Abstract
Active detection of asymptomatic malaria cases and resolution of associated factors are essential for malaria elimination. There are no nationwide estimates for asymptomatic malaria and associated factors in Ethiopia. Therefore, this study aims to generate comprehensive and conclusive evidence from various studies conducted in Ethiopia. Published articles from various electronic databases such as PubMed, Google Scholar, CINAHL, Scopes, Cochrane Library, the Web of Science, and African Journals Online were accessed. Also, unpublished studies from Addis Ababa digital library were identified. All observational study designs were included in the search. Data were extracted on the Microsoft Excel spreadsheet and analyzed using STATA version 14.1. A random-effects model was fitted to estimate the pooled prevalence of asymptomatic malaria. A meta-regression and subgroup analysis was computed to see heterogeneity. The publication bias was assessed by the funnel plots and Egger’s statistical tests. The analysis found that the pooled burden of asymptomatic malaria was 6.7 (95% confidence interval = 4.60, 8.79). The pooled prevalence of Plasmodium falciparum was 3.75 (95% confidence interval = 2.25, 5.18), and that of Plasmodium vivax was 2.22 (95% confidence interval = 1.46, 2.99). Factors such indoor residual spray service (odds ratio = 0.46; 95% confidence interval = 0.26, 0.81), never used insecticide-treated nets (odds ratio = 6.36; 95% confidence interval = 4.01, 10.09), and presence of stagnant water in the vicinity (odds ratio = 3.24; 95% confidence interval = 1.20, 8.71) were found to have a significant association with asymptomatic malaria. This study highlighted that pooled prevalence of asymptomatic malaria is high and varied by population groups. Prevalence of asymptomatic malaria was increased among those who never used insecticide-treated nets and were living near stagnant water by six and three times, respectively. The use of more sensitive diagnostic methods could yield a higher burden of the disease. Furthermore, active case detection is recommended for effective intervention toward elimination.
Introduction
Malaria is a mosquito-borne, life-threatening disease for which there are both preventive and therapeutic measures. Malaria continues to cause unacceptably high levels of disease and mortality decades after the first malaria eradication effort in 1955. The top objective for the international community is to reduce morbidity and mortality while maintaining the long-term goal of eradicating malaria. Although the second global campaign, Roll Back Malaria, was a massive success in reducing malaria morbidity and mortality, the illness continues to be a serious public health issue in resource-poor nations.1,2
In 87 malaria-endemic countries, there were estimated 229 million malaria cases and 409,000 malaria fatalities in 2019. With estimated 215 million malaria infections and 384,000 malaria deaths in 2019, the WHO Africa region accounts for roughly 94% of the worldwide malaria burden. The WHO African region is home to 27 of the 29 nations that account for 95% of all malaria cases worldwide. The global change in malaria cases has not been consistent over the last two decades, and malaria incidence has been dropping at a slower rate in recent years, by only 2% between 2015 and 2019, compared to 27% between 2000 and 2015. In Africa, there were fewer malaria cases in 2019 (204 million) than in 2000, while the malaria case incidence fell from 363 to 225 cases per 1000 people at risk throughout this time, according to data from both the global and African regions. 3
Large-scale interventions that target the vector (i.e. insecticide-treated nets (ITNs) and indoor residual spraying (IRS)) and that target the parasite (i.e. Artemisinin-based Combination Therapy (ACT) have made tremendous success in reducing malaria incidence and mortality. Because of this success in lowering malaria incidence and mortality, several countries now have low or moderate malaria transmission. Consequently, the desire to consider malaria elimination as the next malaria control strategy is gaining attention. As a result, the World Health Organization (WHO)4,5 has developed a strategic framework for malaria elimination from 2016 to 2030 to reduce malaria morbidity and mortality by 90% and eliminate malaria in 35 countries by 2030.
However, asymptomatic malaria poses a growing threat to this goal in all malaria-endemic nations. Information on asymptomatic malaria is critical to improve malaria prevalence estimates and to enhance malaria elimination efforts. Many studies have found that the prevalence of asymptomatic malaria is greater than that of symptomatic malaria, particularly when considering the asymptomatic subpatent infection. Asymptomatic malaria parasitemia, the presence of asexual parasites in the blood without symptoms of illness, may result from naturally acquired immunity in endemic areas due to genetic factors and malaria infection resulting in low parasite density.6–12 The lower gametocyte density that may result from asymptomatic malaria infection can still result in malaria transmission.8,13 Therefore, to achieve malaria elimination, it is critical to understand the epidemiology of malaria infection and particularly interrupt the ongoing malaria transmission from the asymptomatic reservoir, which is even more effective than mass drug administration in low transmission settings.13,14
The study conducted so far indicated that the burden of asymptomatic malaria varies by population group, transmission setting, and study setup or diagnosis method.10,15–18 Because malaria risk mapping is shrinking and malaria incidence and mortality are reducing dramatically, Ethiopia has planned to eliminate malaria nationwide by 2030.19,20 It is critical to understand the local malaria situation, including the amount of asymptomatic malaria and associated risk factors, in order to meet this goal. Although a few small-scale studies have evaluated the prevalence of asymptomatic malaria and associated variables, no national-level estimate exists. Therefore, this systematic review and meta-analysis were aimed to estimate the pooled prevalence of asymptomatic malaria and associated factors among different population groups in Ethiopia.
