Abstract
Background
The local injection of novel cardioprotective study drugs prior to percutaneous coronary intervention could cause embolisation of thrombus, resulting in increased reperfusion injury and subsequent infarct size. The aim of this study was to assess the safety of the delivery of an intracoronary therapy delivered during primary percutaneous coronary intervention for acute myocardial infarction prior to the re-establishment of thrombolysis in myocardial infarction III flow.
Methods
One hundred sixty-seven patients with acute myocardial infarction successfully reperfused through primary percutaneous coronary intervention and undergoing Cardiac MRI within the first week after reperfusion were studied. Patients either underwent the delivery of an intracoronary agent (IMP or placebo) prior to balloon dilatation (n = 80) or standard primary percutaneous coronary intervention procedure (n = 117).
Results
Baseline characteristics were similar between the two groups. There were a similar number of successful procedures (IC IMP 75 (93.8%) vs. No IMP 114, (97.4%), p = 0.374), rates of no-reflow (IC IMP 1 (1.3%) vs. No IMP 2 (1.7%), p = 0.99) and levels of ST segment resolution (88.5% IC IMP vs. No IC IMP 87.0%, p = 0.669) between the two groups. Similar levels of microvascular obstruction were seen between the two groups with a trend to reduced infarct size, and improved ejection fractions in the IMP group. Lower MACE rates were seen in the IMP group.
Conclusion
The local intracoronary infusion of potential cardioprotective agents prior to the restoration of TIMI flow in patients undergoing primary percutaneous coronary intervention for acute myocardial infarction appears to be safe and does not increase microvascular damage. This route should be considered when testing novel cardioprotective agents.
Keywords
Introduction
Timely delivered primary percutaneous coronary intervention (PCI) has become the favoured reperfusion therapy for ST-elevation myocardial infarction (STEMI) in the US and Europe. 1 However, this interventional technique has not abolished the unpredictable phenomenon of no-reflow, reperfusion injury and the underappreciated, but potentially equally important, syndrome of microvascular obstruction (MVO). 2 MVO has been associated with adverse prognosis, adverse LV remodeling and diminished recovery of LV function, independently of infarct size.3,4
Compared with the systemic administration of intravenous pharmacotherapies, highly localized administration of intracoronary pharmacotherapy may be associated with a several-hundred-fold increase in the local concentration of an agent in the epicardial artery/microcirculation with associated beneficial effects. However, despite the potential benefit of this technique, there is limited evidence for the delivery of therapies locally prior to balloon dilatation and restoration of thrombolysis in myocardial infarction (TIMI) flow. Potentially, infarct size may be increased as injection of the study drugs prior to PCI could cause embolisation of thrombus to the microcirculation, resulting in increased MVO, reperfusion injury and subsequent infarct size. Several studies have opted to infuse novel therapies such as adenosine after thrombus aspiration to minimise the chances of this occurrence.5,6
The aim of this study was to assess the safety of the delivery of intracoronary therapies delivered during primary PCI for AMI prior to the re-establishment of TIMI III flow.
Methods
Between April 2008 and November 2012, 197 patients with ST-segment elevation MI successfully reperfused through primary percutaneous coronary intervention (PCI) and undergoing CMR within the first week after reperfusion were studied. All of these patients had been consented into interventional clinical trials including stem cell trial and pharmacological intervention trials (REGENERATE- AMI (NCT00765453), NITRITE-AMI (NCT01584453) and myocardial oedema in acute myocardial infarction (AMI) (NCT00987259)). These studies were approved by local ethics committee and performed in accordance with the Declaration of Helsinki (1996) and the principles of the International Conference on Harmonization–Good Clinical Practice (ICH-GCP) guidelines.
NITRITE-AMI was a double-blind, randomised, single-centre, placebo-controlled trial to determine whether the intra-coronary injection of sodium nitrite reduces infarct size in patients with acute STEMI undergoing primary PCI.7,8 After coronary angiography, patients were randomised (1:1) to a high-dose bolus injection of intra-coronary sodium nitrite (1.8 µmol in 10 mL of 0.9% NaCl) or placebo (10 mL of 0.9% NaCl) administered via an over the wire (OTW) balloon just prior to balloon inflation. Both REGENERATE-AMI (which recruited 100 patients with patients presenting with anterior STEMI) and myocardial oedema in AMI studies underwent standard primary PCI for AMI with no administration of intracoronary therapy through an OTW. Use of adjunctive therapies such as aspiration thrombectomy was at the discretion of the operators in all three studies.
