Abstract
This study investigated the effects of altitude on occurrence of mid-term negative events among patients presenting with ST elevation myocardial infarction (STEMI). This study enrolled 492 patients with STEMI. 119 of those patients were living at an intermediate altitude (1960 m, Group I) and 373 were living at sea level (0 m, Group II). There was no significant difference between the different altitude groups in terms of the incidence of cardiac death, urgent target vessel revascularization (TVR), stroke/transient ischemic attack (TIA) and composite endpoints. However, Group I had a significantly higher reinfarction incidence when compared with the Group II. Independent predictors of 6-month composite endpoints were history of statin use, presentation with acute stent thrombosis, peak CK-MB level, success of procedure, Killip classification, and left ventricular ejection fraction. In conclusion, altitude status and altitude-related hematologic changes had no influence over the mid-term outcomes in STEMI patients treated with percutaneous intervention.
Introduction
High altitude is reported to be associated with high incidence of cardiovascular event. 1 The hypoxic environment of the high altitude primarily affects the respiratory and hematologic systems in the human adaptive system. One of the most desired hematologic adaptations while living at high altitude is the elevated red blood cell count and hemoglobin concentration, leading to increased capacity for oxygen transportation to body tissues. 2 Previous studies have shown that coagulability increases in higher altitudes. 3 –5 The resulting coagulation is attributed to the elevated factor levels and change in hematologic indices triggered by hypoxia. 3
Hypercoagulability occurs rapidly after exposure to high altitude and peaks at the end of the first week. However, this rising trend slowly regresses because of transient reduction in clotting factors and platelet dysfunction at the end of the second week. In long-term exposure to high altitude, hypercoagulability continues at peak levels beyond 5 months as a result of the elevated plasma fibrin activity. 6 Although the incidence of cardiovascular (CV) events increases with the rising altitude, there is no data on the prognosis of occurring events. This study investigated the impact of altitude and altitude-related changes in hematologic indices on the development of midterm major adverse cardiac events in patients with ST elevation myocardial infarction (STEMI).
Methods
Patient Selection
This study included all the patients with STEMI that presented to the 2 emergency departments located in different geographic regions and treated by primary percutaneous coronary intervention (PCI) between September 2010 and December 2010. The study inclusion criteria were as follows: detection of STEMI by electrocardiography (ECG), defined as continuous typical chest pain persistent for >30 minutes, and ST-segment elevation ≥2 mm in 2 contiguous ECG leads within 12 hours of symptom onset or up to 18 hours, if there was an evidence of continuing ischemia or hemodynamic instability. Patients who had end-stage renal disease (ESRD) or malignancy as well as history of blood transfusion, menorrhagia, or hematologic disease were excluded from the study. In total, 169 patients were excluded from the study and the underlying reasons were a change of altitude for >1 week within the past 1 year in 54 patients, receiving anticoagulant therapy before or during the transfer in 17 patients, a history of previous myocardial infarction (MI), PCI, coronary artery bypass grafting (CABG), or heart failure (HF) in 44 patients, detection of normal coronary flow in 13 patients, urgent CABG decision in 17 patients, missing or unavailable hematologic parameters in 13 patients, the absence of a follow-up in 6 patients, and other (eg, renal failure) reasons in 5 patients. Thus, the final study population consisted of 492 patients.
There were 119 patients (group I) living at intermediate altitude (1960 m) and 373 patients (group II) living at sea level (0 m). None of the study patients had left their geographic region for more than 1 week within the last year. The study protocol was approved by the local ethics committee.
Data Analysis
Blood samples were collected from all the patients in the emergency department and sent to a laboratory for analysis within 1 hour after collection. Hematologic indices were evaluated from a complete blood count (CBC) analysis obtained by a Coulter LH 780 Hematology Analyzer (Beckman Coulter Ireland Inc, Mervue, Galway, Ireland).
Clinical history of risk factors such as age, sex, diabetes mellitus (DM), hypertension (HTN), hypercholesterolemia, smoking, and a family history of CV disease were noted.
