Abstract
Objective:
The level of immunity against pathogens decreases with old age. As a result, the elderly may be regarded to be at increased risk of malaria morbidity and fatality. There is paucity of studies on malaria among the elderly population in Osun East district, Southwest Nigeria. This study aimed to determine the prevalence of malaria and its association with medical comorbidities among the elderly.
Methods:
A descriptive cross-sectional study was carried out, which involved 972 adult residents of five communities in Osun State, who were selected using a multistage random sampling technique. Data was collected with aid of a structured questionnaire. The medical history of respondents and anthropometric measures were obtained. The presence of malaria parasitaemia in the respondents was determined by rapid diagnostic test (RDT). Appropriate descriptive and inferential analyses were done.
Results:
Out of the 972 respondents, 504 (51.9%) were 60 years and above. The overall prevalence of malaria RDT positivity was 4%. The positivity rate was higher among the elderly (4.6%) compared to those less than 60 years (3.4%), albeit not statistically significant (
Conclusion:
The malaria positivity rate was higher among the elderly in the study area, although not statistically significant. The prevalence was not associated with comorbid medical conditions.
Introduction
Malaria has a huge disease burden globally with about 40% of world population at risk, though it occurs almost exclusively in the tropics and subtropics.1,2 It is endemic and continues to be a major cause of morbidity and mortality in sub-Saharan Africa and Nigeria.1–5 An estimated 241 million malaria cases and 627,000 malaria deaths occurred worldwide in 2020 alone. 1 More than 90% of these malaria deaths occurred in sub-Saharan Africa, 40% of which took place in just two countries: Nigeria and Democratic Republic of Congo.2,4
Several programmes have been designed over time towards the control of malaria. 5 The reduction in the incidence and mortality rates of malaria has been uneven globally in spite of several interventions targeted at its control. In Nigeria, the gains of these control efforts have been demonstrably insignificant, reversed or stalled.1–6 Case management and vector control through the use of long-lasting insecticide-treated nets (LLIN) and indoor residual spraying (IRS) remain important components of malaria control strategies in Nigeria.7–10
Malaria is known to be associated with high morbidity and mortality, predominantly among children and pregnant women.11–13 The elderly population were not considered to be at risk of significant morbidity and mortality due to partial immunity conferred by persistent exposure to malaria over the years.14–16 However, there is also a possibility that old age and the presence of non-communicable diseases (NCDs) lead to the attenuation of this immunity.17,18 These may put elderly persons at a significantly higher risk of malaria and its unacceptable sequelae.15,19,20
Moreover, the burden of NCDs is increasing among older adults in many economically developing countries, including Nigeria.20–22 Hypertension, diabetes and obesity are the most prevalent risk factors for cardiovascular and renal diseases globally.23–26 These NCDs are known to have a potentially negative impact on the body’s immunity against infections.
The potential relationship between malaria and NCDs could however be bidirectional, that is, the chronic effects of repeated exposure to malaria could also predispose individuals to increased risk of NCDs.27,28 These effects and the mediating mechanisms include endothelial injury and dysfunction, release of proinflammatory markers and activation of the Renin-Angiotensin-Aldosterone System (RAAS) leading to increased levels of angiotensin II.27–29 The recurrent and persistent release of inflammatory substances may lead to long-term alteration in the distensibility and compliance of blood vessels. The decrease in the levels of nitric oxide resulting from endothelial dysfunction may also contribute to this process, which may ultimately lead to the development of NCDs such as hypertension.27,28 In the same vein, the activation of the RAAS produces an increase in Angiotensin II which causes vasoconstriction and retention of sodium, thus increasing the risk of high blood pressure (BP) and attendant chronic cardiovascular complications.27,29 These possibilities thus underscore the need for continuous evaluation of the potential nexus between malaria infection and NCDs.