CMR imaging
Cardiac magnetic resonance (CMR) imaging was performed on a 1·5 T Philips Achieva scanner with a cardiac 32-channel phased array coil. Balanced steady-state free precession cine imaging was used to acquire 10–12 short axis slices (8 mm slice thickness, 2 mm gap) with one slice per breath-hold. Sequence parameters were 1.5 ms echo time (TE), 3.1 ms repetition time (TR), and acquired voxel size was 1.8 × 1.86 mm with a typical field of view (FOV) of 360 mm in the phase encode direction. We acquired 45 phases with 25% phase sharing. Parallel imaging (SENSE) was used with an acceleration factor of 2.0.
Delayed enhancement images were acquired 10 min after injection of a dose of 0·2 mmol/kg of gadoterate meglumine (Dotarem) for late gadolinium enhancement. A T1-weighted segmented inversion-recovery gradient echo pulse sequence (TR 3.9 ms TE 1.9 ms, flip angle 15°, voxel size of 2 × 2 mm, typical FOV 360 mm) was used to obtain 10–12 short axis slices (matched with short-axis cine images) with one slice per breath-hold. The inversion time was adjusted individually according to a T1 scout sequence (Look-Locker). Images were acquired every other heart beat with two signal averages.
Myocardial oedema was assessed using fat suppressed T2-weighted triple inversion turbo spin echo STIR (Short tau inversion recovery) imaging (TE 80 ms, TR two heart beats, TSE factor 31, voxel size 1.8 × 1.8 mm); 10–12 slices were obtained (8 mm per slice, 2 mm gap matched to DE/cine slices) with one slice per breath-hold. This sequence has previously been used and validated for assessment of myocardial oedema and MSI.9–11
Images were anonymised, batched and analysed in blinded fashion by two experienced operators. Scar and oedema volumes were calculated by manually drawing endocardial and epicardial contours followed by semi-automated selection of normal remote myocardium per slice. Myocardial oedema was described as >2SD in signal intensity from remote normal myocardium. Infarct size was calculated using the full-width half maximum method as previously described. 12 In case of discordance between operators, blinded review by a level III accredited CMR reader was performed. Analysis was performed using dedicated software (CVI42, Circle Cardiovascular Imaging Inc., Calgary, Alberta, Canada). Inter and intra-observer variability was calculated.
Statistical analyses
The study population was divided into 2two groups: Intra-Coronary IMP (IC IMP) and no IMP. Primary study end point was the myocardial salvage index on CMR. Secondary endpoints included final TIMI flow ≥ 2 on angiography, rate of ST-segment resolution post primary PCI, the degree of myocardial hemorrhage (T2* measurement) and occurrence of MVO at the first time point of within 48 h post-STEMI, and parameters of left ventricular remodeling including left ventricular end diastolic volume index (LVEDI), left ventricular end systolic volume index (LVESI), left ventricular stroke volume index (LVSVI), left ventricular ejection fraction, and infarct size post STEMI. Continuous data were expressed as mean ± standard deviation and analyzed by the Student t test. Categorical variables were reported as frequencies and percentages and analyzed by the Chi-square or Fisher exact test, as appropriate. All tests were two-tailed, and statistical significance was accepted at p < 0.05.
As this was a post trial comparison between RCTS, a post hoc sample size was calculated for the between-group comparison with regard to the primary endpoint of myocardial salvage index. From the results of published studies and this trial data, a sample size of 198, with a mean MSI of 48 and a SD of 15% gives a statistical power of 80% power, to reject the null hypothesis of equal means between the two groups assuming no less than 4% difference (two-tailed Student t test, 0.05).
Results
Baseline characteristics for study population.

LAD: left anterior descending artery; PCI: percutaneous coronary intervention.
Procedural characteristics were similar between the two groups with rates of thrombus aspiration catheters, GPIIb/IIIa inhibitors and use of drug eluting stents.
Procedural outcomes
There were a similar number of successful procedures in the two cohorts (IC IMP 75 (93.8%) vs. No IMP 114, (97.4%), p = 0.374). Rates of no-reflow were similar between the two groups (IC IMP 1 (1.3%) vs. No IMP 2 (1.7%), p = 0.99). Levels of ST segment resolution (>70% within 90 min of the procedure) were the same in the two groups (88.5% IC IMP vs. No IC IMP 87.0%, p = 0.669).
CMR parameters
Cardiac magnetic resonance data for study population.

Note: Values shown as mean (95% CI) unless otherwise stated.
