Abstract
Despite the increasing trends, reports on long-term follow-up are limited on transitioning from parenteral to oral treprostinil therapy in patients with pulmonary arterial hypertension (PAH). We investigated both the effectiveness of parenteral to oral treprostinil transition and the characteristics associated with transition failure over a duration of two years. The study included 37 Group I functional class I and II patients with PAH on combination therapy. Patients were excluded if cardiac index ≤2.2 L/min/m2, right atrial pressure ≥11 mmHg, or 6-min walk distance ≤250 m. Patients were categorized as successful (
Background
Pulmonary arterial hypertension (PAH) is a serious and life-altering disease. Though not curable, available therapies help to alleviate symptoms and improve quality of life for patients. The treatment options for PAH are rapidly changing, as is the approach to management. Initial management can often include a monotherapy or combination of therapies. Previous work has demonstrated a lower risk of clinical decline with combination therapy in a subset of patients than with monotherapy 1 and profound hemodynamic improvement even in severe cases. 2 Combination therapy with parenteral prostacyclin class agents is typically prescribed for the sickest patients but is associated with increased burden for patients with infusion-related side effects including the inconvenience of a pump and the risk of infection.
Treprostinil, a prostacyclin analog, infusion therapy is one of the cornerstones in PAH management that has shown success with improvement of hemodynamics and symptoms in many patients.3,4 After development as a tablet formulation (known as Orenitram)® in 2013, recent data demonstrate successful transition from subcutaneous parenteral to oral treprostinil therapy for up to 24 weeks in PAH patients who are functional class (FC) I/II with normalized cardiac index (CI) and right atrial pressures (RAP), and have been on stable doses of parenteral treprostinil therapy. 5 With these findings, the transition from parenteral treprostinil to its oral form in more patients may be anticipated.
Given pharmacokinetic data indicate that the area under the curve (AUC) is similar between parenteral and oral formulations of treprostinil, 5 it is possible that clinicians may attempt transition in patients with advanced disease, such as high right ventricular (RV) afterload. Despite similar overall AUC, serum peaks/troughs may be poorly tolerated in a patient with high afterload state and a marginally adapted RV, as opposed to parenteral delivery. This scenario may explain why our anecdotal long-term observations indicate that a subset of patients with initially compensated RV function fail transition after the 24-week period. Although it is understood that some patients can be successfully transitioned to oral therapy, we do not know which subset of patients can be safely targeted for transition. As hemodynamics are important markers of disease severity in PAH patients, 6 our aim was to compare resting hemodynamics in patients who have been successfully and unsuccessfully transitioned to long-term oral treprostinil in an effort to identify safe candidates for transition.
Methods
Study design and patient selection
Thirty-seven patients with Group I PAH provided written informed consent to participate in the study, which was approved by the institutional review board at the University of Arizona (Tucson, AZ, USA) and St. Joseph’s Medical Center (Phoenix, AZ, USA) (Fig. 1). The diagnosis of PAH was established by dedicated PAH providers with invasive confirmation (based on right heart catheterization [RHC]) based on updated guidelines.
7
All patients included in the study were FC IV at diagnosis (Table 3) and therefore were initiated on parenteral therapy. All patients were eventually treated with combination therapy by adding a phosphodiesteras-5 (PDE5) inhibitor and/or endothelin receptor (ERA) antagonist within the first month. All enrolled patients were administered combination therapy for >90 days and on a stable dose for >30 days. All individuals were WHO FC I–II. Participants with FC III–IV, a CI ≤ 2.2 L/min/m2, a RAP ≥ 11 mmHg, and/or a 6-min walking distance (6MWD) ≤250 m were excluded.