Each patient received echocardiography within the first six hours following the procedure. A transthoracic echocardiography was performed using a system V (Vingmed, GE, Horten, Norway) device with a 2.5 MHz phased-array transducer. The left ventricular ejection fraction (LVEF) was measured using the modified Simpson rule. 7
Coronary Angiography, Primary Angioplasty, and Stenting
All patients received chewable acetylsalicylic acid (300 mg, unless contraindicated) and oral clopidogrel (600 mg loading dose) before the coronary angiography. The infarct-related artery (IRA) was graded according to the Thrombolysis in Myocardial Infarction classification (TIMI). 8 Heparin (10 000 U) was administered after the determination of the coronary anatomy. Primary coronary interventions, including balloon angioplasty and/or stent implantation, were performed only on the IRA. After angioplasty, all patients were admitted to the coronary care unit, where 500 U/h of intravenous heparin or 1 mg/kg per d of subcutaneous low-molecular-weight heparin was administered. All patients received a combination antiplatelet regimen of asetil salisilic asit (100 mg) and clopidogrel (75 mg). The use of glycoprotein 2b/3a inhibitors was left to the discretion of the interventional cardiologist. Concomitant medical treatment with β-blockers, angiotensin-converting enzyme inhibitors, and statins were started according to the guidelines of the American College of Cardiology/American Heart Association.
Definitions
Reperfusion time was defined as the time from onset of chest pain to the first balloon inflation. Door-to-balloon time was defined as the interval between hospital admission and balloon inflation. Patients were considered as having ESRD if they were dependent on chronic dialysis. Patients were also evaluated according to the Killip classification. 9 Acute, subacute, and late stent thrombosis were defined according to the Academic Research Consortium. 10 Optimal angiographic success was described as final TIMI grade 3 flow and a stenosis <20% in the IRA at the end of the procedure. 11 Multivessel disease was defined as the presence of a 50% stenosis in at least 2 major epicardial coronary arteries or the left main coronary artery.
Clinical End Points
All of the patients were prospectively followed for 6 months, and the month 6 assessments were conducted either on-site or via telephone. The main end points evaluated in this study were the composite end point of CV mortality, reinfarction (re-MI), urgent target-vessel revascularization (TVR), and stroke/transient ischemic attack (TIA). Cardiovascular mortality was defined as unexplained sudden death and death as a result of acute MI, HF, or arrhythmia. Re-MI was defined as an increase in creatine kinase (CK) that is twice the last value associated with CK-MB ≥10% of the total CK or ST-segment re-elevations. Urgent TVR was defined as the requirement of PCI or coronary surgery because of the thrombosis of the IRA. Stroke/TIA was defined as an acute loss of neurological function due to an abnormal perfusion of brain tissue.
Statistical Analysis
Continuous variables were expressed as mean ± standard deviation (SD) values. Categorical variables are expressed as percentages. Continuous variables were compared with the Independent Student t test and the Mann-Whitney U test, whereas categorical variables were compared with the chi-square test. The incidence of end points over time was analyzed with the Kaplan-Meier method, whereas differences between the altitude groups were evaluated with log-rank tests. Cox regression models were applied to assess the independent predictors of clinical end points. Significant variables were selected in the univariate analysis (P < .05) in order to be used in the multivariate model. The results were presented as adjusted hazard ratios (HRs) with 95% confidence interval (CI). The 2-tailed P values <.05 were considered to indicate statistical significance. All statistical studies were carried out with the SPSS program (version 15.0, SPSS, Chicago, Illinois)
Results
In total, 492 patients were enrolled in the study (56.7 ± 12.4, male: 85.4%). Group I had 119 patients (58.3 ± 12.6, male: 85.7%) and group II had 373 patients (56.2 ± 12.3, male: 85.3%). Group I demonstrated longer reperfusion time (361.8 ± 394.5 vs 172.3 ± 146.4, P < .001) and higher peak CK-MB (234.1 ± 175.1 vs 171.3 ± 122.8, P < .001) values when compared with group II. The other baseline clinical and procedural characteristics as well as the medication history are shown in Table 1 .