Determining the pooled prevalence of asymptomatic malaria infection and associated factors will improve the precision of estimates and examine the source of heterogeneity if there is any. Furthermore, it also allows us to gain insight into different aspects of asymptomatic malaria, which we cannot get from individual studies. This is particularly important to understand the burden of asymptomatic malaria in different parts of the country and specific risk factors to be considered for targeted intervention. The findings of this study can also be used better to understand the status of asymptomatic malaria in Ethiopia and to design national-level initiatives that take into account the local asymptomatic malaria burden. In addition, this research will identify gaps in the present literature to address the issues of asymptomatic malaria in the context of the country’s malaria elimination target.
Method
Search strategy
This systematic review and meta-analysis were conducted to assess the prevalence of asymptomatic malaria and associated factors in Ethiopia. The existence of similar systematic reviews and meta-analyses published on this topic was checked to prevent repetitions. Both published and unpublished studies were searched thoroughly using electronic databases such as Medline, HINARI, PubMed, Cochrane Library, the Web of Science, and Google Scholar using the key terms “prevalence, magnitude, proportion, malaria, asymptomatic malaria,
Selection and eligibility criteria
Inclusion criteria
In this systematic review and meta-analysis, we included all studies conducted on the prevalence of asymptomatic malaria in Ethiopia. All of the studies were original research articles published in English that included basic information on sample size, diagnostic procedures, malaria infection prevalence, and status among adults in various locations of Ethiopia. Furthermore, studies were conducted in health institutions and community-based settings. Studies on the general public, pregnant women, and children were considered in the review. We included cross-sectional studies published in the form of journal articles and available on an institutional repository in the English language. No restriction was made for the year of publication. Conference abstracts and non-human studies were excluded. We tried to contact the primary authors of the articles published only abstract, and we excluded articles that were not accessible after contacting the principal investigator two times via email.
Outcome measurement
This study has two main outcomes. The first outcome was to estimate the prevalence of asymptomatic malaria in Ethiopia. It was calculated using the metan prevalence standard error command after generating prevalence and standard error of prevalence. The asymptomatic malaria was confirmed by microscopy and/or rapid diagnostic test (RDT) in the primary studies. The factors associated with asymptomatic malaria were the study’s second outcome. Data for this outcome were extracted in a format of two by two tables on the Microsoft Excel spreadsheet. Then, the log odds ratio (OR) for each factor was calculated based on the findings of the original studies. Factors included in this study were residence (rural/urban), sex (male/female), education (literate/illiterate), IRS (yes/no), ITN (ever used/never used), and presence of stagnant water (yes/no).
Quality assessment and data extraction
The Newcastle–Ottawa Scale for cross-sectional studies quality assessment tool was adapted and used to assess the quality of the included study. 22 Two data extractors extracted data (A.T. and T.T.) using a standardized data extraction checklist on Microsoft Excel. Reference management software (Endnote version X7.2) was used to combine search results from databases and initially remove duplicate articles. Then, studies were screened and excluded by their titles and abstracts. Full text of the remaining articles was evaluated for eligibility based on the pre-arranged inclusion and exclusion criteria. For the first outcome (prevalence of asymptomatic malaria), the data extraction checklist includes the author name, year of publication, region (an area where the study was conducted), study design, method of outcome measurement, sample size, response rate, and the number of cases with asymptomatic malaria. For the second outcome (factors associated with asymptomatic malaria), data were extracted in a format of two by two tables. Then, the log OR was computed based on the findings of the primary studies. Discrepancies between two independent reviewers were resolved by involving another reviewer (B.R.) after discussion for possible consensus.
Data analysis and synthesis
The necessary information was extracted from each original study using a format prepared in a Microsoft Excel spreadsheet. Then, the data were exported to STATA Windows version 14 for analysis. The prevalence and standard error of prevalence were generated for each study using the “generate” command on STATA for the first outcome variable. Moreover, the logarithm and standard error of the OR for each included study were generated for the second outcome. The pooled magnitude of asymptomatic malaria and associated factors was presented in the form of a forest plot. The heterogeneity among the included studies was checked by Cochran’s
Results
Study selection
Eleven thousand three hundred sixty-five published and four unpublished articles were identified from several electronic databases and Addis Ababa digital library, respectively. Of the identified studies, 11,330 because of the irrelevant title, 15 due to duplication, and 2 articles with no manuscript were excluded. On further screening of the abstract, two articles were excluded for they did not have necessary data in the form of two by two tables. The full text of the remaining 16 studies was assessed and screened for eligibility. Accordingly, two studies were excluded because they did not study factors associated with asymptomatic malaria. Finally, 14 articles that fulfilled the eligibility criteria were included in the final analysis. We used the PRISMA flow diagram to present the study selection process (Figure 1).

