Abstract
Busulfan is widely used to treat malignant diseases, particularly for therapeutic intensification prior to an autologous stem cell graft. Numerous side effects consecutive to busulfan are described, but few descriptions of pulmonary hypertension exist, while bronchiolitis obliterans remains a rare complication. We report the clinical observations of four patients from the French Pulmonary Hypertension Registry who experienced subacute pulmonary hypertension after receiving busulfan as preparation regimen before an autologous stem cell graft for malignancies (Hodgkin’s disease, Ewing’s sarcoma and primary large B cell lymphoma of the brain). Patients experienced severe pulmonary arterial hypertension 2 to 4.5 months after busulfan administration. Pulmonary hypertension improved after treatment with approved drugs for pulmonary arterial hypertension and/or corticosteroids. During the follow-up period, two patients developed chronic respiratory insufficiency due to interstitial lung disease, leading to double lung transplantation. The pathological assessment of explanted lungs revealed interstitial lung fibrosis with advanced bronchiolar lesions and severe pulmonary vascular damage. Three of the four patients were still alive after 36 to 80 months and the fourth died unexpectedly and suddenly after 5 months. In conclusion, PAH is a rare but severe complication associated with busulfan chemotherapy in adults. Histological examinations provide evidence for diffuse pulmonary vascular damage combined with interstitial lung injury in most cases.
Keywords
Introduction
Autologous stem cell transplantation (ASCT) is a therapeutic option that is frequently used to treat various types of hematological cancer or solid tumors. Before the hematopoietic stem cell infusion, a conditioning regimen using high doses of chemotherapy and/or radiation therapy is systematically administered. Busulfan is an alkylating agent that is widely used to treat this specific indication, most commonly in association with other drugs.1–4 Many side effects of busulfan have been already described, including pulmonary toxicity, leading to acute lung injury, 5 chronic interstitial fibrosis 6 or acute alveolar hemorrhage. 7 Limited evidence is available on the pulmonary vascular toxicity induced by busulfan.
Pulmonary arterial hypertension (PAH) is a severe and devastating condition defined by an increase in mean pulmonary arterial pressure (mPAP) > 20 mmHg with a normal pulmonary artery wedge pressure (PAWP) ≤ 15 mmHg and increased pulmonary vascular resistance (PVR) > 3 Wood units (WU), leading to a progressively worsening right-sided heart failure. 8 PAH may be idiopathic or develops a result of an associated risk factor, such as drug or toxin exposure. 9 Among these factors, alkylating agents have been identified as a possible risk factor for the development of drug-induced PAH. 10 A pediatric study of ASCT has shown that PAH is not a rare complication in children receiving busulfan/melphalan and may occur in 19% of cases. 11 PAH was not reported in children receiving a combination of carboplatin/etoposide/melphalan, suggesting that PAH may represent a specific side effect of busulfan. 11 Nevertheless, the diagnosis of pulmonary hypertension (PH) was suspected by echocardiography without performing right heart catheterization (RHC) to confirm the diagnosis and determine its mechanisms.
The main objective of the present study was to describe clinical cases of severe precapillary PH confirmed by RHC occurring after ASCT preceded by a conditioning regimen including busulfan. We also analyzed the histological findings of lung explants from two of these patients who received a transplant. Finally, we briefly reviewed and discussed other cases reported in the literature.
Methods
Patients
Data from four patients admitted to the French reference center for precapillary PH after busulfan exposure between March 2014 and July 2019 were analyzed. This retrospective study complied with the Declaration of Helsinki. Although French law does not require ethics committee approval or informed consent for retrospective data collection, the data were anonymized and complied with the requirements of the Commission Nationale Informatique et Liberté, the organization dedicated to privacy, information technology and civil rights in France. The committee approved the methods used to collect and analyze data on 24 May 2003 (approval number 842063).
PH was screened using transthoracic echocardiography and precapillary PH was systematically confirmed by RHC showing an mPAP > 20 mmHg, a normal PAWP ≤ 15 mm Hg and PVR ≥ 3 WU. Acute vasodilator testing was performed during RHC using inhaled nitric oxide (NO). Consistent with current guidelines, all patients in whom the diagnosis of PH was confirmed by RHC underwent extensive testing to identify additional possible risk factors for PH, including history of potential offending drugs, serology testing for HIV and connective tissue diseases. Ventilation/perfusion scintigraphy and high-resolution computerized tomography (HRCT) excluded both chronic thromboembolic PH and diffuse interstitial lung disease. Details of chemotherapy exposure (busulfan and/or others) were collected by recording a detailed clinical history at the time of PH diagnosis. Symptomatic treatment, medical therapies approved for PAH and corticosteroids were administered to patients according to the clinical judgment and discretion of treating physicians. Clinical, functional and hemodynamic follow-up data were collected for each patient, and main patient characteristics are presented in Table 1.
Characteristics of the four patients with pulmonary hypertension associated with busulfan.