Baseline Clinical and Angiographic Characteristics.a
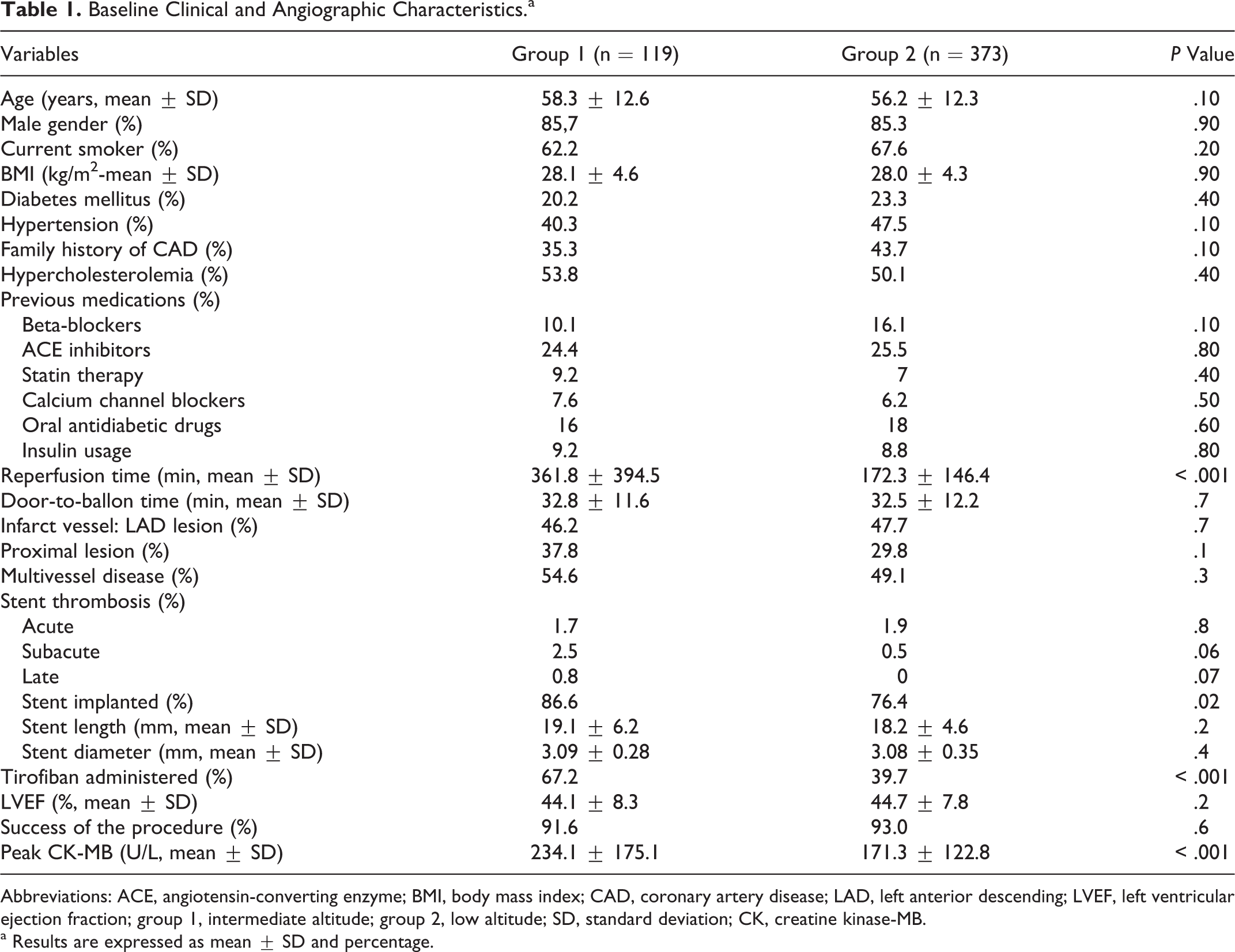
Abbreviations: ACE, angiotensin-converting enzyme; BMI, body mass index; CAD, coronary artery disease; LAD, left anterior descending; LVEF, left ventricular ejection fraction; group 1, intermediate altitude; group 2, low altitude; SD, standard deviation; CK, creatine kinase-MB.
a Results are expressed as mean ± SD and percentage.