Disposition of study cohorts relative to transition group. Of the patients initially screened, six were excluded due to protocol deviation, poor drug adherence, and/or clinical instability. Timescales in the margins represent mean months between assessments by parenteral comparator (cParenteral), left margin, and successful transition (STransition)/unsuccessful transition (UTransition), right margin. Patients are assigned here to STransition or UTransition here based on assessment at each visit (as opposed to final assignment at last assessment). All patients had a RHC at enrollment and at least one follow-up RHC. A subset had more than one follow-up RHC.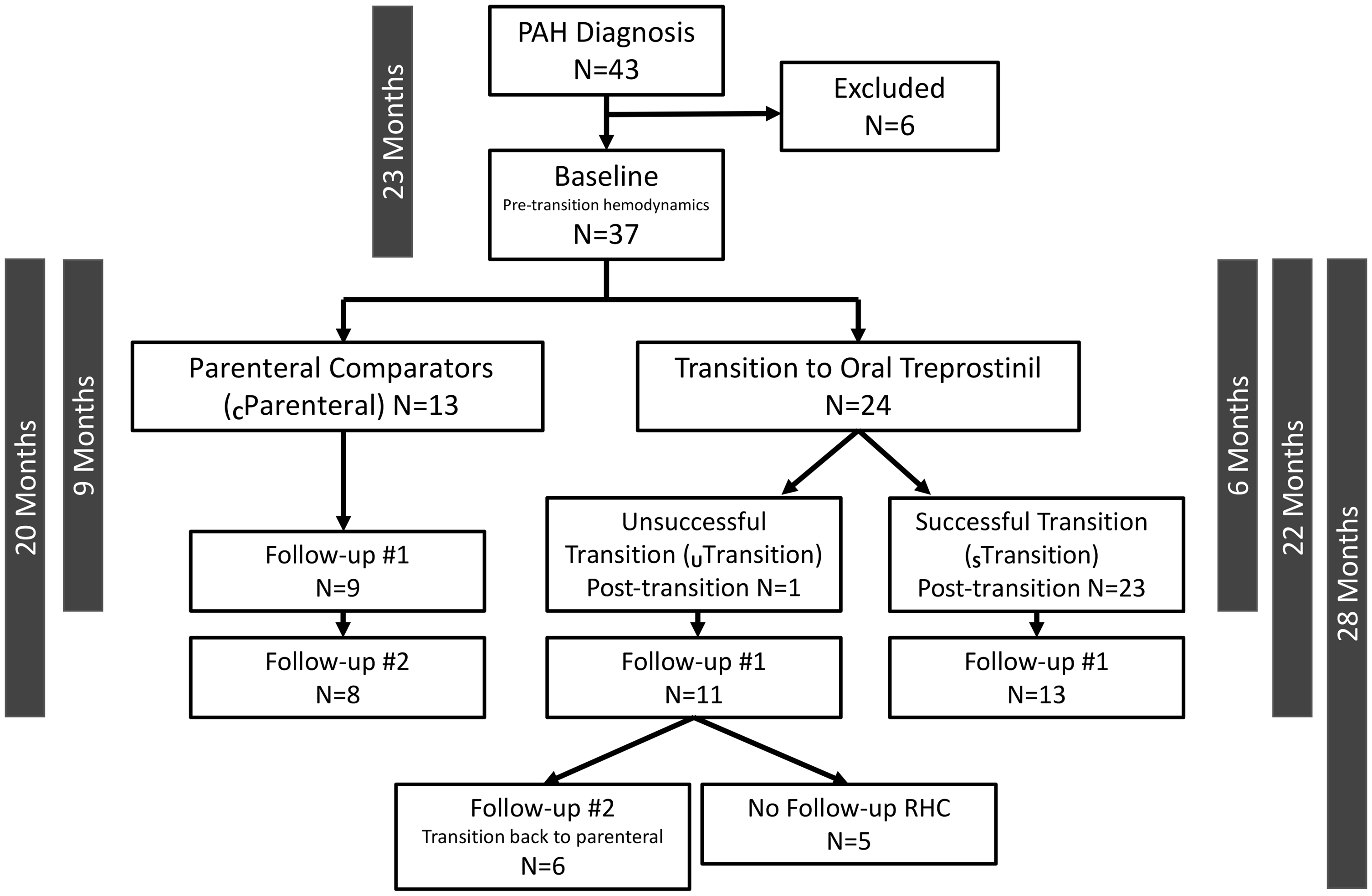
Only patients who underwent parenteral to oral treprostinil transition following a previously reported transition protocol
5
were included (see transition protocol below). Patients were classified by the principal investigator (PI) at the time last assessment as:
‐ Successful transition (STransition): Continued FC I-II AND unchanged hemodynamics >24 weeks from transition; ‐ Unsuccessful transition (UTransition): FC deterioration to III/IV and hemodynamic deterioration or unable to tolerate sufficient oral trerpostinil to maintain disease stability; ‐ Parenteral comparator (CParenteral): Individuals eligible to enroll (see inclusion/exclusion criteria) but did not transition due to early trial closure or personal choice to remain on parenteral treprostinil. The purpose of this group was to “control” for disease progression (vs. transition failure).
Upon assessment of transition failure, it has been our standard of care to transition patients back to parenteral treprostinil before initiation of a second oral, if the patient was only on one oral agent.
