Abstract
Tuberculosis (TB) is the leading cause of death in people living with HIV (PLHIV) globally, causing 208,000 deaths in PLHIV in 2019. PLHIV have an 18-fold higher risk of TB, and HIV/TB mortality is highest in inpatient facilities, compared with primary care and community settings. Here we discuss challenges and potential mitigating solutions to address TB-related mortality in adults with HIV. Key factors that affect healthcare engagement are stigma, knowledge, and socioeconomic constraints, which are compounded in people with HIV/TB co-infection. Innovative approaches to improve healthcare engagement include optimizing HIV/TB care integration and interventions to reduce stigma. While early diagnosis of both HIV and TB can reduce mortality, barriers to early diagnosis of TB in PLHIV include difficulty producing sputum specimens, lower sensitivity of TB diagnostic tests in PLHIV, and higher rates of extra pulmonary TB. There is an urgent need to develop higher sensitivity biomarker-based tests that can be used for point-of-care diagnosis. Nonetheless, the implementation and scale-up of existing tests including molecular World Health Organization (WHO)-recommended diagnostic tests and urine lipoarabinomannan (LAM) should be optimized along with expanded TB screening with tools such as C-reactive protein and digital chest radiography. Decreased survival of PLHIV with TB disease is more likely with late HIV diagnosis and delayed start of antiretroviral (ART) treatment. The WHO now recommends starting ART within 2 weeks of initiating TB treatment in the majority of PLHIV, aside from those with TB meningitis. Dedicated TB treatment trials focused on PLHIV are needed, including interventions to improve TB meningitis outcomes given its high mortality, such as the use of intensified regimens using high-dose rifampin, new and repurposed drugs such as linezolid, and immunomodulatory therapy. Ultimately holistic, high-quality, person-centered care is needed for PLHIV with TB throughout the cascade of care, which should address biomedical, socioeconomic, and psychological barriers.
Introduction
Tuberculosis (TB) is the leading cause of death in people living with HIV (PLHIV) globally, causing 208,000 deaths in PLHIV in 2019. 1 The World Health Organization (WHO) estimates that in 2019, 456,426 PLHIV were diagnosed with TB (see Figure 1 for global HIV TB prevalence estimates); yet 44% of TB disease goes undiagnosed in PLHIV. 1 This is a major concern as PLHIV are 18 times as likely to develop TB, compared with those without HIV. 1 TB risk increases in PLHIV as CD4 T cell count decreases, with data from a systematic review and meta-analysis of 164 full-text articles reporting that the odds of getting TB in PLHIV was 1.43 times higher [95% confidence interval (CI): 1.16–1.88] for every 100 cells per mm3 decrease in CD4 cell count. 2 Post-mortem studies demonstrate that TB is the cause of approximately 40% of facility-based deaths in PLHIV, 3 and that TB is undiagnosed at the time of death in almost half of these decedents. 4 TB prevalence and mortality are considerably higher for PLHIV seen in inpatient facilities (36.4% and 22.6%, respectively) and in primary care settings (20.5% and 3.1%, respectively) compared with community settings (6.9% and 1.6%, respectively). 5 Table 1 compares the differences in TB mortality between PLHIV and people without HIV, demonstrating a marked increase in TB mortality in PLHIV in countries with a high HIV prevalence such as Lesotho and South Africa. The WHO’s End TB Strategy aims to reduce all TB deaths by 95% by 2035, compared to 2015. 6 There is an urgent need to understand the drivers of TB mortality in PLHIV and develop interventions to decrease these largely preventable deaths.