Group I showed higher hemoglobin ([Hb] 15.2 + 1.8 vs 14.2 + 1.5, P < .001), hematocrit ([Htc] 44.4 + 5.7 vs 40.6 + 4.6; P < .001), and platelet distribution width (PDW; 16.8 + 0.7 vs 16.6 + 0.8, P = .02). Group 2 demonstrated higher mean platelet volume ([MPV] 8.4 + 1.0 vs 8.0 + 1.0; P < .001), mean corpuscular hemoglobin ([MCH] 31.5 + 2.0 vs 30.8 + 2.3, P < .001), and MCH concentration ([MCHC] 35.0 + 1.0 vs 34.3 + 2.0, P < .001) values. There was no difference between the groups in regard to other hematologic indices (Table 2 ).
Baseline Hematologic Indices.a
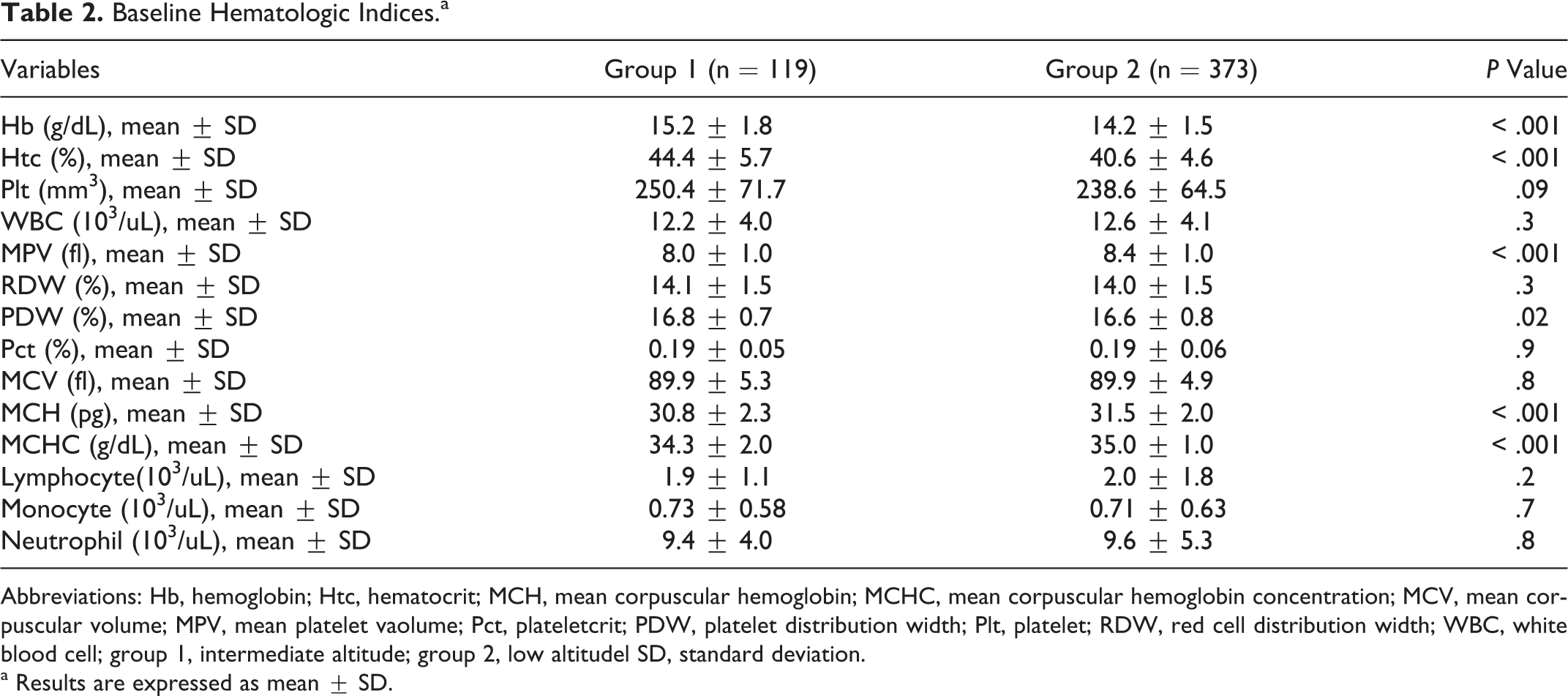
Abbreviations: Hb, hemoglobin; Htc, hematocrit; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MPV, mean platelet vaolume; Pct, plateletcrit; PDW, platelet distribution width; Plt, platelet; RDW, red cell distribution width; WBC, white blood cell; group 1, intermediate altitude; group 2, low altitudel SD, standard deviation.