Transition protocol
Baseline pre-transition hemodynamics and RV function were assessed with RHC, echocardiography, 6MWD, and measurement of brain natriuretic peptide (BNP) and were collected at the time of enrollment. Participants who transitioned were hospitalized for supervised transition from parenteral to oral treprostinil for <5 days. Titration and transition of parenteral and oral treprostinil were performed based on study protocol. 5 The patients were transitioned gradually in a stepwise fashion reducing parenteral treprostinil while simultaneously increasing oral treprostinil. Goal dose of oral treprostinil was based on previous experience that 1 mg oral treprostinil TID is equivalent to 6 ng/kg/min parenteral treprostinil. 5 Relatively small dose adjustments were made after discharge based on tolerance and side effect mitigation.
Assessments
In addition to baseline RHC measurements, transition individuals were evaluated with clinic visits monthly for three months then every three months for 12 months. Transition patients were also followed with a RHC on at least two separate occasions defined as follow-up visits 1 and 2 (Fig. 1). These latter RHC evaluations were clinically indicated occurring at varying times. For the transition groups (
Pulmonary vascular resistance (PVR) was calculated based on measurements derived during RHC and defined as ([mean pulmonary artery pressures (mPAP) – wedged PAP (PCWP)]/cardiac output [CO]). Pulmonary artery capacitance was defined as stroke volume/systolic PAP-diastolic PAP. Echocardiography was performed by technicians specifically trained in right heart analysis. Images were obtained using commercially available equipment (GE, Philips) with a 3.5-MHz transducer in standard views with the patient in the left decubitus position. Standard M-mode and two-dimensional images were obtained during a breath-hold according to American Society of Echocardiography Guidelines. 8 RV end-diastolic area (RVEDA), RV end-systolic area (RVESA), RV fractional area change, % (RVFAC=(RVEDA−RVESA)/RVEDA×100), and tricuspid annular plane systolic excursion (TAPSE) were obtained as echocardiographic measures of RV function.
Statistical analysis
All analyses were conducted in program R (version 3.3.2
9
). For continuous variables, summary statistics are given as mean ± 1 standard deviation (SD) or mean (range), unless otherwise noted. Comparisons among the three groups were conducted with Kruskal–Wallis rank-sum tests with P values adjusted for false discovery rate (FRD) using the method of Benjamini and Hochberg.
10
Where there were statistically significant differences among the three groups (α = 0.05), Wilcoxon rank-sum tests were used to test for differences between each pair of groups (these post-hoc tests were not adjusted for multiple comparisons). A Cox regression analysis was done to analyze the relationship of baseline hemodynamic and RV function variable to clinical failure. Receiver operating characteristic (ROC) curves were used to compare the variables with the highest hazard ratios from the Cox analysis for their utility to predict successful transition. Only individual in the
Results
Demographics, clinical characteristics, and treprostinil dosing
Demographics and clinical characteristics of the study cohort.

Values are presented as n (%) or n (range) where applicable.
STransition, successful transition; UTransition, unsuccessful transition; CParenteral, parenteral comparator; BSA, body surface area; iPAH, idiopathic PAH; CTD, connective tissue disease-associated PAH; CHD, congenital heart disease-associated PAH; ERA, endothelin antagonist; PDE5, phosphodiesterase 5 inhibitor; SC, subcutaneous; IV, intravenous.
Treprostinil dosing characteristics of the transition cohort.
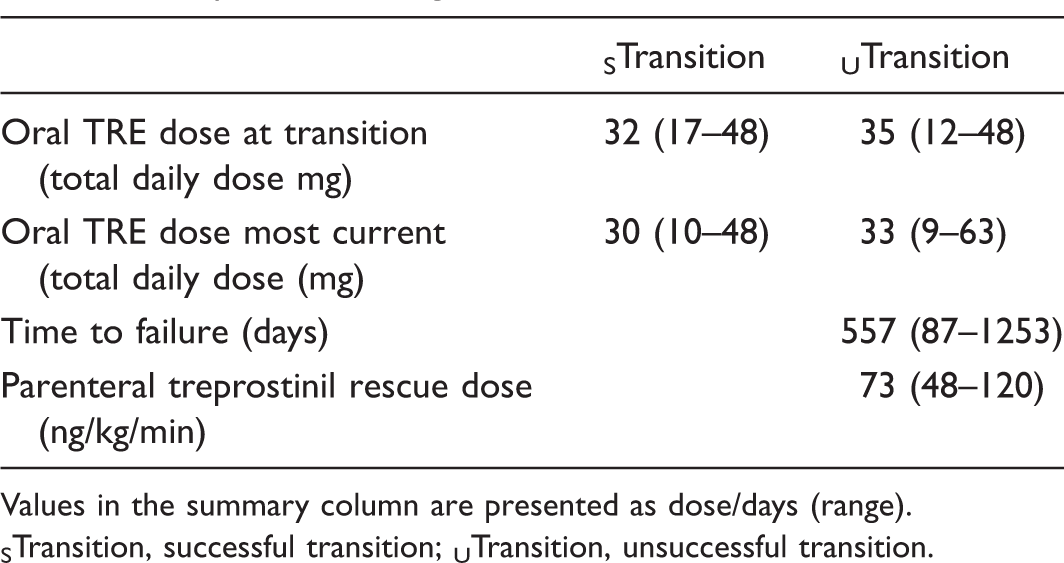
Values in the summary column are presented as dose/days (range).