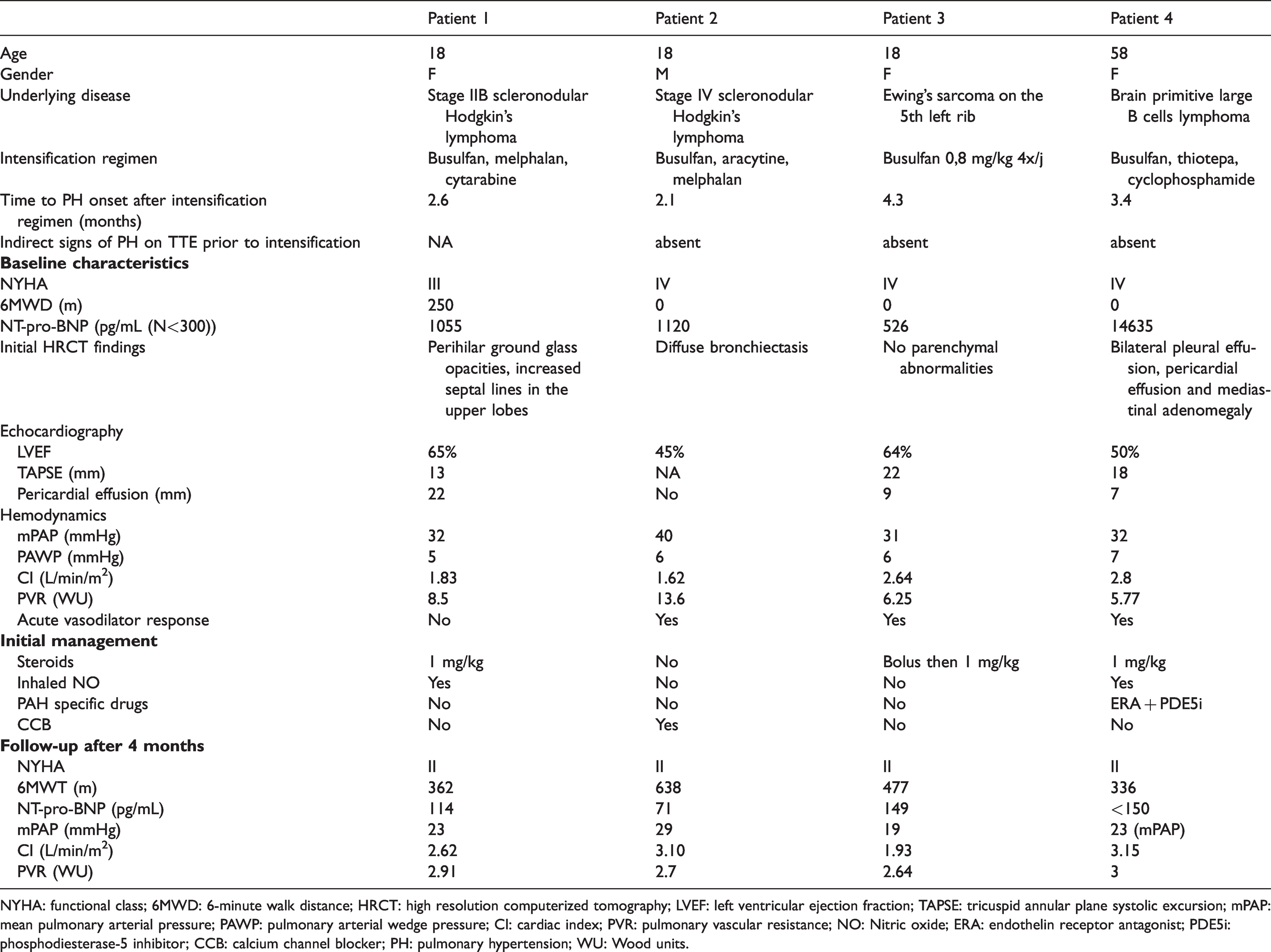
NYHA: functional class; 6MWD: 6-minute walk distance; HRCT: high resolution computerized tomography; LVEF: left ventricular ejection fraction; TAPSE: tricuspid annular plane systolic excursion; mPAP: mean pulmonary arterial pressure; PAWP: pulmonary arterial wedge pressure; CI: cardiac index; PVR: pulmonary vascular resistance; NO: Nitric oxide; ERA: endothelin receptor antagonist; PDE5i: phosphodiesterase-5 inhibitor; CCB: calcium channel blocker; PH: pulmonary hypertension; WU: Wood units.
Pathological assessment
Explanted lungs from two patients who received a transplant were available for the pathological assessment. Lung explants were fixed with formalin (10% neutral buffer), and parenchymal samples were collected from all lobes. Hematoxylin and eosin-stained slides were subjected to a histopathological assessment.
Results
Description of clinical cases
Patient 1, a 28-year-old woman with a scleronodular Hodgkin’s stage IIB lymphoma, was successively treated with first-line chemotherapy combining doxorubicin, bleomycin, vinblastine and dacarbazine, then an external radiotherapy treatment, followed by a second-line treatment combining dexamethasone, cytarabine and cisplatin and a third-line treatment combining doxorubicin, ifosfamide and etoposide. Due to a relapse of the disease, a first ACST was performed after the administration of a conditioning regimen including carmustine, etoposide, aracytine and melphalan. Finally, a second ACST was performed two months later after the administration of a conditioning regimen including 4 mg/kg busulfan, cytarabine and melphalan. Seventy-nine days after the second ASCT, she was referred to our center for rapidly progressive dyspnea and clinical signs of acute right heart failure. Echocardiography showed a dilated right ventricle, an estimated systolic pulmonary artery pressure (sPAP) of 57 mmHg and signs of severe right ventricular dysfunction, including a tricuspid annular plane systolic excursion (TAPSE) at 13 mm, an inferior vena cava dilatation at 19 mm and a moderate pericardial effusion. RHC confirmed the diagnosis of severe precapillary PH with mPAP 32 mmHg, a PAWP 5 mmHg, a cardiac index (CI) 1.8 L/min/m2 and PVR 8.5 WU. The arterial blood gas analysis showed severe hypoxemia with an arterial pressure of oxygen of 43 mmHg in room air, and the NT-pro-BNP level was elevated at 1055 pg/mL. HRCT of the chest showed perihilar ground glass opacities and thickening of the septal lines in the upper lobes. A diagnosis of acute pulmonary embolism was excluded (Fig. 1a and b). Initial management in the intensive care unit (ICU) combined inotropic support with dobutamine, inhaled NO and corticosteroids at the dose of 1 mg ⋅ kg−1. Her short-term evolution was marked by a progressive improvement in right ventricular function and gas exchange. NO and dobutamine were thus discontinued. One month after admission, the patient was in NYHA functional class II, and the 6-minute walk distance (6MWD) was 362 m. RHC showed a hemodynamic improvement with a mPAP of 32 mmHg, right atrial pressure (RAP) of 4 mmHg, PAWP of 3 mmHg, CI of 3.1 L/min/m2 and PVR of 5.5 WU. The arterial pressure of oxygen in room air was 72 mmHg. No use of specific treatments for PAH was required.