There is however a paucity of studies in Nigeria with regards to the epidemiology of malaria and its association with NCDs among elderly persons. In a community-based study, the prevalence of malaria parasitaemia among people older than 60 years was found to be 16.2%, 14 while a facility-based study by Chukwuocha et al. 15 among patients older than 50 years in Southeast Nigeria showed a very high prevalence of 72.8% and the prevalence was high among persons with NCD comorbidities. In another study by Ibrahim et al. among adult febrile patients, there was also a significant association between the prevalence of malaria infection and certain NCDs such as hypertension and diabetes. 30 It is important to continually evaluate the dynamics of malaria among older persons, particularly in light of the aforementioned considerations, as it is the most common infectious disease in Nigeria. This study compared the malaria prevalence among elderly persons (⩾60 years) with those who were less than 60 years. It also assessed the association between malaria infection and medical comorbidities among the elderlies.
Methods
Study design and area
This cross-sectional community-based survey was carried out in five selected communities of Osun East senatorial district in Osun State, Southwest Nigeria, between November 2021 and January 2022. The communities were selected from five local government areas (LGAs) in the district (Figure 1). Osun State, with an estimated population of 5.5 million people, is one of the 36 states in Nigeria. It is subdivided into 3 senatorial districts, each of which comprise 10 LGAs, making a total of 30 LGAs in the state.

Map of Osun State – Nigeria showing the study areas.
Study population and selection criteria
All adult respondents aged 18 years and above who attended a community health outreach programme in each of the selected towns were included in the survey. Those who did not give consent or were too ill to participate in the study were excluded.
Sample size and technique
The minimum sample size of 280 was calculated using Fischer’s formula31,32 with the following assumptions: 95% confidence level, nationwide prevalence of malaria among geriatric population = 24%, desired precision (
Using multistage sampling technique, the Osun East senatorial district was purposively selected for the study among the three districts in Osun State. Out of the 10 LGAs in the district, 5 LGAs (Ife East, Ife central, Ife South, Oriade and Ilesha East) were randomly selected. One community was selected from each of the LGAs as follows: Ogbon Oya (Ife East), Enuwa-Ife (Ife central), Garage Olode (Ife South), Igando-Ilesha (Ilesha East) and Iloko Ijesha (Oriade LG). In each of the aforementioned localities, a health outreach programme was done. It was advertised in the media, made known at religious gatherings (churches and mosques) and discussed at community association meetings. All those who attended the outreach sessions for adults were invited to participate in the study.
