Abstract
Recent advancements in intravascular imaging technologies have transformed our capacity to precisely identify and assess the culprit lesions responsible for acute coronary syndrome in clinical practice. Acute coronary syndrome commonly results from atherosclerotic plaque rupture, causing intracoronary thrombosis and acute myocardial ischemia, frequently presenting with chest discomfort and dyspnea. Intravascular imaging techniques, including near-infrared spectroscopy, optical coherence tomography, and intravascular ultrasound, provide more detailed insights into high-risk coronary lesions, overcoming the limitations of traditional coronary angiography. There is a growing clinical interest in the use of in vivo intravascular imaging to precisely characterize the morphology of atherosclerotic plaques, which are influenced by local hemodynamic flow. This article aims to offer a thorough examination of intravascular imaging technologies and physiological assessments, emphasizing their clinical utility in diagnosing and managing acute coronary syndrome, while also exploring their potential in shaping personalized treatment strategies to enhance patient outcomes.
Introduction
Although substantial advancements have been made in pharmacological and interventional treatments, coronary artery disease (CAD) continues to be the main cause of death throughout the world. 1 The most prevalent clinical symptom of CAD is acute coronary syndrome (ACS), which includes unstable angina, non-ST-elevation myocardial infarction (NSTEMI), and ST-elevation myocardial infarction (STEMI). CAD involves pathophysiological changes in the coronary arteries, including inflammation, lipid, calcium, and cellular waste buildup, and endothelial dysfunction. ACS’s primary underlying mechanisms include plaque erosion (30%-35%), plaque rupture (55%-60%), and calcified nodules (2%-7%). 2
While diagnostic coronary angiogram is commonly considered the benchmark technique, it has shortcomings in offering detailed information about bioactive processes and structural components of atherosclerotic plaques. 1 As a result, current advancements in intravascular imaging techniques have facilitated the detailed assessment of the lesions responsible for ACS. In order to visualize the histological features of plaque rupture, erosion, and calcified nodules in ACS patients, the development of intracoronary imaging technologies such as intravascular ultrasonography (IVUS), optical coherence tomography (OCT), and near-infrared spectroscopy (NIRS) has been essential.
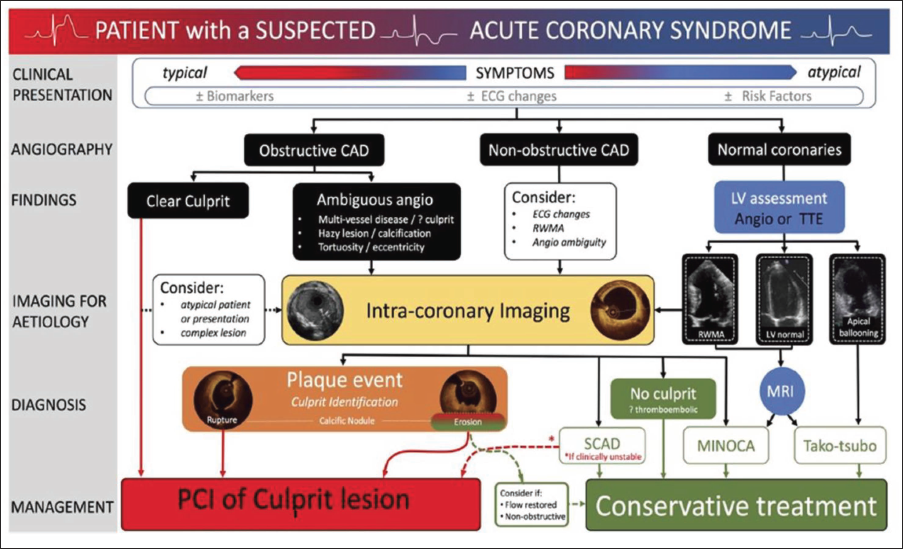
Percutaneous coronary intervention (PCI) in patients with ACS should be guided by intravascular imaging, especially OCT, as per the 2023 European Society of Cardiology (ESC) guidelines. This recommendation is Level IIa (Class A). When the culprit lesions are ambiguous, the use of intravascular imaging, such as OCT, is recommended, though with a lower Level IIb recommendation (Class C). 3 The 2024 ESC guidelines for chronic coronary syndrome emphasize the use of IVUS or OCT (Class IA) guidance during PCI for complex lesions, such as true bifurcations, left main stem, and long lesions. 4
Intravascular Imaging Techniques
Intravascular Ultrasound
IVUS is the most utilized technique for real-time, high-resolution imaging within coronary arteries, providing detailed cross-sectional images. IVUS provides cross-sectional views of deeper plaque components and a detailed evaluation of the vessel wall, including the adventitia and media. Additionally, IVUS aids in assessing vascular remodeling. IVUS can effectively penetrate plaques up to a depth of 10 mm; however, the presence of calcium can obstruct the ultrasound signal, limiting its visibility. 5 Challenges like acoustic shadowing in calcified plaques and difficulty identifying plaque components have driven the development of advanced techniques such as virtual histology intravascular ultrasonography (VH-IVUS) and OCT.
Lipid-rich plaques, which make up around two-thirds of all plaques, are difficult for IVUS to identify because they lack the spatial resolution needed for a thorough examination of plaque properties, such as determining the thickness of the fibrous cap. Moreover, IVUS is not a reliable tool for the detection of thrombus. Other limitations of IVUS include its lower resolution of the longitudinal view, making it difficult to assess strut malapposition. IVUS requires anticoagulation, longer procedural time, and expertise for accurate image interpretation. IVUS has limitations in penetrating calcium and evaluating thrombi, and it is relatively costly. Furthermore, it cannot assess stent-strut tissue coverage due to low resolution. Overall, thrombus detection and tissue characterization are challenging with IVUS.
