Abstract
The feasibility and usefulness of routine cardiac magnetic resonance imaging (CMR) in the management of idiopathic pulmonary arterial hypertension (IPAH) is unknown. The aims of the study were: 1) to study whether a decrease in CMR-derived right ventricular ejection fraction (RVEF) coincides with clinical deterioration; 2) to determine whether RVEF is responsive to early escalation of pulmonary arterial hypertension (PAH)-specific therapy. This was a prospective study including 30 incident IPAH patients. Patients underwent right heart catheterization and CMR at regular follow-up visits (baseline, four, eight, 12, 24 months; no right heart catheterization at eight months). New York Heart Association (NYHA) functional class II patients started with monotherapy (endothelin receptor antagonist or phosphodiesterase-5-inhibitor) and NYHA III patients with combination therapy (endothelin receptor antagonist plus phosphodiesterase-5-inhibitor). In the case of a deterioration in RVEF of more than 3% compared with the previous measurement, PAH-specific therapy was added (i.e. treatment escalation). In 11 patients without signs of clinical deterioration, a greater than 3% decrease in RVEF occurred. After treatment escalation, RVEF significantly improved (average improvement of 7%, p = 0.009) whereas right ventricle volumes, N-terminal pro-brain natriuretic peptide and six-minute walking distance remained stable. Clinical worsening did not occur after escalating therapy. Throughout the study, four patients presented with clinical worsening, despite a stable RVEF. Three of these four patients had a baseline RVEF <35%. In IPAH patients presenting with an early decrease in RVEF but otherwise stable disease, progressive right ventricle failure and subsequent clinical worsening did not occur when therapy was escalated. Nevertheless, clinical worsening did occur in patients with a low baseline RVEF.
Keywords
Introduction
Idiopathic pulmonary arterial hypertension (IPAH) is a haemodynamic condition in which increased pulmonary artery pressures (PAPs) and pulmonary vascular resistance (PVR) ultimately lead to right ventricular (RV) failure and death.1–3 Multiple drugs are available that all successfully reduce PVR but overall survival in IPAH remains poor.4–6 Preserving RV function is considered as the optimal treatment goal to improve survival. 7 In fact, treatment goals stated in the guidelines are reflective of RV function and include right atrial pressure, cardiac index and six-minute walking distance. 1
The gold standard for quantifying RV function is RV ejection fraction (RVEF), measured by cardiac magnetic resonance imaging (CMR). Previous work has shown that CMR-derived RVEF gradually worsens over time in patients showing late disease progression. 8 As such, it may be hypothesized that routine monitoring of RVEF by CMR may allow early therapeutic interventions before clinical deterioration becomes manifest. However, the role of CMR in the disease management for patients with IPAH is unknown. Therefore, we designed and implemented a prospective standardized CMR-based treatment strategy. Here, we present the clinical data of a cohort of IPAH patients in whom therapy was rapidly escalated upon a deterioration in RVEF. We aimed to explore the feasibility and usefulness of this CMR-based strategy by studying: 1) whether a decrease in RVEF coincides with clinical deterioration and 2) whether adjusting pulmonary arterial hypertension (PAH)-specific therapy on the basis of early changes in RVEF allows long term preservation of RV function.
Methods
Patients and study design
In this prospective study 30 treatment-naïve patients referred to the VU University Medical Centre between January 2012 and September 2015 were included. Inclusion criteria were: diagnosis of IPAH or hereditary PAH (HPAH), age 18–80 years and New York Heart Association (NYHA) functional class II and III. A diagnosis of IPAH/HPAH was established in patients with a mean PAP (mPAP) above or equal to 25 mmHg and a pulmonary arterial wedge pressure (PAWP) below 15 mmHg, after the exclusion of other conditions associated with pulmonary hypertension. 1 Exclusion criteria were contra-indications for CMR.
Throughout the study, therapy was escalated by prescribing additional PAH-specific therapy upon a deteriorating RVEF. Clinical stability was defined by the absence of clinical worsening (defined as either hospitalization for worsening PAH or NYHA III–IV symptoms combined with a decrease in six-minute walking distance (6MWD) >15% 9 ).