STransition, successful transition; UTransition, unsuccessful transition.
Hemodynamics and RV function by time point and transition cohort.
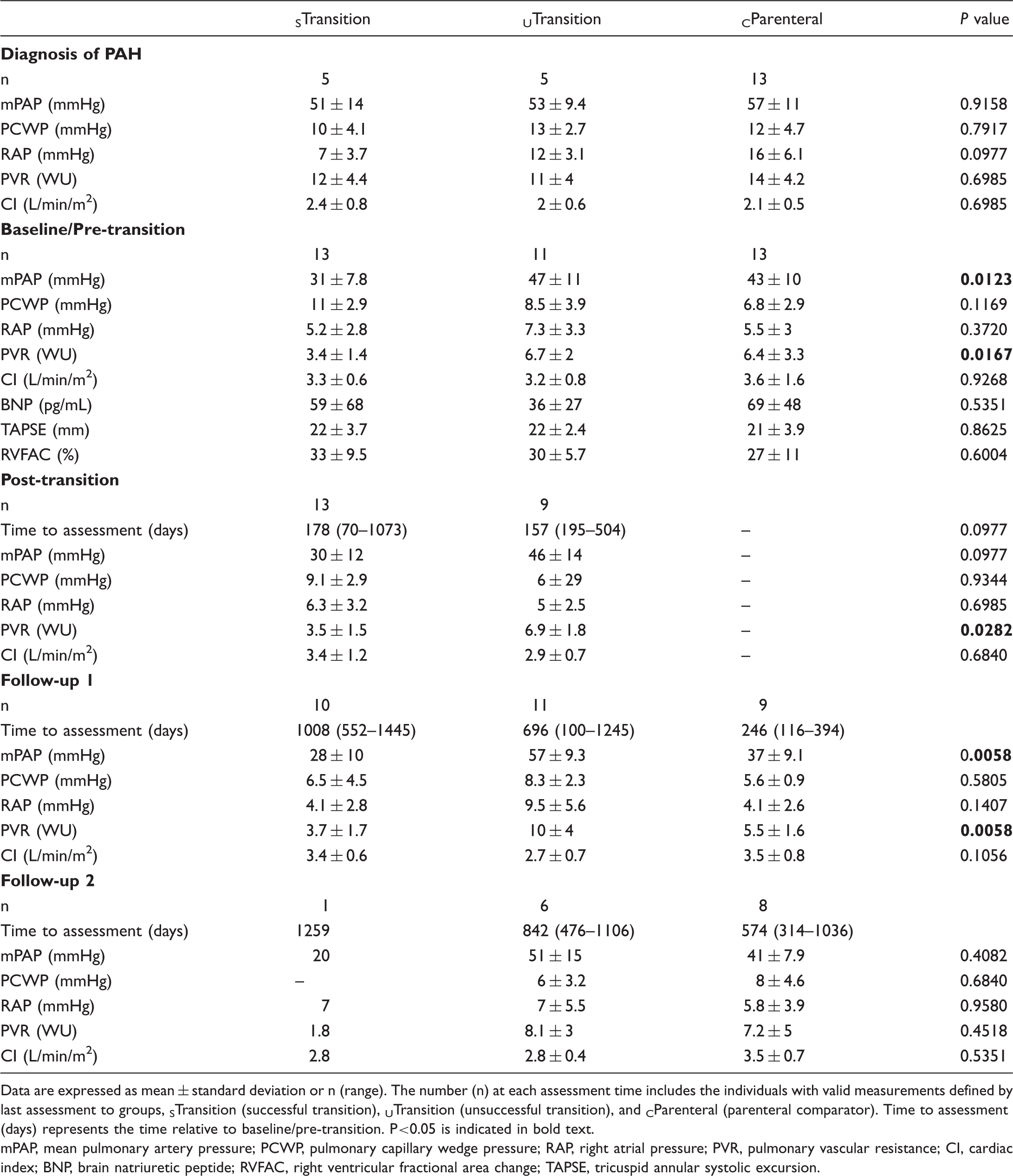
Data are expressed as mean ± standard deviation or n (range). The number (n) at each assessment time includes the individuals with valid measurements defined by last assessment to groups, STransition (successful transition), UTransition (unsuccessful transition), and CParenteral (parenteral comparator). Time to assessment (days) represents the time relative to baseline/pre-transition. P<0.05 is indicated in bold text.