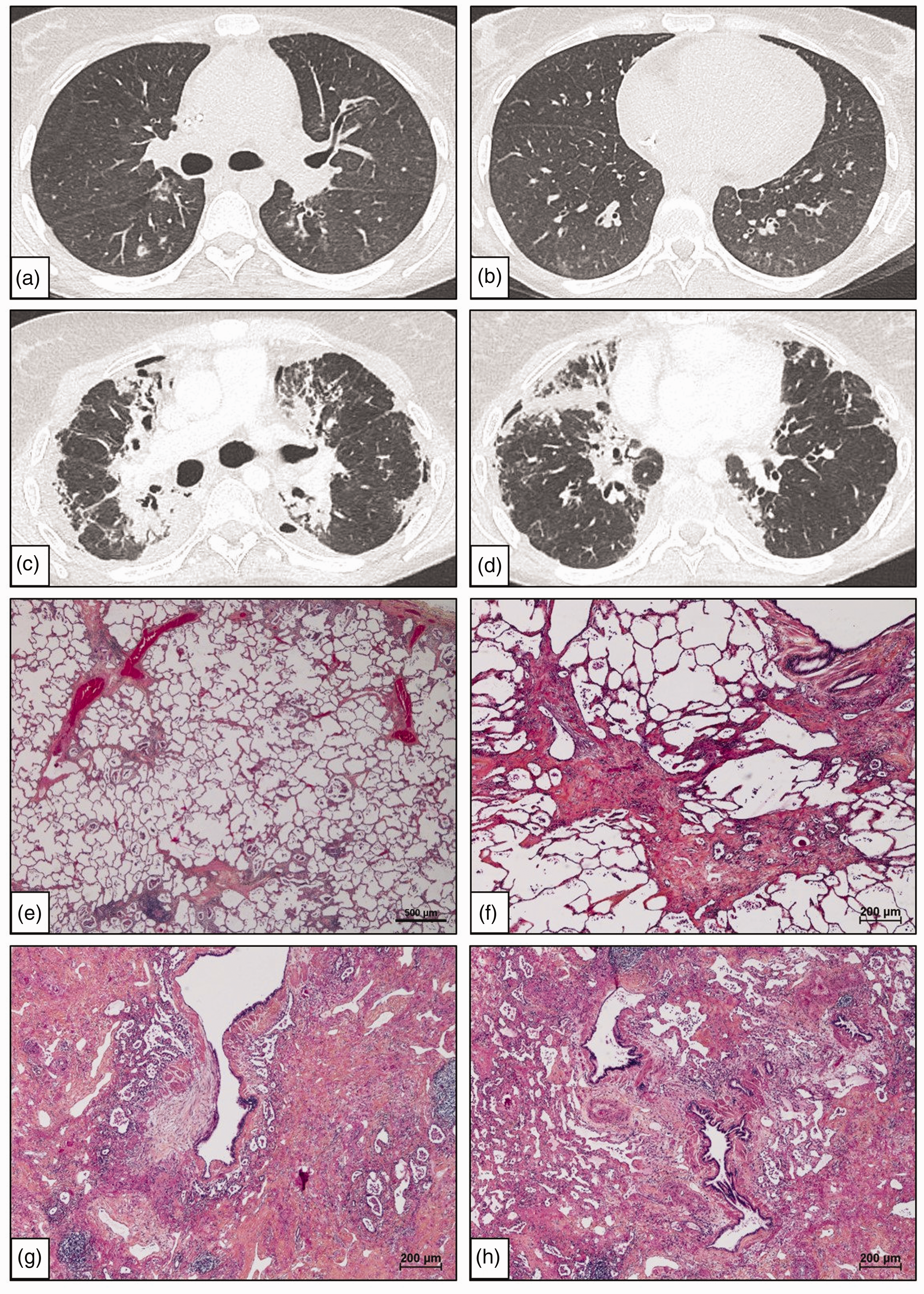
(a and b) The baseline CT of the chest performed at our center showed an enlargement of the pulmonary artery, a diffuse ground glass aspect and neither septal lines nor mediastinal adenomegaly. (c and d) At 47 months after the baseline scan, the CT of the chest showed a severe fibrotic parenchymal disease associated with an architectural distortion. (e) Non-fibrotic lung parenchyma. Alveolar septa are thin or slightly enlarged by congestion and inflammation. No capillary hemangiomatosis is observed. Peri-bronchiolar alveolar spaces contain inflammatory exudate that are constituted of macrophages and lymphocytes surrounding cholesterol clefts (arrowhead). Some bronchioles are obliterated by fibrous tissue (star). (f) Dense fibrous tissue surrounding small airways. Fibrotic lesions show temporal heterogeneity: “old” collagen-rich fibrotic lesions predominated, while “new” fibrotic lesions are visible within the bronchiolar wall. (g) Scarred lung parenchyma showing dense hyaline fibrosis and fibroblastic foci next to the bronchiolar lumen. Fibroblastic foci are characterized by aggregates of actively proliferating myofibroblasts within a myxoid matrix that are oriented with the long axis parallel to the long axis of the bronchial lumen. (h) Centrilobular fibrosis with vascular remodeling. Small muscular arteries appear to be markedly remodeled with an increase in the thickness of the intimal and smooth muscle layer.
After 47 months, the patient experienced a worsening of symptoms. The RHC showed a hemodynamic improvement (mPAPm of 16 mmHg, PAWP of 3 mmHg, RAP of 1 mmHg, CI of 2.2 L/min/m2 and PVR of 4.49 WU). However, pulmonary function tests showed a severe restrictive syndrome with a forced vital capacity (FVC) of 22%, a forced expiratory volume in 1 second (FEV1) of 26% and a total lung capacity (TLC) of 42% of the theoretical values with reduced diffusion capacity for carbon monoxide (DLCO) of 38% of the theoretical value. The HRCT showed severe fibrotic parenchymal disease, with architectural distortion (Fig. 1c and d). The patient was listed and received a double lung transplant 52 months after the diagnosis of PAH. She was still alive 25 months after transplantation.
Patient 2, an 18-year-old man with stage IV scleronodular Hodgkin’s lymphoma at diagnosis, was referred to our ICU for rapidly progressive dyspnea and signs of acute right heart failure. The patient was successively treated with several lines of chemotherapy including doxorubicin, bleomycin, vinblastine and dacarbazine as the first-line protocol, followed by ifosfamide, navelbine and etoposide. Finally, a first ASCT was performed after the administration of a conditioning regimen combining carmustine, etoposide, cytosine-arabinosine and melphalan. The second ASCT was performed two months later after the administration of a conditioning regimen including busulfan, aracytine and melphalan. Myocardial scintigraphy performed two months before the second ASCT showed a low left ventricular ejection fraction of approximately 45% that was related to the previous anthracycline treatment. Two months after the second ASCT, he developed clinical signs of right heart failure. At admission, the systemic blood pressure was 80/60 mmHg and the heart rate was 155 bpm. The NT-pro-BNP level was 1120 pg/mL. HRCT ruled out an acute pulmonary embolism and showed previously existing diffuse bronchiectasis and an enlarged pulmonary artery with a size of 33 mm (Fig. 2a). Echocardiography showed a dilated right ventricle, an estimated sPAP of 71 mmHg and signs of severe right ventricular dysfunction. RHC confirmed the diagnosis of severe precapillary PH with an mPAP of 40 mmHg, a PAWP of 6 mmHg, RAP of 13 mmHg, a CI of 1.6 L/min/m2 and PVR of 13.6 WU.