LVEDVi: indexed left ventricle end-diastolic volume; LVESVi: indexed left ventricle end-systolic volume; LVMi: indexed left ventricle mass; LVEF: left ventricle ejection fraction; AAR: area at risk; MSI: myocardial salvage index; MVO: microvascular obstruction.
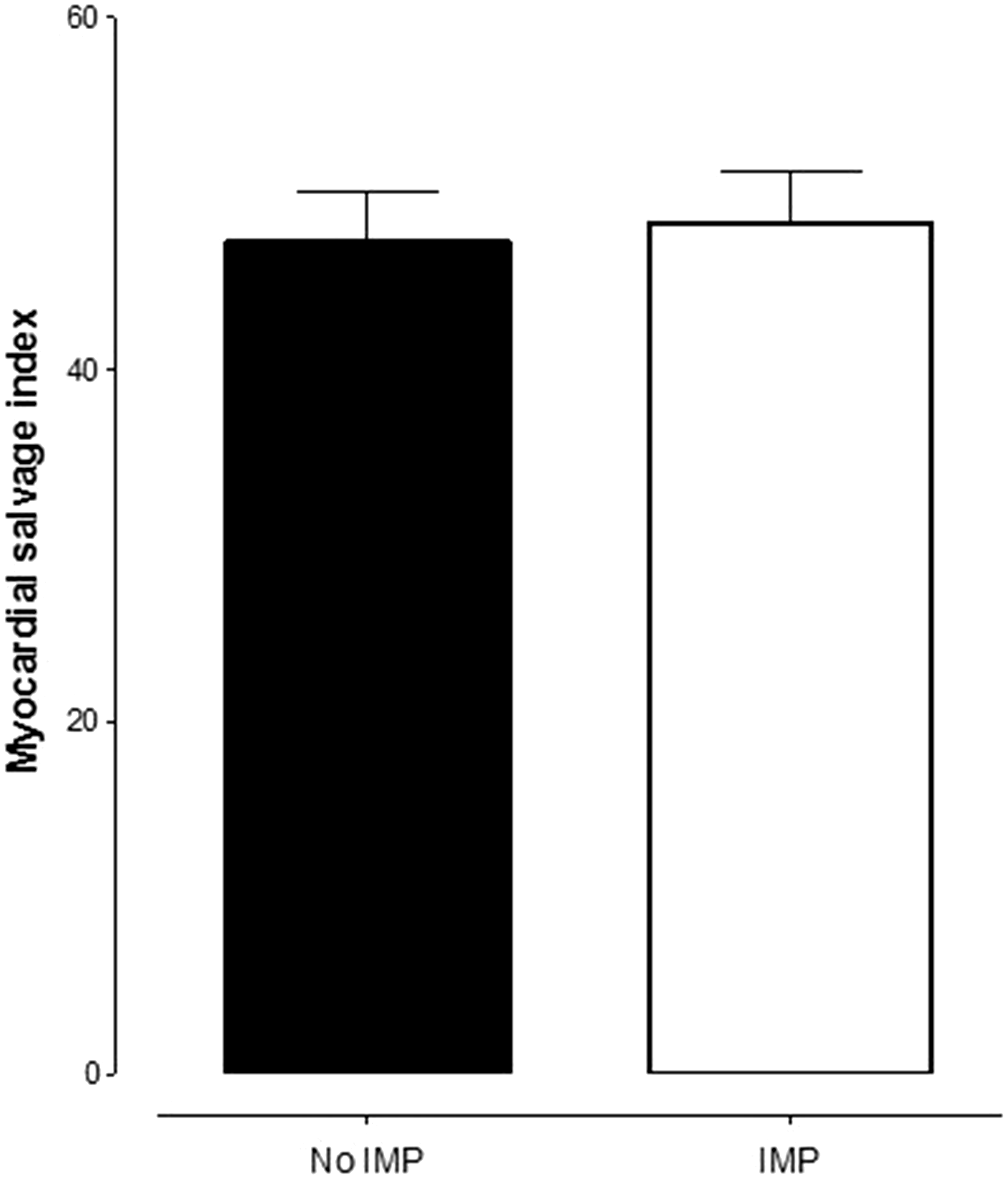
Assessment of myocardial salvage index (MSI) on CMR. Amount of MSI on CMR is presented for 117 patients in the no IMP (control) group and 68 patients in the IMP group. There was no difference in the MSI as assessed by CMR between the two groups (p = 0.8396). Significance evaluated using unpaired t test and data shown as mean ± SEM.