mPAP, mean pulmonary artery pressure; PCWP, pulmonary capillary wedge pressure; RAP, right atrial pressure; PVR, pulmonary vascular resistance; CI, cardiac index; BNP, brain natriuretic peptide; RVFAC, right ventricular fractional area change; TAPSE, tricuspid annular systolic excursion.
Hemodynamic and RV function measurements
At diagnosis, there were no significant differences in mPAP, PVR, CI, or RAP between groups (P > 0.05) although all patients generally had very elevated mPAP, depressed CI, and elevated RAP (see Table 3). mPAP and PVR at enrollment discriminated
The median time from enrollment/pre-transition to post-transition assessment were similar, about 24 weeks, between the Change from pre-transition PVR after transition, follow-up 1, and follow-up 2 by transition cohort. The high PVR in the unsuccessful transition (UT) group at enrollment was also accompanied by increasing PVR at follow-up 1. This change in PVR was not reversed by follow-up 2 by “rescue” therapy. *Denotes that mean PVR by follow-up 1 was significantly higher in the UT vs. successful transition (ST) (P = 0.0001) and the UT vs. parenteral comparator (CP) groups (P = 0.003).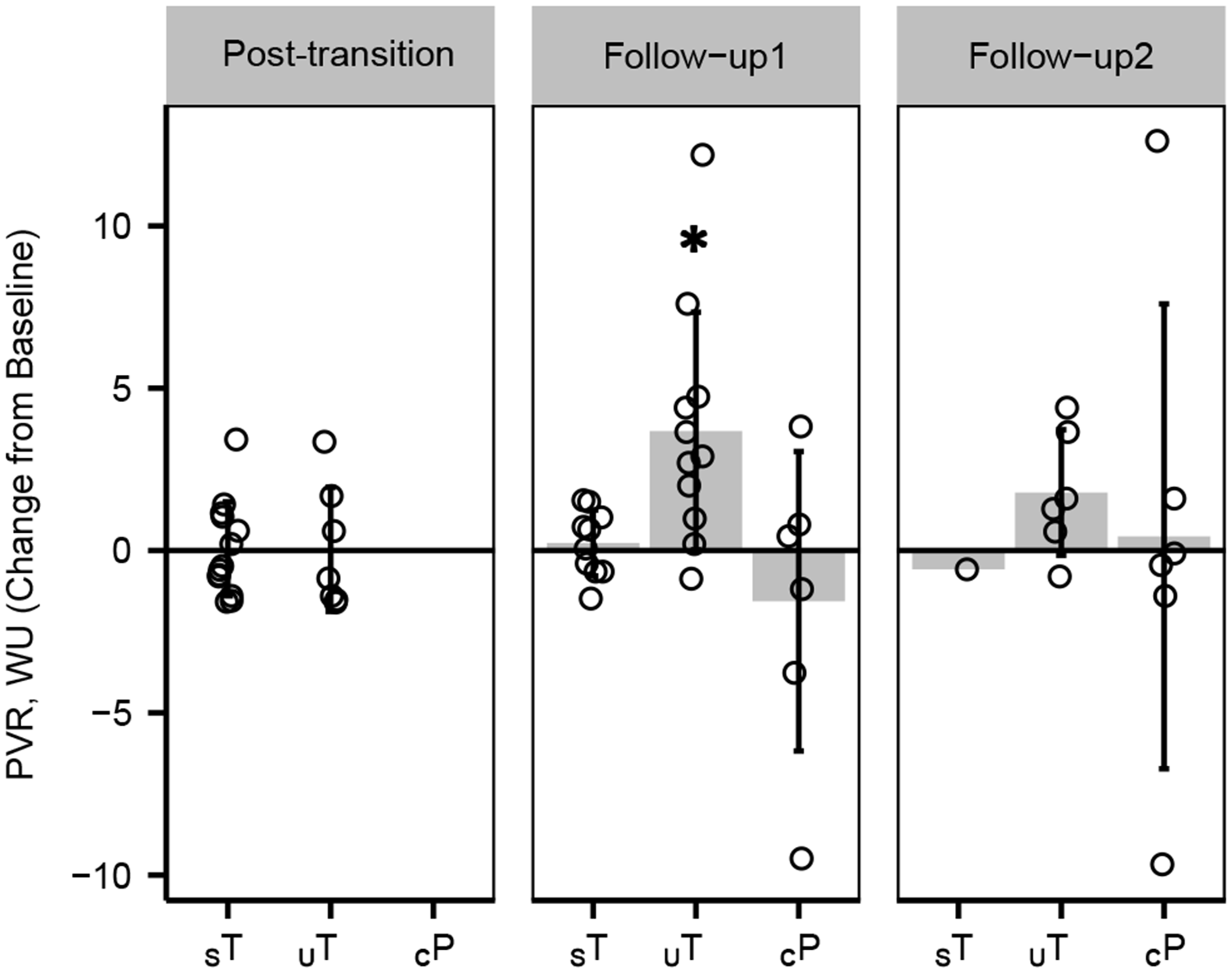
Pre-transition prediction of transition success and failure
Cox regression analysis using baseline hemodynamic, RV function, and treprostinil dosing variables to predict transition failure.