PRISMA flow diagram: prevalence of asymptomatic malaria and associated factors in Ethiopia, systematic review and meta-analysis, 2021.
Features of included studies
From the total of 14 studies included in this review, 13 were published articles, while one was preprint from the research square and all used a cross-sectional study design. Three studies were conducted on children, five were conducted on pregnant women, and six were conducted on the general public. The total number of people who took part in the investigations was 5924, with the smallest sample size of 230 in Southern Nations, Nationalities, and Peoples’ Region (SNNPR) 24 and the largest sample size of 1094 in Oromia. 25 The age group used for analysis was not consistent across the studies. Of the 14 studies included in the final analysis, 7 were conducted in Amhara Region,26–32 4 in SNNPR,24,33–35 2 in Oromia,25,36 and 1 in Benishangul-Gumuz. 37 Fifty percent of the studies were community-based.27–29,32,33,35,36 The outcome measure was only RDT for two studies,27,37 only microscope for five studies,24,28,29,31,35 and both microscopy and RDT for seven studies.25,26,30,32–34,36 The studies done by microscopy have identified the species, while two of the RDT result did not (Table 1).
Summary of included studies regarding the prevalence of asymptomatic malaria and associated factors in Ethiopia, 2021.

Description of study sites and study period
Areas included in the studies were considered due to their malaria distribution. Accordingly, five studies were conducted during the major malaria transmission season (September to November).25–28,37 There were three studies in the minor malaria transmission season (April to May).33–35 The rest six studies did not follow these transmission patterns.24,29–32,36 Except for one 26 where malaria is endemic, malaria is seasonal and unstable in the rest areas.
Prevalence of asymptomatic malaria
In this meta-analysis, we found significant heterogeneity across studies (

Forest plot of prevalence of asymptomatic malaria by microscopy in Ethiopia, systematic review and meta-analysis, 2021.

Forest plot of prevalence of asymptomatic malaria by RDT in Ethiopia, systematic review and meta-analysis, 2021.

Forest plot of prevalence of asymptomatic malaria due to

Forest plot of prevalence of asymptomatic malaria due to
Meta-regression was computed to see underlying sources of heterogeneity using sample size and year of publication, but none of them showed a statistically significant presence of heterogeneity (Table 2).
Meta-regression using sample size and year of publication to observe related heterogeneity on the prevalence of symptomatic malaria in Ethiopia, 2021.

Moreover, to minimize potential heterogeneity, subgroup analysis was conducted based on the type of population and setting where the studies were conducted. The subgroup analysis indicated the highest prevalence of asymptomatic malaria in the study done in the general population 8.57 (95% CI = 4.67, 12.48) while the lowest prevalence was observed in a study conducted among pregnant women 6.13 (95% CI = 3.85, 8.41) (Figure 6). The subgroup analysis also revealed the presence of inconsistent prevalence between study settings, whereby the highest prevalence was observed in community-based study 8.59 (95% CI = 5.12, 12.06) (Figure 7).

Subgroup analysis of prevalence of asymptomatic malaria by population group in Ethiopia, systematic review and meta-analysis, 2021.