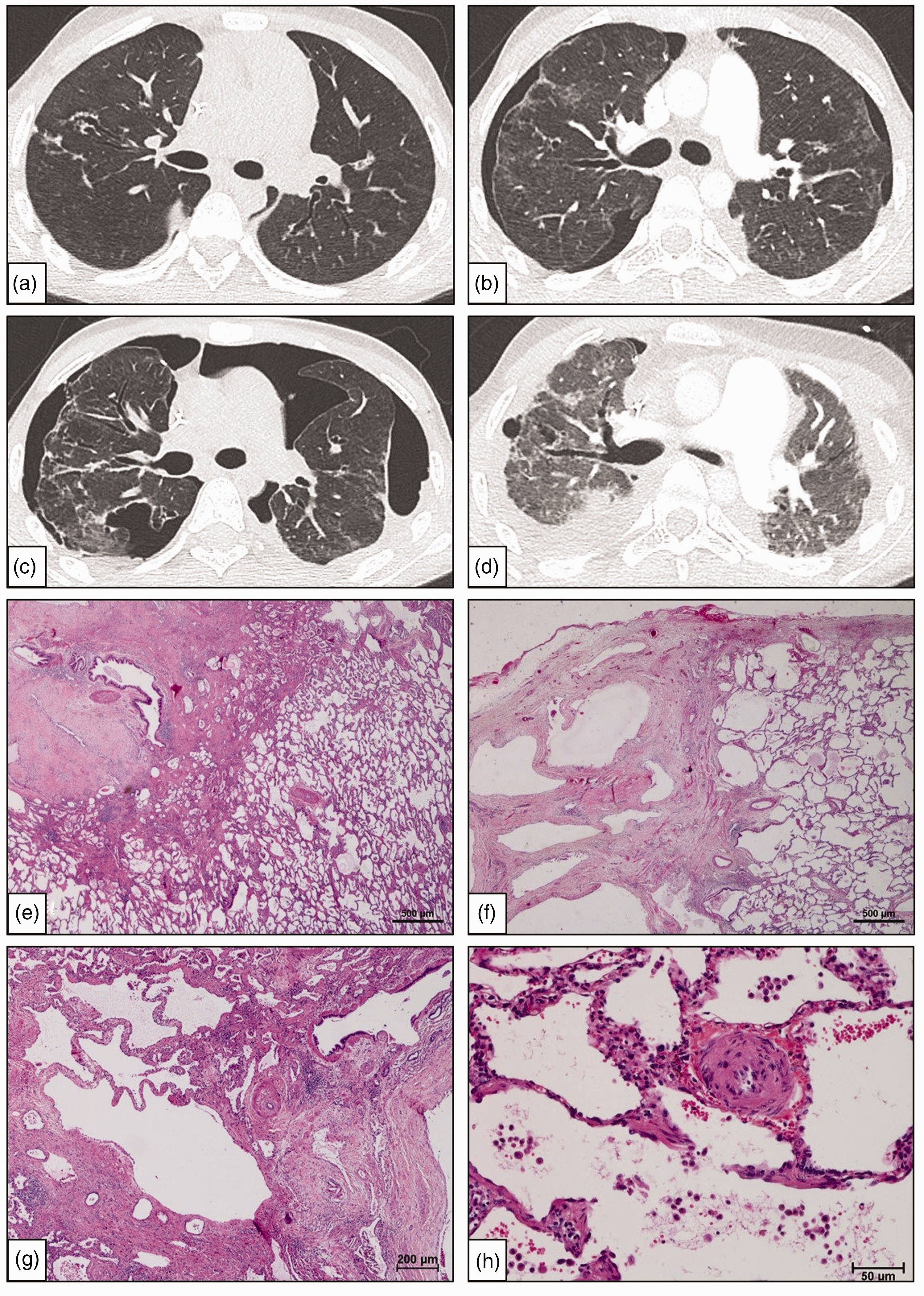
(a) At the baseline, the CT of the chest showed the stability of the lung parenchymal signs of the malignant hematological disease and bronchiectasis, without signs of other parenchymal abnormalities. (b) Eight months later, the CT showed ground glass images, an increase in bronchiectasis and subpleural bullous images. (c) A relapse of pneumothorax occurred, despite surgical treatment of the first occurrence. (d) During the follow-up period, an increase in the bronchiectasis, subpleural bullous effusions and a ground glass aspect were observed. (e) Peribronchiolar dense fibrosis distorting the bronchial lumen (black arrowhead) and vascular remodeling are observed. Note that arterial remodeling is also apparent outside the fibrotic foci. (f) Peripheral honeycomb changes. Note the abrupt transition between the fibrotic area and the lung. (g) Honeycomb changes showing airspaces surrounded by interstitial fibrosis. A markedly remodeled small pulmonary artery is easily identified. (h) Microvessel remodeling. Marked thickening of the vascular wall is observed and constituted of proliferating smooth muscle cells and fibrosis.
A first-line symptomatic treatment combining NO and a dobutamine infusion was introduced, leading to a rapid hemodynamic improvement. A second RHC was performed five days later, resulting in a mPAP of 40 mmHg, PAWP of 2 mmHg, CI of 3.6 L/min/m2 and PVR of 8.1 WU, as well as an acute vasodilatory response to NO with a mPAP of 21 mmHg, PAWP of 2 mmHg, CI of 3.6 L/min/m2 and PVR of 3.8 WU. Treatment with calcium channel blockers (diltiazem) was introduced. Four months later, the patient presented with NYHA functional class II, the 6MWD was 638 m and echocardiography revealed a 45–50% left ventricular ejection fraction and an estimated sPAP of 40 mmHg, without signs of right ventricular dysfunction.