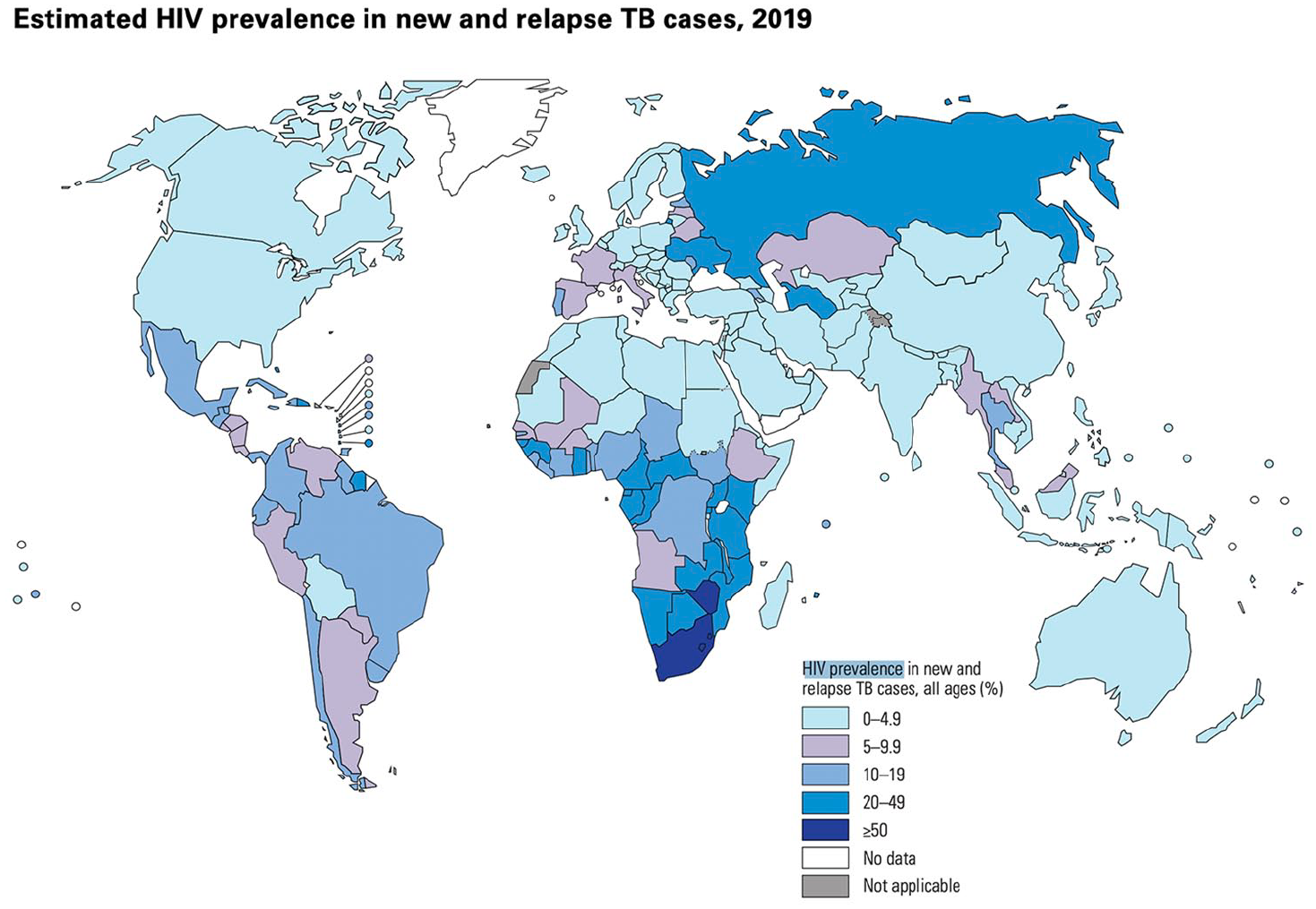
Global estimation of HIV prevalence in new and relapsed TB cases in percentage, 2019. Figure credit: WHO Global Tuberculosis Report 2020, WHO.
Estimated global burden of TB and comparison of TB mortality in people with and without HIV co-infection globally and in high TB burden countries with differing HIV prevalence a , 2019.

TB, tuberculosis; HIV, human immunodeficiency virus.
Per the Global Tuberculosis Report 2020, World Health Organization (WHO).
Based on the 2020 estimated list of top 30 high-burden HIV/TB countries.
High TB case fatality in PLHIV has been attributed to the combination of the following biological factors: rapid progression of disease due to failure of the immune system to control
The cascade of care, which maps the stages and gaps in HIV care delivery, has become a useful model to evaluate the quality of HIV care and has driven policy guidance on targets for HIV diagnosis, treatment, and viral suppression. 10 More recently, this model has been developed in the context of TB and is helpful to understand the extent of different gaps within local and national TB care cascades, with a view to designing targeted interventions that may vary by setting. 11 This narrative review seeks to explore how different aspects of TB care delivery, organized according to the stages of the TB cascade of care, impact the risk of mortality due to drug-susceptible TB disease in adults with HIV, and consider potential interventions that may mitigate this risk.
Challenges that drive TB-related mortality in PLHIV
Epidemiological risk factors for HIV/TB infection and mortality
Several studies have investigated epidemiological risk factors for increased TB mortality in PLHIV. Delayed initiation of ART is a critical risk factor for TB-related mortality in PLHIV (see section on Timing of treatment). 12 Other risk factors reported to be associated with HIV/TB mortality include low CD4 count, advanced HIV, not receiving ART, not being on co-trimoxazole preventive therapy, older age, male sex, being a female sex worker, incarceration, use of intravenous drugs, low weight, and being bed-ridden.13–17 Several studies have demonstrated that socioeconomic factors such as being illiterate, 18 and having a poor nutritional status 19 also increase the risk of mortality in people with HIV/TB co-infection.