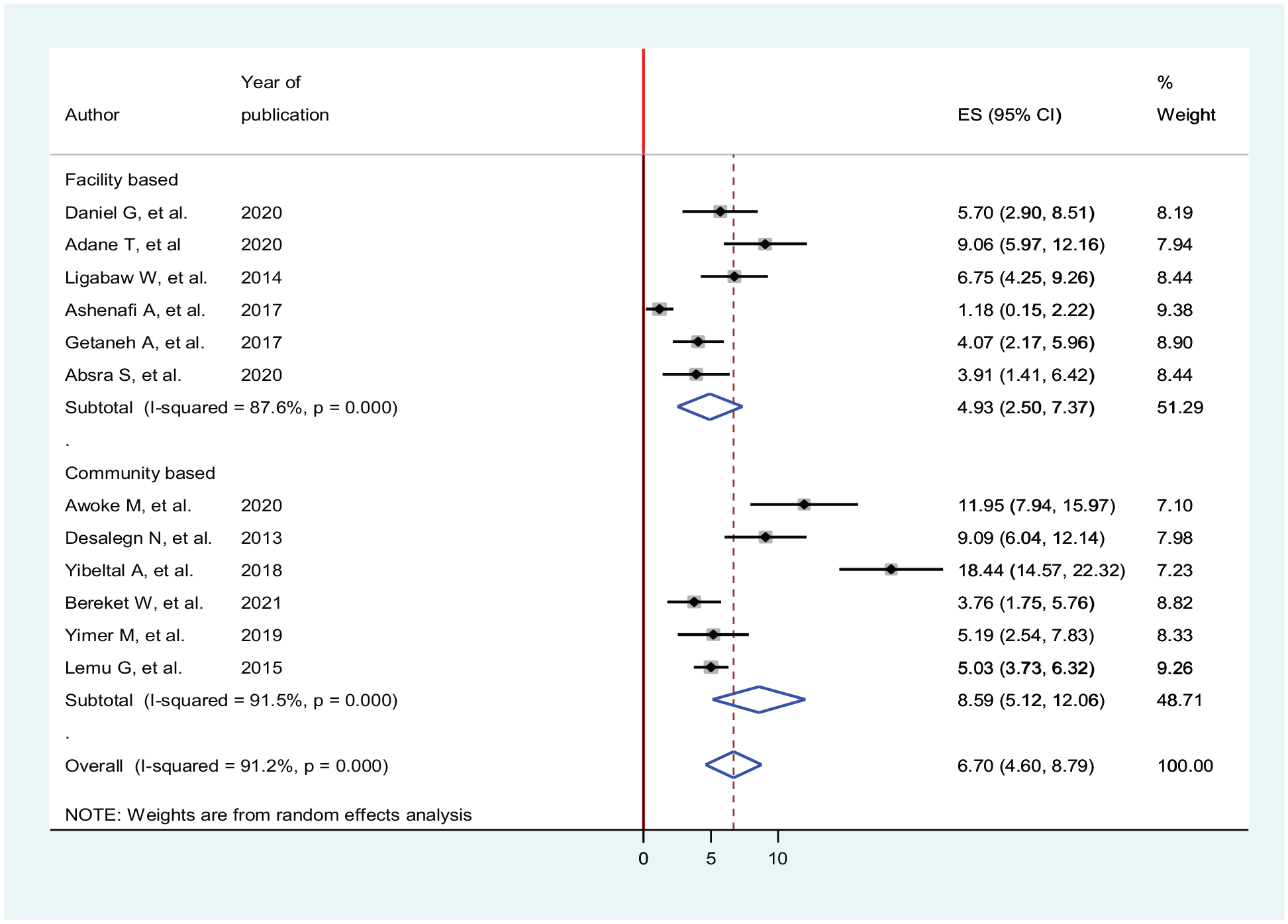
Subgroup analysis of prevalence of asymptomatic malaria by study setting in Ethiopia, systematic review and meta-analysis, 2021.
To see for the presence of publication bias, a graphical funnel plot and Egger’s test at a 5% significance level were computed (Figure 8). The asymmetric funnel plot indicates the presence of publication bias. In addition, Egger’s test showed there was a statistically significant presence of publication bias (
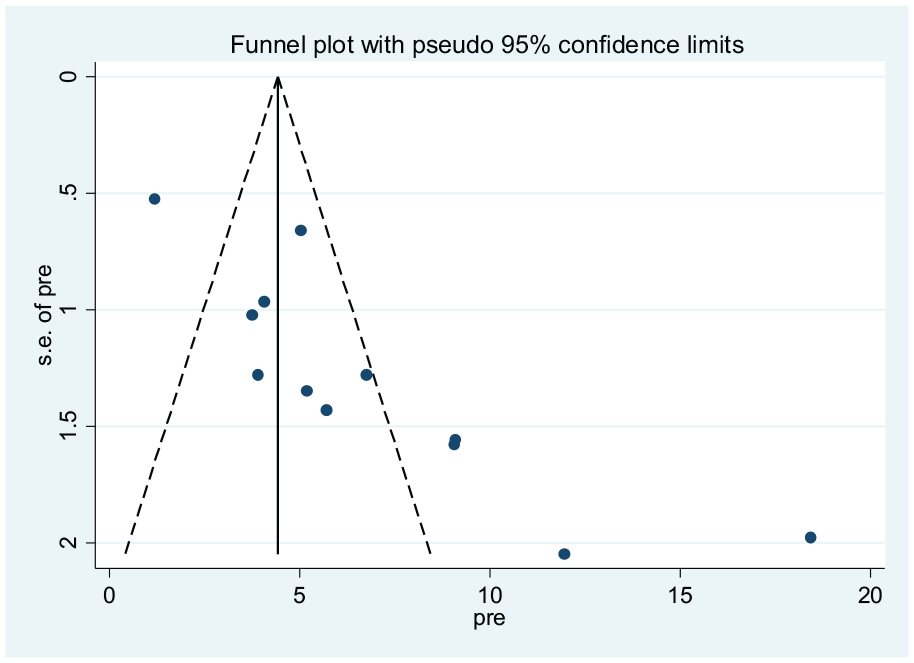
Funnel plot indicating presence of publication bias, prevalence of asymptomatic malaria by microscopy in Ethiopia, systematic review and meta-analysis, 2021.