Angioscopy
Coronary angioscopy utilizes high-resolution fiber optics (10-50 µm) to visualize arterial intima and plaque surface. Despite its ability to assess superficial characteristics (such as color), recognizing red and white thrombus, and spotting localized damage to the artery wall (such as ulceration, fissure, and flaps), angioscopy delivers constrained information regarding the structure of plaques. Moreover, angioscopy necessitates a blood-free field and is greatly reliant on the expertise of the operator, reducing its widespread clinical use. Consequently, the use of coronary angioscopy is largely confined to research settings. 1
Virtual Histology Intravascular Ultrasonography
Virtual histology uses IVUS radiofrequency backscatter signals to create color-coded images, each representing a distinct component of tissue. This enhances grayscale IVUS images, providing additional information for a more comprehensive analysis. VH-IVUS demonstrates high predictive accuracy when compared to histopathology samples. However, multiple studies using porcine models have raised concerns regarding their consistent reliability.
Optical Coherence Tomography
Optical coherence tomography uses near-infrared light from a rapidly rotating single fiber optic wire during withdrawal through the vessel to provide extremely high-resolution pictures. OCT holds a special place among imaging techniques because of its remarkable lateral and axial resolution, which are 10 and 70 µm, respectively. OCT’s main drawback is its limited penetration depth, which is considerably less in lipid-rich plaques (around 0.2 mm). This makes it more difficult to assess plaque burden and precisely gauge the lipid pools’ volume and depth. Additionally, a blood-free area is required for OCT imaging capture. Fourier domain OCT, a second generation of OCT technologies, enables high-speed coronary artery imaging without arterial obstruction during picture capture. It also enables the capture of images from long segments while preserving excellent longitudinal resolution during brief contrast injections. 6 OCT is the most precise technique for evaluating the complex morphology of the ACS causative lesions. Intracoronary contrast injections are one of the intricate procedures used in this imaging technique to eliminate red blood cells.
Optical coherence tomography requires blood blockage or removal in the detection vessel, complicating the procedure and limiting its use in patients with severe coronary ischemia. OCT’s limited penetration depth restricts its ability to assess the entire lesion. OCT struggles with measuring the diameter of larger blood vessels and assessing vascular remodeling. OCT faces challenges in clearly visualizing internal tissue that is obscured by thrombus, particularly red thrombus, within the blood vessel. OCT can clearly visualize the stent intima after implantation, but it cannot distinguish between the histological characteristics of the stent surface that covers the intima. 7
Near-infrared Spectroscopy
By keeping an eye on how infrared light at various wavelengths and intensities is absorbed and scattered, NIRS employs spectroscopic analysis to identify the components of lipid and plaque. NIRS has shown >90% accuracy and specificity in identifying lipid components in human studies. Diagnostic criteria for plaque rupture, erosion, and calcified nodules are displayed in Table 1, and a comparison of IVUS and OCT is shown in Table 2.
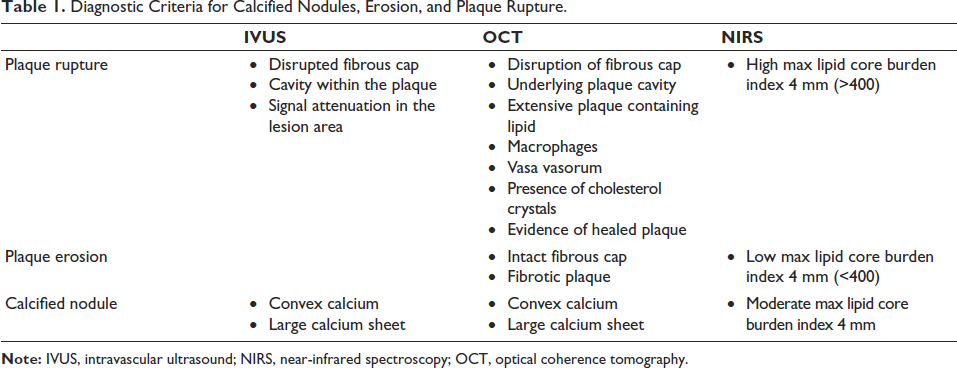
Diagnostic Criteria for Calcified Nodules, Erosion, and Plaque Rupture.