Challenges impacting healthcare engagement
Barriers to healthcare engagement at each stage of the HIV/TB cascade of care impact the quality of care and can adversely affect all outcomes, including mortality. 20 A systematic review found that the main factors that affected initial engagement by Indian patients with TB care were not recognizing symptoms as severe enough to seek medical attention, work pressure, dissatisfaction with nearby health facility, lack of proximity to a healthcare facility, financial constraints, alcohol dependence, and inconvenient working hours. 21 A systematic review reported that barriers to healthcare engagement among Ethiopian presumptive TB patients included short duration of cough, absence of previous TB treatment history, lack of money, old age, low education level, lack of jobs, perceived wellness, and use of traditional healers. 22 A cross-sectional study in the Democratic Republic of Congo found that fear of discrimination and stigmatization, health worker attitudes, availability of both TB and HIV drugs, affected healthcare engagement in people with HIV/TB co-infection. 23 Barriers to healthcare engagement such as stigma are often intersectional and compounded in PLHIV compared with people without HIV (dual stigma for people with HIV/TB co-infection), and also highlight broader health system weaknesses, such as inefficient care delivery processes, gaps in data communication, and infrastructure. 20
TB preventive therapy
Given their increased risk of TB infection, TB preventive therapy (TPT) is recommended for PLHIV to prevent TB infection and reduce mortality. 24 The Temprano randomized controlled trial(RCT) from Côte d’Ivoire found that 6 months of TPT reduced TB mortality by in PLHIV 39%. 25 However, implementation of TPT remains weak with only 21% of eligible PLHIV initiated on TPT globally in 2021. 1 A systematic review and meta-analysis evaluating the latent TB cascade of care found that people with an immune-compromising condition like HIV were more likely to complete screening and receive a result (86%) compared with the general population (62%) and that people with medical conditions including HIV were more likely to complete treatment (50%) than the general population (10%); yet HIV infection was also cited as a risk factor associated with not starting treatment in one of the three included studies that evaluated PLHIV. 26 A retrospective study from Namibia evaluating TPT uptake in PLHIV found that only 20% of eligible patients completed the TPT cascade of care. 27 A cross-sectional study from Zambia also found low TPT completion rates with only 38% of the 25% of eligible PLHIV who initiated TPT completing treatment. 28
Challenges with the diagnosis of TB in people living with HIV
Interventions to increase the likelihood of earlier diagnoses for both HIV and TB reduce mortality in adults with HIV/TB co-infection, since factors such as having a higher CD4 level and being on ART are associated with lower mortality. 29 However, sputum, which remains the mainstay of TB diagnostic testing, is more difficult to obtain in the adults with HIV/TB co-infection, 30 due to paucibacillary disease and the lack of cavitation, which are associated with a higher rate of smear negative disease. 31 In addition, the accuracy of many TB diagnostic tests is lower in PLHIV, 32 who are also more likely to have extra pulmonary TB. 33
Screening and triage
There is an urgent need for improved TB screening or triage tests, due to their potential impact on facilitating earlier diagnosis and reducing the number of confirmatory molecular tests needed. 34 Chest radiography (CXR) has a sensitivity of 89–96% compared to 82–93% for symptom-based screening. 35 The use of computer-aided detection (CAD) software to enable automated CXR analysis is now conditionally recommended by WHO as an alternative to human readers. 35 However, a recent study of PLHIV with presumptive TB with negative smear microscopy in Uganda demonstrated that while CXR interpretation in conjunction with Xpert MTB/RIF testing increased the overall sensitivity for TB diagnosis (88%), specificity was decreased (52%) compared to Xpert MTB/RIF alone (96%), highlighting the diagnostic limitations of CXR in this patient population. 36
A prospective cohort study from 2019 conducted in PLHIV with a cell count of ⩽350 cells/μL from two clinics in Kampala, Uganda found that those with elevated C-reactive protein (CRP) levels (⩾8 mg/L) were 1.95 times as likely to develop an opportunistic infection like TB over the course of 3 months as compared to those with lower CRP levels (⩽8 mg/L).34,37 A prospective study of PLHIV in Uganda found that the sensitivity and specificity of CRP as a screening test were 89% and 72%, respectively.
38
A cross-sectional study from South Africa in PLHIV found that compared with TB symptom screening, CRP screening had the same sensitivity (90.5%) but a higher specificity (58.5%
Diagnosis – molecular tests
Molecular WHO-recommended rapid diagnostic tests (mWRDs) should be used, whenever possible, as the initial diagnostic test for pulmonary TB and certain forms of extrapulmonary TB such as TB meningitis since they not only detect
Diagnosis – microbiological tests
Diagnosis – biomarker-based tests
Other challenges associated with diagnosis in high HIV/TB incidence settings
Access to currently WHO-recommended diagnostic tests remains a major limitation for PLHIV undergoing TB evaluation in high TB incidence settings. 30 The relatively high cost and necessary infrastructure associated with mWRDs prohibit wider scale-up. 51 Optimizing implementation of mWRDs like Xpert also requires consideration of multiple factors beyond diagnostic accuracy alone, including comprehensive training, quality assurance, ensuring adequate supply chains, maintenance, and technical support. 52
Timing of TB and HIV treatment and treatment adherence on HIV/TB mortality
Decreased survival of people with HIV/TB co-infection is more likely with late HIV diagnosis and delayed start of ART treatment, with lower mortality risks reported in PLHIV diagnosed at CD4 > 350 cells/mm3 compared to PLHIV diagnosed at CD4 < 350 cells/mm3.