Eight months after the diagnosis of PAH, pleuroparenchymal complications occurred with a bilateral spontaneous pneumothorax (Fig. 2b and c). The HRCT showed ground glass opacities, an increase in bronchiectasis and subpleural bullous images (Fig. 2d). The patient was listed for a rapidly progressive restrictive pulmonary insufficiency and received a successful heart-double lung transplant 16 months after the diagnosis of PAH. The patient was still alive 64 months after transplantation.
Patient 3, an 18-year-old woman with Ewing’s sarcoma on the fifth left rib, was admitted to our ICU for right heart failure. She received two lines of chemotherapy, including vincristine, ifosfamide, doxorubicin and etoposide as the first-line treatment, followed by vincristine, actinomycin A and ifosfamide as the second-line treatment. After chemotherapy, resection of the fifth left rib with partial resection of the left upper lobe and partial resection of the diaphragm was performed. Histology showed no tumor invasion of the lung or the diaphragm, but a microscopic tumor invasion of the surgical margins was observed. Due to this residual disease, a preparation regimen composed of 0.8 mg/kg busulfan four times a day was organized, followed by an ASCT and an external radiotherapy treatment. She developed rapid progressive dyspnea and was admitted to the ICU 130 days after the ASCT. Echocardiography showed a dilated right ventricle, an estimated sPAP of 60 mmHg and signs of severe right ventricular dysfunction, including pericardial effusion. RHC confirmed the diagnosis of severe precapillary PH with an mPAP of 31 mmHg, PAWP of 6 mmHg, RAP of 7 mmHg, CI of 2.6 L/min/m2 and PVR of 6.3 WU. During the acute vasoreactivity test with NO, the mPAP decreased to 21 mmHg with a PAWP of 7 mmHg, a CI of 2.9 L/min/m2 and RVP of 3.2 WU, suggesting an acute vasodilatory response to NO. The initial HRCT ruled out a pulmonary embolism and showed no specific signs of pulmonary vascular disease (Fig. 3a). The diagnosis of busulfan-induced PAH was suggested and treatment with a systemic corticosteroid bolus for three days was initiated at a dose of 1 mg⋅kg−1. Twelve months later, the patient presented with NYHA functional class I, the 6MWT was 475 m and the NT-pro-BNP level was 147 pg/mL. Echocardiography showed an absence of right ventricular dysfunction, a tricuspid regurgitant velocity (TRV) of 3 m/s and a left ventricular ejection fraction of 50% with a diffuse hypokinesia of the left ventricle, which was compatible with the diagnosis of anthracycline-related cardiotoxicity. HRCT showed signs of bronchiolitis obliterans with subpleural reticulations, localized fibrotic lesions in the left lower lobe and localized bronchiectasis without mediastinal adenomegaly (Fig. 3b). The patient was still alive 36 months after the diagnosis of PAH.
Patient 4, a 58-year-old woman with primary large B cell lymphoma in the brain was also referred to our center after the onset of rapidly progressive dyspnea due to right heart failure. She reported a history of ischemic cardiopathy with a left ventricular ejection fraction of 62%, ischemic arteritis in the lower limbs and the discontinuation of tobacco use at approximately 45 pack-years. Her hematological malignancy has been previously treated with two chemotherapeutic protocols, including methotrexate, etoposide, methylprednisolone, and carmustine in the first line and rituximab and aracytine in the second line. Due to the relapse of the disease, she received a preparation regimen of 8 mg/kg busulfan, thiotepa and cyclophosphamide prior to ASCT. She was admitted to the ICU 105 days after the ASCT. The NT-pro-BNP level was 14,635 pg/mL. Echocardiography showed a left ventricular ejection fraction of 50%, an estimated sPAP of 40 mmHg and a TAPSE of 18 mm. The HRCT showed bilateral pleural effusion, pericardial effusion and mediastinal adenomegaly, without any signs of pulmonary embolism or ground glass opacities (Fig. 3c). RHC was performed and showed a precapillary PH with a mPAP of 32 mmHg, PAWP of 7 mmHg, RAP of 5 mmHg, CI of 2.8 L/min/m2 and PVR of 5.8 WU, and an acute vasodilatory response to NO with a mPAP of 22 mmHg, PAWP of 7 mmHg, CI of 2.9 L/min/m2 and PVR of 3.4 WU. Pulmonary function testing showed an FVC of 88%, FEV1 of 79%, TLC of 103% and DLCO of 40% of the theoretical values. A corticosteroid treatment (1 mg/kg) was introduced in association with inhaled NO, rapidly switched to bosentan and tadalafil. During the follow-up, the clinical and hemodynamic situation improved rapidly and two months later the patient was on NYHA functional class II, 6MWD was 336 m, NT-pro-BNP was normal. RHC showed mPAP 23 mmHg, PAWP 8 mmHg, RAP 4 mmHg, CI 3.2 L/min/m2, PVR at 3 WU. Pulmonary function testing showed FVC at 100%, FEV1 at 90% and DLCO of 32% of the theoretical values. HRCT showed no signs of parenchymal lesions and a partial regression of the mediastinal adenomegaly (Fig. 3d). The patient died suddenly five months after the diagnosis of PH.
Histological findings from heart and lung explants
The histological examination of the lung explant from patient 1 revealed interstitial fibrosis centered on and extending around the airways with bridges from one airway to another, resulting in a distortion of the lung architecture (Fig. 1e and f). Fibroblastic foci were almost always identified within the bronchial walls. Advanced bronchiolar lesions resulted in bronchiolar lumen occlusion (Fig. 1e). Alveolar spaces next to the fibrotic bronchi contained inflammatory exudates that were constituted of macrophages, lymphocytes and cholesterol clefts. The scarred lung parenchyma showed dense hyaline fibrosis without elastosis (Fig. 1g). Vascular remodeling was mainly identified within the fibrotic fields, where small muscular arteries were remodeled with intimal thickening overcoming the smooth muscle media (Fig. 1h). No plexiform lesion was identified. Venous remodeling was mild and no capillary hemangiomatosis was observed. Paraseptal and fibrotic thickening of the visceral pleura was also observed.
