Abstract
Introduction
Glycoprotein IIb/IIIa inhibitors are recommended by guidelines in patients with ST-segment elevation myocardial infarction treated with primary percutaneous coronary intervention. There are few studies directly comparing these agents. The aim of this study was to assess whether eptifibatide is a safe and cost-effective alternative to abciximab in the treatment of primary percutaneous coronary intervention for ST-segment elevation myocardial infarction.
Methods
This was an observational cohort study of 3863 patients who received a GPIIb/IIIa inhibitor whilst undergoing primary percutaneous coronary intervention from 2007 to 2014. Patients who did not receive a GPIIb/IIIa inhibitor were excluded. Time to first major adverse cardiac event defined as death, non-fatal myocardial infarction, stroke or target vessel revascularization, and total hospital costs were compared between the groups.
Results
In all, 1741 patients received abciximab with 2122 receiving eptifibatide. Patients who received eptifibatide had higher rates of previous MI/percutaneous coronary intervention and were more likely to undergo a procedure from the radial route. Unadjusted Kaplan–Meier analysis revealed no significant difference in the 1-year event rates between patients given eptifibatide versus abciximab (p = 0.201). Age-adjusted Cox analysis demonstrated no difference in 1-year outcome between abciximab and eptifibatide (hazard ratio: 0.83; 95% confidence interval: 0.73–1.39), which persisted after multivariate adjustment (hazard ratio: 0.92; 95% confidence interval: 0.79–1.56) including the incorporation of a propensity score (hazard ratio: 0.88; 95% confidence interval: 0.71–1.44). Eptifbatide was associated with significant cost savings being 87% cheaper overall compared to abciximab (on average £650 cheaper per patient and saving approximately £950,000).
Conclusion
This observational data suggest that eptifibatide is associated with similar outcomes and significant cost savings compared to abciximab when used in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention.
Background
Advances in anti-platelet treatment for acute myocardial infarction (AMI) with the introduction of ticagrelor 1 and prasugrel 2 have improved outcomes after primary percutaneous coronary intervention (PCI) with guidelines recommending their use. 3 However, recent emerging data suggest that the administration of morphine, a commonly used analgesic during AMI can impair the absorption and delay the onset of action of ADP receptor antagonist drugs.4,5 This has lead to the suggestion of the need for strategies such as adjunctive pharmacology to overcome or at least diminish the negative impact of morphine on these anti-platelet effects of oral P2Y12 receptor inhibitors. 6 Glycoprotein IIb/IIIa inhibitors have been used as adjuncts for primary PCI for many years but have fallen out of fashion throughout the UK and Europe due to the use of these newer anti-platelet drugs. The most commonly used GPIIb/IIIa inhibitor is abciximab,7,8 with some contemporary data supporting its use as a bridging strategy in the morphine era; however, it is still relatively expensive and emerging data suggest comparability of eptifibatide.9–13 We therefore conducted a quality improvement study at our institution switching from abciximab to eptifibatide in order to determine whether eptifibatide provides a safe, effective and cost-effective alternative to abciximab in the treatment of primary PCI for ST-segment elevation myocardial infarction (STEMI) and providing further data for its use as a bridging agent if morphine is used.
Methods
Study design
The study population was derived from a high-volume, single centre registry of all patients undergoing primary PCI for acute STEMI between January 2006 and December 2014. In 2010, we switched from abciximab to eptifibatide as the first line GPIIb/IIIa inhibitor administered at our institution. This was done following a thorough literature review of available randomized/observational data, discussion with management and based upon the significantly lower cost of eptifibatide compared to abciximab. This study was performed following this switch between GPIIb/IIIa inhibitors. It therefore included 3863 consecutive patients receiving either abciximab (n = 1741) (pre-2010) or eptifibatide (n = 2122) (post-2010) as adjunctive therapy between 2006 and 2014. Patients who did not receive a GPIIb/III inhibitor were excluded. Barts Health Heart Attack Centre is the only tertiary centre for the North-east region of London and took all patients with AMI for primary PCI in an unselected manner. This included patients with cardiogenic shock and post cardiac arrest, including intubated and ventilated patients.