53
A systematic review and meta-analysis from 2020 evaluating the effect and timing of ART initiation in eight RCTs found that early ART initiation (1–4 weeks following TB treatment initiation) was associated with a lower overall mortality rate as compared to those with later ART initiation [8 weeks to 6 months following TB treatment initiation; risk ratio (RR) = 0.81; 95% CI: 0.66–0.99,
Challenges with TB and HIV treatment adherence and completion
Following diagnosis and treatment initiation, ensuring adherence to and completion of TB treatment remain critical challenges to preventing TB mortality in PLHIV. A systematic review from 2015 evaluating TB treatment non-adherence and loss to follow-up found that the main factors associated with these outcomes were socio-economic factors (lack of food, transportation costs, unemployment, etc.), individual behavior (lack of knowledge, medication side effects, etc.), healthcare workers (poor communication, stigma, etc.), and HIV status.
55
The increased financial burden of paying for both TB and HIV treatment is also a recognized barrier, along with the lack of social support and dual disease stigma.
55
A review article from 2019 looked at the gaps in the cascade of TB care for PLHIV and found that healthcare system level gaps pertaining to TB treatment completion included interrupted supply chain management of TB drugs and diagnostics, poor implementation of TB treatment guidelines, and poor quality of care.
20
Virological failure, either due to poor adherence to ART or the development of HIV drug resistance, is also a risk factor for mortality in PLHIV. A nested observational cohort study evaluating unselected hospitalized PLHIV in Malawi found that 32% of those on ART for at least 6 months had virological failure, the majority of whom (83%) had resistance to at least two ART drugs, which was associated with increased mortality (adjusted hazard ratio 1.7, 95% CI: 1.2 to 2.4;
TB treatment in special situations – TB meningitis
TB meningitis is a form of central nervous system (CNS) TB that accounts for 5% of all extrapulmonary TB.
57
This form of TB is typically the most severe, as up to 50% of people die or suffer neurological complications.
57
In PLHIV, in whom 40% of TB is extrapulmonary (compared to the 10% in HIV-negative patients), there is a fivefold increase of the likelihood of CNS involvement.
58
PLHIV who get TB meningitis will commonly present with extrameningeal involvement and are more likely to suffer from altered mental status.
58
Diagnosis of TB meningitis is particularly challenging as the clinical manifestations may be nonspecific
59
and the accuracy of TB diagnostic tests (including smear, microscopy, culture, and NAAT) is lower on cerebrospinal fluid samples, compared with sputum.32,59,60 Treatment of TB meningitis is challenging because mortality is high at both 6 and 9 months into treatment (27.9% and 22.4%, respectively), with 53.4% of co-infected TB meningitis patients dying during treatment compared to 17.4% of HIV-negative patients.
54
Furthermore, mortality risk after relapse of TB meningitis in PLHIV is higher than in those without HIV (83%
TB treatment in special situations – TB-IRIS
TB-IRIS is a severe inflammatory syndrome associated with HIV/TB co-infection and occurs following the rapid reconstitution of the immune system after ART initiation. 62 The likelihood of TB-IRIS increases with the severity of CD4+ T-cell deficiency prior to ART initiation, with data from China demonstrating that patients with initial CD4 cell counts less than 50 cells/mm3 were at a 4.6-fold increased likelihood of getting TB-IRIS as compared to those with CD4 cell counts greater than 100 cells/mm3 [odds ratio (OR): 4.6, 95% CI (1.033–20.238)].62,63 After ART initiation, there was a 2.3-fold increased likelihood of getting TB-IRIS among patients who had a fourfold or greater increased in CD4 cell count as compared with those who had less than fourfold increase in CD4 cell count. 63 There are three types of TB-IRIS: paradoxical, unmasking, and CNS. 62 Both paradoxical and unmasking TB-IRIS typically occur within 3 months of ART initiation. Paradoxical TB-IRIS occurs when patients with known TB experience worse symptoms post-ART initiation while unmasking TB-IRIS occurs in patients without a prior TB diagnosis and experience marked inflammatory symptoms. 62 CNS TB-IRIS occurs in patients with TB meningitis 62 and is a contributor to poor outcomes including mortality in this high-risk population. 64
High rates of recurrent TB and post-TB disease sequelae
A recent systematic review reports higher rates of recurrent TB in high HIV prevalence areas,
65
consistent with a prior systematic review that reported a TB recurrence rate of 4.5 (3.2–5.8) in PLHIV compared to 1.9 (1.2–2.7) in people without HIV per 100 person-years.