For patient 2, the histological examination showed a lung architecture that was markedly altered by collagen-rich interstitial fibrosis. Compared to patient 1, fibrosis appeared more advanced and destructive, resulting in honeycomb changes (Fig. 2e and f). Fibrotic lesions were rich in collagen, without elastosis and few fibroblastic foci. Vascular remodeling was also accentuated within fibrotic areas, involving mainly smooth muscle pulmonary arteries (Fig. 2g). Intimal thickening appeared to be prominent. Outside fibrosis alveolar walls were altered by inflammation, capillary dilation and cellular desquamation. Remodeling of pulmonary microvessels was also present (Fig. 2h).
The histological examination of the heart showed mild interstitial myocardial edema that was particularly visible around the small intramyocardial vessels (Fig. 4). Inflammation was rarely detected, but mainly located around the small vessels. Neither interstitial fibrosis nor cardiomyocyte necrosis was observed. Coronary vessels were patent without atheromatous lesions.
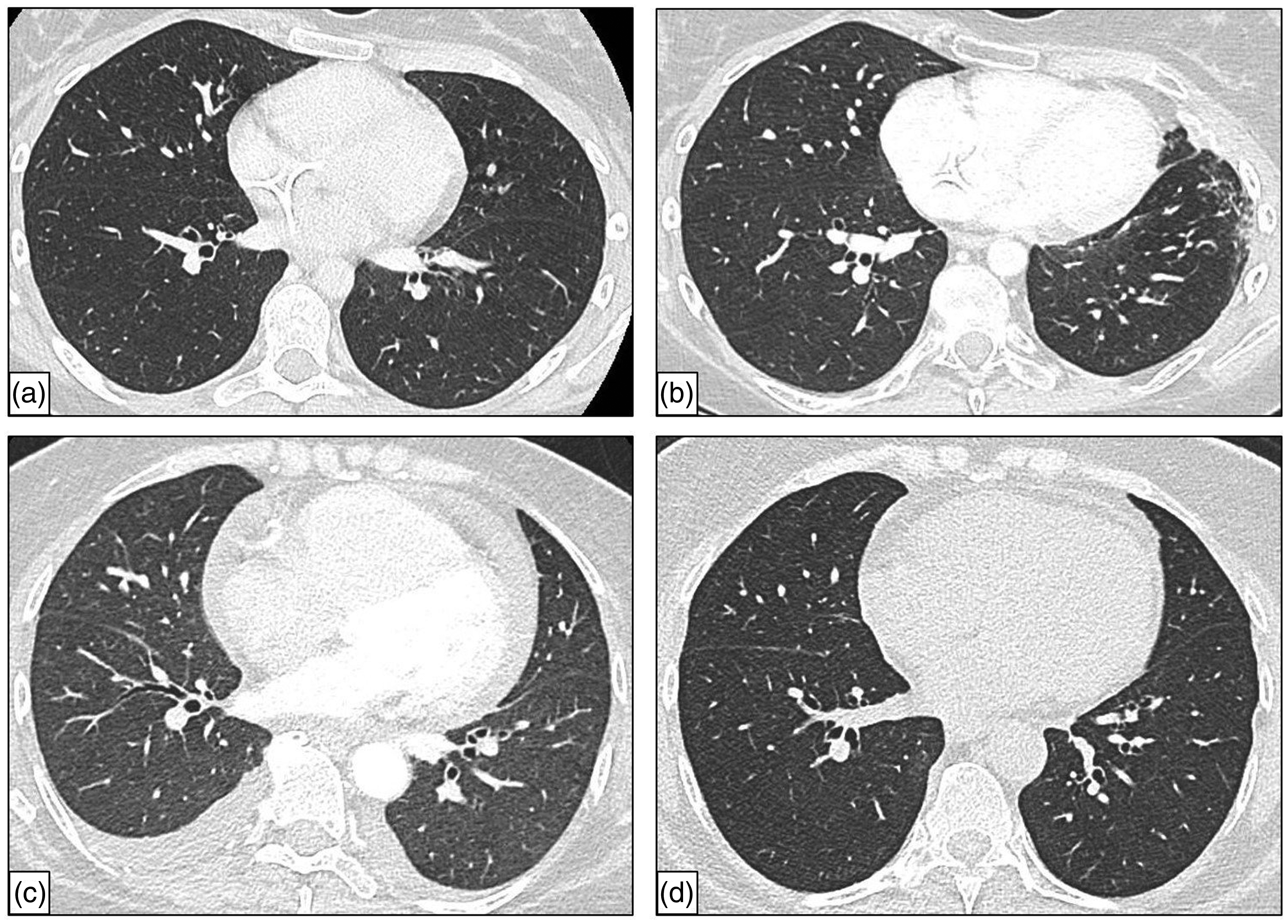
(a) At the baseline, no signs of PVOD or any parenchymal lesions are observed. (b) During follow-up, bronchiolitis obliterans signs appear with subpleural reticulations and localized fibrotic lesions in the left lower lobe and localized bronchiectasis and no mediastinal adenomegaly. (c) The CT of the chest revealed bilateral pleural effusion and pericardial effusion and a mediastinal adenomegaly. (d) The CT of the chest showed no evidence of a parenchymal disease and a partial regression of the mediastinal adenomegaly.