The study was approved by the Ethics and Research Committee of Obafemi Awolowo University Teaching Hospitals Complex, Ile-Ife (protocol number: ERC/2022/03/04). A written consent was obtained from each respondent, and confidentiality was maintained.
Data collection
A structured questionnaire adapted from a previous study by Ibrahim et al. 30 was used to obtain the socio-demographic variables and past medical history of the respondents. The self-reported medical history was based on previous diagnosis by qualified medical professionals. The anthropometric measures, BP, rapid diagnostic test (RDT) for malaria parasite and random blood glucose were measured.
The respondent’s BP was taken with the Accosson® Mercury sphygmomanometer. It was ascertained that they had not taken coffee, alcohol or cigarettes nor engaged in physical exercises for at least half-an-hour prior to the assessment. After applying the cuff, the stethoscope’s bell was positioned a little above the antecubital fossa. The systolic BP (SBP) was assessed using the palpation method and the cuff was thereafter inflated to about 20 mmHg beyond the SBP. The cuff was then gently deflated, and the first sound (Korotkoff phase 1) and its disappearance (phase 5) were noted and recorded. A respondent was characterized as having hypertension if he/she was a known hypertensive or had any of the following measures: SBP ⩾ 140 mmHg or diastolic BP ⩾ 90 mmHg. 33
The weight (measured to the nearest 0.1 kg) and height were taken in the absence of any body accessories such as shoes, purses and cell phones, and the measuring instruments were regularly adjusted to zero. In order to calculate the body mass index (BMI), the weight was divided by the square of height in metres. To measure the random blood glucose, a drop of capillary blood sample was obtained by pin prick and dropped on the test strip of a glucometer (Accu-Check Active,® Roche Diabetes Care GmbH, Mannheim, Germany), pre-calibrated and standardized by the manufacturer, Roche Diagnostics GmBH, Germany. The SD Bioline Malaria Ag Pf® RDT kits (Standard Diagnostics, Kyonggi-do, South Korea)were used to test the blood for
Statistical analysis
Data was analysed using SPSS Version 17. Simple descriptive and inferential statistics were performed, and the results were presented in tables. The association between categorical variables was determined with the use of the Pearson Chi-square test. The level of significance was set at
Results
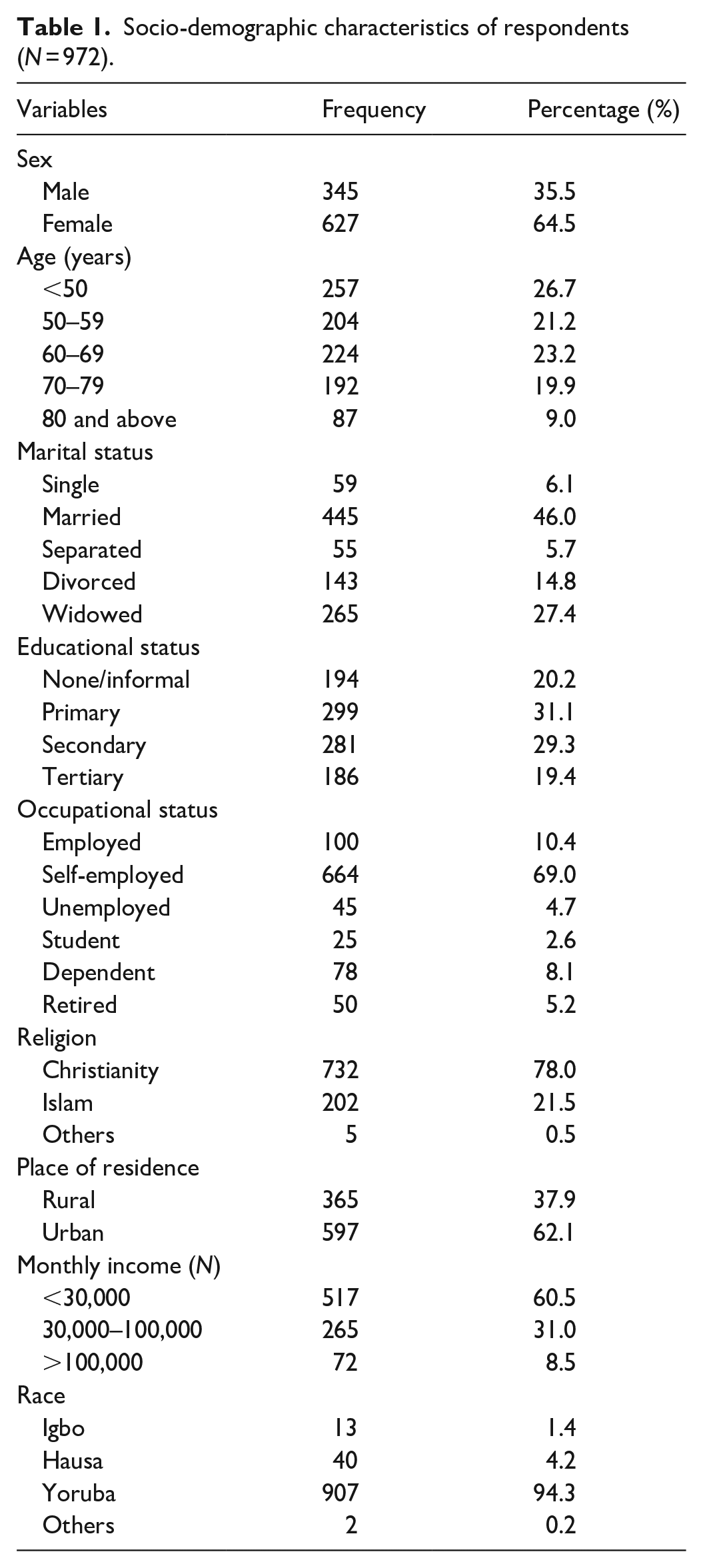
A total of 972 respondents were recruited out of which 504 (51.9%) were 60 years and above. There were 627 (64.5%) females and 345 (35.5%) males. They were grouped based on their educational status, and 281 (21.3%) attained secondary school education while 196 (19.4%) had a tertiary level of education; a majority of respondents, 445 (46.0%), were married. Table 1 shows other socio-demographic features.