a Results are expressed as mean ± SD.
At month 6 (postprocedure), the incidence for composite end point of CV mortality, re-MI, urgent TVR, and stroke/TIA was 18.7% for the overall population (Table 3 ). There was no significant difference between group I and group II in regard to the incidence of cardiac death, urgent TVR, stroke/TIA, and composite end points. However, Group I had a significantly higher re-MI incidence than group II (Figure 1 A-E). Cox regression analysis showed that the independent predictors of 6-month composite end points were history of statin use, presentation with acute stent thrombosis, peak CK-MB level, success of procedure, Killip classification, and LVEF (Table 4 ). Altitude was not observed to be an independent predictor of the composite endpoints of the cohorts.
The Incidence of Composite End Point of Cardiovascular Mortality, Reinfarction, Urgent Target-Vessel Revascularization and Stroke/TIA Between Altitude Cohorts.a
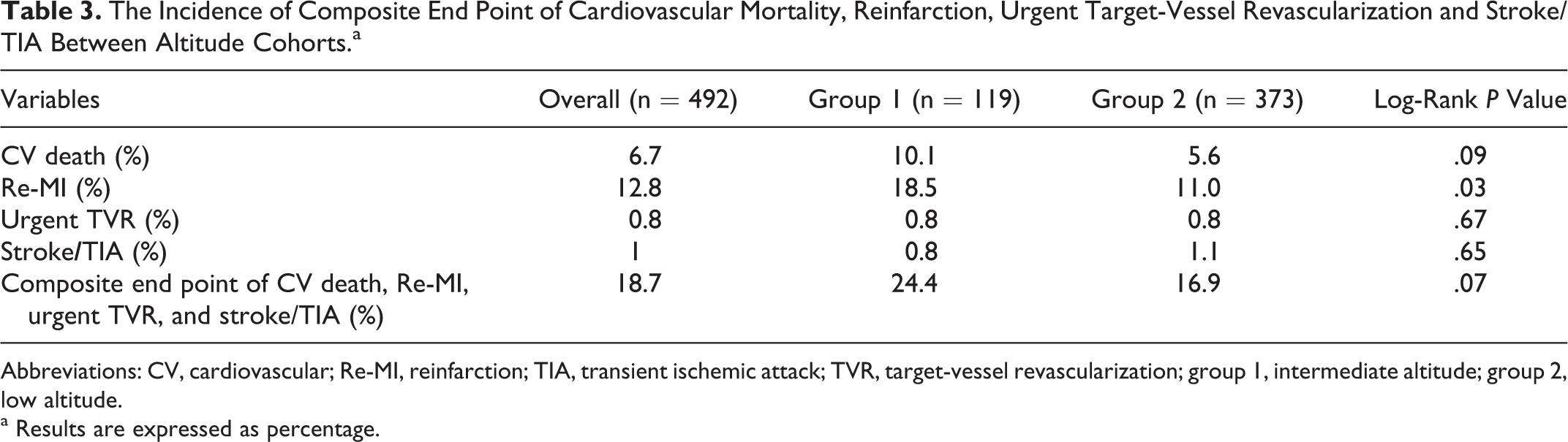
Abbreviations: CV, cardiovascular; Re-MI, reinfarction; TIA, transient ischemic attack; TVR, target-vessel revascularization; group 1, intermediate altitude; group 2, low altitude.
a Results are expressed as percentage.