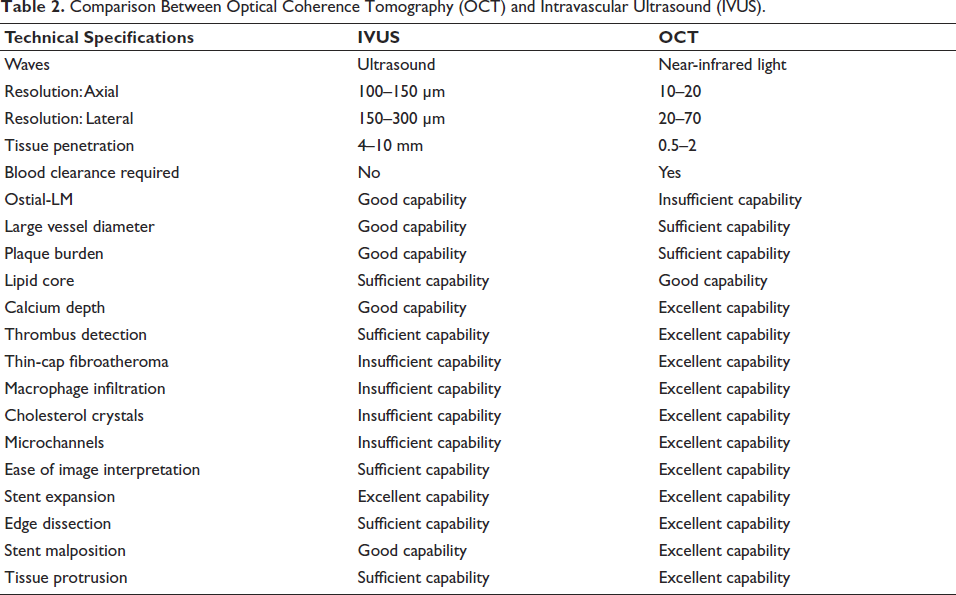
Comparison Between Optical Coherence Tomography (OCT) and Intravascular Ultrasound (IVUS).
NIRS–IVUS’s invasiveness is a key limitation, restricting its application of primary prevention in patients with preclinical coronary disease who do not exhibit any symptoms. NIRS has drawbacks as well, such as its inability to provide comprehensive details regarding the vascular lumen, plaque structure, and fibrous cap properties.
Artificial Intelligence-based Intravascular Imaging Tools
Recent advancements in artificial intelligence (AI)-driven tools have been incorporated into modern clinical intravascular OCT techniques to enhance and improve human interaction, analysis, and making decisions. 8 Nowadays, intravascular OCT programs like Aptivue, Lunawave, and Ultreon can show the recrossing point in three dimensions by using “stent enhancement” and allow for real-time co-registration with intravascular OCT pictures that have been reconstructed. Imaging-guided recrossing reduces carinal and side branch strut burden while enhancing vessel geometry. Ultreon, the latest intravascular OCT software, precisely outlines calcific arcs and measures calcium deposit thickness throughout the target vessel. However, it is important to note that the accuracy of calcium assessment relies on adequate blood clearance, which is facilitated by the use of contrast media. 8 The only AI-certified 2.0 version of cardiovascular OCT software is Abbott’s Ultreon™ 2.0.
Intracoronary Evaluation in Coronary Artery Disease Diagnosis and Treatment
Identifying Plaques
Intracoronary imaging helps identify lesions accurately by providing a thorough image of the arterial lumen, plaque disruption, and associated thrombus in patients who present with characteristic acute chest pain and ST-segment elevation on electrocardiography (ECG). Coronary atherosclerosis, the primary cause of ACS, is characterized by the loss of the artery wall’s three-layered architecture as observed by intracoronary imaging. Figures 1 and 2 show lipid plaque on IVUS, OCT, and NIRS. OCT, owing to its high resolution, allows detailed visualization and distinction of atherosclerotic plaque components, such as lipid cores (appearing as low-signal- regions with indistinct borders), thin fibrous caps (seen as uniform, high-intensity signal layers), and calcifications (characterized by sharply defined, signal-poor zones). Lipid content can significantly attenuate the light signal, making it challenging to clearly visualize the posterior margin of the core of lipid accumulation and underlying tissue structures. An intravascular imaging-guided algorithm for managing ACS patients is depicted in Figure 3.
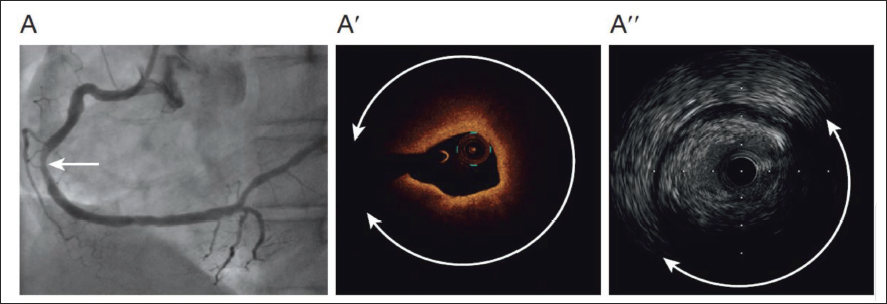
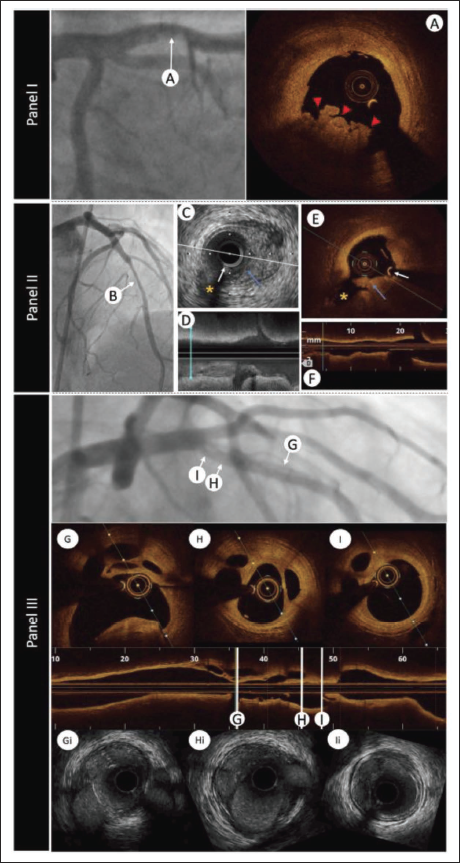
Although stationary imaging at the level of the suspected thrombus plus a little contrast injection to improve the luminal shape can help, IVUS detection of thrombus is more challenging (Figure 4). High-quality IVUS technology enhances diagnostic skills and precision by offering greater resolution. Plaque erosion, plaque rupture, and calcified nodules are the most frequent underlying causes of coronary thrombosis that result in ACS. 2
Plaque Rupture
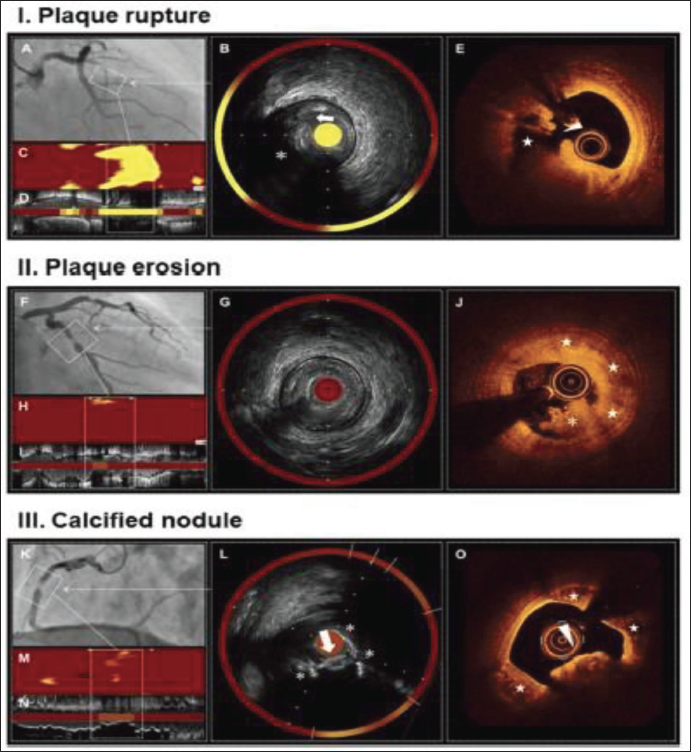