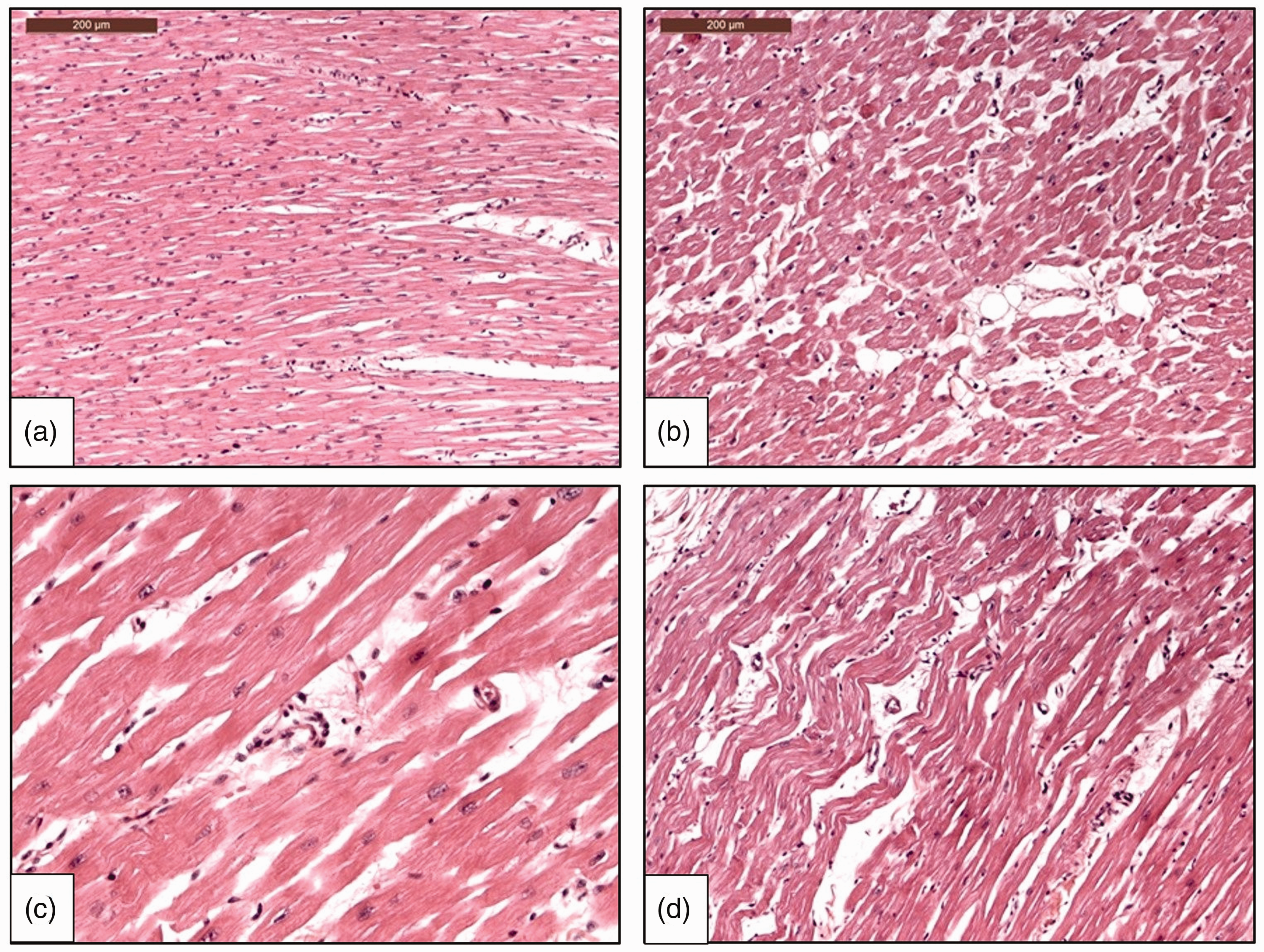
In the cardiac explant, the spacing between myocardial muscle fibers has been expanded by edema and a few adipose cells. The edema is more visible around the small intramyocardial vessels. Inflammation is seldom observed but is mainly located around the small vessels. Neither interstitial fibrosis nor cardiomyocytes necrosis are present.
Discussion
We described four case reports of patients who developed PH after ASCT preceded by conditioning regimen including busulfan. All patients developed subacute right heart failure requiring a management in an ICU 2 to 4.5 months after the administration of the conditioning regimen. In all cases, we observed a progressive improvement of PH after symptomatic management of right ventricular insufficiency and the administration of PAH-specific therapies and/or corticosteroids. However, two of the four patients developed a second case of progressive respiratory insufficiency due to severe interstitial lung fibrosis, requiring a double lung transplant. Our study is the first to report histological findings of severe pulmonary vascular and lung parenchymal toxicity induced by busulfan.
The four patients received at least two different lines of busulfan-free chemotherapy before the intensification regimen. The only drug in common to the four cases during the intensification regimen is busulfan. None of the four patients had any signs of right heart failure prior to the intensification regimen. Patient 2 had a reduced LVEF of 45% consecutively to anthracycline chemotherapy. Patient 4 had a history of ischemic cardiopathy with LVEF at 62%. None of the four patients had any parenchymal lung disease before the intensification regimen, except for the second patient who had previously existing bronchiectasis, without associated PH. Concerning the two patients who underwent double lung transplantation (Patients 1 and 2), radiological signs of interstitial lung disease appeared, respectively, 47 and 8 months after the diagnosis of PH. Right heart failure occurred rapidly after the intensification regimen. The specific chronology led us to consider busulfan as the most convincing candidate drug explaining PH occurrence.
Moreover, hemodynamic response of the four cases, after busulfan discontinuation and medical management, provides a strong argument towards a group 1 PH, when compared to the usual non-reversible evolution of other forms of PH.
Alkylating agents have been identified as the predominant chemotherapeutic class of drugs likely to induce toxic PH.8,10,12 Many drugs of this class, such as mitomycin C or cyclophosphamide, induce PH with venular involvement in human and animal models. Pulmonary vascular side effects of busulfan are less well known and have been poorly described. Recently, Levy et al. reported 22 cases of PH after bone marrow transplantation in children, and 21 of these patients were treated with busulfan, suggesting that this specific drug might be mainly involved in the development of the pulmonary vascular side effects described for this cohort. 13 Indeed, busulfan is an alkylsulfonate and alkylating agent. The main indication for this drug therapy remains chemotherapeutic intensification before ASCT. 14 Busulfan has been already identified as a major risk factor for the development of another form of severe vascular damage after hematopoietic stem cell transplantation, such as hepatic veno-occlusive disease. 15 Indeed, busulfan has been suspected to be associated with PAH in a pediatric cohort with ASCT. 11
Precapillary PH is characterized by the loss and obliteration of small precapillary pulmonary arteries, abnormal muscularization and perivascular inflammation. Pulmonary vascular remodeling results in a right-sided heart, a functional decline and shorter survival. Pulmonary endothelial dysfunction plays a primary role in vascular remodeling, leading to impaired endothelial-dependent vasodilatation, reduced anticoagulant properties, reactive oxygen species production, increased expression of adhesion molecules and the release of inflammatory cytokines and growth factors. 16 Busulfan has been described to induce acute endothelial injury through many pathophysiological pathways. This drug might induce a hypercoagulation state mediated by members of TGF-ß1 family and antifibrinolytic activity through plasminogen activator inhibitor (PAI)-1. 17 Moreover, busulfan induces an increase in the numbers of circulating endothelial cells and their progenitors. 18 In addition, the renewal of endothelial cell upon the occurrence of endothelial dysfunction appears to depend on resident progenitor cells, without the therapeutic effect of bone marrow transplantation. 19 In the four cases reported in our study, the patients developed a subacute form of PH within the four months following busulfan exposure. The clinical presentation was not specific and various signs of right heart failure were reported, including dyspnea and circulatory insufficiency, similar to the study by Levy performed in a cohort of children. 13 Interestingly, some patients had a positive vasoreactivity test during the first RHC. This is likely related to the severe and acute component of endothelial toxicity related to busulfan exposure.