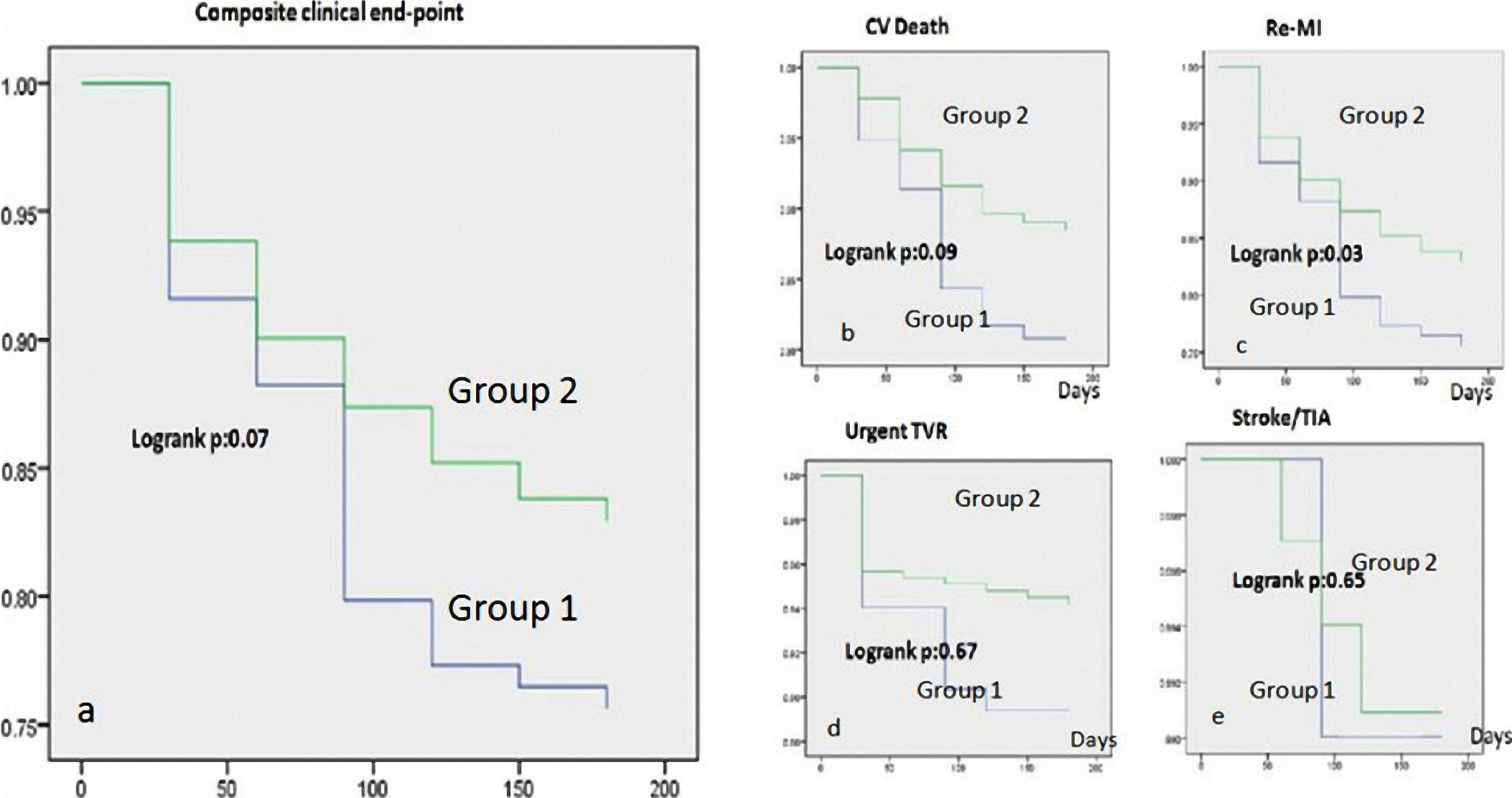
Kaplan-Meier of low and intermediate altitude cohorts. For composite end points (A), cardiovascular death (B), reinfarction (C), urgent target vessel revascularization (D) and stroke/TIA (E). TIA indicates transient ischemic attack; group 1, intermediate altitude; Group 2, low altitude.