Plaque rupture is the predominant substrate in ACS, found in 55%-80% of cases.2, 9 The hallmark of plaque rupture is the breakdown of the thin fibrous cap, leading to the formation of a separate cavity in the plaque. The development of a thrombus, mainly composed of red blood cells, is facilitated by these factors and results in a notable reduction in the lumen area. Vasoconstriction and thrombosis may cause myocardial ischemia in this situation by abruptly stopping coronary blood flow. The clinical approach usually involves stent placement for ruptured plaques. Plaque rupture, with a higher lipid content, is linked to a worse prognosis than other plaque types. 9 Plaque rupture is typically easy to identify in OCT imaging due to several distinctive features. These features comprise macrophages, vasa vasorum, cholesterol crystals, and healed plaques. In contrast, IVUS detects plaque rupture as an ulcer-like indentation beginning at the junction between the lumen and the intimal layer. Plaque rupture lesions look hypoechoic on IVUS because of their significant plaque burden and positive vascular remodeling. Often referred to as an “attenuated plaque,” this type of plaque creates an auditory shadow that comes from the lipid core. 10
Plaque Erosion
Plaque erosion is the second most common lesion type, accounting for 30%-35% of ACS cases.2, 9 Plaque erosion is characterized by a preserved, thick fibrous cap and the presence of a localized, platelet-rich white thrombus, without surface lipid accumulation or calcification. OCT, with its superior resolution, is the only imaging tool capable of detecting plaque erosion, while IVUS (100-200 µm resolution) cannot identify minor ruptures. OCT-derived criteria for plaque erosion are different from conventional pathological definitions, hence the following characteristics are used to diagnose plaque erosion using OCT: (a) “Definite” plaque erosion is defined by disruption of the fibrous covering within a lesion predominantly made up of tissue that is fibrous, accompanied by a white thrombus on the luminal surface; and (b) “Possible” plaque erosion is indicated by an irregular luminal contour without a visible thrombus, or by a thrombus that obscures the underlying plaque, either upstream or downstream from the thrombus location without the presence of surface lipid or calcification. In order to overcome the shortcomings of IVUS alone in detecting plaque erosion, NIRS is currently combined with IVUS in a single catheter. 2
According to recent results from the EROSION III research, which contrasted PCI guided by OCT with angiography, the OCT group had 15% fewer stents inserted than the angiography group, and their prognoses were similar. This study showed that a non-stenting approach is safe and highlighted the crucial role of OCT in refining PCI strategies for patients with ACS. 11
Calcified Nodule
Calcified nodules, while relatively rare in causing coronary thrombosis, generally occur in areas with significant calcification, making stent deployment and optimization more challenging. Calcified nodules affect 2%-8% of patients with ACS; they are more common in older men, among diabetes, tortuous coronary arteries, and people with chronic renal failure.2, 9 These lesions demonstrate a rupture in the calcified plaque, resulting in the disruption of the fibrous cap, which is then covered by thrombus. Erupted calcified nodules can be identified using either IVUS or OCT. OCT can accurately identify the calcification’s angle and boundary; at sharp margins, thin calcification appears as signal-poor areas. OCT categorizes calcification into three types: eruptive calcified nodules, superficial calcific sheets, and calcified protrusions.2, 9 According to clinical OCT, an eruptive calcified nodule is a lesion with fibrous cap discontinuity and/or thrombus that is located above a calcified plaque that has significant calcium both proximally and distally, as well as projecting calcification into the lumen. Multimodal imaging of calcified nodules, plaque erosion, and plaque rupture is shown in Figure 5.