66
Post-TB lung disease following TB completion is an additional concern. A prospective cohort study in Malawi found that after successful treatment of pulmonary TB, one-third of the cohort had abnormal spirometry and 40% had bronchiectasis. Although PLHIV were less likely to experience an acute respiratory event 1 year post-TB treatment (12.9%
Potential solutions to mitigate TB-related mortality in PLHIV
Addressing challenges with healthcare engagement
Addressing challenges with healthcare engagement in people with HIV/TB co-infection requires a multifaceted approach. Health workers in Namibia who received training in TB infection control, and patients’ rights and confidentiality, were more willing to report discrimination in the healthcare setting [adjusted odds ratio (aOR) 2.1 with 95% CI: 1.03–4.39% and 2.2 with 95% CI: 1.11–4.47]. 69 In addition, since many HIV patients avoid local ART centers due to anticipated stigma, including fear of recognition, ensuring anonymity is essential, along with reassurance from doctors and patient support networks that could encourage patients to seek care. 70 Internal stigma is also common, with patients expressing embarrassment and shame regarding both HIV and TB diagnoses. 71 Given that stigma is associated with a lack of knowledge about TB, 72 community-informed education campaigns may help reduce stigma on a larger scale. Financial support can reduce the socioeconomic barriers that impact engagement in care,73,74 although data on strategies such as financial incentives for populations that include PLHIV are limited. 75 There is an urgent need to increase health system resources to improve healthcare infrastructure, integration of TB and HIV healthcare, and enable person-centered care that addresses socioeconomic constraints such as food insecurity and lack of transportation. 20
Optimizing HIV/TB integrated care
A descriptive cross-sectional study conducted at Mulago National Referral Hospital in Kampala, Uganda investigating the hospital’s efforts to integrate TB and HIV care found that the main challenges were lack of program coordination (providers being late for appointments and poor instruction provided to patients) and lack of treatment for other opportunistic infections.
76
A retrospective cohort study in Nigeria found that the difficulties in running an HIV/TB integrated healthcare facility included missing data due to poor documentation, which led to miscommunication and lost referrals from the clinic to other care facilities (X-ray/lab units) that limited the ability of patients to complete treatment.
77
Given the importance of integrated HIV/TB care, we sought examples of changes in healthcare systems and policies to facilitate its implementation. A programmatic evaluation of almost 120,000 patients treated by the ART program in South Africa from 2009 to 2013 demonstrated increased uptake of ART in ART-naïve patients from 37.0% to 77.7% and reduced TB case fatality from 7.4% to 5.2% (
Implementing TB preventive therapy
Alternative shorter TPT regimens (instead of 9 months of isoniazid) are now recommended by WHO
24
based on several recent trials. An RCT that enrolled patients from the United States, Spain, Canada, and Brazil found that the use of rifapentine plus isoniazid for 3 months was as effective at preventing TB as the standard 9 months of isoniazid, with higher completion rates (82.1%
Improving TB screening and diagnosis
Wider implementation of TB screening in PLHIV is essential to reducing TB-related mortality. The WHO consolidated guidelines on tuberculosis recommend that PLHIV should be screened systematically for TB at every visit to a healthcare facility. 35 An RCT to be completed in 2023 will be conducted in PLHIV in Ugandan clinics to test the effect of CRP results on uptake of TPT. 85 The WHO also recommends that further clinical trials should be undertaken to evaluate other screening tools including CXR with CAD in PLHIV. 35
There is an urgent need for rapid, accurate biomarker-based tests for TB diagnosis in PLHIV. A diagnostic accuracy study from 2019 using frozen urine PLHIV samples from FIND Specimen Bank and the University of Cape Town Biobank found that the sensitivity of Fujifilm SILVAMP TB LAM (FujiLAM), a next-generation LAM assay, was 70.4% (95% CI: 53.0, 83.1) compared to 42.3% (95% CI: 31.7, 51.8) for AlereLAM in PLHIV, without a statistically significant difference in specificity between the two tests (FujiLAM: 90.8%, 95% CI: 86.0–94.4; AlereLAM: 95.0%, 95% CI: 87.7–98.8) 86 Trials evaluating the impact of this higher sensitivity next-generation LAM assay are underway. 87
Given the higher risk of mixed-strain infections and drug-resistant TB in PLHIV, improving access to whole-genome sequencing and next-generation sequencing, as strategies that include drug resistance profiling and enable the identification of multiple strains will also improve TB care and public health surveillance efforts.30,44 Such initiatives will require greater funding to be dedicated TB diagnostics research and development, as well as building laboratory capacity in low- and middle-income countries. 88
Impact of TB diagnostic tests on HIV/TB patient important outcomes including mortality
Xpert and mortality
A recent Cochrane Review from 2021 analyzing data from five RCTs with 9932 participants judged to represent moderate-certainty evidence reported that it was not possible to determine the impact of the use of Xpert MTB/RIF compared with smear microscopy on all-cause mortality (RR: 0.9, 95% CI: 0.8–1.1). 61 They did, however, report that there was probably a reduction in mortality in PLHIV (OR: 0.8, 95% CI: 0.67–0.96), which emphasizes the importance of access to Xpert and other mWRDs in PLHIV being evaluated for TB. 61 Given the increased sensitivity of Xpert Ultra, including on CSF samples, wider scale-up of Xpert Ultra can facilitate TB diagnosis in PLHIV. 41
LAM and mortality
Urine LAM is the only currently recommended biomarker-based TB diagnostic test and has been demonstrated to have an impact on mortality.