All patients underwent baseline CMR, right heart catheterization (RHC), blood sampling (N-terminal pro-brain natriuretic peptide (NTproBNP)) and a 6MWD. These measurements were repeated after four, eight, 12 and 24 months of follow-up. At eight months of follow-up, no RHC was performed. Patients with a deterioration in RVEF at 24 months underwent one additional CMR within the first year after their last study visit.
This study conforms to the Declaration of Helsinki ethical guidelines and was approved by the local Medical Ethics Committee (approval number 2013/58, CCMO number NL4187802913). Written informed consent was obtained for all patients.
Treatment regimens
Patients with NYHA functional class II started with upfront oral monotherapy (phosphodiesterase-5-inhibitor PDE5-inhibitor (PDE5I) or an endothelin receptor agonist (ERA)). Patients with NYHA class III started with upfront combination therapy (PDE5I plus an ERA). During follow-up, changes in treatment regimens were made based on RVEF, measured by CMR. In the case of a stable/improved RVEF during follow-up (i.e. decrease in RVEF of less than 3%), treatment remained unchanged. In the case of a deterioration in RVEF of more than 3% (based on a previous repeatability study 10 ), therapy was escalated, leading to either combination or triple therapy.
Study procedures
Right heart catheterization
Haemodynamic assessment was performed with a 7F balloon tipped, flow directed Swan-Ganz catheter as described previously. 3
Cardiac magnetic resonance imaging
CMR was performed on a Siemens 1.5-Tesla Avanto scanner (Siemens, Medical Solutions, Erlangen, Germany). CMR data acquisition and postprocessing were all performed according to standard protocols. 11 In short, during postprocessing, short-axis images were manually delineated to obtain ventricular volumes using the MASS-software package (MEDIS, Medical Imaging Systems, Leiden, The Netherlands). On end-diastolic images (first cine image after the R-wave trigger) and end-systolic images (cine image with visually the smallest cavity area), endocardial contours of the RV and left ventricle were manually traced by three experienced and independent observers, unaware of patients’ clinical status. RVEF was calculated as stroke volume (SV) divided by end-diastolic volume (EDV) and multiplied by 100%, SV was calculated as EDV minus end-systolic volume. Ventricular volumes were estimated using the Simpson rule, excluding papillary muscles and trabeculae.
Clinical data
Data on hospitalization, signs of RV failure and potential side effects were monitored and treated as in our daily clinical practice. We also performed a post-hoc analysis to assess whether patients were at a low or high risk of clinical deterioration at the time of a deteriorating RVEF. In order to do this, we retrospectively identified three non-invasive criteria derived from the risk assessment table from the guidelines (NTproBNP <300 ng/ml, NYHA class I–II and 6MWD >440 m). 12
Statistical analysis
Data are presented as mean (±SD) for normal distributed data and absolute numbers (%) or median (min–max) for non-normal distributed or categorical variables. Comparisons of CMR variables and haemodynamics obtained after four, 12 and 24 months of follow-up were made using repeated measures analysis of variance for normally distributed variables and after log-transformation for non-normally distributed data, with Bonferroni post hoc tests. Changes in RVEF, RV volumes, 6MWD and NTproBNP levels after escalating therapy were assessed using paired T-tests. Analyses were performed using SPSS 20.0. Throughout the analyses a p-value <0.05 was considered statistically significant.
Results
Baseline parameters and treatment
Baseline characteristics for the total cohort, patients who were stable during follow-up and patients with a decrease in RVEF>3% during follow-up (unstable RVEF).