Non-obstructive Myocardial Infarction
Another possible cause of ACS is non-obstructive myocardial infarction (MINOCA), which usually happens when coronary stenosis is <50%. Plaque erosion, rupture, coronary artery spasm, microvascular spasm, spontaneous coronary artery dissection, and microcirculatory dysfunction are the main causes of MINOCA. 12 OCT and other tools for intracoronary imaging are crucial for detecting thrombosis in the absence of prominent atherosclerotic plaques. These techniques help detect thromboembolism, vasospasm, and other non-atherosclerotic lesions. Intracoronary imaging plays a crucial role in diagnosing patients with atypical ACS presentations, enabling accurate identification of the underlying cause. 13 Antiplatelet and anticoagulant medications are used less frequently when OCT is used to differentiate between culprit and non-culprit lesions.
Precise Identification of Vulnerable Non-culprit Lesions
In ACS patients, the recurrence of adverse events following surgery is frequently linked to vulnerable plaques. The rapid growth of plaques with a big lipid core, thin fibrous top, and many macrophages can have serious short- and long-term consequences, including cardiac mortality. The common characteristics of susceptible plaques as determined by intracoronary imaging are shown in Figure 6. In patients with ACS, areas of the epicardial coronary arteries with severe stenosis are frequently home to vulnerable plaques. OCT studies in patients with ACS demonstrate that plaques with a fibrous cap thickness <65 µm and a lipid angle >180° are more likely to be severe, with STEMI patients frequently exhibiting more severe lesions in thin fibrous cap lesions. 14
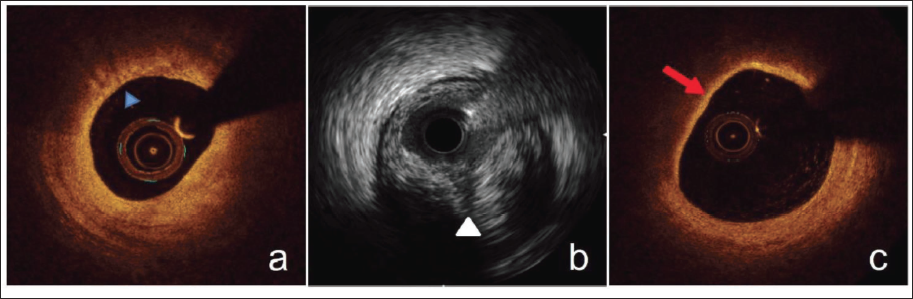
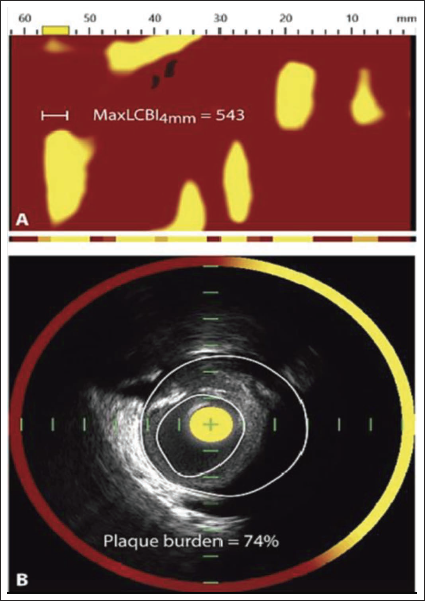
Over the past decade, IVUS has been widely used for in vivo studies of vulnerable plaques. The development of high-resolution 60-MHz IVUS has enhanced plaque detection sensitivity, although IVUS still has a lower lateral resolution than OCT. When assessing susceptible plaques with NIRS, the lipid core burden index (LCBI), and in particular Max LCBI4mm, is a crucial parameter. A 100-unit increase in LCBI was linked to a 19% increase in major adverse cardiac events (MACE) in a study of 117 ACS patients using both NIRS and IVUS, highlighting the important prognostic role of lipid burden in bad cardiac outcomes. 15
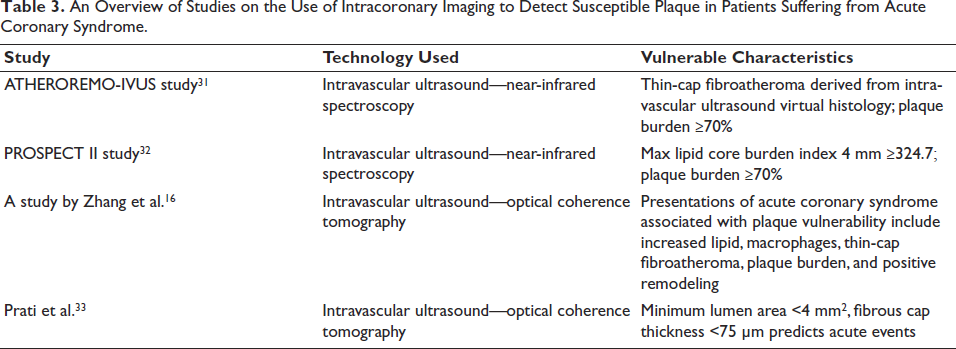
Several studies have combined multiple intracoronary imaging tools, going beyond the use of single modalities. The findings of research using intracoronary imaging to identify susceptible plaques in ACS patients are shown in Table 2. 16 Early treatment of vulnerable plaques may lead to better clinical outcomes, according to the findings. Intracoronary imaging enables personalized treatment approaches, such as enhanced follow-up and intensified pharmacotherapy, for high-risk individuals with vulnerable plaques. However, several extensive tests in this area are still needed to verify this superiority. Numerous investigations used OCT and IVUS together to gain deeper insights into the pathology of ACS patients. 17 Table 3 provides a summary of studies that highlight the detection of vulnerable plaques in ACS patients using intracoronary imaging techniques.