Socio-demographic characteristics of respondents (
The overall prevalence of malaria parasitaemia was 4.0%. However, as shown in Table 2, the prevalence of malaria RDT positivity among the elderly was higher (4.6%) compared to that of those less than 60 years (3.4%), albeit not statistically significant (
Association between rapid diagnostic test positivity and age (

Table 3 shows that 52.6% and 16.1% of elderly population used insecticide-treated nets and insecticide sprays, respectively. Over two-thirds (62%) of the elderly population had window nets installed in their homes. Malaria positivity rate was not significantly associated with the use of insecticide-treated nets (
Association between malaria RDT positivity and preventive measures against malaria among elderly respondents (

RDT, rapid diagnostic test.
Association between use of preventive measures and respondents’ residence and education (

As shown in Table 5, there was no association between prevalence of malaria positivity and comorbid conditions such as overweight/obesity (
Relationship of malaria with comorbidities among elderly respondents (

BMI = body mass index, RDT = rapid diagnostic test.
Fischer’s exact test.
Discussion
The geriatric population in malaria endemic areas is not considered to be seriously vulnerable to the disease.14–16 The greater bulk of malaria management and control interventions is focused on pregnant women and children.5–8 Higher prevalence of malaria RDT positivity in the elderly (4.6%) in this study, though not statistically significantly different from others (3.4%), suggests the need to refocus local malaria case management and control intervention strategies to accommodate elderly persons.3,14,15 This prevalence was lower to that obtained in other studies in Southwest and Southeast Nigeria probably due to variations in study population structure and timing of survey.14,15
It is expected that having acquired significant immunity due to repeated exposure to malaria infection over the course of their life, people of the older age group are more resistant to the infection. It is however also possible that there may be a reduction in the capacity of the ageing immune system to mount a robust defence against the infection.11,34 Elderly patients are more susceptible to having complications from various infectious diseases. This tends to be a result of multimorbidity and a diminished capacity to establish sufficient immune response.11,35
The findings from various studies have shown that as people age, their cellular response decreases as a result of a transition from a Th1 to a Th2 response. 35 Ageing has also been shown to have impact on a number of human haematology parameters and peripheral blood lymphocyte.35,36 This state of diminishing age-specific immunity to malaria has however not been conclusively proven in older individuals in highly endemic areas, who are regularly exposed to plasmodia.11,34,37 This is indeed an area for further research, considering that the primary mechanisms of the host response and immunity to plasmodial infection are not fully understood.
Despite the fact that comorbidities have a potentially deleterious impact on host immunity and thus increase in predisposition to infectious disease, the present study did not however find an association between malaria parasitaemia and the common comorbidities among the respondents. It is possible that, unlike immunity to other infectious diseases, malaria immunity – the mechanisms of which are not yet fully elucidated – is not significantly impacted by comorbidity among people living in malarial endemic zones such as Nigeria. This finding is however contrary to the reports by Chukwuocha et al. 15 and Ibrahim et al. 30 who, respectively, demonstrated statistical association between malaria infection and certain NCDs. Also, studies in non-endemic regions among patients, who returned from endemic areas, showed significant association between NCDs and the risk of malaria infection as well as its severity.37,38
The pathophysiological nexus between malaria and NCDs invariably involve many different mechanisms and factors. These factors may vary among individuals. For example, the oxidative stress that results from malaria infection has been proposed as one of the mechanisms that potentially increase the risk of NCDs.27,28 This hypothesis therefore suggests that the routine use of antioxidants or intake of foods rich in antioxidants, especially during malaria infection, will attenuate this effect. 28 This history of intake of antioxidants was not obtained from the respondents in this study and may be an important confounder that might explain the lack of association between malaria parasitaemia and NCDs. Furthermore, most of the proposed mechanisms are related to the long-term cumulative effect of repeated malaria infection.27,28 Hence, with regards to the malaria–NCD connection, the real issue is repeated malaria infection over time and not necessarily the singular impact of the malaria parasitaemia that was ongoing at the time of the study. In view of the cross-sectional nature of this study, the association that was tested was between the prevalence of parasitaemia at the time of the study and that of NCDs. Other factors such as the differences in study population and methods might account for the association reported by other authors. The studies by Chukwuocha et al. 15 and Ibrahim et al., 30 for example, were facility-based studies among symptomatic patients who presented for care and are more likely to have more severe forms of malaria than the respondents in our study, which was community based.