Cox Regression Analysis to Predict Midterm Predictors of Composite End Points.
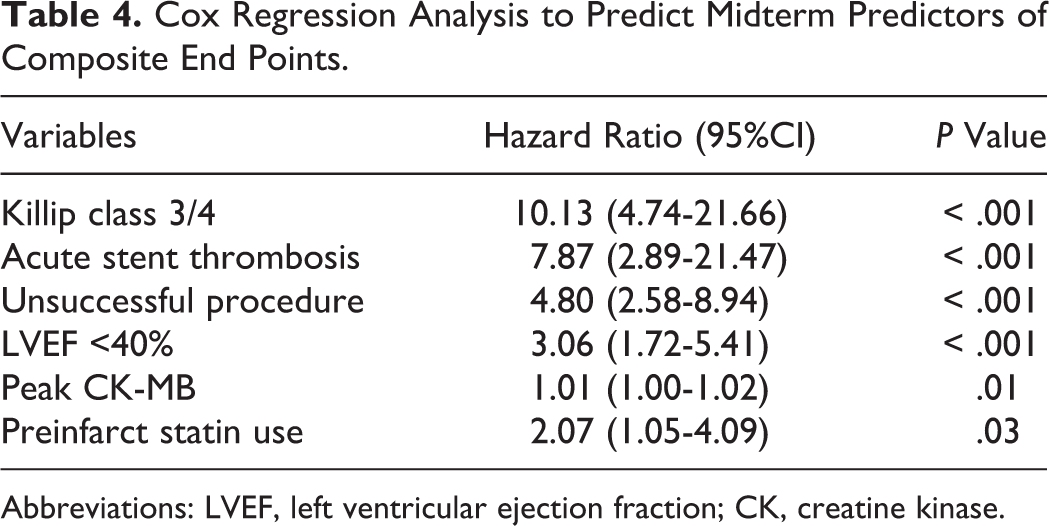
Abbreviations: LVEF, left ventricular ejection fraction; CK, creatine kinase.
Discussion
In this study, altitude was found to have an influence over some hematologic parameters, but neither altitude nor altitude-related hematologic indices had an influence over the midterm outcomes of STEMI patients treated with primary PCI.
Many of the previous studies have shown that altitude generates a thrombogenic environment and that CV events are more common at high altitudes. 1,3 –5 However, since this study was not a prospective incidence-prevalence study, it cannot point out the influence of altitude over STEMI frequency. To our knowledge, the current review bears importance by being the first study in the literature, which provides some insights into the relationship between altitude and the prognosis of STEMI patients. In this regard, all the patients presenting to the selected 2 study centers between certain dates were enrolled in order to make the study more reliable and reduce the bias.