89
A Cochrane review analyzing two RCTs in inpatient settings and one RCT in outpatient settings, all in sub-Saharan Africa, reported that the use of LAM as part of a TB diagnostic strategy likely reduces mortality at 8 weeks and probably results in a slight increase in TB treatment initiation in PLHIV in inpatient settings, and that the use of LAM as part of a TB diagnostic strategy may reduce mortality at 6 months and may result in a large increase in TB treatment initiation in PLHIV in outpatient settings.
90
LAM is hypothesized to indirectly lead to increased mortality risk in people with HIV/TB co-infection, as increased LAM levels were found to contribute to the survival of
Improving the treatment of HIV/TB co-infection
Timing of ART in adults with HIV/TB co-infection
In 2021, the WHO updated their guidelines to recommend that ART should be started within 2 weeks regardless of CD4 cell count following initiation of antituberculosis treatment in people with HIV/TB co-infection.
94
A systematic review and meta-analysis from 2021 looked into nine clinical trials from over 2000 screened articles to determine the best timeline for starting ART in people with HIV/TB co-infection.
95
While there was no difference in mortality in earlier (⩽4 weeks) ART initiation
While implementation outside of clinical trials is often more challenging, a number of studies have looked at the implementation of strategies to improve time to ART initiation in real-world situations. A prospective cohort study in South Africa looked at the implementation of a collaborative learning network, where representatives from each sector of healthcare meet to learn about quality improvement methods and to discuss methods of improvement in HIV healthcare, to HIV healthcare and found that monthly ART initiation increased from 179 patients per month to 511 patients per month following the intervention. 96 A prospective cohort in Siberia looked at ART initiation in a TB referral clinic following the implementation of increased education, HIV testing, and prioritization of HIV referrals and found that ART initiation rates increased from 17% to 54%. 97
Duration of TB treatment
There is considerable interest in shortening TB treatment regimens to minimize toxicity, drug interactions, impact on patient quality of life, and facilitating TB care from a programmatic standpoint. Typically, longer durations of treatment may be recommended in PLHIV, as the Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines states that an additional 3 months can be added on to the standard 6 months of TB in PLHIV due to their increased risk of TB relapse.
98
Observational data has demonstrated higher risk of unsuccessful TB treatment outcomes in PLHIV treated for less than 6 months. A recent RCT compared the standard 6-month TB treatment to two different 4-month regimens, one where RIF was replaced with rifapentine and the other where RIF was replaced with rifapentine and ethambutol with moxifloxacin in 2343 patients with culture-positive pulmonary TB in 13 countries.
99
The 4-month regimen containing rifapentine and moxifloxacin was noninferior to the standard regimen (15.5%
Role of empiric TB treatment in PLHIV
Empiric treatment of TB without definitive microbiological confirmation of disease is often pursued given the challenges of TB diagnosis. 101 A prospective cohort study conducted in Kenya found that there was no difference in risk of mortality for PLHIV who were treated for TB empirically compared with those treated based on microbiological confirmation. 102 Similarly, an RCT conducted in the Ivory Coast, Uganda, Vietnam, and Cambodia that compared the risk of mortality at 48 months for empiric TB treatment with test-guided TB treatment in HIV-infected adults who had not previously received ART and had CD4+ T-cell counts below 100 cells/mm3 found that empiric TB treatment did not reduce the risk of mortality (aHR: 0.97; 95% CI: 0.67–1.40). 101 However, an observational cohort study of TB/HIV co-infected patients conducted in South Africa found that in-hospital mortality was lower in patients with microbiologically confirmed TB, compared with patients treated empirically (HR: 0.5; 95% CI: 0.3–0.9), emphasizing the importance of trying to obtain a microbiological diagnosis of TB in PLHIV. 46
Optimal treatment for TB meningitis
There is increasing evidence that we are underdosing RIF in many people with TB and that higher RIF doses are safe and well tolerated.
103
A pharmacokinetic analysis of a prior placebo-controlled trial comparing immediate
Reducing the risk of TB-IRIS
Current recommendations for treatment of TB-IRIS depend on the type of TB-IRIS presentation but most commonly will include corticosteroid therapy to reduce the aberrant inflammation that drives TB-IRIS.
62
An RCT demonstrating PLHIV with TB who received prednisone during the first 4 weeks after ART initiation had a reduced TB-IRIS incidence of 32.5% compared with 45.7% who received placebo (RR = 0.7, 95% CI: 0.51, 0.96;
Ensuring that care does not end at TB treatment completion
A recent modeling analysis estimated that 155 million TB survivors were alive in 2020, of whom 8% were estimated to have HIV at the time they were diagnosed, highlighting the importance of interventions to prevent ongoing morbidity, recurrent TB, and reduce the impact of ongoing stigma. 114 This call has been echoed by TB survivors, who emphasize the importance of mental health support and services to mitigate the long-term negative impacts of TB. 115 Secondary preventive therapy has been shown to reduce the risk of recurrent TB in PLHIV. 116 Currently there are no evidence-based guidelines for the management of post-TB lung disease or other complications of TB. 68 The scope of TB care delivery needs to be expanded such that we can ensure patients will have a stable and successful life post-TB treatment and address adverse health effects like post-TB lung disease, along with the socioeconomic and psychological burden.