To reduce and adjust for the observed publication bias in the studies, the trim and fill analysis was performed for estimation of the number of missing studies that might exist. Trim and fill analysis is a nonparametric method for estimating the number of missing studies that might exist and it helps in reducing and adjusting publication bias in meta-analysis. In trim and fill analysis, seven studies were imputed for missing studies and after adjustment for publication bias, the estimated pooled prevalence of asymptomatic malaria was 3.41 (95% CI = 1.25, 5.58) (Figure 9).
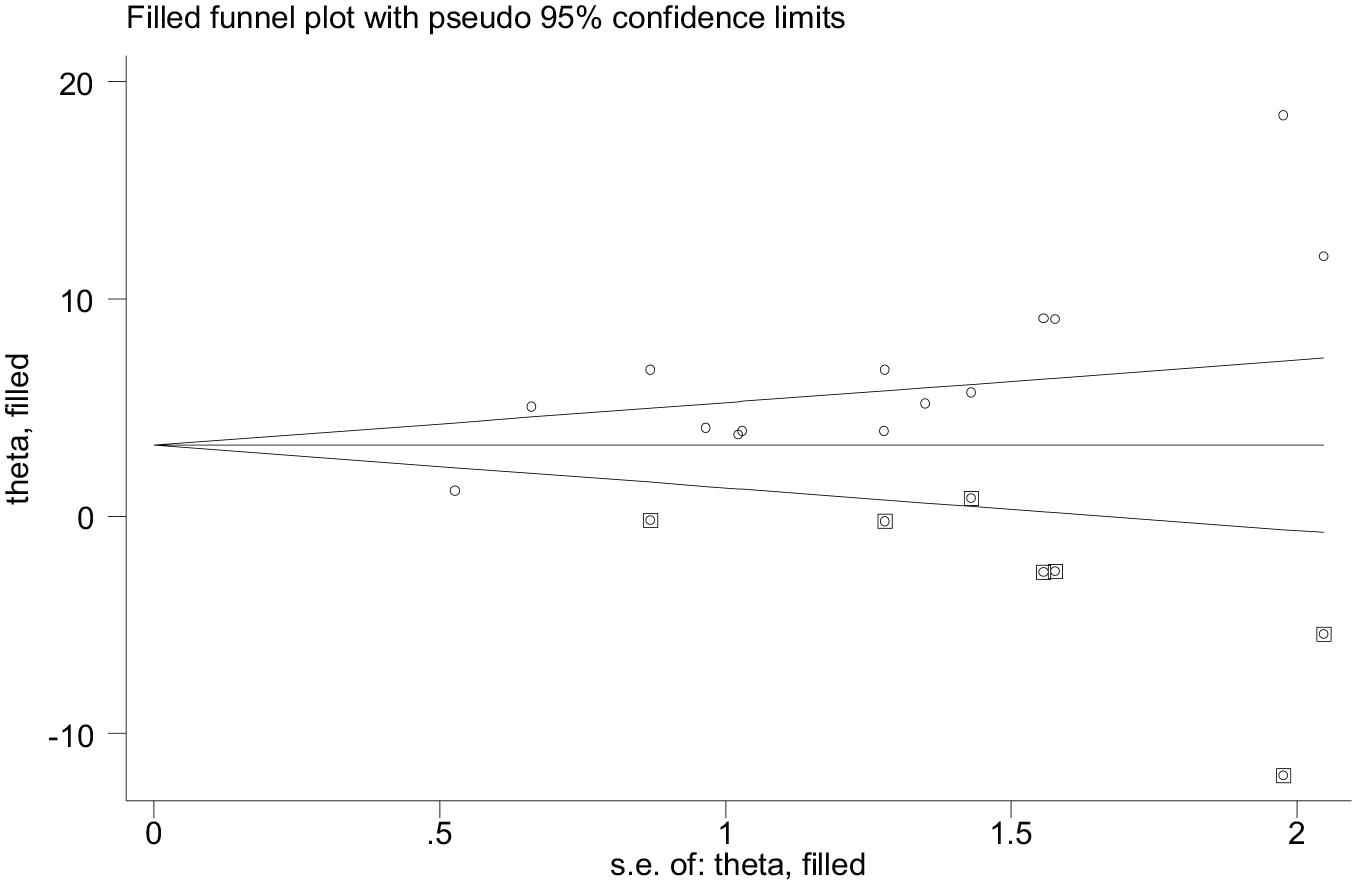
Filled funnel plot, prevalence of asymptomatic malaria by microscopy in Ethiopia, systematic review and meta-analysis, 2021.
Sensitivity analyses of the studies were done to test the effect of a single study on the pooled result of remaining studies using a random-effect model. We found no strong evidence for the influence of individual study on remaining studies (Figure 10).

