbic acid, ketones, urobilinogen and bilirubin. The urine was tested shortly after collection. The reagent strip was dipped into the urine for 2 s (ensuring that all the test areas were fully immersed) before being drawn across the rim of the urine container to remove any excess. The colours on the dipstick strip were compared with those supplied on a colour chart on the test kit container after they were placed on a flat horizontal surface for 1 min. The colour changes seen after 2 min or on the test pad edges were not noteworthy.
Statistical analysis
Entry of the data and its analysis was done using Statistical Package for Social Sciences (SPSS) version 20.0. Differences in proportion were evaluated using the Chi-square test or by Fisher’s exact test where figures were small. Statistical significance was obtained if p value < 0.05.
Results
The socio-demographic profile in Table 1 showed that a total of 100 female medical students aged between 18 and 28 years were enrolled on the study, with a mean age of 22.28 ± 2.349 years. In all, 42 respondents (42.0%) were in the age group 21–23 years; 53 (53.0%) resided in the hostel, while 47 (47%) stayed off-campus.
Socio-demographic characteristics of respondents.

SD: standard deviation.
Abnormalities were noted in 54 (54%) urine samples. Trace proteinuria was the most common abnormality found in 33 (33%) of the subjects, followed by abnormal urine appearance in 17 subjects (17%). A minority of the respondents had nitrite and leucocyte in their urine samples: 8 (8%) and 14 (14%), respectively. These are subtle incidental findings that would warrant further investigations as shown in Table 2.
Prevalence of abnormal dipstick urine abnormalities.

Second year medical students were noted to have a slightly higher number of urine abnormalities as seen in 17 samples (Table 3).
Distribution of the levels of study of subjects with dipstick urine abnormalities (%).

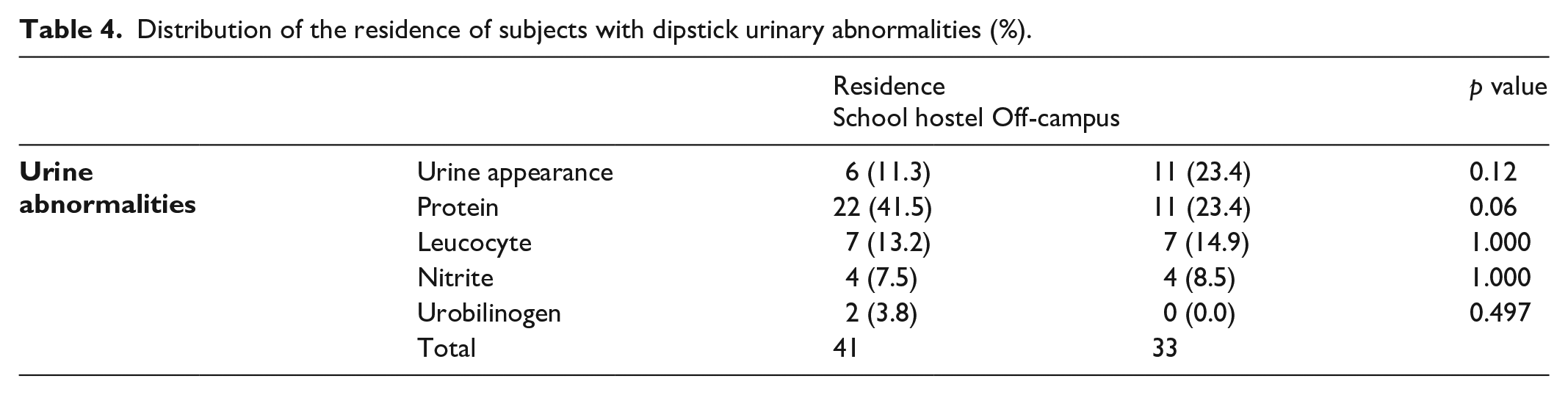
In all, 41 subjects with urine abnormalities were noted to reside in the school hostel as shown in Table 4. Despite this, there was no significant association between the residence of the subjects and their urine abnormalities.
Distribution of the residence of subjects with dipstick urinary abnormalities (%).
Five subjects (62.5%) with abnormal urine appearance were also noted to have nitrites in their urine samples. This association is statistically significant (p value = 0.003). Table 5 shows that subjects with abnormal urine appearance also had nitrites and leucocytes in their urine samples. This was found to be statistically significant (p values = 0.003 and 0.001 respectively).