Abciximab was administered as an intravenous bolus of 0.25 mg/kg before/at the start of primary PCI followed by a continuous infusion of 0.125 µg/kg/min (to a maximum of 10 µg/min) for 12 h. Eptifibatide was administered as an intravenous bolus of 180 µg/kg at the start of primary PCI followed by a continuous infusion of 2.0 µg/kg/min for 12 h following the procedure. Contraindications for glycoprotein IIb/IIIa inhibitor were in accordance with summary of product characteristics14,15 and included previous major gastrointestinal bleeding, previous haemorrhagic stroke, chronic renal impairment, thrombocytopenia and concurrent anticoagulant use. Glycoprotein IIb/IIa inhibitor infusions were stopped if there was an adverse reaction, reduction in platelet count from baseline of >50%, minor or major bleeding according to the TIMI bleeding criteria. Standard PPCI protocol for our institution includes pre-loading with 300 mg aspirin, 300 mg (pre-2007) or 600 mg clopidogrel (post-2007) and a bolus of GPIIb/IIIA inhibitors unless contraindicated. All patients were prescribed 75 mg aspirin and 75 mg clopidogrel maintenance therapy. Clopidogrel maintenance therapy was recommended for 1-year post primary PCI. Aspiration thrombectomy was performed at the operator’s discretion. Successful primary PCI result was defined as final TIMI (Thrombolysis In Myocardial Infarction) flow grade 3 and residual stenosis <20% in the infarct-related artery at the end of the procedure.
Data were prospectively entered onto the local database at the time of primary PCI. Data collected included patient characteristics (age, sex, history of hypertension, hypercholesterolaemia, diabetes mellitus, smoking status, previous myocardial infarction, previous PCI, previous coronary artery bypass graft (CABG), left ventricular ejection fraction and baseline cardiogenic shock), procedural factors included access site, culprit vessel, number of diseased vessels, number of vessels treated, stent type, TIMI flow pre-procedure, TIMI flow post-procedure and procedural complications (including myocardial infarction, emergency CABG, arterial complications and arrthymias requiring DC cardioversion). Further inpatient complications, post-discharge complications and further revascularisation were documented retrospectively using the electronic patient record.
The primary effectiveness end-point was the first major adverse cardiac event (MACE) defined as death, non-fatal myocardial infarction or target vessel revascularization.
Secondary end-points were in-hospital PCI-related bleeding, categorised by the TIMI bleeding criteria and cost per patient. MACE events (identified from patient notes and electronic records) were adjudicated by three independent physicians who were not involved in the procedure and were unaware of the patient’s GPIIb/IIIa type. All-cause mortality data were recorded as of the 12 September 2015 and obtained via the British Cardiovascular Intervention Society-UK Central Cardiac Audit Database (CCAD). This national database is periodically linked to the UK Office of National Statistics and provides live/death status of treated patients. Only patients who had complete database records and National Health Service unique numbers (allowing live/death status to be assessed) were included in the analysis. A retrospective data quality audit of 100 randomly selected medical records established that 94.8% of data fields, including complications, were entered correctly into the database.
Cost-effectiveness
The cost of eptifibatide and abciximab per patient were calculated based on the standard drug dosages and protocols from Epistent, Pursuit and PrismPlus trials, respectively (Table 1) and based on the unit prices given in the British National Formulary (BNF) (Table 2). The average cost per patient and the total savings made by the switch were calculated.
Standard drug dosages and protocols from Epistent, Pursuit, and PrismPlus trials, respectively.

aBased on unit prices given in Table 3 below.
PCI: percutaneous coronary intervention.
GP2B3A drug cost per unit.