Sensitivity analysis, prevalence of asymptomatic malaria by microscopy in Ethiopia, systematic review and meta-analysis, 2021.
Factors associated with asymptomatic malaria in Ethiopia
Sex and asymptomatic malaria
There were five studies27,29,31,34,35 identified to assess for an association between sex and asymptomatic malaria infection of which only one study
27
identified a significant association (OR = 5.63; 95% CI = 2.80, 11.31). The pooled result identified no significant association between sex and asymptomatic malaria infection (OR = 1.38; 95% CI = 0.56, 3.38). For this finding, random-effect model was used as heterogeneity was observed (

Association between asymptomatic malaria and sex in Ethiopia, systematic review and meta-analysis, 2021.
Education level and asymptomatic malaria
Seven studies were selected to assess the association between educational level and asymptomatic malaria infection.26–28,31–33,35 A random-effect model was used for this analysis as there is heterogeneity (

Association between asymptomatic malaria and educational level in Ethiopia, systematic review and meta-analysis, 2021.
IRS service and asymptomatic malaria
There were seven studies selected to assess the association between IRS service and asymptomatic malaria.26,27,29,32,33,36,37 Three studies27,29,37 showed no statistically significant association while four studies,26,32,33,36 indicated statistically significant association. The pooled analysis indicated a significant association between IRS service and asymptomatic malaria infection. Utilization of IRS decreases the odds of infecting with asymptomatic malaria by 54% as than their counterparts (OR = 0.46; 95% CI = 0.26, 0.81). For this finding, random-effect model was used as heterogeneity was observed (

Association between asymptomatic malaria and indoor residual spray in Ethiopia, systematic review and meta-analysis, 2021.
ITN utilization and asymptomatic malaria
Nine studies were selected to assess the association between ITN utilization and asymptomatic malaria infection.26–29,31,33,35–37 All selected studies revealed the presence of a statistically significant association between ITN utilization and asymptomatic malaria infection. The pooled finding also indicated that there is a statistically significant association between ITN utilization and asymptomatic malaria infection. The odds of infecting with asymptomatic malaria were 6.36 times higher among the population who had not used ITN as compared to those who had used ITN (OR = 6.36; 95% CI = 4.01, 10.09). For this finding, random-effect model was used as heterogeneity was observed (

Association between asymptomatic malaria and insecticide-treated net utilization in Ethiopia, systematic review and meta-analysis, 2021.
Presence of stagnant water and asymptomatic malaria
One of the four studies27,31,36,37 selected to assess the association between presence of stagnant water and asymptomatic malaria infection indicated there is no statistically significant association with OR = 0.49 (95% CI = 0.11, 2.14).
31
The pooled analysis indicated that there is a statistically significant association between the presence of stagnant water nearby the home and asymptomatic malaria. The odds of developing asymptomatic malaria infection were 3.24 times higher among the population residing near stagnant water than their counterparts (OR = 3.24; 95% CI = 1.20, 8.71). For this finding, random-effect model was used as heterogeneity was observed (
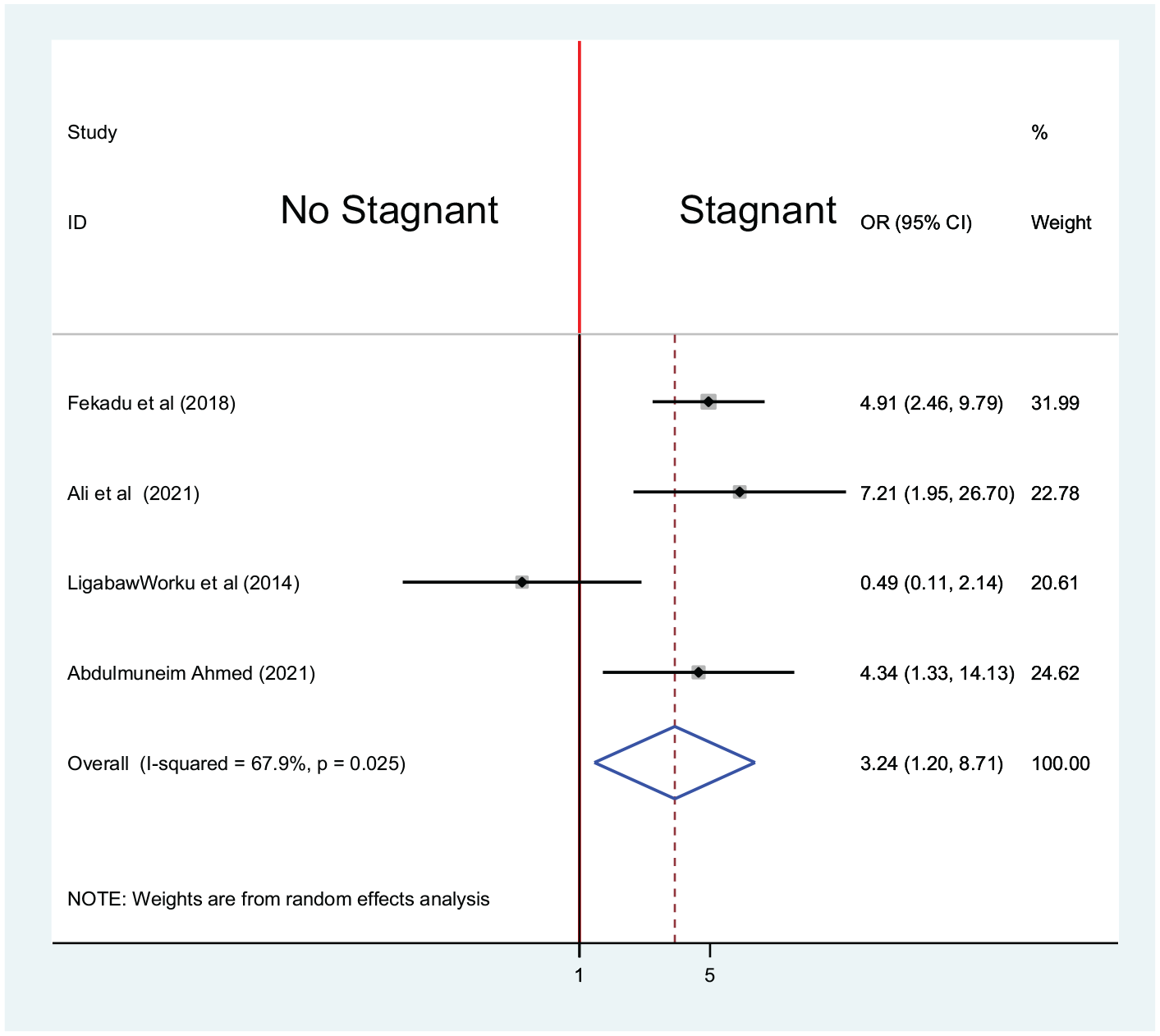
Association between asymptomatic malaria and presence of stagnant water in Ethiopia, systematic review and meta-analysis, 2021.
Place of residency and asymptomatic malaria infection
There were three studies selected to assess the association between place of residency and asymptomatic malaria infection.31,32,36 Only one study showed a statistically significant association.
32
The pooled result indicated there is no statistically significant association between place of residency (OR = 1.38; 95% CI = 0.52, 3.67). For this finding, random-effect model was used as heterogeneity was observed (

Association between asymptomatic malaria and residence place in Ethiopia, systematic review and meta-analysis, 2021.
Discussion