The study noted usage rates of LLIN/IRS that were less than desirable, suggesting a need to scale up programmes and interventions targeting improved effective use of such.10,39,40 On the other hand, the lack of association between malaria RDT positivity rate and the usage of LLIN/IRS suggests a need for review of the effectiveness of the strategy. 41 This may be due to the fact that the effectiveness of vector control strategies such as LLIN depends on certain vector parameters which may be different across regions or may change over time within the same region. These parameters include biting seasonality, resting behaviour, feeding behaviour (e.g. whether the vectors are endophagic with preference for biting indoors or exophagic who bites outdoors), peak biting time and insecticide resistance, among others. Entomological surveillance studies have shown that these vector behaviours and bionomics do change with time.42–45 Routine surveillance and evaluation are thus required to monitor these changes, which will then inform corresponding modifications of the malaria control and elimination strategies. For example, because LLIN provide protection during the sleeping hours, their effectiveness is contingent upon a vector behaviour characterized by late-night indoor feeding. When there is change or difference in biting and feeding behaviour, the effectiveness of LLIN is attenuated.43–45 It is thus crucial that the control programmes and tools are continuously evaluated and realigned with the potential changes in vector behaviour.
Limitations of study
The results of this study must be cautiously interpreted within the ambits of certain limitations. The questionnaire which was developed for the study was not previously validated nor pilot-tested in the study area. It was however adapted from a previous work by Ibrahim et al.
30
The lack of validation or pilot-testing before its use in this study potentially limits the validity and reliability. The presence of malaria parasitaemia was determined with the use of the RDT kit instead of microscopy, which is the gold standard. The histidine-rich protein 2 (HRP2)-based RDT kit used could only detect the presence of
Conclusion
The malaria RDT positivity rate was higher among those older than 60 years compared to other adults in the study area. Although the observed difference was not statistically significant, it may indicate the need to view older persons as being more vulnerable to malaria infection.
With regards to the clinical implication, there is a need to pay more attention to the opportunistic diagnosis and treatment of malaria among elderly persons, even when they are asymptomatic. This requires the inclusion of malaria test as part of routine medical screening. Attending physicians should also have a high index of suspicion for malaria when older persons present with non-specific complaints of ill health.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231164259 – Supplemental material for Malaria among the elderly in five communities of Osun East district, Southwest Nigeria: Prevalence and association with non-communicable diseases
Supplemental material, sj-docx-1-smo-10.1177_20503121231164259 for Malaria among the elderly in five communities of Osun East district, Southwest Nigeria: Prevalence and association with non-communicable diseases by Ibrahim Sebutu Bello, Temitope Oluwafemi Olajubu, Olusegun Olumuyiwa Osundiya, Oluwasina Tajudeen Salami, Azeez Oyemomi Ibrahim and Abdulakeem Ahmed in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to thank the leaders and residents of the selected communities where the surveys took place. Also, the staff of the Family Medicine department and management of the Obafemi Awolowo University Teaching Hospital Ile-Ife, who facilitated the outreach, are greatly appreciated.
Authors’ contribution
Availability of data and materials
The datasets for this study would be made available by the correspondence author on a reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Ethics and Research Committee of the Obafemi Awolowo University Teaching Hospital, Ile-Ife (protocol number: ERC/2022/03/04). The respondents gave written consents. The reporting of this study was guided by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Informed consent
Written informed consent was obtained from all subjects before the study. Yes
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