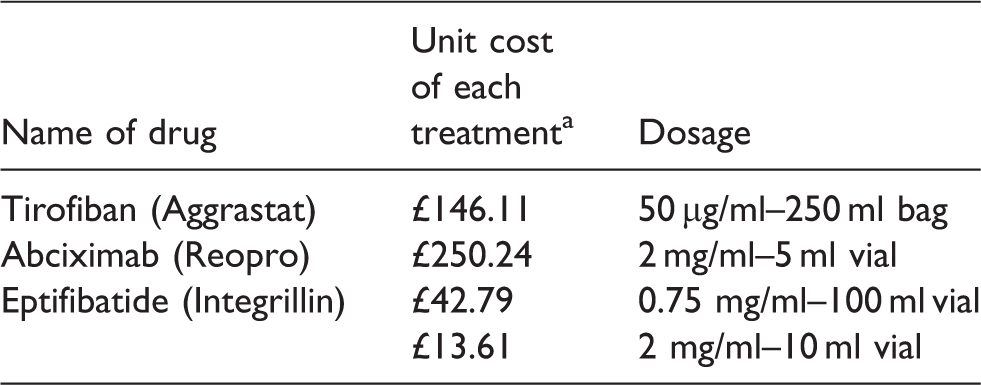
aBNF September 2014–March 2015.
BNF: British National Formulary.
Baseline characteristics according to treatment group.
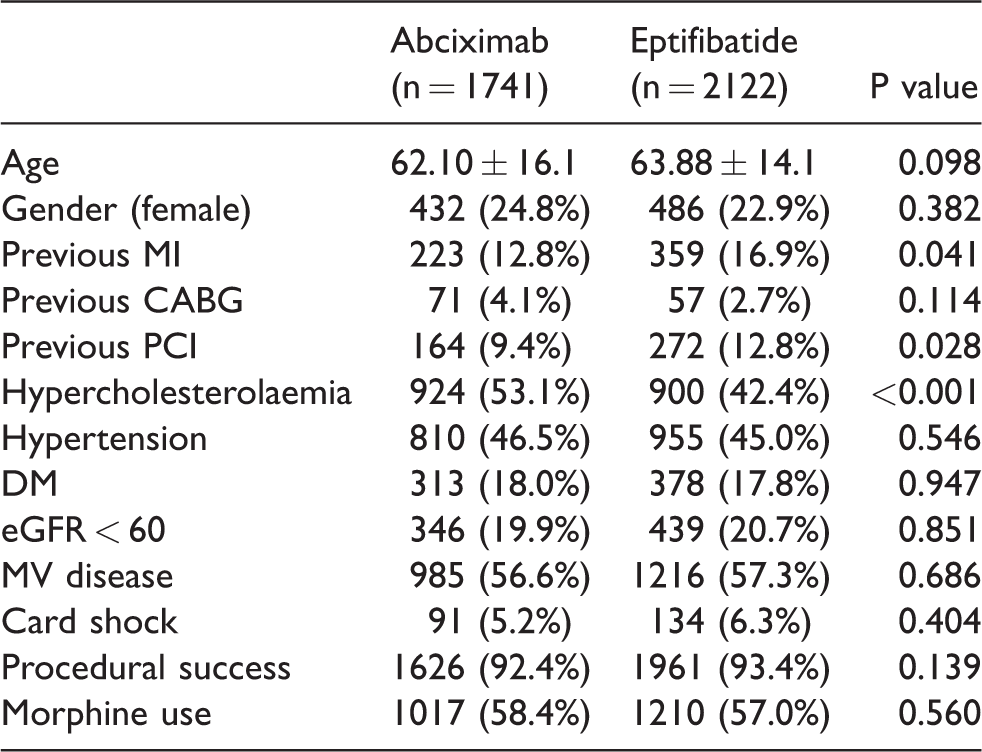
MI: myocardial infarction; CABG: coronary artery bypass graft; PCI: percutaneous coronary intervention; DM: diabetes mellitus; eGFR: estimated glomerular filtration rate; MV: multivessel.
*p value < 0.05.
Procedural characteristics according to GPIIb/IIIa group (P < 0.05).

LAD: left anterior descending.
In-hospital outcomes post primary PCI comparing GPIIb/IIIa type.