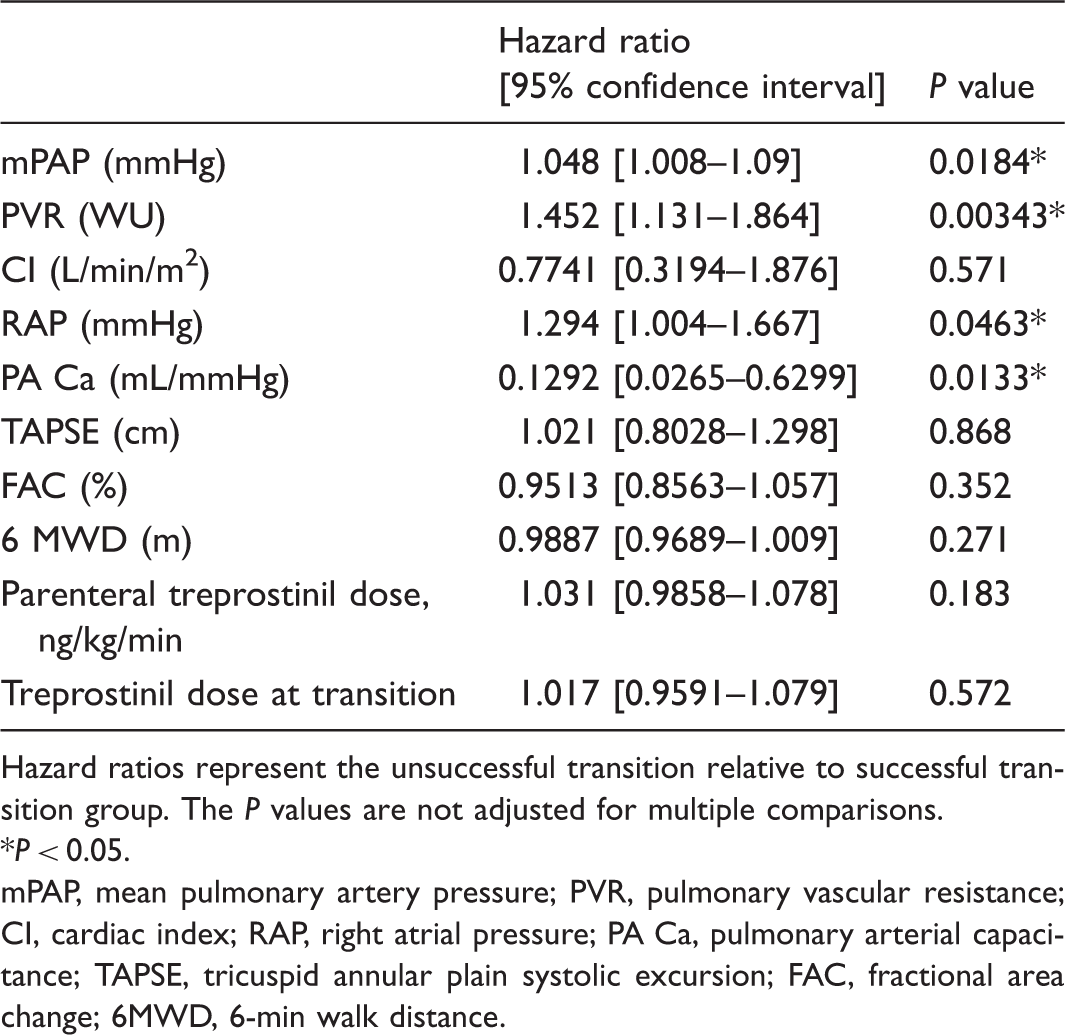
Hazard ratios represent the unsuccessful transition relative to successful transition group. The P values are not adjusted for multiple comparisons.