There are discussions about altitude and the accompanying changes in hematologic indices. The Hb concentration that is adequate for transportation of oxygen at low amplitudes becomes inadequate at higher altitudes, which leads to the adaptive response of increased red cell mass and Hb levels. 2 This response stems from the higher amount of heme production that is triggered by mitochondrial activation induced by elevated erythropoiesis. Mitochondrial activation is stimulated by physiological activator peroxisome proliferator-activated receptor-gamma coactivator 1alpha (PGC-1α), leading to the stabilization of HIF-1α, which plays a role in the induction of factors involved in erythropoiesis. 12 Hudson et al showed a significant and sustained elevation in platelet count parallel to the rising altitude. 13 Previous studies have shown that large platelets contain more dense granules and are more thrombogenic when compared with the small platelets. 14,15 Contrary to this study, another study showed that MPV was increased in acute coronary syndrome patients living at high altitudes, and elevated MPV was claimed to be one of the causes underlying the thrombogenic environment induced by the high altitude. 16 Sowade et al showed that recombinant human (rh) erythropoietin (Epo) treatment increases Htc with an MPV drop and a PDW rise. 17 Fabris et al with rhEpo-treated children showed a drop in MPV and a rise in PDW. It may be assumed that this finding was the result of the known, probably stochastic, effect of Epo on megakaryocytes. 18,19 These changes in the hematologic parameters were attributed to the occurrence of reduced plasma volume as well as alterations in Epo and thrombopoietin (Tpo) levels induced by hypoxia. 20,21 Similarly, this study showed significantly raised Hb, Htc, and PDW values parallel to the rising altitude, while exhibiting significant decreases in MPV levels.
Elevated Hb concentration has been shown to cause raised CV event incidence by creating an environment with higher viscosity. 22 The AMORIS study demonstrated that elevated Hb levels were associated with acute MI. 23 Similarly, raised Plt and RDW have been shown to increase the incidence of CV events and to worsen the prognosis. 24 –26 Although there are ongoing discussions about MPV being predictive of stroke, acute MI, and restenosis, it has also been shown to have a prognostic significance in patients with stroke and acute MI. 27
There is a positive correlation between increasing altitude and thrombosis of deep venous, cerebral, coronary, and mesenteric arteries as well as pulmonary and portal system thrombosis. 3,28,29 Acute coronary syndromes have been shown to occur more frequently and at earlier ages at higher altitudes. 1 Moreover, at high altitudes the infarct in patients with stroke is more massive. 30 The resultant thrombotic environment is attributed to the changes in the coagulative factor levels that are induced by altitude. Increasing altitude is associated with elevated factor 10-12 levels, shortened prothrombin time, and impairment of clot retraction. 3 If the exposure to high altitude continues for long, this hypercoagulative environment persists. 5,6
Previous studies have demonstrated many factors affecting long-term prognosis of STEMI patients treated with primary PCI. Halkin et al showed that an age >65 years, Killip class 2/3, baseline LVEF <40%, anemia, renal insufficiency, triple-vessel disease, and postprocedural grade 0 to 2 TIMI flow were all predictors of CV mortality in STEMI cases without cardiogenic shock. 31 In the study of Ergelen et al, Killip class 2/3, unsuccessful procedure, contrast-induced nephropathy, DM, and an age >70 years were found to be the predictors of CV mortality. 32 In this study, the history of previous statin use, Killip class 3/4, baseline LVEF <40%, high peak CK-MB level, unsuccessful procedure, and admission with acute stent thrombosis raised the composite end point risk. We observed that altitude status and altitude-related hematologic indices had no influence over the midterm composite end point.
Moreover, one of the remarkable results of this study was that despite the presence of longer reperfusion time and higher peak CK-MB levels in group I patients with STEMI due to geographic limitations, there was no difference between the groups with regard to door-to-balloon time.
Limitations
We did not have the chance to investigate how patients that had been excluded from the study due to the delivery of anticoagulant agents as well as the presence of MI, PCI, CABG, and HF, could have been influenced from the altitude and changing hematologic parameters.
Microvascular perfusion assessment (eg, resolution of ST segment elevation and myocardial blush) and previous primary PCI risk score models were also absent in this study. Studies benefiting from those evaluations may further improve prognostication beyond postprocedural TIMI flow grade. 33,34
Conclusion
This study shows that altitude status and altitude-related hematologic changes do not have an influence over midterm outcomes in STEMI patients treated with percutaneous intervention.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