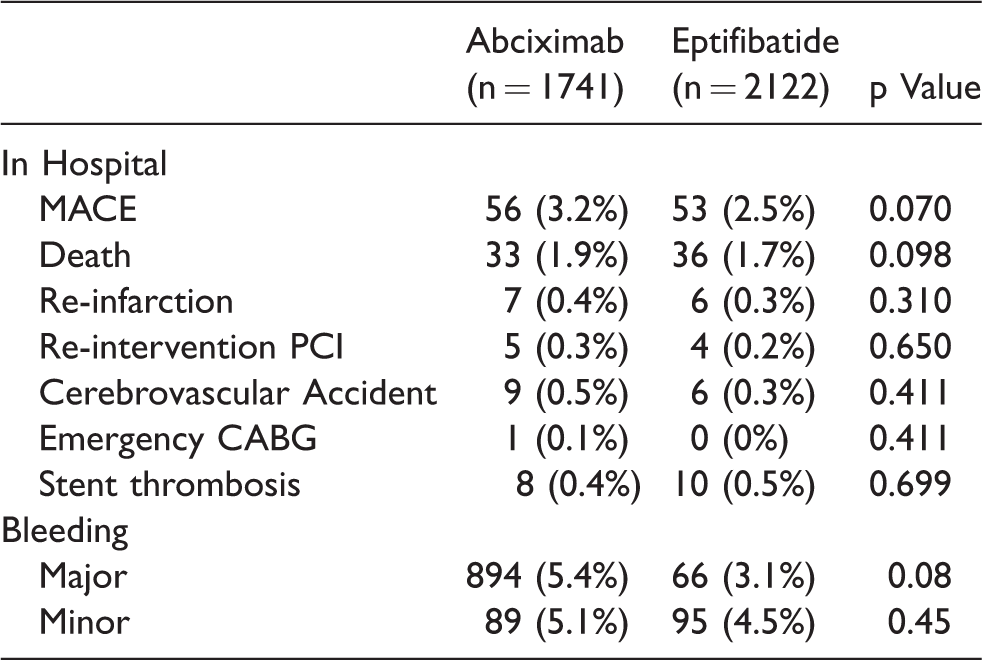
MACE: major adverse cardiac event; PCI: percutaneous coronary intervention; CABG: coronary artery bypass graft.
Ethics
The data were collected as part of a mandatory national cardiac audit and all patient identifiable fields were removed prior to analysis. The local ethics committee advised us that formal ethical approval was not required.
Statistical analysis
Clinical characteristics of eptifibatide- versus abciximab-treated patients were compared using the Pearson Chi Square test for categorical variables and Student t test for continuous variables. Normality of distribution was assessed using the Shapiro–Wilks test. We calculated Kaplan–Meier product limits for cumulative probability of reaching an end point and used the log rank test for evidence of a statistically significant difference between the groups. Time was measured from the first admission for a procedure to outcome (all cause mortality). Cox regression analysis was used to estimate hazard ratios (HRs) for the effect of GPIIb/IIIa inhibitor type in age-adjusted and fully adjusted models, based on covariates (p < 0.05) associated with the outcome. The proportional hazards assumption was evaluated by examining log (−log) survival curves and additionally was tested with Schoenfield’s residuals. The proportional hazard assumption was satisfied for all outcomes evaluated. Cox analysis was performed both with and without year of procedure as a covariate to correct for any time-related effect during the study period.
A propensity score analysis was carried out using a non-parsimonious logistic regression model comparing abciximab and eptifibatide patients. Multiple variables were included in the model, including age, gender, diabetes, hypertension, hypercholesterolaemia, previous CABG, previous PCI, previous MI, multivessel disease, chronic renal failure, pre-procedure TIMI flow, ejection fraction and procedural success. C-Score was 0.81 indicating good discrimination. After ranking propensity score in an ascending order, a nearest neighbour 1:1 matching algorithm was used with callipers of 0.2 standard deviations of the logit of the propensity score. Each abciximab and eptifibatide patient was used in at most one matched pair to create a matched sample with similar distribution of baseline characteristics between observed groups. Based on the matched samples, cox proportional hazard model was used to determine the impact of GPIIb/IIIa inhibitor on mortality over follow-up. STATA version 10 and Graphpad Prism version 5 were used for all analysis.
Results
During the study period, there were 4829 patients presenting with a STEMI. Of these patients, 3863 (80.0%) received a GPIIb/IIIa inhibitor as adjunctive therapy for primary PCI. In all, 1741 patients received abciximab and 2122 patients received eptifibatide.