An Overview of Studies on the Use of Intracoronary Imaging to Detect Susceptible Plaque in Patients Suffering from Acute Coronary Syndrome.
Stent Optimization Criteria for Optical Coherence Tomography
Optical coherence tomography-guided PCI has been linked to better results than standard angiography-guided PCI, according to several studies. However, the OCT adoption in real-world clinical practice remains limited. One contributing factor is the absence of standardized quantitative OCT criteria for stent optimization. Given that the minimum stent area (MSA) is influenced by vessel size, relative stent expansion may serve as a more practical predictor of optimal outcomes. A relative stent expansion greater than 70%, as demonstrated in the study by Kubo and Nakazawa, is considered an attainable target in everyday clinical practice. Other studies have adopted relative expansion thresholds of over 80% or even 90% as benchmarks for optimal stent deployment under OCT guidance. However, achieving these criteria has been difficult in clinical practice. 18
Stent Optimization Criteria for Intravascular Ultrasound
Operators with limited IVUS experience may face challenges with online analysis and stent optimization, leading to longer procedure durations. To date, there is no consensus or standardized method for IVUS-guided stent optimization, and the procedure is still carried out at the operator’s discretion. By optimizing the mid- and proximal stent segments and rounding the stent diameter by 0-0.25 mm, the expert consensus of the European Association of PCI recommends stent sizing based on the distal reference lumen diameter. The ULTIMATE trial defined stent under-expansion (MSA <90% of the distal reference lumen area or 5.4 mm2) and addressed it with post-dilatation using a noncompliant balloon, reduced by 0.25 mm to match the distal reference external elastic membrane diameter. Furthermore, stenting was used to treat edge dissection and a plaque burden of more than 50% at the stent edges. The ULTIMATE trial utilized these simple, standard steps for optimal stenting. Nevertheless, more research is needed to validate and standardize IVUS-guided stent optimization methods. 19
Intravascular Imaging Studies in Acute Coronary Syndrome Patients
Intravascular ultrasound-guided PCI significantly reduced the risk of in-hospital mortality in ACS patients (odds ratio 0.74, 95% CI 0.64-0.85, P < .01), according to a study by Shafi et al. Application of IVUS increased from 2.64% to 4.10% in 2016 to 2019, respectively (P < .001). 20 A recent trial by Li et al., which included ACS patients, found that IVUS-guided stent implantation resulted in a lower 1-year rate of cardiac death, target vessel myocardial infarction, and clinically driven revascularization compared to angiography guidance alone. 21
Additionally, the OCCUPI trial demonstrated that, in comparison to angiography guiding alone, OCT guidance dramatically decreased 1-year MACE rates. These results demonstrate OCT’s potential therapeutic benefit as an intravascular imaging technique for directing PCI in ACS patients. When Abdelmonaem et al. evaluated the procedural and short-term results of PCI in ACS patients guided by IVUS and OCT, they found no discernible variations in important parameters. 17 In contrast use, edge dissection, tissue prolapse, and no-reflow occurrences, there were no appreciable changes between the IVUS and OCT groups. But at 6 months, the MACE rate was much higher in the IVUS-guided group. 17
Coronary Physiology’s Function in Acute Coronary Syndrome Patients
Fractional Flow Reserve
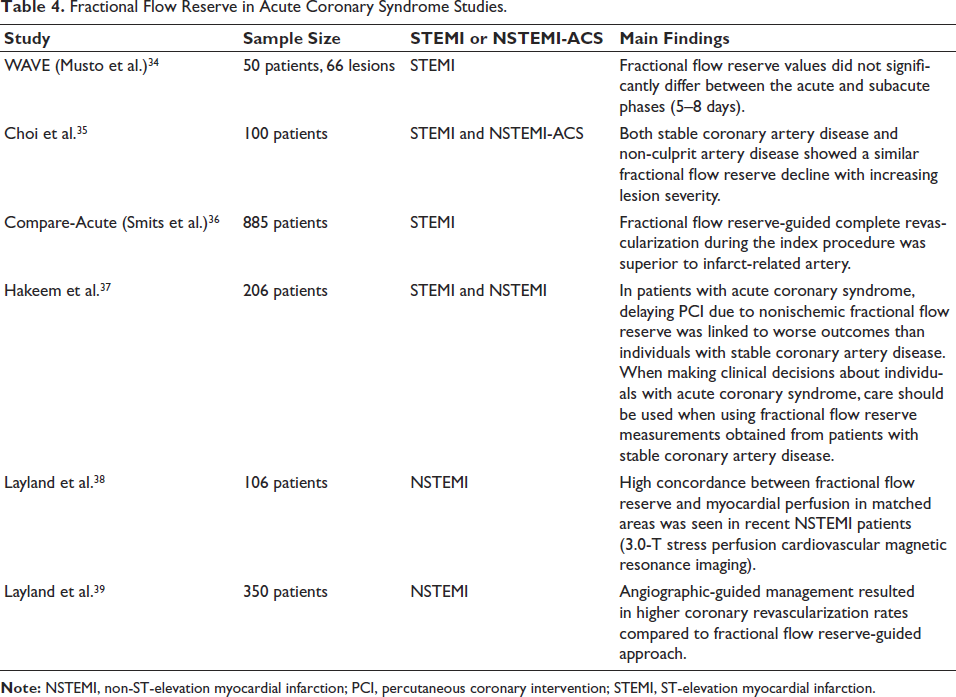
Fractional flow reserve (FFR) is frequently used to direct PCI in patients with diffuse coronary lesions or simple and complex multivessel lesions. The ratio of the mean distal to proximal coronary pressure (Pd/Pa) under the greatest myocardial hyperemia brought on by intracoronary adenosine is known as the FFR. A positive lesion is suggested by an FFR <0.75, whereas a negative lesion is generally indicated by an FFR >0.8. The gray zone, where interpretation may be less certain, is defined as values between 0.75 and 0.8. 9 A summary of research that has assessed the utility of FFR in ACS patients is included in Table 4.