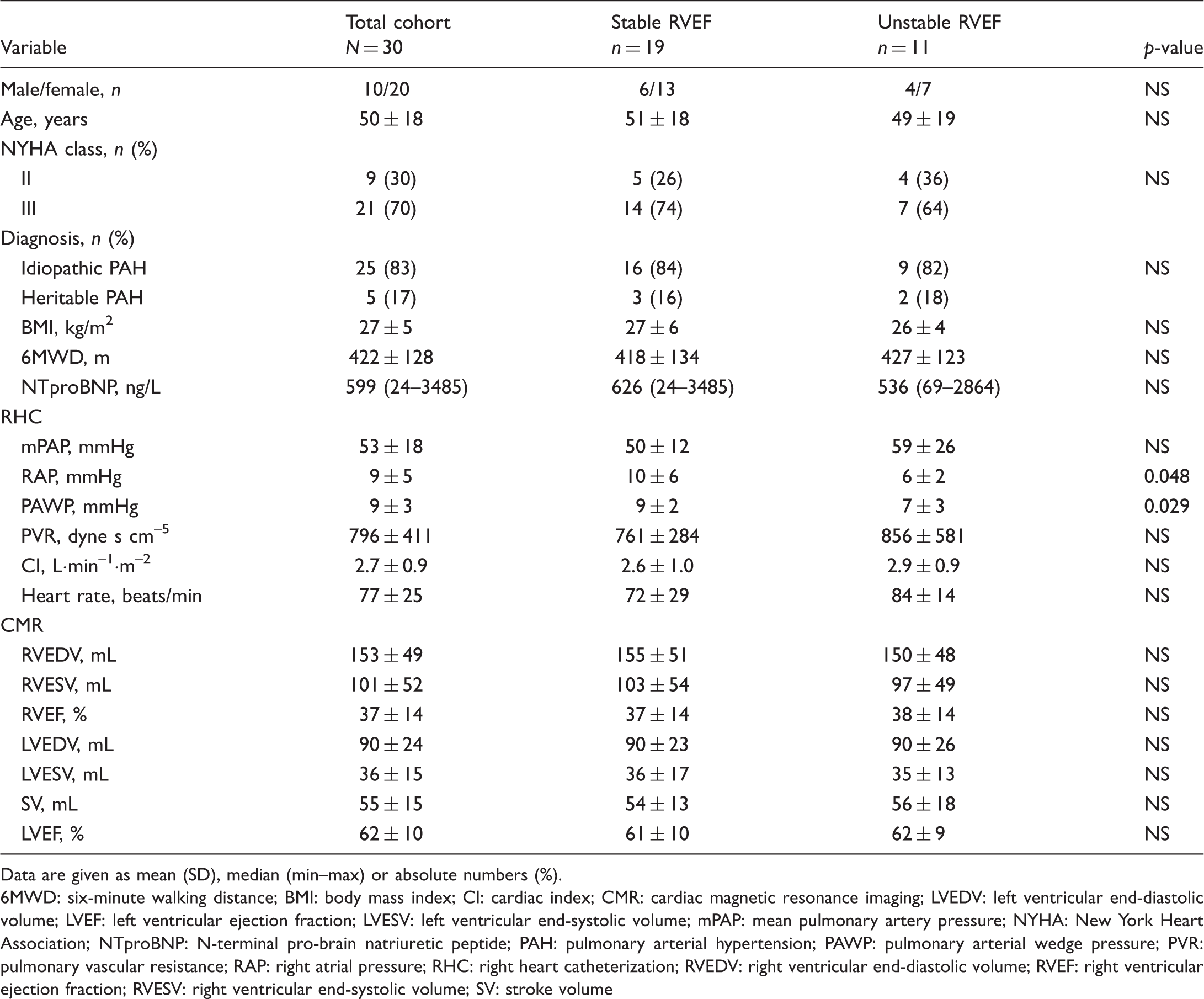
Data are given as mean (SD), median (min–max) or absolute numbers (%).
6MWD: six-minute walking distance; BMI: body mass index; CI: cardiac index; CMR: cardiac magnetic resonance imaging; LVEDV: left ventricular end-diastolic volume; LVEF: left ventricular ejection fraction; LVESV: left ventricular end-systolic volume; mPAP: mean pulmonary artery pressure; NYHA: New York Heart Association; NTproBNP: N-terminal pro-brain natriuretic peptide; PAH: pulmonary arterial hypertension; PAWP: pulmonary arterial wedge pressure; PVR: pulmonary vascular resistance; RAP: right atrial pressure; RHC: right heart catheterization; RVEDV: right ventricular end-diastolic volume; RVEF: right ventricular ejection fraction; RVESV: right ventricular end-systolic volume; SV: stroke volume
Clinical stability and the effect of treatment escalation on RVEF
During 24 months of follow-up, 19 patients had a stable RVEF and thus treatment was not escalated. Eleven patients (37%) had a decrease in RVEF >3% (four (44%) on initial monotherapy and seven (33%) on initial combination therapy). A decline in RVEF was not necessarily observed at the same time point for every patient (Supplementary Fig. B); a decline occurred in five patients after eight months of follow-up, in three patients after 12 months of follow-up and in two patients after 24 months of follow-up. One patient had a decrease in RVEF at both eight months and 24 months of follow-up, resulting in 12 decreases in RVEF in total. A decline in RVEF was observed in patients with otherwise stable disease as no patients with a decline in RVEF presented with signs of clinical worsening.