P < 0.05.
mPAP, mean pulmonary artery pressure; PVR, pulmonary vascular resistance; CI, cardiac index; RAP, right atrial pressure; PA Ca, pulmonary arterial capacitance; TAPSE, tricuspid annular plain systolic excursion; FAC, fractional area change; 6MWD, 6-min walk distance.
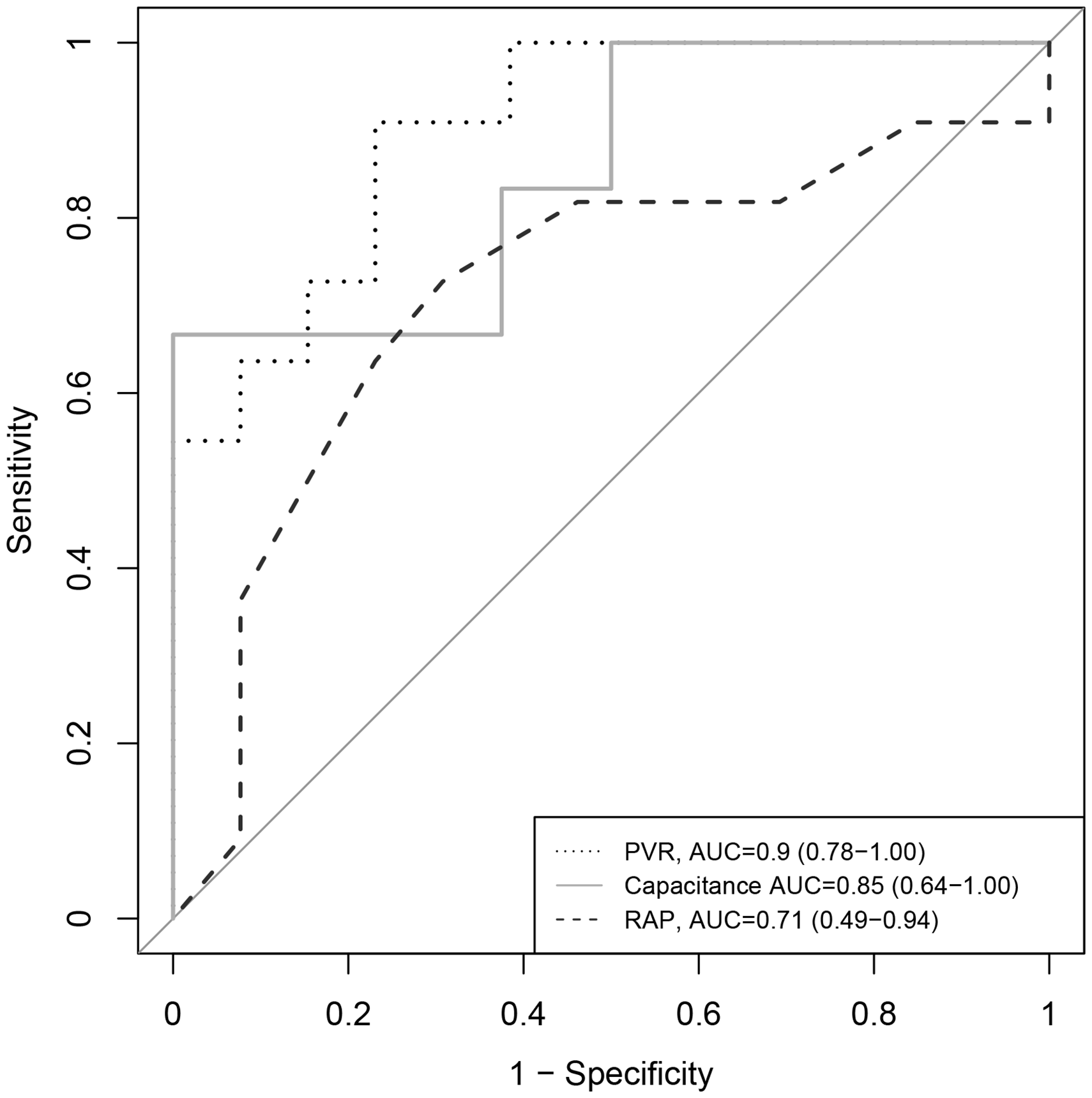
ROC curves on RV afterload and performance at pre-transition enrollment. PVR demonstrated superior (AUC 0.9 [range = 0.78–1.0], P = 0.0001) accuracy in prediction of transition success than do capacitance and RAP. The optimal PVR cut-off was 4.16 WU, 2.2 mL/mmHg for capacitance, and 6.5 mmHg for RAP.