Fractional Flow Reserve in Acute Coronary Syndrome Studies.
Instantaneous Wave-free Ratio
The mean Pd/Pa ratio during the mid-diastolic wave-free period, which lasts from 25% of diastole to 5 ms prior to systole, is used to compute the instantaneous wave-free ratio (iFR). This ensures stable conditions for pressure measurement, with coronary microvascular resistance being minimal and consistent during this period. Based on various clinical trials, both American and European guidelines for chronic CAD endorse the utilization of iFR to assess intermediate coronary stenoses, with a recommendation for revascularization if iFR is ≤0.89. It is interesting to note that iFR has been demonstrated in some situations to more accurately represent epicardial blood flow than FFR, with multiple studies indicating its superiority. Furthermore, iFR does not require adenosine. 22
Resting Full-cycle Ratio
Resting full-cycle ratio (RFR) is a novel non-hyperemic pressure ratio (NHPR) that measures the lowest Pd/Pa value observed throughout the entire cardiac cycle. RFR has demonstrated high diagnostic accuracy, exhibiting strong concordance with both iFR and FFR measurements. The VALIDATE RFR study indicates that RFR is diagnostically equivalent to iFR and has the benefit of identifying the lowest Pd/Pa throughout the cardiac cycle, which could reveal substantial stenosis that phase-specific assessments miss. 23
Quantitative Flow Ratio
An angiographic technique called quantitative flow ratio (QFR) calculates FFR without the need for guidewire insertion or artificially generated hyperemia. It is ascertained by applying computational fluid dynamics methods to quantitative coronary angiography 3D reconstructions. Research shows that QFR has good diagnostic accuracy for identifying myocardial ischemia and has a strong correlation with FFR, especially when FFR is used as the gold standard. QFR-guided PCI outperformed angiography-guided PCI in the FAVOR III China study, significantly reducing the incidence of myocardial infarction and revascularization. These results are consistent with the FAME (Fractional Flow Reserve versus Angiography for Multivessel Evaluation) study and provide more evidence that, regardless of the physiological assessment technique employed, physiology-guided PCI produces better results than angiography-guided PCI. 24 A comparison among QFR, FFR, and iFR is illustrated in Table 5.
Comparison Between Quantitative Flow Ratio (QFR), Fractional Flow Reserve (FFR), and Instantaneous Wave-free Ratio (iFR).

Utilization of Coronary Physiological Tools
An increasing number of ACS patients are being evaluated for the hemodynamic significance of intermediate-severity non-infarct-related artery (IRA) stenosis using intracoronary physiology. The IRA may experience varying degrees of microvascular obstruction in patients with ACS. Small observational studies have utilized intracoronary physiology to evaluate the success of myocardial reperfusion through the measurement of microcirculatory resistance.3, 25 Figure 7 illustrates the application of coronary physiology in different clinical scenarios for ACS.