After escalating therapy, all patients had a stable or improved RVEF at the subsequent follow-up measurement (average RVEF improvement of 7%, p = 0.009; Fig. 1). In addition, no clinical worsening occurred after escalation of treatment.
Changes in right ventricular ejection fraction (RVEF) in patients with an unstable RVEF.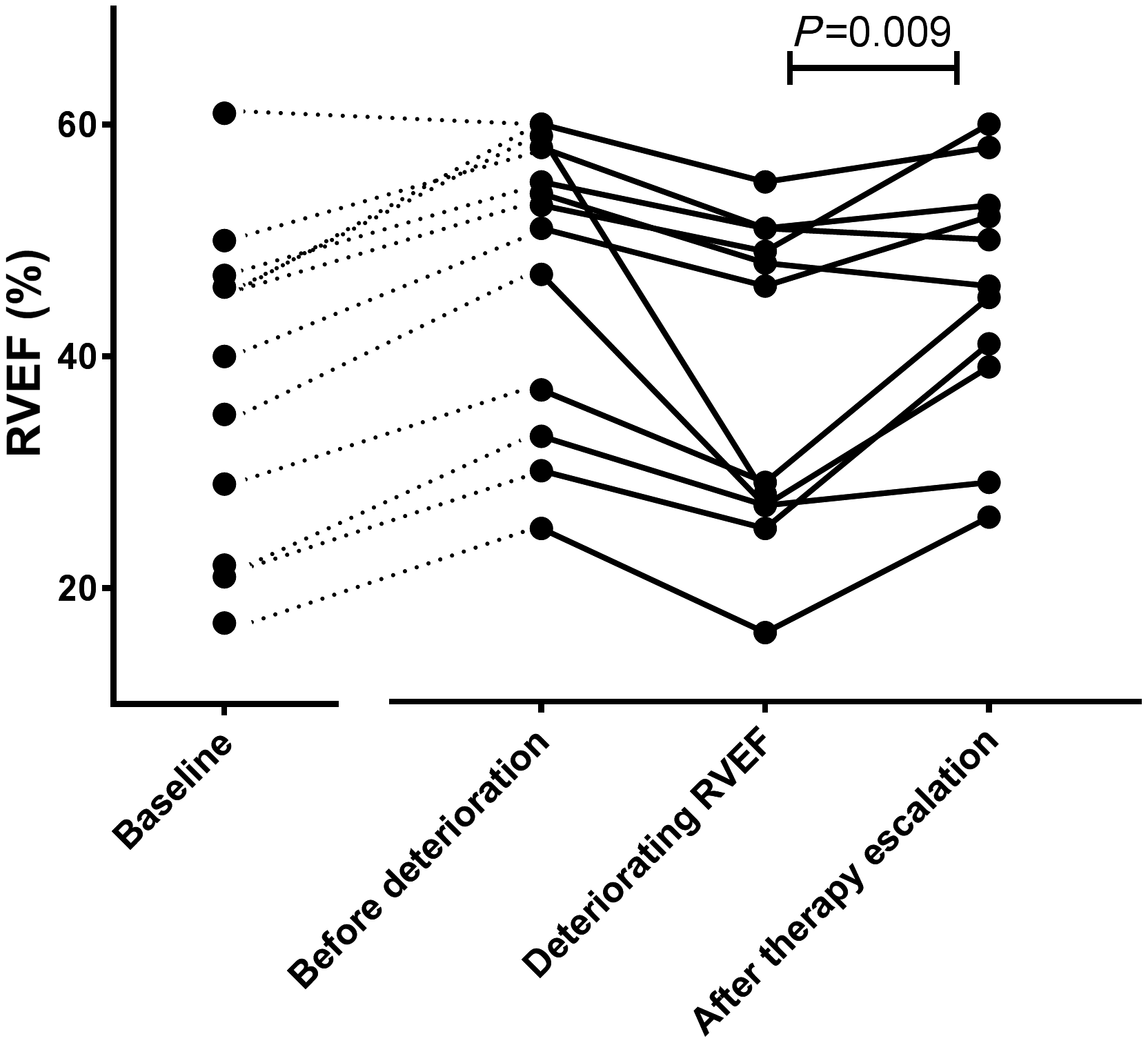
Average improvement in RVEF in the monotherapy group after treatment escalation was 6% versus 10% in the combination therapy group after adding intravenous therapy. After escalating therapy, no significant changes in RV volumes were observed but there was a significant increase in SV (p = 0.018, Fig. 2). Both NTproBNP levels (median value 526 ng/L versus 184 ng/L after treatment escalation) and 6MWD (467 (±102) m versus 514 (±69) m after escalation of therapy) did not significantly change. In addition, 83% of patients were in NYHA functional class I or II after treatment escalation compared with 67% at the time of deteriorating RVEF.
Changes in right ventricular (RV) volumes and stroke volume in patients with an unstable RV ejection fraction (RVEF). No change in RV volumes (end diastolic: RVEDV; end systolic: RVESV) but improvements in left ventricular stroke volume (LV SV) were observed.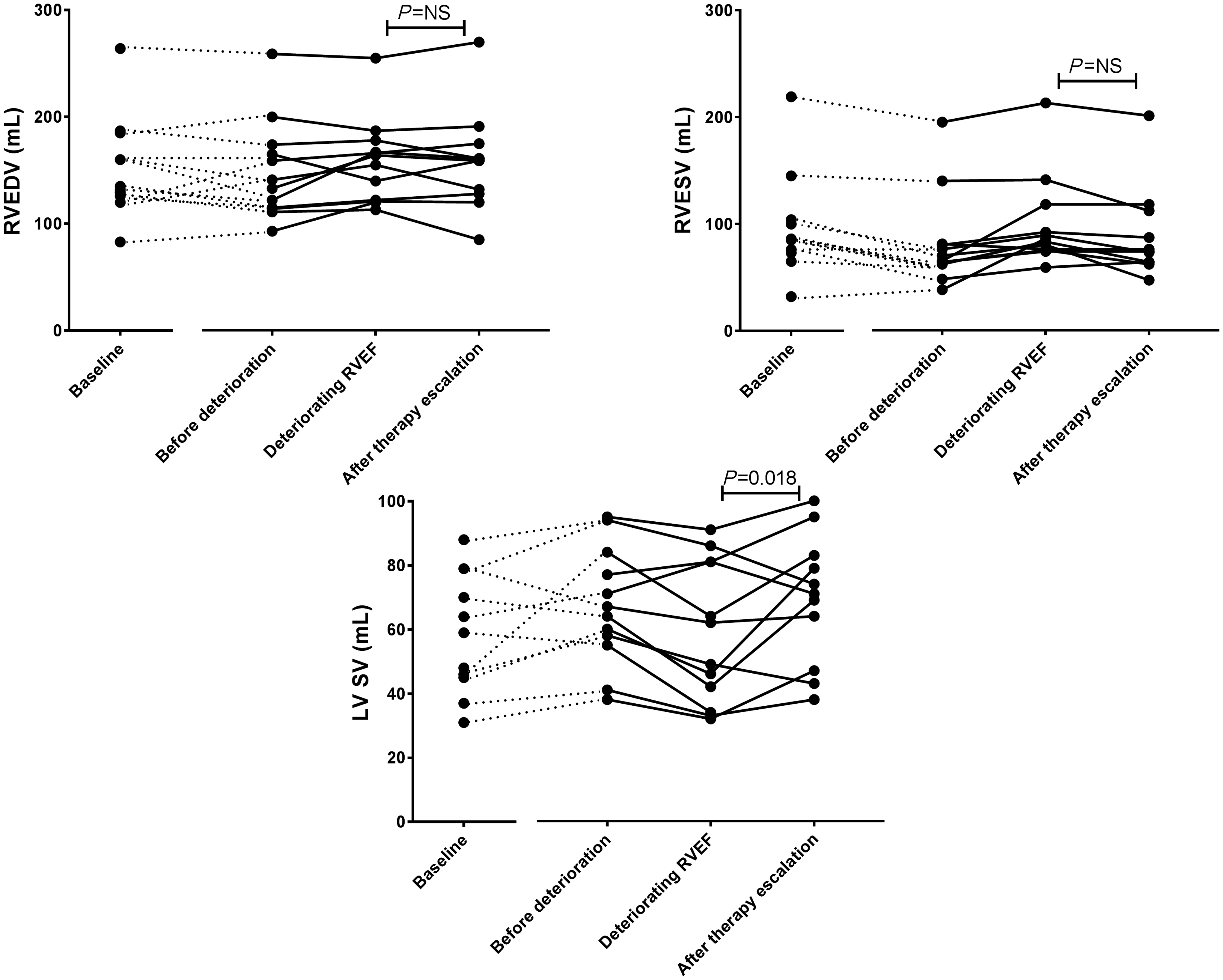
Individual patient data on RV function and volumes and follow-up data on haemodynamics, NYHA functional class and 6MWD for the total cohort can be found in the Supplementary Material.
Clinical worsening in patients with a stable RVEF