We observed a rapid improvement of the PH after a corticosteroid treatment in three patients and the initiation of PAH therapies in two patients, but without complete normalization of hemodynamics. Busulfan is involved in overexpression of eNOS 20 and in increasing the level of circulating endothelial cells and their progenitors, 18 which can possibly explain the therapeutic effect of inhaled-NO in three out of four patients and the long-term responder profile of the patient 2 to calcium-channel blockers.
Three out of the four cases were treated with corticosteroids. One known property of corticosteroids is their action to inhibit the pulmonary arteries smooth muscle cells proliferation 21 through a reverse remodeling action 22 and several examples have attempt to prove that corticosteroids can reverse the inflammatory component of certain types of PH.23–25
We observed the persistence of severe vascular damage in lung explants from patients who underwent a double lung transplant with different patterns of lesions. The remodeling of muscular arteries with intimal thickening was the main feature, while venous remodeling was mild and no capillary hemangiomatosis was observed. Importantly, vascular lesions predominated in areas of pulmonary fibrosis, and a significant inflammatory component was observed in both patients.
Furthermore, busulfan is also involved in enhancing cellular aging 26 which might increase the levels of proinflammatory cytokines, matrix metalloproteases and epithelial growth factor, which participate in the pathogenesis of fibrosis, particularly in lung fibrosis. This finding might explain the onset of late lung fibrosis, e.g., bronchiolitis obliterans, in patients treated with busulfan. 27 Bronchiolitis obliterans is a less rare complication of ASCT,28–39 particularly after the administration of a busulfan chemotherapy as a unique risk factor,31,32,34,37 and is associated with a circulating immunoglobulin deficiency. 40 In patients with bronchiolitis obliterans, a good correlation with the pulmonary function testing36,38 is usually described, although contradictory findings have been reported. 41 The histological examination of the lung explants from our patients showed lesions of bronchiolitis obliterans that were mainly centered on the bronchiolo-vascular axes and interstitial and subepithelial fibrosis with a heterogeneous topography (patchy lesions) at two different stages in both of our patients. These descriptions are congruent with other available descriptions of busulfan-induced bronchiolitis obliterans,34,42,43 particularly the patchy distribution of the lesions. 34 The outcome of lung fibrosis after a busulfan chemotherapy remains a rare long-term complication that has been already reported.27,44–48
Vascular remodeling is often delicate to characterize in fibrotic area and our histopathological findings alone cannot confirm the vascular toxicity caused by busulfan. However, these findings further emphasize the precapillary nature of PH and are taken together with clinical histories and outcomes to associate PH occurrence with busulfan exposure.
Not all patients who receive busulfan chemotherapy experience PAH or bronchiolitis obliterans as complications, and different factors may explain this interindividual variability. These factors include age, 49 the route of administration,7,50,51 the precedence of other chemotherapies,52,53 the concomitant administration of other chemotherapies 52 and the interval between the administration of two different chemotherapies. 53 All our patients received a polychemotherapy regimen prior to ASCT that included melphalan. The busulfan-melphalan combination is attributed to increasing the incidence of PAH compared with other chemotherapy combinations. 11 Based on this findings, busulfan chemotherapy might be a necessary but not sufficient condition to induce the occurrence of PAH after the administration of a preparation regimen before ASCT, and busulfan acts synergistically with other alkylating agents such as melphalan in promoting the occurrence of PAH.
The main limitations of this report are the administration of multiple chemotherapies to all of our patients, and thus the precise role of busulfan in the onset of PH and bronchiolitis obliterans is complex. Additionally, ASCT itself is known to be responsible for the late onset of bronchiolitis obliterans, with specific pathophysiological hypotheses. 34 The main strengths of our study are the exhaustive histological description of the parenchymal, vascular and myocardial lesions related to busulfan and the description of two different stages of the same disease in two different patients.
In conclusion, acute precapillary PH associated with busulfan in the context of a preparation regimen administered prior to ASCT is a rare but severe and probably underestimated complication of this chemotherapy. This type of PH appears to be partially reversible in most cases by an appropriate treatment, including high-dose corticosteroids, but not necessarily specific medications. We provided histological evidence for diffuse pulmonary vascular damage. In addition, we provided an exhaustive histological description of bronchiolitis obliterans that occurred in two patients. These histological findings may improve our understanding of the still unclear mechanisms of the drug-related PAH pathophysiology and might lead to the consideration of alkylating agents as definite risk factors for PAH in future classifications, as this drug class is listed as a possible risk factor for PAH in the 2015 ESC/ERS Guidelines.10,54
Since PH remains a rare complication associated with busulfan, it is currently difficult to definitely confirm the direct toxicity of this agent on pulmonary vessels. Further in vitro and in vivo studies are needed to reinforce this hypothesis and possibly decipher the underlying mechanisms of vascular toxicity caused by alkylating agents such as busulfan.
Our report further highlights the necessity to raise clinical suspicion when PH occurs after busulfan-based chemotherapy. International collaborative databases covering all putative drug-associated PH exist and their importance is further emphasized by these clinical cases. A systematic referral of all suspicious cases would promote larger cohort studies.
Footnotes
Acknowledgements
We thank all physicians from the French Pulmonary Hypertension Network.
Conflicts of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
Conception and design: D Montani, L Savale. Data collection: D Montani, L Savale, J Hagenburg, B Lechartier, MR Ghigna. Data analysis and interpretation: J Hagenburg, L Savale, B Lechartier, MR Ghigna, MC Chaumais, X Jaïs, O Sitbon, M Humbert, D Montani. All authors approve and revise the manuscript.