Conclusion
Addressing the high mortality faced by PLHIV with TB is an urgent priority. Although many of the drivers of this increased mortality require further research, such as optimizing treatment regimens, including the use of adjunctive immunomodulatory therapy beyond steroids alone, to reduce morbidity and mortality from TB meningitis, there is clearly much more that can be done with existing tools. Improving the delivery of integrated person-centered HIV/TB care should incorporate innovative approaches to reduce stigma and improve healthcare engagement, as well as ensure access to quality-assured WHO-recommended screening and diagnostic tests including CRP, CXR, mWRDs, and urine LAM, with prompt linkage to care and close follow-up to ensure initiation of effective treatment and management of drug interactions and side effects. Clinicians should consider the possibility of TB-IRIS and whether steroids should be used pre-emptively to reduce this risk. Table 2 provides a summary of evidence-based interventions that can decrease HIV/TB mortality and provides an overview of ongoing challenges and research gaps. High-quality implementation research is urgently needed to evaluate how best to implement evidence-based TB interventions using contextually acceptable strategies, 20 which should include key populations such as PLHIV who have histor-ically often been excluded. 100 Implementers and clinicians need to ensure the holistic care of people with HIV/TB co-infection throughout the cascade of care (Figure 2) to ensure prompt diagnosis and treatment of both infections, with comprehensive and supportive care that does not end at treatment completion and that addresses the biomedical, socioeconomic and psychosocial barriers faced by affected individuals and communities.
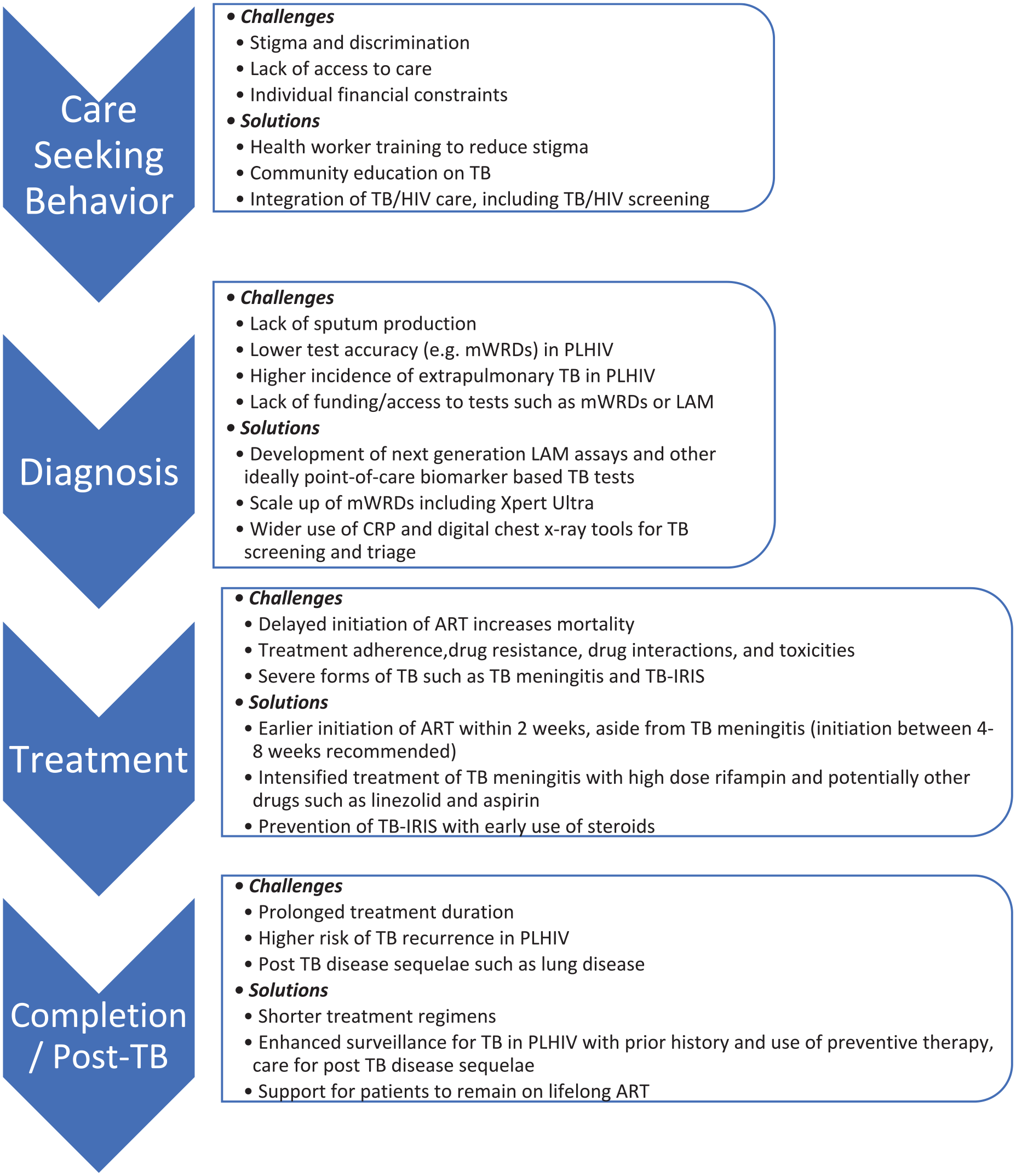
Challenges and potential solutions to improve TB HIV mortality organized according to the stages of the cascade of TB care for PLHIV.