In total four patients presented with clinical worsening: three showed clinical worsening within the first four months and one patient at 24 months of follow-up. All four patients presented with clinical worsening despite a stable RVEF but three out of four patients had a low baseline RVEF (<35%). The clinical worsening events consisted of worsening NYHA functional class and a decrease in 6MWD of >15% in two patients (baseline RVEF 15% and 27%). The other two patients were admitted for signs and symptoms of RV failure. Of these, one patient showed a stable RVEF (decrease of 2%, baseline RVEF 61%) one month before hospitalization and eventually died, after refusing all PAH-related therapy. The other patient failed to improve after the initiation of combination therapy at baseline and was admitted for RV failure before four months of follow-up and subsequently triple therapy was started (baseline and four months RVEF 28%).
Low-risk criteria during unstable RVEF
Number of patients presenting with low-risk criteria at the time of an unstable right ventricular ejection fraction (decrease >3%).

Data are given as absolute numbers (%).
6MWD: six-minute walking distance; NTproBNP: N-terminal pro-brain natriuretic peptide
Protocol violations
Two patients who had no decrease in RVEF received add-on therapy due to an unsatisfactory clinical response. One patient had a low RVEF both at baseline and first follow-up (RVEF = 28%) and therapy was escalated due to signs and symptoms of RV failure at four month follow-up (previously described patient with clinical worsening). The other patient had no improvement in pulmonary hypertension-related symptoms after four months of follow-up (RVEF = 42%). In addition, one patient refused triple therapy despite a decline in RVEF (protocol violations) and on patient discontinued triple therapy due to significant side effects (treatment failure).
Discussion
In this study we showed that: 1) a decrease in RVEF can occur in seemingly clinically stable patients and 2) treatment escalation upon early decreases in RVEF can result in RV functional improvement and subsequent clinical stability. However, when RVEF is low at baseline but remains subsequently stable, clinical stability is not guaranteed.
To our knowledge, this is the first study exploring the effect of CMR-based treatment decisions on RV function and its impact on clinical worsening. RV function determines clinical outcome and survival in patients with IPAH.3,13–15 In an attempt to improve patient outcomes by preserving RV function, it therefore makes great sense to escalate therapy upon the detection of a decrease in RVEF, even if other clinical signs suggest disease stability. In order to explore the feasibility of using RVEF as a tool to guide treatment decisions we first had to assess whether changes in RVEF indeed occur in patients with otherwise stable disease and whether the course of RV failure can be altered by escalating therapy. Here we have shown that early decreases in RVEF indeed can occur at a time of apparent clinical stability. We have also shown that by adding PAH-specific drugs in these patients, RV function can be improved. In fact, escalating therapy guided by a deterioration in RVEF was associated with a favourable outcome, as no clinical worsening occurred in patients receiving additional therapy throughout the study.