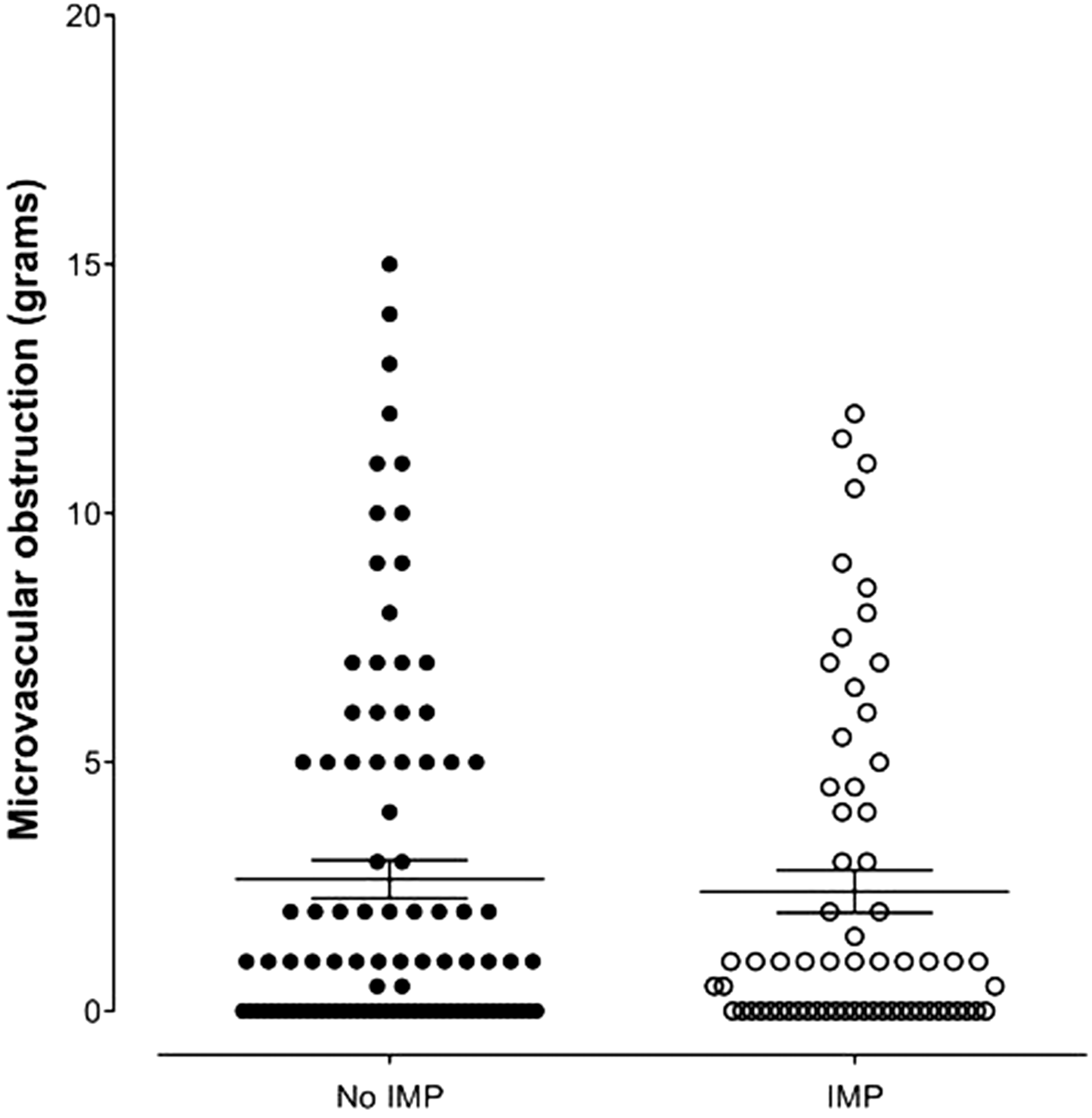
Assessment of microvascular obstruction on CMR. Amount of microvascular obstruction on CMR is presented for 117 patients in the no IMP (control) group and 68 patients in the IMP group. There was no difference in the amount of microvascular obstruction as assessed by CMR between the two groups (p = 0.8648). Significance evaluated using unpaired t test and data shown as mean ± SEM.
Inter-observer and intra-observer variability
Measurements of LV function, T2 AAR, MVO and LV scar parameters were evaluated by interclass correlation coefficient (ICC) with 95% CI. Inter-observer agreement for LVEF, IS, MVO, AAR and MSI was excellent, with ICCs of 0.992, 0.925, 0.968, 0.908 and 0.951, respectively. Intra-observer agreement was also excellent, with ICC of LVEF: 0.982, IS 0.980, MVO 0.991, AAR 0.982, and MSI of 0.931.