Despite the declining pattern of the prevalence of malaria in Ethiopia, the disease is still becoming one of the major public health problems. 38 The increasing trends of the asymptomatic nature of the disease are highly influencing and challenging to the elimination plan of malaria at the intended time as of the globe in general and in Ethiopia as well.4,15,19,39,40 This review and meta-analysis was the first of its kind targeting asymptomatic malaria in Ethiopia. There are a relatively small number of available and eligible studies for review, especially when compared to several eligible studies conducted in developed countries. Those available studies were vacillating to uncover the complete prevalence and the associated factors. Thus, this study was mainly intended to find out the pooled prevalence of asymptomatic malaria and its associated factors in Ethiopia. Asymptomatic malaria infection plays a key role in the transmission of malaria, and the intervention to target this parasite reservoir may be needed to achieve malaria elimination in both low- and high-transmission areas. 8
In this systematic review and meta-analysis, the pooled prevalence of asymptomatic malaria as examined by microscopy and RDT was found to be 6.29% (95% CI = 4.31, 8.27) and 5.07% (95% CI = 3.50, 6.65), respectively. This is about 13 times for microscopy and 4 times for RDT more than that of the 2015 malaria indicator survey. 41 The subgroup analysis of this study revealed that the prevalence in the general population, pregnant women, and children was 8.57%, 6.13%, and 6.70%, respectively. The findings, when compared to the prevalence of asymptomatic malaria among pregnant women as a global which was 10.8%, 10 were relatively lower. Another finding from Ethiopia was also reported as the pooled prevalence of asymptomatic malaria 11.99%. 42 The difference could be because of the variation in the study size, diagnostic approach, and the difference in endemicity class. The study was conducted in a malarious endemic area where the residents experienced hyperendemic malaria transmission. Again, it was conducted during a major malaria transmission period in Ethiopia. 28 Other studies used RDT as a diagnostic method that could increase the prevalence. 27 For instance, the global prevalence reported included all studies from all over the globe, and the greatest share among the reported from subgroup analysis prevalence was covered by Nigeria, one of the malarious countries in Africa and as the world in general. 10
The pooled prevalence of the present findings was also lower than the findings from pocket studies of different African countries (Kenya and Burkina Faso) and Ethiopia.27,28 However, the difference could be due to the population group whereby the study in Kenya and Burkina Faso were conducted among under-five children and testing method used which was RDT in case of the studies conducted in Ethiopia. The unprecedented investment in antimalarial preventive (LLINs/IRS) interventions and improvement in diagnostic capacity has led to marked reduction of malaria morbidity. Moreover, the evidence is supported by the overall health sector development, particularly the health extension program that strengthened the primary health care unit.15,19 Above all, the Ethiopian government’s determination to implement a malaria elimination plan could contribute to a dramatic reduction in malaria prevalence through a variety of techniques, which include providing technical and material assistance to regions across the country, coordinating overall regional capacity building in manpower, logistics, and finance, and strong monitoring and evaluation. 43
When compared to research conducted at the global or Africa level, this study contained fewer studies due to the restricted sources of the study’s field, namely, asymptomatic malaria. However, there is no guarantee that the findings are comparable as the parasite prevalence surveys are often opportunistic, prone to observer bias, non-standardized, and could be affected by a multitude of factors including the age structure of the sample, the timing of sampling with local malaria transmission seasons, and the methodology applied to parasite detection.15,43