Discussion
To our knowledge, this study is the first protocolized parenteral transition trial with long-term follow-up in the combination treatment era. The findings demonstrate, in contrast to the initial 24-week trial,
5
a higher rate of transition failure. Transition failure is accurately predicted by PVR at enrollment which increases on follow-up beyond a similar group of patients remaining on parenteral therapy. Transition appeared successful in nearly all patients with enrollment PVR ≤ 4.16 WU to a mean follow-up period of 1008 days (range = 552–1445 days; nearly three years). Additionally, “rescue” therapy was unable to bring PVR back to baseline levels in a subset of patients in the
All patients in this trial adhered to the initial trial transition protocol. 5 During the initial trial, one patient (3%) and three more (11%) required parenteral “rescue” during the 24-week and extension periods (median = 214 days follow-up), respectively. What is unique to this trial cohort is that the patients participated in follow-up over twice as long as the 24-week and extension periods combined. The average enrollment PVR of our transition groups (approximately 5.0 WU) was very similar to the baseline mean PVR (5.2 WU) in the initial trial. Time since diagnosis, RV function, and parenteral and oral treprostinil dosing at enrollment were also similar between the trials. Although the pharmacokinetic AUC was similar to parenteral to oral in that study, 5 small perturbations may be poorly tolerated in a marginally adapted RVPA system. Therefore, we theorize that the higher failure rate in our trial was due to other pharmacokinetic factors (such as greater peaks and troughs), between oral and parenteral treprostinil, in the context of high RV afterload.
Our trial is also unique including a parenteral comparator (
Previous attempts at transitioning from parenteral to oral therapy in patients with PAH have met limited success.
13
Both prospective14–16 and retrospective17–20 studies evaluating the transition from inter-class parenteral to oral medications have found similarly that baseline hemodynamics generally predict probability of success. The study with the longest follow-up by Escolar et al.
20
demonstrated a similar 50% success rate after a mean >30-month (>900 days) follow-up from parenteral treprostinil to oral PDE5i/ERA. In this trial, both mPAP and PVR were predictive of transition success similar to our trial. Given that the successful transitions in the above study had a similar initial response to parenteral therapy as our
Our trial has several limitations. It is retrospective in design and therefore many measures such as 6MWD, BNP, and echocardiography data are incomplete. Another limitation is the small number of participants enrolled in this trial. However, the exclusionary nature of the enrollees was necessary to maintain control of many factors including dosing and follow-up testing. Next, due to study design, it was challenging to quantify medication-related adverse events. Therefore, it was difficult to exclude the possibility that some of the transition failures may also be in part due to side effects limiting optimal dosing. Lastly, given recent data demonstrating significant effects of triple upfront therapy 2 in FC IV patients and superior efficacy of upfront ERA/PDE5i therapy, 1 it is possible that if all our patients were on this combination that additional failures could have been avoided.
In conclusion, our data suggest that in a subset of PAH patients on combination background therapy, intra-class transition from parenteral to oral treprostinil is successful in the long term in 54% of participants. Pre-transition PVR can accurately predict failure which occurs gradually >24 weeks later. This deterioration in hemodynamics appears greater than that if they had stayed on parenteral treprostinil and may not be reversed by “rescue” therapy. Therefore, these authors recommend extreme caution when transitioning patients with greatly elevated PVR. Future directions in transition research may reveal measurable differences in imaging or biomarkers that will obviate the need for catheterization to predict success.
Footnotes
Conflict of interest
FPR has received grant funding and consultancy and advisory board compensation from Gilead, Actelion Pharmaceuticals, Bayer and United Therapeutics. JF serves as consultant to United Therapeutics, Gilead, Actelion, Bayer and Reata. He serves as speaker for Gilead, Bayer, and United Therapeutics.
Funding
FPR is funded by the National Heart Lung and Blood Institute (NHLBI U01 grant RFA-HL-14-027). AAD is funded by the NHLBI R01 HL 136603. SA has received advisory board compensation from Bayer.