Comparison between the dipstick urine abnormalities of the study subjects (%).

Statistically significant.
Discussion
Urinalysis is a simple and cheap test that can aid the diagnosis of many health conditions. Our study was carried out among asymptomatic female medical students, and thus differs from most previous studies which were conducted in out-patient clinics, hospitals and laboratories.
A total of 100 female medical students participated in this study. This is closer to that obtained in a study by Amali et al. (213) 8 and Ugwuja et al. (250) 9 but is much lower than that obtained in studies by Fouad et al. (2500) 10 and Silverberg et al. (23,427). 11 The reason for the variations could be due to the differences in the size of the study population.
In our study, the mean age of female medical students was 22.28 ± 2.349 years (18–28 years). The majority of students in our study (42%) were within the age group 21–23 years, followed by 30% in the age group 18–20 years. This is in congruence with the mean age of 20.08 years (13–38 years) obtained in a study by Khallid et al. 12 Our findings were also in line with the mean age of 20.2 ± 1.2 years (18–25 years) obtained in a study by El-Tayeb et al. 13
In all, 53 (53%) of our study participants resided in the school hostel, while 43(43%) lived off-campus. This is in contrast to the study by Amali et al. 8 and El-Tayeb et al., 13 where all the study participants resided in the school hostel. Our study subjects were grouped according to their residence into two – school hostel and off-campus. Students who reside in the university hostel share amenities including the toilets, while those who stay off-campus (outside the school premises) live in rented self-contained apartments. Hostel girls may hover over the toilet seat (rather than sitting on it) when urinating to avoid contact with germs. They also tend to ‘hold in pee’ longer than necessary, which causes urinary retention that promotes the growth of bacteria that can cause UTIs. On the other hand, girls who live off-campus typically don’t share restrooms because they have their private toilets. We studied them to determine if there was any significant relationship between their places of residence and presence of abnormal urine parameters. Our study showed that 41 subjects with urine abnormalities resided in the school hostel as shown in Table 4. Despite this, no significant association was found between the subjects’ residences and their urine abnormalities.
The prevalence of dipstick urinary abnormalities in our study was 54%. This is much higher to values obtained by Ugwuja et al. (20.7%), 9 Khallid et al. (15.4%) 12 and Oviasu et al. (5.25%), 7 but closer to the values obtained by Tjale (35%) 14 and Isezuo et al. (29.8%). 15 The prevalence of urine abnormality in our study appears to be significantly high because of the possibility of transient abnormalities as seen by Fouad et al. 10 Thus, the persistent abnormality rate could be much lower.
In our study, the dipstick urine abnormalities in our study were the following: trace proteinuria (33%), abnormal urine appearance (17%), leucocytes (14%), nitrites (8%) and urobilinogen (2%). Our findings can be compared with that of Oviasu et al., 7 in which the prevalence of urine abnormality for both sexes was 5.25% with proteinuria and haematuria accounting for 4.7% and 0.55%, respectively. Oviasu et al. also noted that proteinuria and haematuria were more frequent in girls being 4.72% and 0.79%, respectively, as against 4.68% and 0.29% for boys, though the difference was not statistically significant; significantly, 6 of the girls with proteinuria (0.52%) had nephrotic range proteinuria though oedema-free. Our results are also comparable to that of Silverberg et al., 11 in which the prevalence was 1.6% proteinuria of 2+ or more on two occasions, 0.6% slight haematuria or greater on two occasions, and 0.06% slight or more glycosuria on two occasions. Our findings contrasted that of Khallid et al. 12 in which 8.1% had pyuria, 6.1% had haematuria and 4.8% had albuminuria. In general, results of urine components from most studies show low values, unlike our higher values. We believe this may be due to differences in the demographic characteristics of the subjects, prevalent urinary diseases in the various study areas and/or the methodology used.
False positives in females could be caused by exposure to cold, extended recumbence, exercise, dehydration or contamination of urine samples with menstrual blood. However, we endeavoured to keep urine samples free of menstrual blood contamination by excluding females who were menstruating or had just finished menstruating throughout the study period.