The authors of this systematic review and meta-analysis would want to judge the decreased pooled prevalence in this scenario with utmost circumspection when compared to other empirical studies. The prevalence of asymptomatic malaria reported by small-scale studies so far in Ethiopia surprisingly ranges from 1.18% to 18.44% which could not be ruled out for the possible presence of different factors mentioned above. Again, we were shilly-shally to conclude the burden of malaria in practice would fit the pooled prevalence reported in this study. This could be because RDT and microscopy-based diagnoses are prone to underestimate the prevalence of asymptomatic malaria. Apart from this, asymptomatic malaria was also defined differently in different articles.43,44
In this systematic and meta-analysis, sex, educational status, and residence of the individuals were not found to be significantly associated with asymptomatic malaria in Ethiopia. This finding was incongruent with other empirical studies,12,28,31,45,46 but agreed with small-scale studies in Ghana and Tanzania.47,48 The difference could be related to the age structure of the sample, methodology (study area, sample size and diagnostic approach), and timing of the sample with the local malarial transmission seasons. 15 The distribution of malaria in Ethiopia is varying based on altitude due to its direct effect on temperature and it reaches its peak bi-annually. 49 Furthermore, the authors could not found similar studies done using a comprehensive approach to compare and contrast the identified associated factors. Kim A Lindblade et al. 8 suggested that the level of immunity of the individuals to infection, co-infection with soil-transmitted helminths, and the method used to diagnose the infection are the main associated factors.
On the other side, IRS in the last 12 months reduced the odds of asymptomatic malaria by about 54% (OR = 0.46; 95% CI = 0.26, 0.81) than their counterparts. Moreover, never using ITN and the presence of stagnant water nearby houses are among the independent predictors of asymptomatic malaria in Ethiopia. This finding agrees with other literature. This is because these methods use as a preventive method by creating physical barriers, repelling or killing the mosquitoes that have the potential to persist for some other times without needing to be re-treat with insecticide. 49
Limitations
There was an exhaustive literature search conducted for both published and unpublished studies and the quality of each study was assessed using the Newcastle–Ottawa Scale for cross-sectional studies quality assessment tool. This will contribute to the quality of the review to produce conclusive findings. The results from this study are important for the country striving for nationwide malaria elimination by 2030. However, one should use the findings with caution due to some limitations of this review. First, the diagnostic tests used in the studies, microscopy and RDT, are limited in detecting all asymptomatic malaria infections. This might pose a wrong conclusion in generalizing the reduced pooled prevalence reported in this study. Second, most of the studies were conducted at the facility level which is not the ideal setting for assessing asymptomatic malaria.
Conclusion
In conclusion, this study highlighted that pooled prevalence of asymptomatic malaria is high and varied by population groups. Prevalence of asymptomatic malaria was increased among those who never used ITNs and were living near stagnant water by six and three times, respectively. We recommend improved use of ITNs and environmental management. The use of more sensitive diagnostic methods could yield a higher burden of the disease. A study that considers factors on the actual causal pathway is required to generate more informative factors. Furthermore, active case detection is recommended for effective intervention toward the elimination.
Footnotes
Acknowledgements
The authors thank all authors of the studies included in this systematic review and meta-analysis.
Author contributions
A.T. was involved in developing the proposal. A.T. and T.T. were involved in the design, selection of articles, and data extraction and statistical analysis. A.T., T.T., and B.R. were involved in developing the initial drafts of the manuscript. AT, T.T., B.R., and G.M. participated in the final preparation of the manuscript and they approved the final draft of the manuscript for submission.
Availability of data and materials
All data analyzed during this study are included in the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