Patient characteristics (Table 3)
With respect to baseline characteristics, the two groups were well balanced. The mean age in the eptifibatide group was 63.88 ± 14.1 and 62.10 ± 16.1 in the abciximab group. The majority of the patients were male in both groups (76.2% in the eptifibatide group and 77.1% in the abciximab group). Patients in the eptifibatide group were more likely to have had a previous myocardial infarction (16.9% vs 12.8% and undergone previous PCI compared with patients receiving abciximab (12.8% and 9.4%, p = 0.028). There was also a higher prevalence of hypercholesterolaemia in the eptifibatide group compared with the abciximab group (53.1% versus 42.4%, respectively, p < 0.001). Similar rates of morphine use were seen in the two groups.
Procedural characteristics (Table 4)
Patients treated with eptifibatide were significantly more likely to undergo the procedure via the radial route receive intervention to the LAD and have multi-vessel intervention. They were also more likely to undergo PCI with drug-eluting stents.
Procedural outcome (Table 5)
There were similar rates of procedural success achieved in the two groups (93.4% of the eptifibatide group vs 92.4% of the abciximab group). There were no significant differences between the groups for in-hospital MACE or any of its components (MI/emergency bypass surgery/death). Additionally, there was no difference in the rates of any procedural complication between the groups.
Bleeding (Table 5)
No difference in rates of either major (3.1% of the eptifibatide group versus 5.4% of the abciximab group, p = 0.08) or minor bleeding (4.5% of the eptifibatide group versus 5.1% of the abciximab group, p = 0.45) were seen between the two groups.
Follow-up (Figure 1)
Unadjusted Kaplan–Meier analysis revealed no significant difference in MACE rates over the follow-up period between patients given eptifibatide versus abciximab (18.4% vs 19.2%, p = 0.121). The pattern was similar for all MACE components including all cause mortality, recurrent myocardial infarction and target vessel revascularisation.
Unadjusted and multivariate analysis
Age-adjusted Cox analysis demonstrated no difference in outcomes after the follow-up period between abciximab and eptifibatide (HR: 0.83; 95% confidence interval (CI): 0.73–1.39), which persisted after multivariate adjustment (HR: 0.92; 95% CI: 0.79–1.26). In addition, after regression adjustment incorporating a propensity score (age, gender, ethnicity, previous MI, PCI or CABG, diabetes, hypertension, hypercholesterolaemia, smoking status, presence or absence of shock and ejection fraction) into the hazards model as a covariate, no difference in outcome emerged (HR: 0.88; 95% CI: 0.71–1.24) (Figure 2).

Kaplan–Meier curves showing cumulative probability of major adverse cardiac events after primary PCI according to GPIIb/IIIa type.PCI: percutaneous coronary intervention.

Multivariate Cox regression analysis for hazard of all-cause mortality after percutaneous coronary intervention.
The above Cox proportional hazard model was repeated with the year of procedure included as a categorical variable to allow for improvements in PCI technique and technology over the long study period. This confirmed no association between GPIIb/IIIa inhibitor type and MACE (HR: 0.85; 95% CI: 0.73–1.22).
Propensity score
To further account for confounding variables and bias, propensity score matching was performed to adjust for differences in demographic and procedural variables producing a total of 2882 patients (1441 in the eptifibatide group and 1441 in the abciximab group). Following matching the baseline demographics and procedural variables were well balanced in the two propensity-matched cohorts. In the propensity-matched cohorts, Cox regression analysis revealed that GPIIb/IIIa inhibitor type was not a predictor of MACE (HR: 0.82; 95% CI: 0.60–1.13).
Cost data
Looking at the cost comparisons between the two agents, the average cost per patient treated with abciximab based on a unit cost of each treatment (£250.24) is £750.72. The average cost per patient treated with eptifibatide based on a unit cost of each treatment (£42.79/13.61) is £99.19. No difference was seen in clinical outcomes or length of stay between the two agents resulting in a cost comparison based on the price of the agent. For the propensity-matched cohorts of 1441 patients, the total cost of abciximab was £1,081,787 compared to £142,933 for eptifibatide meaning an 87% cost saving of £938,854 made with the switch to eptifibatide over the study period.