Adverse events
One year after infarction, seven patients (8.75%) in the IC IMP group suffered MACE (two hospitalisations for heart failure, one recurrent myocardial infarction and four unscheduled revascularizations) compared to 17 patients (14.5%) (1 mortality, 4 recurrent MI, 10 unscheduled revascularization, 2 hospitalisations for heart failure) in the no IC IMP group (p = 0.1685) (Figure 3).
Kaplan–Meier curves showing cumulative probability of major adverse cardiac events (MACE) after primary PCI according to treatment group.
Discussion
This study demonstrates that the local intracoronary infusion of a novel cardioprotective agent prior to the restoration of TIMI flow in patients undergoing primary PCI for AMI appears to be safe and does not increase microvascular injury compared to standard therapy. Intracoronary therapies have been shown to be safe in two previous published studies8,13 including NITRITE-AMI when delivered prior to reperfusion in the context of primary PCI; however, this is the first study to have a control arm with no IC agent to demonstrate no harmful effect on the microvasculature. Concerns over distal embolization and increased microvascular damage with the delivery of local intra-coronary therapies prior to reperfusion have been raised but this study shows when compared to standard therapy (i.e no IC agent), there is no evidence of such an effect.
In the NITRITE-AMI study, 8 MVO was shown to be reduced in patients treated with nitrite compared to those who received placebo with the incidence of MVO reduced by ∼20% and 35% in the nitrite-treated group in the whole cohort and the TIMI flow<1 subgroup, respectively. However, this comparison was between two groups who both received an IC infusion prior to reperfusion which itself may have increased the amount of reperfusion injury and microvascular injury. This study has demonstrated that compared to standard therapy this is not the case. It is worth noting that factors that impact on MVO, such as comorbid conditions and the use of antiplatelet and anticoagulant therapy, were similar between the two treatment groups. MVO has been implicated in worse clinical outcomes due to poor myocardial perfusion despite epicardial coronary artery revascularization. 14
The timing of the cardio-protective strategy is critical in the design of clinical studies. The time window of protection is quite narrow because the main part of lethal reperfusion injury takes place within the first minutes of reflow after prolonged ischemia. Protective interventions must be applied either intravenously prior to the opening of the culprit coronary artery or locally at the onset of reperfusion.
Compared with the systemic administration of intravenous pharmacotherapies, the highly localized administration of intracoronary pharmacotherapy may be associated with a several-hundred-fold increase in the local concentration of an agent; however, despite the potential benefit of this technique, there is limited evidence for the delivery of therapies locally prior to balloon dilatation and restoration of TIMI Flow. Potentially infarct size may be increased as injection of the study drugs prior to PCI could cause embolisation of thrombus and other particles to the microcirculation, resulting in increased MVO, reperfusion injury and subsequent infarct size. Several studies have therefore adopted to infuse novel therapies to reduce reperfusion injury after reperfusion (restored with thrombus aspiration) to minimise the chances of this occurrence.5,15,16 The importance of the state of the microcirculation after primary PCI and its association with outcomes is being increasingly recognised 17 and understood and this study demonstrating the safety of local drugs administered prior to reperfusion is important for the design of future studies aimed at cardioprotection.
Limitations
This is a combination of several RCTS recruiting patient population who have suffered STEMI and underwent successful primary PCI. One of the studies included in the analysis recruited only patients with anterior infarcts due to occlusions of the LAD, which may have lead to bias in the results.
Conclusion
The local intracoronary infusion of potential cardioprotective agents prior to the restoration of TIMI flow in patients undergoing primary PCI for AMI appears to be safe and does not increase microvascular damage. This route and the methodology described here should be considered when testing novel cardioprotective agents.
Footnotes
Acknowledgements
We thank the Barts Heart Centre Interventional Group at St. Bartholomew's Hospital for recruiting patients. We also thank the nursing staff of the cardiac catheterisation laboratory, MRI and coronary care units at Barts Heart Centre.
Contribution
KSR, SH and DAJ drafted the manuscript, SH, RSK and MA assisted in data collection, MW, AM and AA critically reviewed the manuscript and DAJ had the original concept for the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author would like to thank National Institute for Health Research, UK and Heart Cells Foundation for the financial support.
Ethical approval
The ethical approval was Provided by two committees for the three RCTs: NRES Committee London - West London and, NRES Committee London – East London.
Guarantor
Dr. Daniel Jones (NIHR Academic Clinical Lecturer).