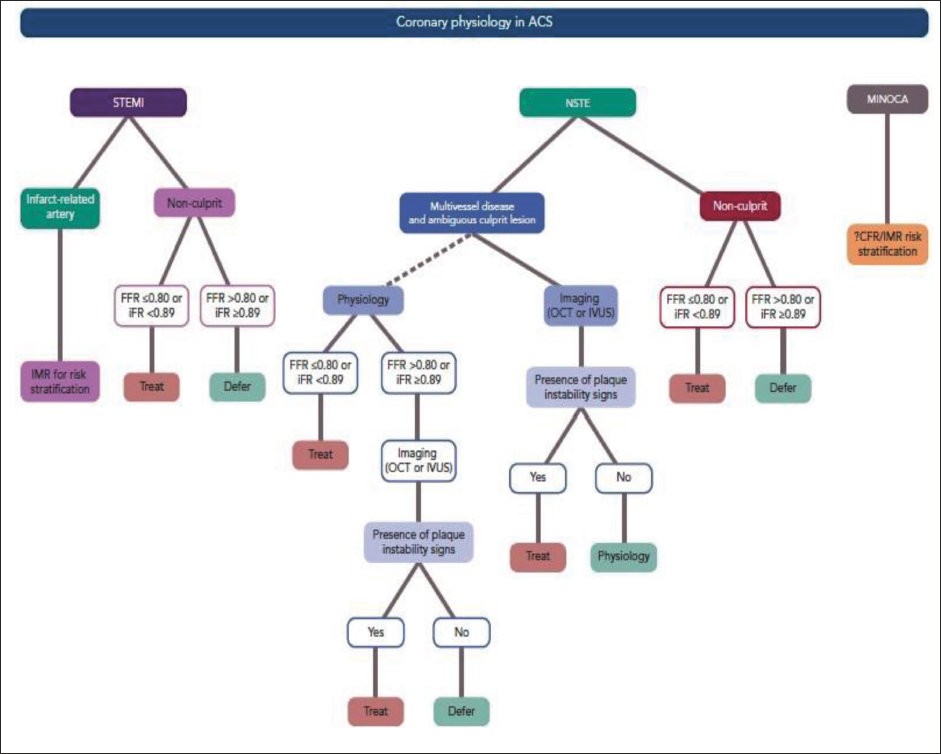
Fractional flow reserve’s usage in clinical settings is restricted by its invasive and time-consuming nature. The accuracy of the results can also be impacted by microcirculatory variables and adenosine intolerance in certain patients. As a result, new methods that employ NHPRs are being researched as possible substitutes for FFR. These include iFR, QFR (contrast-enhanced FFR), OFR (OCT-based FFR), UFR (IVUS-based FFR), and CT-FFR (coronary angiography-derived flow reserve). Compared to FFR, most of the emerging techniques demonstrate excellent accuracy and reproducibility. 26 Among these technologies, CT-FFR, although noninvasive and more informative than computed tomography angiography, is less accurate than FFR in patients with ACS. 26
Pitfalls of Physiology in Acute Coronary Syndrome
Physiologic instability in ACS can compromise FFR accuracy by altering stenosis geometry, microcirculatory conditions, and adenosine responsiveness. NHPRs may be affected by increased adrenergic activity in the ACS setting. However, these effects are short-lived and vary in magnitude based on the patient’s hemodynamic instability.
Recent evidence suggests that in STEMI patients, FFR and NHPR measurements in non-culprit vessels may vary as the condition evolves from the acute to the subacute phase. Due to significant dynamic changes in the coronary microcirculation distal to the infarct-related lesion, FFR and other invasive physiological assessments may be unreliable for guiding PCI of the culprit lesion during the acute phase. Consequently, after the acute and subacute stages, FFR can be used to assess the functional relevance of the offending lesion. 27 FFR estimates may be off in the early stages of ACS because the maximal hyperemic response may be dampened by the underestimating of lesion severity brought on by malfunction in the infarct-related coronary bed. Aortic stenosis can also affect FFR estimation by diminishing the effect of adenosine. Finally, by competitively inhibiting its receptors, tolerance to vasodilators, such as stimulants like caffeine, might counteract the pharmacologic activity of adenosine and perhaps lead to false-negative readings. 28
Enhancing Risk Stratification Through the Integration of Multimodal Diagnostic Approaches
Relying solely on intracoronary imaging or coronary physiological tools to guide revascularization in ACS patients is insufficient. At present, multiple studies are underway that combine plaque vulnerability characteristics with coronary physiology to enhance prognostic risk stratification in patients. 9 PCI on angiographically mild lesions was shown to be safe and to significantly enhance minimum lumen area (MLA) during follow-up in the PROSPECT ABSORB research, which included 182 ACS patients with a 25-month follow-up. 29 According to the COMBINE trial, which followed up with 547 patients with diabetes mellitus and either stable angina or ACS for 18 months, 25% of them had at least one FFR-negative lesion with thin-cap fibroatheroma (TCFA) positivity; the incidence of MACE was five times greater in this subgroup. 30
Future Perspectives
Ensuring adequate education in imaging interpretation remains a key challenge for the broader clinical adoption of intravascular imaging. Recent initiatives have focused on enhancing data fusion methods and developing hybrid dual-probe catheter technologies. By combining the deep tissue penetration of IVUS with the high-resolution surface viewing of OCT, physicians can create fused pictures that provide a more comprehensive assessment of coronary disease. This integration allows for a detailed and comprehensive visualization of atherosclerotic coronary plaque. By combining OCT with NIRS, lipid‑rich cores can be spectroscopically identified while OCT simultaneously provides high‑resolution structural assessment such as fibrous cap thickness. Such an integrated strategy improves the detection of thin‑capped fibroatheromas, well‑established precursors to plaque rupture. Continued technological advancements such as improved spatial resolution, accelerated image acquisition, integrated IVUS or OCT catheters, and analysis of advanced co-registered images are expected to enhance usability and interpretation, promoting wider clinical adoption. While intracoronary imaging-guided PCI holds tremendous promise, randomized trials remain essential to validate its benefits and support widespread clinical implementation.
Summary
While coronary physiology evaluations have expanded our understanding of lesion features, atherosclerosis development, and the underlying processes of ACS, intracoronary imaging has transformed the evaluation and treatment of ACS over the last 10 years. The “one-size-fits-all” approach to modern coronary intervention has given way to a precision-guided plan that is tailored to the patient’s features and the problem. Integrating multiple imaging modalities delivers a comprehensive characterization of atherosclerotic plaque morphology and composition. When combined with intracoronary imaging, the integrated technique presents a possible avenue for individualized ACS treatment in the future.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