Whilst a decrease in RVEF was found in several patients without other signs of clinical failure, we observed clinical worsening in four patients with a stable RVEF, three of whom initially presented with a RVEF <35%. This implies that a treatment strategy based on preventing a decrease in RVEF alone is insufficient to prevent clinical worsening when RV function is severely reduced at baseline. We argue that in order to prevent clinical worsening in those patients, treatment should be escalated not only upon a decrease in RVEF, but also when baseline RVEF is below a certain threshold. For this, RVEF thresholds that best predict clinical stability during follow up should be explored in future studies.
Clinical implications and future directions
The findings in the present study can be regarded as a first step towards the use of imaging techniques in monitoring and optimizing treatment for patients with PAH. However, before serial assessments of CMR-derived RVEF can be propagated to guide therapeutic decisions, the effectiveness of our treatment strategy should be prospectively compared with clinical judgment/available risk-assessment tables in an experimental setting.1,16,17 In addition, optimal timing of CMR performance in the follow-up of patients with PAH needs to be addressed in future studies.
The comparison between our CMR-based treatment strategy and existing risk-assessment strategies is especially relevant given the availability of several recent cohort studies demonstrating the clinical and prognostic relevance of guideline-derived risk assessment.12,18,19 These studies have consistently shown that achieving a ‘low-risk’ status confers survival benefits. Studies from the French registry and the COMPERA registry have used three non-invasive parameters (NYHA functional class, NTproBNP and 6MWD) and showed that these parameters could accurately identify patients at low risk of clinical failure both at baseline and at follow-up. 20 Of interest, 64% of our patients presented with ≥2 of these low-risk criteria at the time of a deterioration in RVEF.
From previous work we know that patients with a deteriorating RVEF are at risk for late disease progression, despite apparent clinical stability.8,13 However, whether patients with a deterioration in RVEF in this study would have eventually deteriorated clinically and thus whether our treatment strategy truly prevented clinical worsening cannot be ascertained form the present data and merits further exploration.
Study limitations
Although the majority of patients adhered to the study protocol, two patients received additional therapy without the presence of a deterioration in RVEF (protocol violations). However, these patients failed to achieve a clinically satisfactory response at four months of follow-up (clinical signs of RV failure or lack of improvement of PAH related symptoms) and therefore withholding additional PAH-specific therapy was not deemed ethical. Furthermore, in two patients therapy was not escalated despite a deterioration in RVEF due to either side effects or refusal of additional therapy. We did, however, consider those patients as having undergone escalation of therapy (intention to treat).
Also, although CMR-derived RVEF is currently considered the gold standard method for assessing RV function, we acknowledge that performing serial CMR evaluations in routine clinical practice may not be feasible for every pulmonary hypertension centre.
Conclusion
In conclusion, the present study showed that a deterioration of RVEF can occur in patients who appear clinically stable by conventional standards. Moreover, early escalation of therapy on the basis of CMR criteria results in a stabilization of RVEF. However, our treatment strategy was not sufficient to prevent clinical worsening in patients with a low baseline RVEF. Thus, an imaging derived treatment strategy seems feasible but should include specific thresholds below which treatment escalation is required.
Footnotes
Acknowledgements
All authors made substantial contribution to the concept, design, acquisition of data and data interpretation. All authors revised the article critically for important intellectual content and approved the version to be published. Ethical approval: see Methods section. Role of the sponsors: none of the sponsors had any role in the development, conduction or writing of this work nor in the decision to submit for publication. Prior to submission, United Therapeutics and Pfizer were given access to the results but could not make any contributions.
Declaration of conflicting interests
AEH, MCV, OAS, JAG, NW, JTM declare that there is no conflict of interest. HJB and AV received unrestricted research support outside this work from Actelion, Bayer and GSK. AV was supported by NWO-VICI (2002406) and the Dutch Heart Foundation.
Funding
This work was supported by United Therapeutics and Pfizer.