Interventions to reduce HIV/TB co-infection mortality: current evidence-based approaches and research gaps.
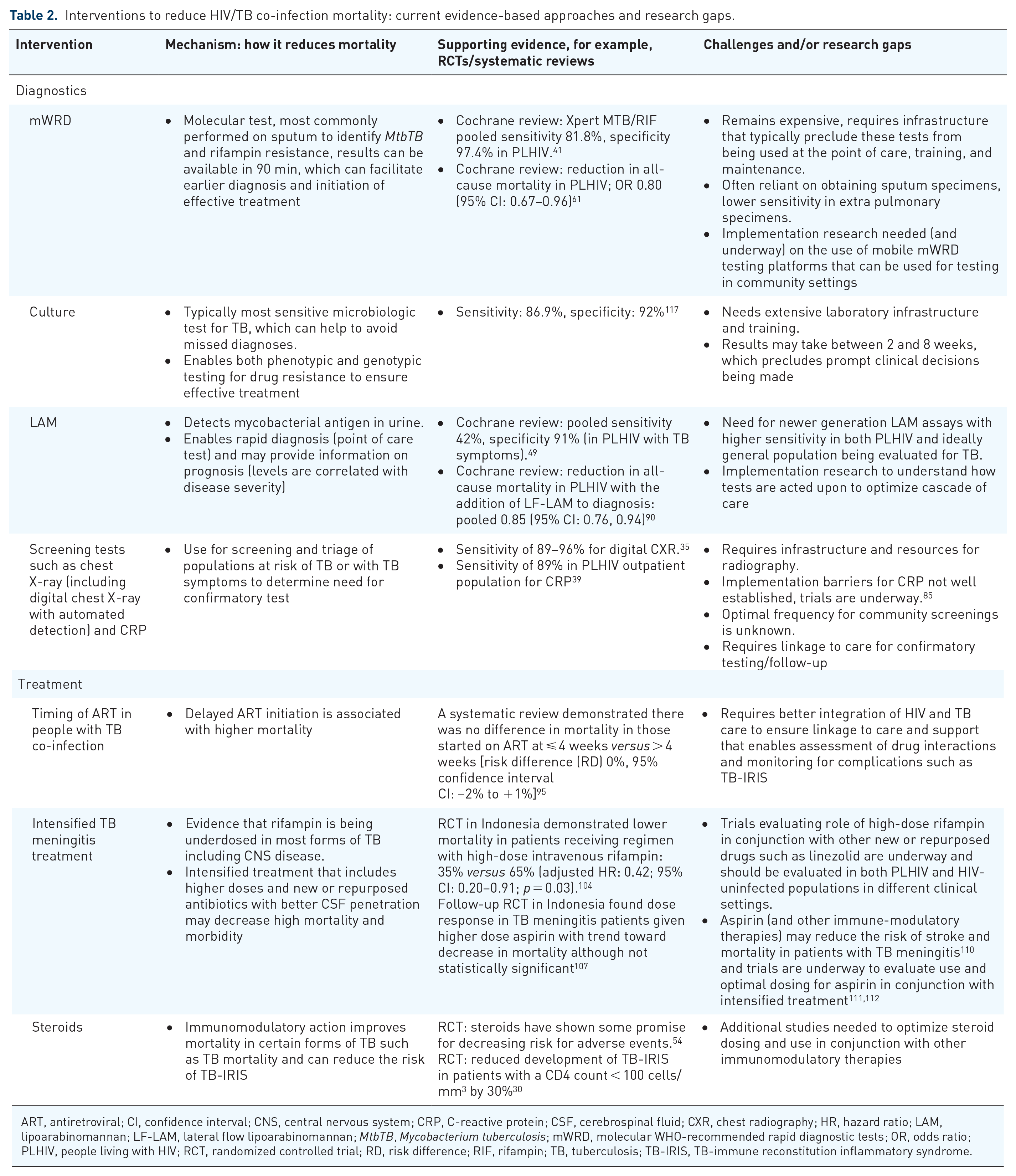
ART, antiretroviral; CI, confidence interval; CNS, central nervous system; CRP, C-reactive protein; CSF, cerebrospinal fluid; CXR, chest radiography; HR, hazard ratio; LAM, lipoarabinomannan; LF-LAM, lateral flow lipoarabinomannan;
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RRN is supported by a National Institutes of Health Career Development Award (NIAID K23 AI132648-04) and an American Society of Tropical Medicine and Hygiene Burroughs Wellcome Fellowship.