Proteinuria was the commonest urine abnormality in our study. It was the commonest in the studies conducted by Silverberg et al., 11 and Oviasu et al. 7 It was also the third commonest in studies conducted by Fouad et al. 10 and Khallid et al. 12 Proteinuria in female medical students can be a sign of underlying renal illness or a one-time occurrence. Mucus, pus, blood, or strongly alkaline (pH > 8) and highly concentrated urine can induce false-positive results; dilute urine can cause false-negative results. 16 In our investigation, taking the first urine sample in the morning helped rule out orthostatic proteinuria as a cause of isolated proteinuria in our subjects. Proteinuria is a mediator as well as a sign of progressive glomerular damage, and investigations have demonstrated that it is a mediator as well as a marker of progressive glomerular damage. 17 It is one of the most powerful predictors of renal function decline. 18 Asymptomatic proteinuria should be investigated extensively to detect and possibly prevent end-stage renal disease (ESRD). 19
The prevalence of leucocyte esterase was 14%. This is similar to findings by Isezuo et al., 15 who found leukocyte esterase to be present in only female subjects. It was also common in female adolescents as reported by Hanif et al. 19
In our study, the prevalence of nitrite was 8%. This is similar to nitrite findings of 6.4% by Uneke et al. 20 Normal urine sample does not contain nitrites. They occur when certain bacteria breakdown urine nitrates into nitrites. Examples of such bacteria include escherichia coli, enterobacter species, klebsiella species and proteus species. Worthy of note is the fact that not all bacteria are capable of converting nitrate to nitrite. Thus a negative nitrite in urine does not rule out UTI.
We noted that 62.5% of the study participants with nitrites in their urine samples had abnormal urine appearance. This association is statistically significant (0.003). Abnormal urine appearance was also noted in participants with leucocytes in their urine samples (64.3%), and this association was found to be statistically significant (0.001). UTIs are a well-known cause of cloudy urine. Some other causes include kidney stones, sexually transmitted infections (STIs) and vulvovaginitis.
All subjects who had urine abnormalities were promptly informed and referred to Nnamdi Azikiwe University Teaching Hospital, Nnewi for further investigations and treatment.
Limitations of the study
Due to limited resources,
We were unable to conduct a pilot study prior to onset of data collection.
A second screening could not be carried out to rule out the presence of transient abnormalities.
A sample size increase would be ideal in future studies to increase the power of the results.
Future studies would include renal function tests to assist in making a definitive diagnosis.
Conclusion
Urine dipstick abnormalities are common in female medical students of Nnamdi Azikiwe University, Nnewi Campus. All students should have routine screening for the early detection and treatment of renal diseases. Repeat screening is beneficial to rule out transient urine abnormalities. A widespread dipstick urinalysis screening programme should be implemented to determine the exact prevalence of dipstick urinary abnormalities among female medical students in Nigeria.
Recommendation
The cheap and inexpensive urine dipstick urinalysis is a veritable test for early detection of renal pathology. This simple non-invasive procedure can save chronic debilitating renal diseases if done routinely. The changes noted early enough if routinely done will act as a pointer for more detailed laboratory investigations to be done on the patient including blood chemistry. It is therefore recommended for all organizations to include free routine dipstick urinalysis for its students both as admission screening and routine screening for health maintenance.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221135575 – Supplemental material for Dipstick urinalysis profile of an asymptomatic female group in south-east Nigeria
Supplemental material, sj-docx-1-smo-10.1177_20503121221135575 for Dipstick urinalysis profile of an asymptomatic female group in south-east Nigeria by Ijeoma Cornelia Mmoh, Chukwuanugo Nkemakonam Ogbuagu, Ifeoma Ann Modebe, Ekenechukwu Nkolika Ogbuagu, Chimdindu Michael Ogbuagu, Obiageli Fidelia Emelumadu, Uzoma Chidi Okereke, George Uchenna Eleje and Obinna Ikechukwu Ekwunife in SAGE Open Medicine
Footnotes
Acknowledgements
The authors thank the study participants, as well as Iloh Jesse and Okechukwu Chioma for their help during this research, especially during data collection.
Author contributions
I.C.M, C.N. O, and I.A.M. contributed to the concept design. I.C. M conducted the data collection & experiment, statistical analysis and wrote the manuscript with supervision from C.N.O, I.A.M and O.F. E, E.N.O, C.M.O, and U.C.O contributed to the literature review. G.U.E and O.I.E contributed to the study analyses and grammatical revision of the manuscript. All authors reviewed and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical clearance was obtained from Nnamdi Azikiwe University Teaching Hospital Ethical Board before commencement of the study. Written informed consent was also obtained from the participants and anonymity maintained before, during and after the study. Confidentiality and data safety was maintained in the course of the study.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects prior to the onset of the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