Discussion
This is the largest observational study performed to date comparing the cost-effectiveness and use of eptifibatide as an alternative to abciximab in primary PCI for STEMI. The results demonstrated the usefulness and safety of these agents in primary PCI demonstrating similar effectiveness for both therapies with comparable procedural success rates, unadjusted and adjusted outcomes at 1 year and over the follow-up period. Importantly, the switch from abciximab to eptifibatide resulted in significant cost savings for the institution with an average of £650 per patient and a total saving of approximately £950,000. This provides important data to guide the choice of GPIIb/IIIa inhibitor to be used in current practice, especially giving increasing concerns over the absorption of newer drugs.
Abciximab is by far the best studied GP IIb/IIIa inhibitor in patients with primary PCI, with studies demonstrating reduced rates of death, MI and TVR compared to placebo,16,17 with its use in contemporary practice demonstrating negation of the effects of morphine on platelets. 6 Eptifibatide also has proven clinical benefit, although this agent has been mostly studied in patients with stable coronary artery disease undergoing PCI. 18 Until recently, there were few direct comparisons between abciximab and eptifibatide in the setting of primary PCI. In the Eptifibatide Versus Abciximab in Primary PCI for Acute Myocardial Infarction (EVA-AMI) trial, 427 patients who underwent primary PCI were randomized to receive eptifibatide or abciximab showed no statistically significant difference between the two agents with respect to the primary outcome of ST-segment resolution, with the study not powered to compare clinical end points. 10 Similar findings were found in a sub-group analysis of HORIZONS-AMI, three registries and in a meta-analysis comparing eptifibatide and abciximab.9,11,19
There are key pharmacological differences between abciximab and eptifibatide, which may result in differences in outcome. Abciximab is an irreversibly inhibiting monoclonal antibody Fab fragment, which has a short plasma half-life (30 min), but a disassociation half-time of 18–24 h meaning abciximab can result in increased bleeding due to persistent platelet inhibition several days after medication discontinuation. 20 However, eptifibatide, which provides reversible, competitive, dose-dependent inhibition of GP IIb/IIIa receptors with a short half-life of about 2.5 h, and normalization of platelet aggregation approximately 4 h after drug cessation.21,22 These factors suggest that eptifibatide may be associated with lower bleeding rates or at least less severe bleeding rates than abciximab due to this quicker resolution of platelet inhibition. This was supported by data in our study where a trend to lower bleeding rates were seen in the eptifibatide group. Furthermore, it is worth noting that although this lower bleeding rate in the eptifibatide group was not statistically significant, there was a significantly higher rate of radial access in the abciximab group and it is possible that if the rates of femoral access were similar between the two groups, this bleeding difference could be significant. These features make eptifibatide an ideal agent to use in conjunction with newer P2Y12 inhibitors whilst waiting levels to reach steady state.
Limitations of the current study
Our study is a consecutive but retrospective observational analysis from a single centre’s experience. As this was an observational study, the findings may have been subject to confounding factors that we have been unable to control for. This may include adherence to evidence based therapies, e.g. medication, which has been shown to be associated with outcome. However, our dataset includes all major clinical variables known to affect outcome, which would support the validity of our results.
Conclusion
This observational study suggests that eptifibatide is associated with similar clinical outcomes and is associated with significant cost savings compared to abciximab in patients with STEMI undergoing primary PCI. These agents should still be considered as adjunctive therapies for primary PCI especially if concerns exist over the absorption of oral anti-platelet drugs.
Footnotes
Acknowledgements
We thank the Barts Heart Centre Interventional Group at St. Bartholomew’s Hospital for recruiting patients. We also thank the nursing staff of the cardiac catheterisation laboratory, pharmacy staff and coronary care units at Barts Heart Centre.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The data were collected as part of a mandatory national cardiac audit and all patient identifiable fields were removed prior to analysis. The local ethics committee advised us that formal ethical approval was not required.
Guarantor
Dr. Daniel Jones (NIHR Academic Clinical Lecturer).
Contributorship
KSR and DAJ drafted the manuscript; SA, PA, NG and PW assisted in data collection; CK, AKJ, AM, EJS, RW and AW critically reviewed the manuscript and DAJ had the original concept for the study.
